International Journal of Ophthalmology and Clinical Research
Supratentorial Anaplastic Ependymoma with Bilateral Papilledema
Meghan Berkenstock1*, Matthew Keisling2, Jinglan Liu2, Judy Mae Pascasio2, Ayman Samkari3, Prithvi Narayan4, Melandee Brown4, Erica Poletto5, Christos D Katsetos2,6 and Myron Yanoff1
1Department of Ophthalmology, Drexel University College of Medicine, USA
2Department of Pathology and Laboratory Medicine, Drexel University College of Medicine, St. Christopher's Hospital for Children, USA
3Department of Pediatrics, Section of Oncology, Drexel University College of Medicine, St. Christopher's Hospital for Children, USA
4Department of Neurosurgery, Drexel University College of Medicine, St. Christopher's Hospital for Children, USA
5Department of Radiology, Drexel University College of Medicine, St. Christopher's Hospital for Children, USA
6Department of Pediatrics, Section of Neurology, Drexel University College of Medicine, St. Christopher's Hospital for Children, USA
*Corresponding author: Meghan Berkenstock, M.D, Department of Ophthalmology, Drexel University College of Medicine, 219 N. Broad St., Third Floor, Philadelphia, PA 19107, USA, Tel: 215-762-5626, Fax: 215-762-5600, E-mail: Meghan.Berkenstock@drexelmed.edu
Int J Ophthalmol Clin Res, IJOCR-2-025, (Volume 2, Issue 3), Case Report; ISSN: 2378-346X
Received: April 16, 2015 | Accepted: May 25, 2015 | Published: June 01, 2015
Citation: Berkenstock M, Keisling M, Liu J, Pascasio JM, Samkari A, et al. (2015) Supratentorial Anaplastic Ependymoma with Bilateral Papilledema. Int J Ophthalmol Clin Res 2:025. 10.23937/2378-346X/1410025
Copyright: © 2015 Berkenstock M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Ependymomas constitute the third most common histological type of CNS tumor in children. Compared to classic cellular ependymoma (WHO grade II), anaplastic ependymoma is a more aggressive and less common subtype of ependymal neoplasm. Here we report a case of a supratentorial anaplastic ependymoma in a child presenting with a right sixth nerve palsy with bilateral papilledema. To our knowledge, the ophthalmic findings have not been previously reported in the context of this tumor type.
Keywords
Ependymoma, Papilledema, Sixth nerve palsy
Case Report
An 11-year-old Caucasian male with no past medical history presented to his pediatrician with a three-week history of daily, morning headaches accompanied by vomiting. The patient was also noted to have asymmetric eye movements and began to hold books close to his face to read. The patient experienced diplopia when looking to the right and needed guidance to walk in unfamiliar places. His ocular history was remarkable for astigmatism corrected with spectacles. The patient was referred to an outside hospital for a CT of the head and was subsequently transferred to our facility for treatment.
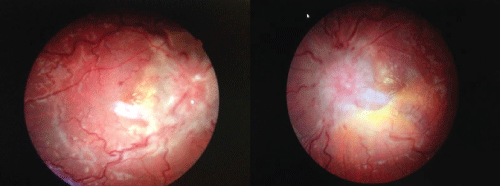
On examination, his best corrected visual acuity was 20/80 and 20/100, and pupils were 8 mm and largely unreactive without an afferent pupillary defect. Intraocular pressures were within normal limits. On extraocular motility testing, no abduction was noted in the right eye, while all other versions and ductions were full. Confrontation visual fields showed a right homonymous hemianopia. His refraction showed a hyperopic shift. The anterior chamber exam was within normal limits. On dilated fundoscopy, florid papilledema, peripapillary hemorrhages, cotton wool spots, and a macular star were noted bilaterally (Figure 1). All retinal veins were dilated and tortuous bilaterally.
.
Figure 1: Extensive bilateral papilledema due to mass effect. Note macular-star exudates and tortuous, dilated vessels due to elevated caval venous pressure.
View Figure 1
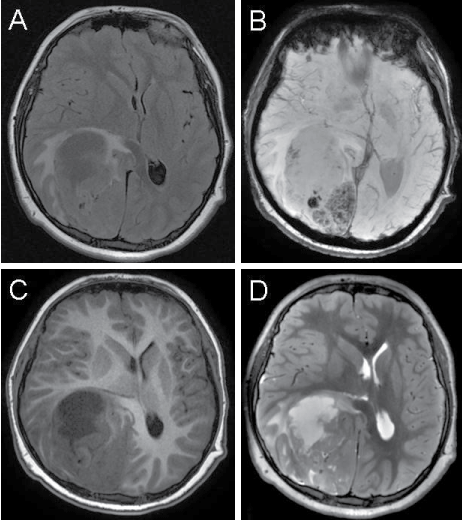
An MRI of the brain with and without contrast showed a large 8.5 � 5.4 � 5.8cm heterogeneous, centrally necrotic mass. The lesion involved the majority of the right temporo-parieto-occipital lobes with significant surrounding edema (Figure 2). The posterior horn of the right lateral ventricle and the third ventricle were compressed, with anterior displacement of the corpus callosum and the adjacent midbrain. A 1.6 cm midline shift to the left was noted without evidence of overt herniation. The patient underwent occipital craniotomy with gross-total resection of the tumor followed by subsequent chemoradiation. Post-operative brain MRI with and without contrast showed post-operative changes with good decompression and improved midline shift and no evidence of residual tumor. Clinically, the patient's visual acuity improved to 20/20 and 20/30; however a bitemporal hemianopsia remained.
.
Figure 2: MRI of the brain showing 8.5 � 5.4 � 5.8cm heterogeneous, centrally necrotic mass at the right temporo-parieto-occipital junction. Panel A depicts extensive vasogenic edema surrounding the mass, with resulting midline shift, seen on fluid-attenuated inversion recovery (FLAIR). Panel B depicts foci of susceptibility artifact seen within the posterior aspect of the lesion on susceptibility weighted imaging (SWI), which can be seen in the setting of hemorrhage or lesional mineralization. Panels C and D depict the centrally necrotic nature of the anterior aspect of the lesion on T1-weighted (C) and T2-weighted (D) imaging. Panel E depicts the rim enhancement of the centrally necrotic anterior aspect and diffuse, heterogeneous enhancement of the posterior aspect of the lesion on post-contrast T1-weighted imaging.
View Figure 2
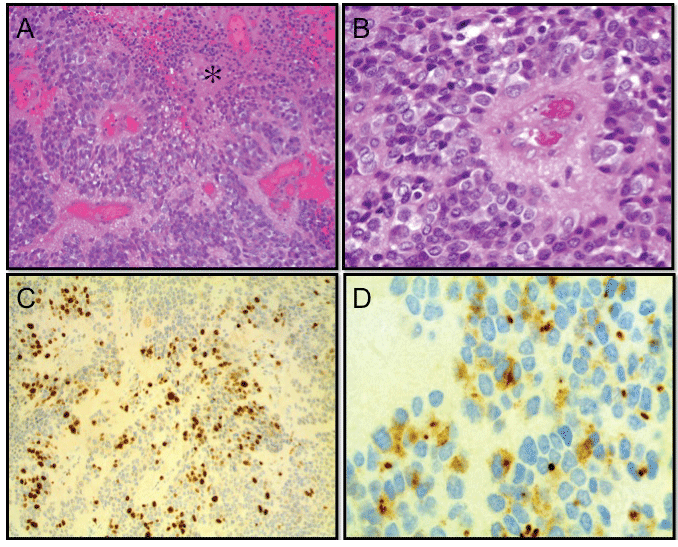
Histopathological evaluation of the surgically excised lesional tissue revealed a poorly differentiated, highly cellular neuroepithelial neoplasm with perivascular pseudorosettes, lack of true rosettes, high mitotic rate, microvascular proliferation, and necrosis consistent with an anaplastic ependymoma (WHO grade III, ICD-O 9392/3) (Figure 3). The Ki-67 proliferative index was high (≥30%) (Figure 3C). Ependymal tumor phenotype was confirmed by intra-cytoplasmic dot-like staining for epithelial membrane antigen (Figure 3D) [1]. Collectively, immunohistochemical studies did not support the diagnosis of a CNS primitive neuroectodermal tumor (CNS-PNET), an embryonal tumor with abundant neuropil and true rosettes (ETANTR), or an atypical teratoid/rhabdoid tumor (AT/RT) [1]. Genetic evaluation revealed genomic changes XX[cp12] similar to those previously reported in three anaplastic ependymoma cases (two infratentorial and one supratentorial) [2].
.
Figure 3: Panels A and B depict low and high power fields featuring poorly differentiated, highly cellular tumor with perivascular pseudorosettes, vascular proliferation, and geographic necrosis (asterisk in panel A). Panel C shows widespread Ki-67 labeling (brown) of tumor cell nuclei (Ki-67 proliferative index 30%). Panel D depicts cytoplasmic dot-like staining (brown) for epithelial membrane antigen consistent with an ependymal tumor cell phenotype. A, B: Hematoxylin and eosin staining. C, D: Avdin-biotin comple peroxidase with hematoxylin counterstain. Images in panels A and C were taken with a 20x objective and images in panels B and D were obtained using 40 � and 100 � objectives respectively.
View Figure 3
Discussion
This case report illustrates an instance of a supratentorial anaplastic ependymoma in a pre-adolescent male presenting with a right sixth nerve palsy with bilateral papilledema. To our knowledge, this ophthalmological presentation has not been previously reported in this clinicopathological setting.
Ependymomas account for 5-12% of primary CNS tumors and are the third most common CNS tumor in children [3,4]. The World Health Organization (WHO) grading scale for ependymal tumors is based on histological features corresponding to WHO grades I through III), with higher stages showing less differentiation, higher mitotic activity, and a tendency to recur after excision [5]. Anaplastic ependymoma is the least common and most aggressive ependymal neoplasm, being considered as high grade (WHO Grade III) [5]. The diagnosis of anaplastic ependymoma (WHO grade III) in the index case was made with the caveat that (a) necrosis of the non-palisading type is also common in grade II ependymomas and that the distinction between non-palisading and palisading necrosis may be subjective; (b) microvascular proliferation is not a required criterion for grade III tumors; and (c) there is no precise cut-off of mitotic counts that would distinguish grade II from grade III tumors of the ependymoma group [1]. Unlike the present case, most anaplastic ependymomas involve the posterior fossa.
Ependymomas comprise multiple genetically distinct disease phenotypes and it is thought that supratentorial ependymomas are largely driven by EPHB2 gene amplification [6]. Presenting symptoms are secondary to obstructive hydrocephalus. Treatment focuses on achieving near total gross surgical excision and on adjuvant radiation [3,7]. Approximately 65% of patients with ependymoma are cured by surgical resection and adjuvant radiotherapy [8]. Conversely, recurrent disease is not curable in most cases, but prolonged survival disease-free intervals can be achieved following repeat surgical resection and chemotherapy and/or radiotherapy [9,10].
A recent Children's Oncology Group (COG) study based on a large prospective trial for newly diagnosed ependymoma demonstrated that the extent of surgical resection is a crucial and independent determinant of disease control [11]. Patients who underwent either a near-total resection (i.e., <5mm2 residual tumor) or macroscopic gross total resection had a significantly longer 3-year event-free survival compared to those who had only partial surgical resection, irrespective of tumor histological grade or anatomical location [11].
Pathophysiology of Ophthalmic Findings
To the best of our knowledge, the ophthalmic findings have not been previously reported in the context of this tumor type. Our patient displayed signs of increased intracranial pressure, namely an abducens palsy, due to the predominantly right-sided intracranial pressure elevation leading to compression of the homolateral VIth cranial nerve. Concomitantly, the increased intracranial pressure also decreases caval venous drainage, causing stasis retinopathy in the retinal vasculature. With the increased resistance, Starling forces reverse with hydrostatic pressure forcing fluid out of capillaries. Clinically, this results in a macular scar and dot-blot hemorrhages. A final consequence of outflow resistance is papilledema. Cerebrospinal fluid egress into the arachnoid villi, and subsequently into dural veins is also limited. Our current understanding of papilledema is that it represents a transfer of intracranial pressure to the optic nerve through the subarachnoid space in the surrounding meningeal coverings [12]. This causes decreased axoplasmic flow and nerve distention. An alternative theory to explain unilateral papilledema in cases of increased intracranial pressure is a compartment-like syndrome that develops in the meninges [12]. On fundoscopic examination, this translates into flame shaped hemorrhages in the nerve fiber layer, optic nerve head elevation, and cotton-wool spots representing nerve fiber layer infarcts. This report raises awareness of the ophthalmic manifestations and sequelae incurred in the setting of supratentorial ependymomas. It further calls attention to the importance of recognizing the potential effects of intracranial space-occupying lesions and increased intracranial pressure on ocular structures- most commonly papilledema with an ipsilateral sixth nerve palsy [13,14].
References
-
Burger PC, Scheithauer BW (2012) Diagnostic Pathology: Neuropathology. Lippincott Williams & Wilkins (Amirsys edn): 106-107.
-
Vagner-Capodano AM, Zattara-Cannoni H, Gambarelli D, Figarella-Branger D, Lena G, et al. (1999) Cytogenetic study of 33 ependymomas. Cancer Genet Cytogenet 115: 96-99.
-
Venkatramani R, Dhall G, Patel M, Grimm J, Hawkins C, et al. (2012) Supratentorial ependymoma in children: to observe or to treat following gross total resection? Pediatr Blood Cancer 58: 380-383.
-
Agaoglu FY, Ayan I, Dizdar Y, Kebudi R, Gorgun O, et al. (2005) Ependymal tumors in childhood. Pediatr Blood Cancer 45: 298-303.
-
Raghunathan A, Wani K, Armstrong TS, Vera-Bolanos E, Fouladi M, et al. (2013) Histological predictors of outcome in ependymoma are dependent on anatomic site within the central nervous system. Brain Pathol 23: 584-594.
-
Johnson RA, Wright KD, Poppleton H, Mohankumar KM, Finkelstein D, et al. (2010) Cross-species genomics matches driver mutations and cell compartments to model ependymoma. Nature 466: 632-636.
-
Applegate GL, Marymont MH (1998) Intracranial ependymomas: a review. Cancer Invest 16: 588-593.
-
van Veelen-Vincent ML, Pierre-Kahn A, Kalifa C, Sainte-Rose C, Zerah M, et al. (2002) Ependymoma in childhood: prognostic factors, extent of surgery, and adjuvant therapy. J Neurosurg 97: 827-835.
-
Messahel B, Ashley S, Saran F, Ellison D, Ironside J, et al. (2009) Relapsed intracranial ependymoma in children in the UK: patterns of relapse, survival and therapeutic outcome. Eur J Cancer 45: 1815-1823.
-
Zacharoulis S, Ashley S, Moreno L, Gentet JC, Massimino M, et al. (2010) Treatment and outcome of children with relapsed ependymoma: a multi-institutional retrospective analysis. Childs Nerv Syst 26: 905-911.
-
Gajjar A, Packer RJ, Foreman NK, Cohen K, Haas-Kogan D, et al. (2013) COG Brain Tumor Committee. Children's Oncology Group's 2013 blueprint for research: central nervous system tumors. Pediatr Blood Cancer 60: 1022-1026.
-
Killer HE, Jaggi GP, Miller NR (2009) Papilledema revisited: is its pathophysiology really understood? Clin Experiment Ophthalmol 37: 444-447.
-
Alexiou GA, Moschovi M, Stefanaki K, Panagopoulos D, Tsotra M, et al. (2013) Supratentorial ependymomas in children: Analysis of nine cases. J Pediatr Neurosci 8: 15-18.
-
Iddrissu M, Dakurah T, Wepeba G (2005) Anaplastic ependymoma of the fourth ventricle causing obstrictive hydrocephalus. Ghana Med J 39: 33-36.





