International Journal of Ophthalmology and Clinical Research
Spectral Domain Optical Coherence Tomography Based Alterations in Macular Thickness and Inner Segment Ellipsoidare Associated with Severity of Diabetic Retinopathy
Sandeep Saxena1*, Khushboo Srivastav1 and LeventAkduman2
1Retina Service, Department of Ophthalmology King George's Medical University, India
2Vitreoretinal and uveitis service, Department of Ophthalmology, Eye institute, St. Louis University School of Medicine, USA
*Corresponding author: Prof. Sandeep Saxena (M.S., F.R.C.S.Ed), Department of Ophthalmology, King George's Medical University, Lucknow, 226003, India, Tel: +91-9415160528; E-mail: sandeepsaxena2020@yahoo.com
Int J Ophthalmol Clin Res, IJOCR-2-007, (Volume 2, Issue 1), Research Article; ISSN: 2378-346X
Received: September 21, 2014 | Accepted: January 11, 2015 | Published: January 14, 2015
Citation: Saxena S, Srivastav K, Akduman L (2015) Spectral Domain Optical Coherence Tomography Based Alterations in Macular Thickness and Inner Segment Ellipsoidare Associated with Severity of Diabetic Retinopathy. Int J Ophthalmol Clin Res 2:007. 10.23937/2378-346X/1410007
Copyright: ©2015 Saxena S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objective: To study the association of spectral domain optical coherence tomography (SD-OCT) based alterations in macular thickness and inner segment-ellipsoid band (ISel) with severity of retinopathy in cases of type 2 diabetes mellitus.
Methods: Two hundred thirty eight consecutive cases and seventy nine healthy controls were included. Eyes of cases were divided into three groups according to ETDRS classification: diabetes without retinopathy (No DR) (n=79), non-proliferative diabetic retinopathy (NPDR) with macular edema (n=79), and proliferative diabetic retinopathy (PDR) with macular edema (n=80). Visual acuity was assessed using logMAR scale. Fasting and post prandial blood glucose and glycosylated hemoglobin were estimated as per standard protocol. All the study subjects were evaluated using SD-OCT using the macular cube 512�128 feature. Central subfield thickness ( μm), cube average thickness ( μm) and ISel disruption in foveal region were noted. The integrity of ISel was evaluated by two experienced observers masked to the status of diabetic retinopathy. Chi�square test, analysis of variance, unpaired t test and pearson correlation analysis were used to assess association between the study variables.
Results: Statistically significant decrease in visual acuity was found between the study groups (p< 0.0001). Significant negative correlation of visual acuity was found with HbA1c, central sub field thickness and ISel disruption (p< 0.001).Significant increase in central subfield thickness and cube average thickness were found between the study groups (p< 0.001). ISel disruption increased with increase in severity of diabetic retinopathy [(NPDR, n=55) versus (PDR, n=68)] (p< 0.001).Statistically significant difference in the mean central subfield thickness in the cases with intact and disrupted ISel was observed in NPDR and PDR groups respectively.
Conclusion: Diabetic macular edema as assessed by central subfield thickness, cube average thickness and ISel disruption, on SD-OCT, is associated with increased severity of diabetic retinopathy. For the first time, it has been demonstrated that with progression of retinopathy from non-proliferative to proliferative stage, increase in central subfield thickness is associated with ISel disruption.
Keywords
Diabetic retinopathy, Spectral domain optical coherence tomography, Photoreceptor, Inner segment ellipsoid, Central subfield thickness, Cube average thickness
Introduction
The prevalence of diabetes mellitus is attaining epidemic proportions worldwide [1,2]. It is estimated that 382 million people had diabetes mellitus in 2013. This number is expected to rise to 592 million by 2035 [3]. Macular edema is the most common cause of visual loss in diabetic retinopathy [4]. The Wisconsin Epidemiological Study of Diabetic Retinopathy reported a prevalence of diabetic macular edema, with duration of diabetes 20 years or more, as 29% in younger onset diabetics and 28% in older onset [5].
Optical Coherence Tomography (OCT) is a useful investigative tool for quantifying and classifying macular edema [6-8]. Optical coherence tomography allows us to quantify macular thickness in diabetic retinopathy with excellent reproducibility. Optical coherence tomography is able to detect sight-threatening macular edema with great reliability [9]. Diabetic retinopathy can result in structural changes in retina which correlate with severity of retinopathy. Photoreceptor dysfunction may be a significant predictor of visual acuity in such patients [10-13]. Inner segment-ellipsoid band disruption has been demonstrated to be an important predictor of visual acuity in diabetic macular edema [14].
Macular thickness parameters on SD-OCT have been correlated with severity of diabetic retinopathy.9In the present study, association of macular thickness and ISel disruption with severity of diabetic retinopathy was evaluated for the first time.
Materials and Method
Our study had institutional review board clearance and was performed in accordance to the tenets of the Helsinki declaration. In this tertiary care center based prospective cross sectional study, 238 consecutive cases and 79 healthy controls (presenting for refraction) were included after an informed voluntary consent. Cases were divided into three groups: patients with diabetes without retinopathy (No DR) (n=79), non-proliferative diabetic retinopathy (NPDR) with diabetic macular edema (n=79), and proliferative diabetic retinopathy (PDR) with diabetic macular edema (n=80) according to the ETDRS classification [15]. The morphological assessment of diabetic macular edema was done on the basis of presence or absence of clinically significant macular edema, which may be characterized as retinal thickening or adjacent hard exudate that involves or threatens the center of the macula16 [16].
The eye with a more severe form of diabetic retinopathy were included. Ocular diseases which could affect the retinal vascular pathology (hypertensive retinopathy, age-related macular degeneration), any previous ophthalmic surgical or laser interventions, uncontrolled diabetes based upon HbA1c levels (>9%), fluorescein angiography suggestive of ischemic maculopathy, cases with signal strength 5 or below on OCT examination and cases taking any mineral supplements or antioxidants were excluded. The best-corrected visual acuity was recorded on log MAR scale. Information regarding patient�s age, gender, and disease duration, status of retinopathy and glycemic control was recorded. Slit lamp biomicroscopic and dilated ophthalmoscopic examination were performed. Fluorescein angiography was performed in cases with diabetic retinopathy. Subsequently, all the study subjects were evaluated using SD-OCT [Cirrus High Definition OCT (Carl Zeiss Meditec Inc., CA, U.S.A]. Every study subject underwent macular thickness analysis using macular cube 512 � 128 feature. Central subfield thickness ( μm) and cube average thickness ( μm) were noted. Central subfield thickness was defined as thickness of the central circle in the circular map known as the ETDRS Grid. Cube average thickness was defined as an overall average thickness for the internal limiting membrane-retinal pigment epithelium tissue layer over the entire 6 x 6 mm square scanned area. Inner segment-ellipsoid band was defined as an outer highly reflective band next to retinal pigment epithelium located at the inner segment ellipsoids [17]. Inner segment-ellipsoid band disruption was defined as break in its continuity in the foveal region (Figure 1). Inner segment-ellipsoid band was studied using horizontal and vertical SD-OCT scans passing through the fovea. The integrity of this layer was evaluated by two experienced observers masked to the status of diabetic retinopathy.
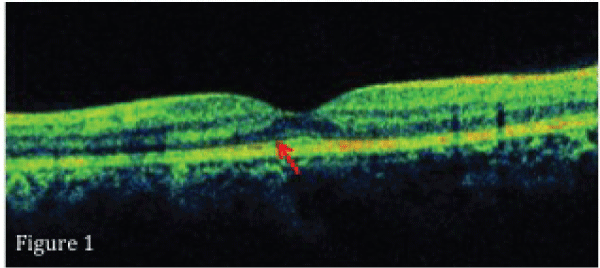
Figure 1: ISel disruption is seen in the foveal region (arrow).
View Figure 1
Fasting and post prandial blood glucose and glycosylated haemoglobin was estimated as per standard protocol.
Data is presented as mean � standard error (SD). The continuous variables (age, central subfield thickness, cube average thickness and visual acuity on logMAR scale) of the study groups - NPDR and PDR were compared by one factor analysis of variance (ANOVA) and the significance of mean difference between the groups was done by Tukey�s test. The discrete (categorical) variables (sex, ISel disruption) were compared by chi-square (χ2) test. Interobserver correlation was computed using analysis of variance. Unpaired t test was used to test the significance of difference between two mean values. Pearson correlation analysis was used to assess association between the study variables. P< 0.05 was considered statistically significant. All analyses were performed on STATISTICA (window version 6.0) software.
Results
There were 163 males and 154 females. The mean age of patients was 52.4 � 4.36 years, 53.5 � 3.68 years and 56.2 � 4.12 years in patients of diabetes without retinopathy, NPDR and PDR groups respectively. No statistical difference was found among the age and sex of the three groups (p>0.05).
Summary of glycosylated hemoglobin (HbA1c), visual acuity (logMAR), central subfield thickness, cube average thickness and ISel disruption have been shown in Table 1.
![]()
Table 1: Summary of glycosylated hemoglobin, visual acuity (logMAR), central
subfield thickness, cube average thickness and inner segment ellipsoid band
(ISel) disruption in the study groups.
View Table 1
The central subfield thickness increased with increase in the severity of retinopathy. From the analysis of variance it was obtained that central subfield thickness was significantly different between study groups (p< 0.001). From multiple comparisons amongst the study groups, it was obtained that the central subfield thickness was significantly different between controls and PDR group (p < 0.001), No DR and NPDR (p< 0.001) and NPDR and PDR (p=0.001). For other pairs of group, no statistically significant difference was obtained.
The cube average thickness increased with increase in the severity of retinopathy. From the analysis of variance, it was obtained that cube average thickness was significantly different between study groups (p=0.001). From multiple comparisons amongst the study groups, it was obtained that cube average thickness was significantly different between controls and PDR (p< 0.001) as well as No DR and PDR group (p=0.019). For other pairs of group, no statistically significant difference was obtained.
An interobserver correlation coefficient for disruption of ISel was 0.94 (95% confidence interval, 0.93 to 0.96). ISel disruption increased with increase in severity of diabetic retinopathy [(NPDR, n=55) versus (PDR, n=68)] (χ2=5.368, p< 0.05). Intact ISel was observed in all the cases of No DR and controls.
In the NPDR group, mean central subfield thickness in the cases with ISel disruption (n=55) and in the cases with intact ISel (n=24) was 328.53 � 90.30 μm and 278.6 � 83.50 μm respectively. Unpaired t test revealed a significant difference between the two mean values (p=0.02).
In the PDR group, mean central subfield thickness in the cases with ISel disruption (n=68) and in the cases with intact ISel (n=12) was 421.36 � 93.55 μm and 338.52 � 90.32 μm respectively. Unpaired t test revealed a significant difference between the two mean values (p=0.005).
Visual acuity decreased with increase in severity of retinopathy. On comparing visual acuity, ANOVA revealed significant difference amongst study groups (F=23.02, p< 0.0001).Pearson correlation analysis revealed statistically significant correlation of visual acuity (log MAR) with HbA1c (r=0.441; p< 0.001), central sub field thickness (r=0.874; p< 0.001) and ISel disruption(r=0.48; p< 0.001).
Discussion
The present study evaluated the correlation of macular thickness and ISel disruption with severity of diabetic retinopathy.
Significant decrease in visual acuity was observed with increase in the severity of retinopathy, similar to studies concluded by Falkensteinet al. [18].
Severity of diabetic retinopathy has been reported to correlate with macular thickness parameters on SD-OCT. Mean macular thickness, retinal thickness, foveal thickness and central macular thickness have been shown to correlate with severity of diabetic retinopathy and visual acuity [19-24]. In the present study, statistically significant association of central subfield macular thickness and cube average thickness with severity of retinopathy was observed.
Importance of vascular endothelial growth factor (VEGF) and intercellular adhesion molecule-1 (ICAM-1) in the development of diabetic complications is well documented. . Our earlier study demonstrated that serum levels of VEGF and ICAM-1 increase significantly with severity of diabetic retinopathy. Increased levels of ICAM-1 cause vascular endothelial cell damage, retinal ischaemia and up regulation of VEGF [10]. Raised levels of VEGF and ICAM-1 lead to blood retinal barrier breakdown with resultant diabetic macular edema.
In the present study, significant increase in ISel disruption was observed with increase in the severity of diabetic retinopathy from non�proliferative to proliferative stage. Significant difference in the mean central subfield thickness in the cases with intact and disrupted ISel was observed in NPDR and PDR groups respectively. It was concluded that increase in central subfield thickness was associated with increase in ISel disruption in diabetic macular edema. This association has been established for the first time.
Ethical statement
Study had institutional review board clearance and was performed in accordance to the tenets of the Helsinki declaration.
References
-
Bjork S, Kapur A, King H, Nair J, Ramachandran A (2003) Global policy: aspects of diabetes in India. Health Policy 66: 61-72.
-
King H, Rewers M (1991) Diabetes in adults is now a Third World problem. The WHO Ad Hoc Diabetes Reporting Group. Bull World Health Organ 69: 643-648.
-
Guariguata L, Whiting DR2, Hambleton I3, Beagley J4, Linnenkamp U4, et al. (2014) Global estimates of diabetes prevalence for 2013 and projections for 2035. Diabetes Res Clin Pract 103: 137-149.
-
Sharma S, Oliver-Fernandez A, Liu W, Buchholz P, Walt J (2005) The impact of diabetic retinopathy on health-related quality of life. Curr Opin Ophthalmol 16: 155-159.
-
Patz A, Schatz H, Berkow JW, Gittelsohn AM, Ticho U (1973) Macular edema--an overlooked complication of diabetic retinopathy. Trans Am Acad Ophthalmol Otolaryngol 77: OP34-42.
-
(1981) Diabetic retinopathy study. Report Number 6. Design, methods, and baseline results. Report Number 7. A modification of the Airlie House classification of diabetic retinopathy. Prepared by the Diabetic Retinopathy.Invest Ophthalmol Vis Sci 21: 1-226.
-
Otani T, Kishi S, Maruyama Y (1999) Patterns of diabetic macular edema with optical coherence tomography.Am J Ophthalmol 127: 688-693.
-
Gupta V, Gupta A, Dogra M (2010) Introduction to OCT, Atlas- OCT of macular diseases and glaucoma. 3-46.
-
Goebel W, Kretzchmar-Gross T (2002) Retinal thickness in diabetic retinopathy: a study using optical coherence tomography (OCT). Retina 22: 759-767.
-
Jain A, Saxena S, Khanna VK, Shukla RK, Meyer CH (2013) Status of serum VEGF and ICAM-1 and its association with external limiting membrane and inner segment-outer segment junction disruption in type 2 diabetes mellitus.Mol Vis 19: 1760-1768.
-
Uji A, Murakami T, Nishijima K, Akagi T, Horii T, et al. (2012) Association between hyperreflective foci in the outer retina, status of photoreceptor layer, and visual acuity in diabetic macular edema. Am J Ophthalmol 153: 710-717, 717.
-
Yanyali A, Bozkurt KT, Macin A, Horozoglu F, Nohutcu AF (2011) Quantitative assessment of photoreceptor layer in eyes with resolved edema after pars planavitrectomy with internal limiting membrane removal for diabetic macular edema. Ophthalmologica 226: 57-63.
-
Shin HJ, Lee SH, Chung H, Kim HC (2012) Association between photoreceptor integrity and visual outcome in diabetic macular edema. Graefes Arch Clin Exp Ophthalmol 250: 61-70.
-
Maheshwary, Oster SF, Yuson RMS, Cheng L, Mojana F, et al. (2010) The association between percent disruption of the photoreceptor inner segment-outer segment junction and visual acuity in diabetic macular edema. Am Jophthalmol 150: 63-67.
-
ETDRS Research Group (1984) Grading Diabetic Retinopathy from Stereoscopic Color Fundus photograph: ETDRS report number 10. Ophthalmology 91:1464-1474.
-
Gangnon RE, Davis MD, Hubbard LD, Aiello LM, Chew EY, et al. (2008) A severity scale for diabetic macular edema developed from ETDRS data. Invest Ophthalmol Vis Sci 49: 5041-5047.
-
Curcio CA, Messinger JD, Sloan KR, Mitra A, McGwin G, et al. (2011) Human chorioretinal layer thicknesses measured in macula-wide, high-resolution histologic sections. Invest Ophthalmol Vis Sci 52: 3943-3954.
-
Falkenstein IA, Cochran DE, Azen SP, Dustin L, Tammewar AM, et al. (2008) Comparison of visual acuity in macular degeneration patients measured with snellen and early treatment diabetic retinopathy study charts. Ophthalmology 115: 319-323.
-
Hee MR, Puliafito CA, Duker JS, Reichel E, Coker JG, et al. (1998) Topography of diabetic macular edema with optical coherence tomography. Ophthalmology 105: 360-370.
-
Oshima Y, Emi K, Yamanishi S, Motokura M (1999) Quantitative assessment of macular thickness in normal subjects and patients with diabetic retinopathy by scanning retinal thickness analyser. Br J Ophthalmol 83: 54-61.
-
Goebel W, Kretzchmar-Gross T (2002) Retinal thickness in diabetic retinopathy: a study using optical coherence tomography (OCT). Retina 22: 759-767.
-
Sanchez-Tocino H, Alvarez-Vidal A, Maldonado MJ, Moreno-Montanes J, Garcia-Layana A (2002) Retinal thickness study with optical coherence tomography in patients with diabetes. Invest Ophthalmol Vis Sci 43: 1588-1594.
-
Alkuraya H, Kangave D, El-Asrar AMA (2005) The correlation between optical coherence tomographic features and severity of retinopathy, macular thickness and visual acuity in diabetic macular edema. Int Ophthalmol26: 93-99.
-
Diabetic Retinopathy Clinical Research Network, Browning DJ, Glassman AR, Aiello LP, Beck RW, et al.(2007) Relationship between optical coherence tomography-measured central retinal thickness and visual acuity in diabetic macular edema. Ophthalmology 114: 525-536.





