International Journal of Neurology and Neurotherapy
Contralateral Basal Ganglia Atrophy in Acquired Hemichorea-Hemiballism
Zaheer F1* , Sudhakar P2, Escott E3,4 and Cambi F2
1Department of Neurology, Baylor College of medicine, Michael E DeBakey VA Medical Center, Houston, USA
2Department of Neurology, University of Kentucky, Lexington, USA
3Department of Radiology, University of Kentucky, Lexington, USA
4Department of Otolaryngology, head and neck surgery, University of Kentucky, Lexington, KY
*Corresponding author:
Fariha Zaheer, Department of Neurology, Baylor College of medicine, Michael E DeBakey VA Medical Center, Houston, TX, USA, E-mail: Fariha.Zaheer@bcm.edu
Int J Neurol Neurother, IJNN-1-004, (Volume 1, Issue 1), Case Report; ISSN: 2378-3001
Received: September 06, 2014 | Accepted: October 17, 2014 | Published: October 20, 2014
Citation: Zaheer F, Sudhakar P, Escott E, Cambi F (2014) Contralateral Basal Ganglia Atrophy in Acquired Hemichorea-Hemiballism. Int J Neurol Neurother 1:004. 10.23937/2378-3001/1/1/1004
Copyright: ©2014 Zaheer F, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Hemichorea-Hemiballism (HCHB) is a hyperkinetic condition characterized by abnormal, migratory, continuous, non-patterned movements of one side of the body. It results from involvement of contralateral basal ganglia that may be affected by metabolic, neoplastic, infectious, autoimmune [1], toxic or neurodegenerative processes [2]. The most common cause is ischemia from a focal vascular lesion [3]. Non-ketotic hyperglycemia has been reported as the second most common cause of HCHB in the Asian population [4]. It is usually seen in the elderly and has a female preponderance. The average reported age is 73 years. HCHB has been reported in patients with chronic diabetes and also in those with new onset hyperglycemia [5-7]. We describe a patient with HCHB in the setting of poorly controlled chronic diabetes, who also demonstrated basal ganglia atrophy on follow-up imaging. Basal ganglia atrophy in HCHB secondary to hyperglycemia is rarely reported.
Case Report
A 58 year old diabetic male was admitted to the neurology service with a 6 weeks history of abnormal left sided involuntary movements. At onset, he experienced mild involuntary movements of the left arm that improved slightly, but then worsened within the next 2 weeks. Then it gradually progressed to involve his left leg. The movements were constant, disabling and persistent even during sleep. On examination he had chorea, ballismus and intermittent dystonic posturing of the left arm and leg.
Unenhanced CT head revealed increased density in the head of right caudate nucleus and subtle increased density in the lateral aspect of the right putamen. The right lentiform nucleus appeared smaller than the left. Contrast enhanced brain MRI revealed T1 shortening (hyperintensity) within the head of the right caudate nucleus and putamen, which was more pronounced along the lateral aspect. The axial T2-weighted image revealed subtle hyper intensity in the putamen while the caudate head had a normal signal. Vascular imaging was unremarkable. His hemoglobin A1C was 12.7 % and plasma osmolality was 301mOsm/kg (checked after he was hydrated in the emergency department). The provisional diagnosis was HBHC due to non-ketotic hyperosmolar hyperglycemia.
He had already failed a trial of muscle relaxants and benzodiazepines. Haloperidol provided no benefit. A combination of benztropine and clonazepam seemed to help but led to gait and cognitive impairment. With tetrabenazine the movements became intermittent and less intense. Eight months after the onset there was significant improvement but not complete resolution. Repeat MRI 5 weeks later showed greater hyperintensity in the globus pallidus on the axial, non-contrast T1-weighted image but otherwise similar hyperintensity within the right caudate head and putamen. On the T2-weighted image, the hyperintensity within the right lentiform nucleus was similar to that seen on the prior study; however there was greater hyperintensity in the head of the right caudate nucleus. The right basal ganglia appeared smaller than the left.
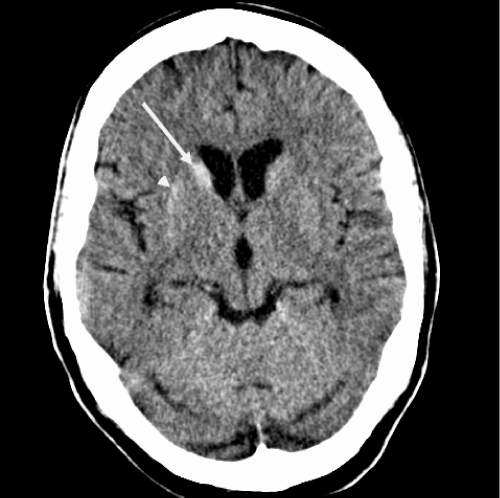
Figure 1: Initial CT scan at presentation to the Emergency Department at our
institution. Axial CT scan at the level of the basal ganglia. There is increased
density of the right caudate nucleus head (long arrow) and subtle increased
density of the lateral aspect of the right putamen (short arrow). The right
lentiform nucleus may be slightly smaller than the left. The hyperdensity could
represent mineralization or possibly blood
View Figure 1
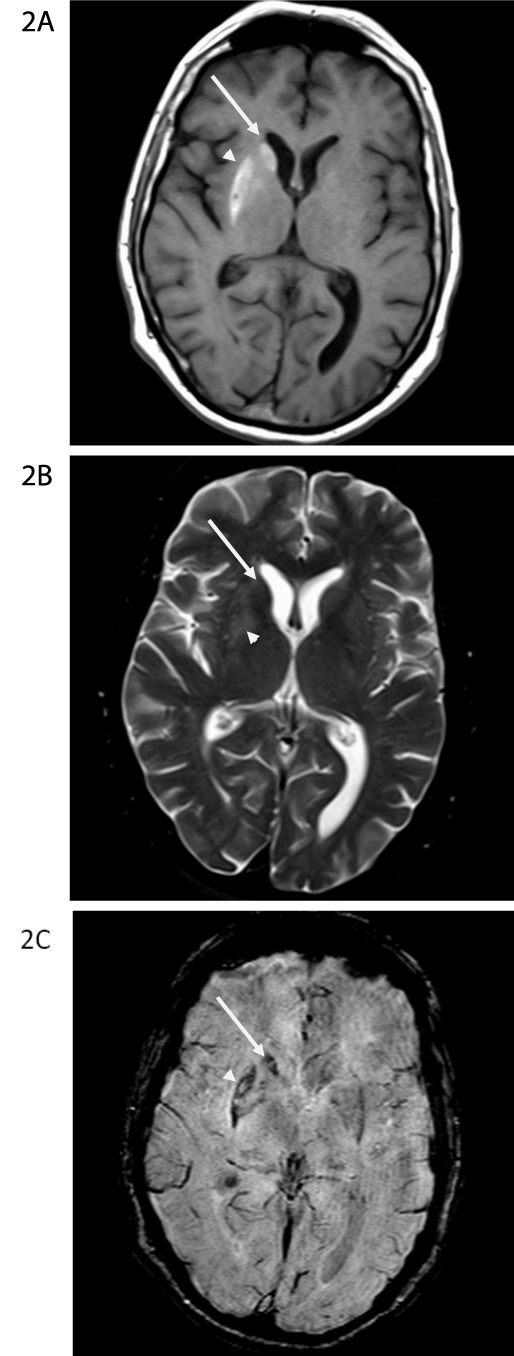
Figure 2A-C: MR scan obtained the day after the CT scan in Figure 1. On the
non-contrast T1-weighted image at the level of the basal ganglia (Figure 2A),
there is T1-shortening (hyperintensity) within the right caudate nucleus head
(long arrow) and putamen, greatest along the lateral aspect (arrowhead). On
the axial T2-weighted image at the same level (Figure 2B), there is subtle
hyperintensity in the putamen (arrowhead). The caudate head has essentially
normal signal (long arrow). On the susceptibility weighted image (Figure 2C),
there is susceptibility artifact in the right caudate nucleus head (long arrow)
and lateral aspect of the right putamen (arrowhead), similar in distribution to
the signal abnormality on the T1-weighted images and the increased density
on the CT scan.
View Figure 2
Discussion
HCHB may occur in chronic poorly controlled diabetes without overt non ketotic hyperosmolar disorder. The condition usually becomes refractory to treatment once a significant damage has resulted from the metabolic derangement. This was clearly illustrated in our case. Basal ganglia atrophy developed over time and led to the persistence of his abnormal movements.
Radiological findings were also characteristic in this case. There was complete sparing of internal capsule, thus pointing towards a metabolic rather than a vascular insult [8,9]. While there was gradual evolution of changes on imaging, there was persistence of T2 hyperintensity in the right lentiform nucleus. Follow-up MRI also showed unilateral atrophy of basal ganglia. Basal ganglia atrophy resulting in hyperkinetic movements is a known feature of various hereditary neurodegenerative disorders, but it is rarely reported in HCHB. A few patients with persistent HCHB due to non-ketotic hyperglycemia were noted to have basal ganglia lacunae. Surprisingly our patient developed unilateral basal ganglia atrophy in the absence of evident ischemic infarcts.
Pathophysiology of HCHB in the setting of hyperglycemia is not well understood. Hyperglycemia can result in breakdown of the blood brain barrier. It can cause regional decrease in cerebral blood flow leading to intracellular acidosis, glutamate excito-toxicity and decreased activity of gamma amino butyric acid [10]. Subsequently brain edema, ischemia, and ultimately gliotic changes may develop [11]. However this does not explain bright signal changes noted on T1 weighted images. But petechial hemorrhages [12], calcification [13], manganese deposition in reactive astrocytes, gliosis and myelin breakdown [14] have been proposed as possible mechanisms underlying T1 shortening seen in the basal ganglia on MRI. Biopsy of a patient with HCHB revealed gliotic brain tissue along with the accumulation of gemistocytes. Gemistocytes are swollen astrocytes, rich with intracellular protein that are reactive to ischemic changes. Protein in the astrocytes restricts motion of water molecules and was thought to explain T1 changes seen in these patients [15]. Ohara et al. found diffuse proliferation of reactive astrocytes and small adjacent recent infarctions in putamen [16].
Treatment options in HCHB are limited and mixed results have been reported [17]. Both typical and atypical neuroleptics have been tried [5]. Successful outcome with tetrabenazine had been reported in some cases [18]. Thalamic stimulation was tried in a refractory case of HCHB from hyperglycemia for symptomatic relief. If symptoms are mild and short lived, medications are not needed. Good control of blood sugar levels should be achieved. In refractory and chronic cases, neuroleptics and tetrabenazine may be tried. Multiple treatment trials may be needed as seen in our case.
Prognosis of these patients is usually good and almost complete resolution of both clinical and radiological findings may be seen. A meta-analysis of 53 patients with HCHB due to hyperglycemia showed that significant improvement in chorea can occur following treatment [19]. In another case series of 10 patients with HCHB, 9 patients improved within 2 days after adequate glycemic control [20]. However, for unclear reasons the duration of symptoms and treatment can be variable. But adequate glycemic control is extremely important as it can effectively shorten the duration of symptoms. The exact risk factors for the development and duration of this rare syndrome needs to be delineated by larger studies.
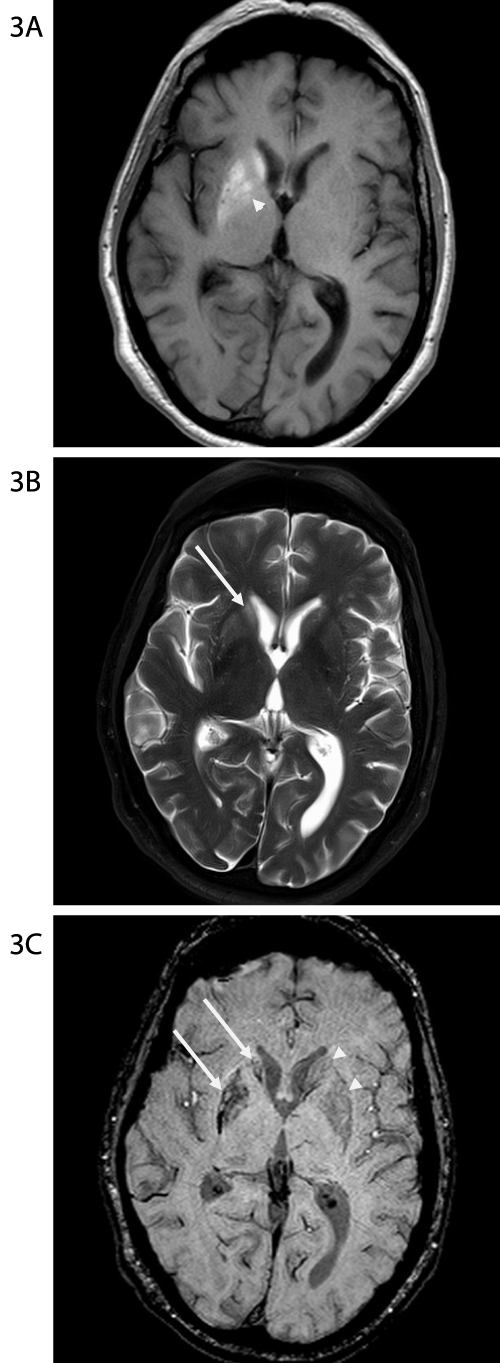
Figure 3A-C: MR scan obtained 5 weeks after the scans in Figures 1 &
2. On the axial non-contrast T1-weighted image at the level of the basal
ganglia (Figure 3A) there is now greater hyperintensity in the globus pallidus
(arrowhead), but there is otherwise similar hyperintensity within the right
caudate head and lentiform nucleus. On the T2-weighted image (Figure 3B),
the extent of hyperintensity within the right lentiform nucleus is reasonably
similar to the appearance on the prior study, however there is now greater
hyperintensity in the right caudate nucleus head (long arrow). On the
susceptibility weighted image (Figure 3C), the extent of the susceptibility
artifact is also not significantly changed. Note that the right basal ganglia (long
arrows) are smaller than the left (arrowheads).
View Figure 3
We have thus described a unique patient with HCHB who had a protracted course with his poorly controlled DM and basal ganglia atrophy that developed overtime. However, reasonable symptom control was achieved with tetrabenazine.
References
-
Kashihara K, Nakashima S, Kohira I, Shohmori T, Fujiwara Y, et al. (1998) Hyperintense basal ganglia on T1-weighted MR images in a patient with central nervous system lupus and chorea. AJNR Am J Neuroradiol 19: 284-286.
-
Dewey RB Jr, Jankovic J (1989) Hemiballism-hemichorea. Clinical and pharmacologic findings in 21 patients. Archives of neurology46: 862-867.
-
Vidakovic A, Dragasevic N, Kostic VS (1994) Hemiballism: report of 25 cases. J Neurol Neurosurg Psychiatry 57: 945-949.
-
Lin JJ, Chang MK, Lee CC, Tsao WL (1995) Hemiballism-hemichorea: clinical study in 23 Chinese patients. Zhonghua yi xue za zhi (Taipei) 55: 156-162.
-
Su CS, Chang YY, Liu KT, Lan MY, Liu JS (2012) Risk factors for prolonged hemichorea-hemiballism caused by hyperglycemia. Parkinsonism Relat Disord 18: 96-98.
-
Cheema H, Federman D, Kam A (2011) Hemichorea-hemiballismus in non-ketotic hyperglycaemia. J Clin Neurosci. 18: 293-294.
-
Ifergane G, Masalha R, Herishanu YO (2001)Transient hemichorea/hemiballismus associated with new onset hyperglycemia. Can J Neurol Sci 28: 365-368.
-
Oh SH, Lee KY, Im JH, Lee MS (2002)Chorea associated with non-ketotic hyperglycemia and hyperintensity basal ganglia lesion on T1-weighted brain MRI study: a meta-analysis of 53 cases including four present cases. J Neurol Sci 200: 57-62.
-
Chu K, Kang DW, Kim DE, Park SH, Roh JK (2002) Diffusion-weighted and gradient echo magnetic resonance findings of hemichorea-hemiballismus associated with diabetic hyperglycemia: a hyperviscosity syndrome?.Arch Neurol 59: 448-452.
-
Cherian A, Thomas B, Baheti NN, Chemmanam T, Kesavadas C (2009) Concepts and controversies in nonketotic hyperglycemia-induced hemichorea: further evidence from susceptibility-weighted MR imaging. J Magn Reson Imaging 29: 699-703.
-
Shan DE (2004) Hemichorea-hemiballism associated with hyperintense putamen on T1-weighted MR images: an update and a hypothesis. Acta Neurol Taiwan 13: 170-177.
-
Srinivas K, Rao VM, Subbulakshmi N, Bhaskaran J (1987) Hemiballism after striatal hemorrhage. Neurology37: 1428-1429.
-
Shan DE (2005) An explanation for putaminal CT, MR, and diffusion abnormalities secondary to nonketotic hyperglycemia. AJNR Am J Neuroradiol 26: 194; author reply 194-195.
-
Nagai C, Kato T, Katagiri T, Sasaki H (1995) Hyperintense putamen on T1-weighted MR images in a case of chorea with hyperglycemia. AJNR Am J Neuroradiol 16: 1243-1246.
-
Shan DE, Ho DM, Chang C, Pan HC, Teng MM (1998) Hemichorea-hemiballism: an explanation for MR signal changes. AJNR Am J Neuroradiol 19: 863-870.
-
Ohara S, Nakagawa S, Tabata K, Hashimoto T (2001) Hemiballism with hyperglycemia and striatal T1-MRI hyperintensity: an autopsy report. Mov Disord 16: 521-525.
-
Ahlskog JE, Nishino H, Evidente VG, Tulloch JW, Forbes GS, et al. (2001) Persistent chorea triggered by hyperglycemic crisis in diabetics. Mov Disord 16: 890-898.
-
Sitburana O, Ondo WG (2006) Tetrabenazine for hyperglycemic-induced hemichorea-hemiballismus. Mov Disord 21: 2023-2025.
-
Nakano N, Uchiyama T, Okuda T, Kitano M, Taneda M (2005) Successful long-term deep brain stimulation for hemichorea-hemiballism in a patient with diabetes. Case report. J Neurosurg 102: 1137-1141.
-
Lai PH, Tien RD, Chang MH, Teng MM, Yang CF, et al. (1996) Chorea-ballismus with nonketotic hyperglycemia in primary diabetes mellitus. AJNR Am J Neuroradiol 17: 1057-1064.





