International Journal of Clinical Cardiology
Spontaneous Coronary Artery Dissection: Report of 6 Cases and Literature Review
Adolfo Ferrero Guadagnoli*, Miguel A Ballarino, Carlos R Leonardi, Alejandro E Contreras, Eduardo J Brenna and Jonathan Miara
Cardiac Catheterization Laboratories, Hospital Privado Centro Médico de Córdoba, Córdoba, Argentina
*Corresponding author: Adolfo Ferrero Guadagnoli, Cardiac Catheterization Laboratories, Hospital Privado Centro Médico de Córdoba, Naciones Unidas 346, C.P X5016KEH, Córdoba, Argentina, E-mail: dradolfoferrero@gmail.com
Int J Clin Cardiol, IJCC-3-070, (Volume 3, Issue 1), Research Article; ISSN: 2378-2951
Received: November 04, 2015 | Accepted: January 23, 2016 | Published: January 27, 2016
Citation: Guadagnoli AF, Ballarino MA, Leonardi CR, Contreras AE, Brenna EJ, et al. (2016) Spontaneous Coronary Artery Dissection: Report of 6 Cases and Literature Review. Int J Clin Cardiol 3:070. 10.23937/2378-2951/1410070
Copyright: © 2016 Guadagnoli AF, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objectives: We present our recent casuistry on such an interesting issue as coronary dissections. Provide data to assist the diagnosis and treatment of this disease.
Background: Spontaneous coronary artery dissection is a non-traumatic and non-iatrogenic separation of the coronary artery walls. It is a rare cause of acute coronary syndrome, mainly affecting women with no significant cardiovascular risk factors and has varying presentations from mild chest pains to sudden cardiac death.
Methods: We identified consecutive patients with confirmed spontaneous coronary artery dissection, all of whom were seen at our hospital between 2005 and 2015 (n = 6). Spontaneous coronary artery dissection was defined as a clinical acute coronary syndrome together with typical diagnostic identified by angiography or with intravascular ultrasound imaging.
Results: There are 6 consecutive patients who were treated at our center between 2005 and 2015, mean age of 41.6 years and 5 were women. Five had ST-segment elevation myocardial infarction and one Non-ST-segment elevation myocardial infarction. One had sudden cardiac arrest. The most affected artery was the anterior descending (83%) and all dissections were type I. Three patients received medical treatment, two percutaneous coronary intervention and one coronary artery bypass grafting. There was no evidence of atherosclerosis on angiography or intracoronary ultrasound in any of the patients. The median follow-up period was 3 years. No patient had recurrence of the disease or died.
Conclusions: In the clinical setting of acute coronary syndrome, spontaneous coronary artery dissection must be considered as a possibility, particularly in young patients without clear risk factors for coronary artery disease and in women in the peripartum period.
Keywords
Stent, Myocardial infarction, Coronary bypass surgery, Coronary angiography
Introduction
Spontaneous coronary artery dissection (SCAD) is defined as a non-traumatic and non-iatrogenic separation of the coronary artery walls, creating a false lumen [1]. SCAD is characterized by the presence of hematoma or thrombus in the false lumen, usually in the outer third of the vessel media, and it can occur primarily or secondarily [2,3]. It results in separation of the layers of the coronary artery wall leading to intramural hematoma (IMH) within the false lumen, which compresses the true lumen and restricts coronary blood flow, leading to acute coronary syndrome (ACS) [3].
Its first angiographic diagnosis was reported almost 50 years after the first description. Treatment with percutaneous coronary intervention (PCI) and surgical bypass were first reported in the mid-1980s [4]. Since this condition was first described by Pretty [5] in 1931, it has been reported as having a low angiographic incidence [6], with greater prevalence in young females. SCAD is a rare cause of ACS and sudden cardiac death (SCD) [7]. Patients may present with diferentclinical scenarios ranging from angina pectoris to cardiogenic shock to SCD [8]. However, its etiology, pathogenesis and therapeutic strategies have not as yet been fully defined.
Materials and Methods
Between 2005 and 2015 we conducted 5634 coronary angiography and 2435 coronary angioplasty in our center. From this we identified and analized consecutive patients with confirmed SCAD, all of whom were seen at our hospital between 2005 and 2015 (n = 6). We collected data from electronic medical records. Spontaneous coronary artery dissection (SCAD) is defined as a non-traumatic and non-iatrogenic separation of the coronary artery walls, creating a false lumen [1], and was also defined as a clinical ACS together with typical diagnostic features (intimal dissection and IMH) identified by angiography or with intravascular ultrasound (IVUS) imaging and complete absence of atherosclerosis. Intimal dissection was defined as the presence of a radiolucent plane, with or without contrast staining. IMH was identified by an abrupt vessel tapering in accordance with the recent proposed classification [9]. Patients with iatrogenic coronary artery dissection, atherosclerotic dissection or chest trauma were excluded. We identified 3 different types of SCAD.
(I) Type 1 (evident artery wall stain): This is the pathognomonic angiographic appearance of SCAD with contrast dye staining of the artery wall with multiple radiolucent lumens.
(II) Type 2 (diffuse stenosis of varying severity): This angiographic appearance is not well appreciated and is often missed or misdiagnosed. It commonly involves the mid to distal segments of coronary arteries, and can be so extensive that it reaches the distal tip. There is an appreciable (often subtle) abrupt change in artery caliber, with demarcation from normal diameter to diffuse narrowing. This diffuse (typically > 20 mm) and usually smooth narrowing can vary in severity from an inconspicuous mild stenosis to complete occlusion.
(III) Type 3 (mimic atherosclerosis): This appearance is the most challenging to differentiate from atherosclerosis and most likely to be misdiagnosed. Angiographic features that favor SCAD are: (I) lack of atherosclerotic changes in other coronary arteries, (II) long lesions (11-20 mm), (III) hazy stenosis and (IV) linear stenosis. The angiographer needs to have a high index of suspicion for SCAD and employ intracoronary imaging for such cases.
Follow-up comprised scheduled (routine or otherwise) review with the primary cardiologist and telephone calls. Demographics, clinical presentation, coronary distribution, treatment modality, early and late events were determined via medical records, and follow-up from the patient’s electronic medical history. Coronary distributions were also determined via angiographic review.
Definition of adverse events
Unstable angina (UA): Ischemic symptoms suggestive of an ACS and no elevation in troponin, with or without electrocardiogram changes indicative of ischemia.
Myocardial infarction (MI): Evidence of myocardial necrosis in a clinical setting consistent with myocardial ischemia. The clinical, electrocardiographic, and cardiac biomarker information was considered to determine whether or not a MI has occurred.
Cardiovascular death (CVD): Includes death resulting from an acute myocardial infarction, sudden cardiac death, death due to heart failure and death due to other cardiovascular causes.
Non-cardiovascular death is defined as any death that is not thought to be due to a cardiovascular cause.
Sudden Cardiac Death (SCD): Refers to a death that occurs unexpectedly, not following an acute MI.
Target Vessel Revascularization (TVR): Target vessel revascularization is any repeat percutaneous intervention or surgical bypass of any segment of the target vessel.
Target Lesion Revascularization (TLR): Target lesion revascularization is any repeat percutaneous intervention of the target lesion (including 5 mm proximal and distal to the target lesion) or surgical bypass of the target vessel performed for restenosis or other complication involving the target lesion.
Target Vessel (TV): A target vessel is any native coronary vessel (e.g. left main coronary artery (LMCA), left anterior descending coronary artery (LAD), left circumflex coronary artery (LCX), or right coronary artery (RCA)). The target vessel includes the target lesion as well as segments of the vessel that are upstream and downstream to the target lesion, including side branches (native vessel).
Target Lesion (TL): A target lesion is any lesion treated or attempted to be treated during the procedure.
Results
The epidemiological characteristics of the series are shown in table 1, while the individual breakdown of clinical presentation and the subsequent course of the disease are shown in table 2.
![]()
Table 1: General Epidemiological Characteristics.
View Table 1
![]()
Table 2: Clinical Characteristics and Patient Outcomes.
View Table 2
Relevant baseline characteristics of our patients: average age of 41.6 years (the youngest 28 and the oldest 56). There were 5 women (83%) and a man. One of them was smokers at the admission, two had arterial hypertension and three had dyslipidemia. Nobody had diabetes. Average body mass index is 25.7 Kg/m2.
Clinical presentation: The majority (83%) appeared as STEMI (ST-segment elevation myocardial infarction) and one as NSTEMI (Non-ST-segment elevation myocardial infarction). One of them (case 5) developed cardiorespiratory arrest and had to be resuscitated, and another had a cardiogenic syncope. The average of left ventricular systolic function (LVSF) by echocardiography (ETT) or ventriculography at admission was 40 %.
Underlying etiologies: One woman was in the immediate postpartum period (30 days after delivery) and had presented preeclampsia during pregnancy. The man was a cocaine addict. Nobody had collagenopathies or intense previous physical stress. One patient had fibromuscular dysplasia (FMD).
Angiographic characteristics: The most affected artery was the left anterior descending artery, and the most frequent (66.5 %) type of dissection was type I.
Treatment: Three patients received only medical therapy, two received coronary angioplasty one with a drug-eluting stent and another with 92 mm of bare metal stents (BMS) (4 BMS 3.0 mm × 32 and 28 mm and 3.5 ° 20 and 12 mm) and another CABG, as a result of multivessel disease (proximal left anterior descending coronary artery and left circumflex coronary artery and distal right coronary artery) and compromise of the left main coronary artery.
Clinical outcomes: The average follow-up is 3 years and all patients are alive. At the follow-up all patients had ETT 6 and 12 months after of the index events and only one has persistence of LVSF impaired measured byETT and gated-Single Photon Emission Computed Tomography (gated-SPECT). Three presented with atypical symptoms at the latest follow-up, one of whom had a new coronary angiography (CAG) performed a month later on suspicion of progression of dissection, but showed partial resolution without progression. No one had CD, SCD, UA, TVR, TLR or any kind of MI.
Discussion
SCAD is a rare entity, the overall incidence in coronary angiographies is around 0.2% [10]. It is difficult to ascertain the true prevalence as it is often under-diagnosed and has presentations varyingfrom mild chest pains to SCD. In patients presenting with ACS, SCAD is noted to occur in 3-4%, diagnosed with optical coherence tomography (OCT) [11-13]. Among stable patients presenting for routine CAG, SCAD was diagnosed in 0.3% [6]. Among women presenting with ACS, prevalence was reported to be higher (8.7%) among those < 50 years old [14]. In the subgroup of women presenting with ST elevation, prevalence of SCAD was even higher at 10.8% [14].
There are two proposed mechanisms for the formation of IMH with SCAD. The first involves an intimal dissection resulting in blood from the endoluminal space entering the intimal space, creating a false lumen filled with blood. The second mechanism of IMH formation is thought to be due to rupture of the vasa vasorum, which are small arterioles within the walls of arteries supplying blood to the walls [15]. When such a rupture occurs, blood can pool within the intramural space, creating a false lumen filled with hematoma. Non-atherosclerotic coronary artery dissection is typically a culmination of disease pathways that predispose arterial beds to injury. These include FMD, multiple pregnancy, systemic inflammation (systemic lupus erythematosus, Crohn’s disease, polyarteritisnodosa and sarcoidosis), connective tissue disorder (Marfan’s syndrome, EhlerDanlos syndrome, and cystic medial necrosis), hormonal therapy, and coronary artery spasm [15]. SCAD has been observed in women who are peripartum or with multiple prior pregnancies, and thus a significant association with pregnancy has been postulated [16,17]. In most cases, the insult occurred a few weeks after delivery, with an average time-delay of about 1 month [18,19]. In addition, hormonal changes during pregnancy are thought to alter normal elastic fibres, impair collagen synthesis and mucopolysaccharide content, causing weakened media. Progesterone is thought to be the culprit hormone, and estrogen, on the other hand, creates a hypercoagulable state. The weakened artery walls at risk of dissection with a prothrombotic state increase the risk of false lumen creation and thrombosis. Eosinophilic infiltrates in arterial adventitia have also been observed in autopsies of peripartum women. It is believed that these eosinophilic granules cause breakdown of the medial-adventitial layer via lytic substances, predisposing the artery to dissection. However, it is unclear if the eosinophilic granules cause SCAD or are a result of SCAD. Interestingly, SCAD in pregnant women often occurs during late pregnancy or the post-partum period [16,20,21]. The hemodynamic changes during late pregnancy can also precipitate SCAD.
In patients with underlying predisposing arteriopathies, there can often be precipitating stressors such as intense exercise or emotional stress, which may trigger the SCAD event [20]. Intense exercise, particularly isometric in type, can increase cardio-circulatory stresses and shear forces against the coronary artery wall. Among our patients, no stressful situation was identified predisposing to SCAD.
Related to arterial integrity, a recent study showed that coronary artery tortuosity is present at a higher proportion in patients with SCAD than in patients with normal coronary arteries [22]. Repeat dissections were common within tortuous segments (80%).
Saw et al. show a strong association between SCAD and FMD, a condition that also predominantly affects women [23]. One of our patients had this pathology. It is a non-inflammatory, non-atherosclerotic disorder of the arterial vasculature that leads to arterial stenosis, occlusion, aneurysm or dissection. It can involve small to medium-sized arterial beds, especially of the renal and internal carotid arteries. The etiology of FMD is unknown, but hormonal influences have been proposed and a small proportion may be genetically inherited. Since 90% of cases affect women [22], sex hormones were thought to influence development of FMD, with some similarities to SCAD.
Patients presenting with SCAD have a spectrum of clinical presentationdepending on the extent and flow-limiting severity of the coronary dissection. Fortunately, the majority appear to present with ACS with good in-hospital prognosis [14]. Commonly found inwomen, asymptomatic presentation of SCAD in men is very rare [24]. It should be suspected in a young patient without major cardiovascular risk factors or in a patient in the peri¬partum period presenting with ACS or SCD [11]. In our series, the most frequent clinical presentation was STEMI, but one had a SCD and another, syncope. In recent retrospective studies, chest pain was the presenting symptom in the majority of SCAD cases [17,19,25,26], as it was in our patients. Ventricular arrhythmia occurred in 8-14% of patients [8,25]. In another series, all cases of SCAD presented with troponin-positive ACS, with 26% presenting with ST-elevation MI, and 3.6% with ventricular arrhythmia [20]. Two older studies showed a higher proportion presenting with ST-elevation MI (80-84%), while non-ST elevation MI were 8-16%, and 4% presented with unstable angina [25,27]. Case series have also noted that patients may have severe left ventricular dysfunction (LVD), often with subsequent full recovery [28-30]. However, left ventricular (LV) recovery may occur with standard forms of MI, but whether there may be more marked LV recovery with SCAD, as postulated by some, is yet unknown. In our experience, four patients had LVD and only one did not recover normal function.
The current widespread availability of CAG enables earlier diagnosis of SCAD. Sawhas characterized three distinct angiographic appearances and patterns of SCAD to aid diagnosis [9]:
(I) Type 1 evident artery wall stain, (II) Type 2 diffuse stenosis of varying severity and (III) Type 3 mimic atherosclerosis. Angiographic features that favor SCAD are: (I) lack of atherosclerotic changes in other coronary arteries; (II) long lesions (11-20 mm); (III) hazy stenosis; and (IV) linear stenosis. The angiographer needs to have a high index of suspicion for SCAD and employ intracoronary imaging for such cases.
There are no apparent differences in clinical presentations with the three angiographic classifications of SCAD. Our patients most commonly had type 1.
The natural history of SCAD appears to entail spontaneous healing in the vast majority of cases [20]. Repeat angiograms in selected patients in previous studies showed variable healing at 1 month (10), < 1 year [13] and full resolution at 40 months [17].
Cardiac computed tomography (CCT) is an ideal noninvasive imaging technique for delineating IMH and assessing luminal narrowing [31]. Furthermore, the follow-up of SCAD without an invasive diagnostic CAG is an important benefit of CCT. The current gold-standard CAG is unfortunately an imperfect tool for the diagnosis of SCAD, because it is only a 2-dimensional luminogram (Figure 1). It is excellent to assess luminal narrowing; however, it is poor in assessing the artery wall, where the key abnormalities occur with SCAD. Dissection may be missed in the absence of an intimal tear or if the true lumen is severely narrowed. The classic angiographic appearance includes contrast media seen in two lumens separated by a radiolucent intimal flap, with persistence of contrast in the false lumen after washout from the remainder of the vessel. Also, hematoma filling the false lumen may simulate intracoronary thrombus [32].
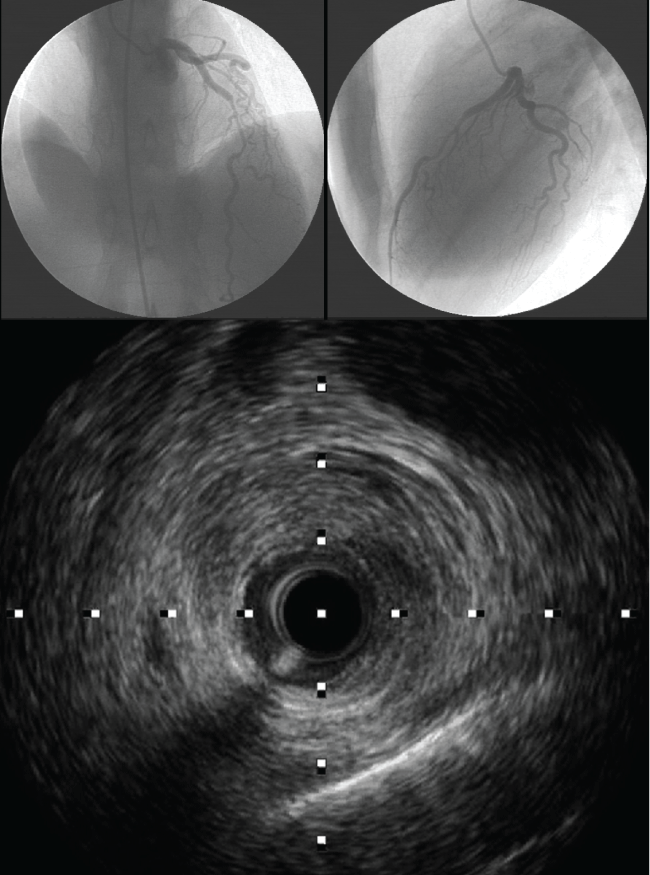
.
Figure 1: Left and right superior images are coronary angiograms imaging, showing a cranial views of left anterior descending coronary artery with long, smooth narrowed lesion extending in mid left anterior descending coronary artery. Inferior image is the intravascular ultrasound view where intima and media are separated from the adventitia and compressed by an echo lucent voluminous huge hematoma. There are two apparent echogenic tram tract like lines, representative of internal and external elastic lamina.
View Figure 1
IVUS and OCT are both tools that allow angiographers to better visualize the artery wall structure and composition. Each modality, IVUS or OCT, has its pitfalls and benefits in detecting pathology in artery walls. IVUS has a lower spatial resolution (150-200 μm) and can delineate true and false lumens and detect IMH, which appears as a homogenous collection behind the intimal-medial membrane (Figure 1). It can also distinguish atherosclerotic stenosis and spasm from hematoma or dissection, and can identify the length of the hematoma or location of the dissection [19]. OCT, on the other hand, is a much higher resolution (10-20 μm) modality and can visualize the true lumen, false lumen, and even intimaltears exceedingly well. Current OCT systems allow a faster interrogation of the target segment but remain limited by the requirement of a blood-free lumen (problematic in vessels with severe stenoses) and by theshadowing caused by red thrombus. Both techniques recognize the position of the guide wire within the true lumen, which remains critical when coronaryinterventions are planned. Although intracoronary techniques provide striking images, they should be used with great care in these patients to avoid further vessel injury in the already disrupted coronary wall [13]. Early diagnosis is important for managing SCAD patients, because thus the use of unnecessary and potentially harmful drug therapies may be avoided.
The optimal treatment for SCAD has not been clearly determinedand there is no consensus as to recommendations. United States and European professional society guidelines advocate early percutaneous revascularization for patients with ACS, based on data from multiple large-scale randomized studies [33-35]. These studies were composed primarily of patients with atherosclerotic coronary artery disease, who derived benefit from intervention by the trapping of plaque and thrombus material behind a stent scaffold in addition to restoration of flow. In this regard, the conventional definition of a successful procedure is the absence of residual stenosis after PCI, conferring lower risks of both acute closure and long-term target vessel revascularization (TVR).
The selection of treatment strategy depends upon the clinical manifestation, locationand extent of dissection, and the amount of ischemic myocardiumat risk [7]. Options include medical therapy, PCI and CABG [14]. Conservative medical therapy is a reasonablefirst option in mid or distal single vessel dissection with a lumendiameter limitation < 50% and TIMI grade 2 or 3 flow in the affected coronary artery [2,10,14].
Considering that a proportion of SCAD involves intimal tear, which is prothrombotic, this would empirically benefit from antiplatelet therapy. Aspirin appears reasonable for acute and long-term SCAD treatment. The role of new P2Y12 antagonists (clopidogrel, prasugrel and ticagrelor) inthe case of SCAD is still unclear. The role of GPIIb/IIIa inhibitors for acute SCAD management has also not been evaluated.
Heparin agents are typically administered for ACS patients on hospital presentation; however, they should be discontinued once SCAD is proven on angiography to avoid extension of IMH.
The role of thrombolytics is controversial as, although these may help re-establish anterograde flow in the true lumen [36], they also carry the risk of increasing flow into the false lumen and propagating the dissection [10], due to increased bleeding in the vessel wall [37] and hemorrhage of the vasa vasorum with bloody effusions inside the vessel wall, causing compression of the true lumen, further lumen reduction, and extension of the dissected area [37]. Thrombolytic treatment should therefore be delayed until CAG is performed.
Beta-blockers reduce arterial shear stress and are presumably beneficial in reducing coronary artery wall stress, similar to the benefits in aortic dissection [38].
Nitroglycerin may be useful in alleviating ischemic symptoms from overlying vasospasm during acute SCAD presentation. Angiotensin-converting enzyme inhibitors are only routinely administered when there is significant LV dysfunction after the myocardial infarction (ejection fraction ≤ 40%; class 1 indication) [39]. The use of statins for non-atherosclerotic SCAD has not been studied, and these should be administered only in patients with preexisting dyslipidemia. However, some studies have demonstrated that early intervention with either PCI or coronary artery bypassgraft (CABG) following the diagnosis of SCAD leads to a betteroutcome [40]. PCI is the therapy of choice in single vessel disease, especially proximal dissection, in which there are ongoing symptoms and persistent restriction of coronary blood flow (TIMI grade 0 or 1 flow). Giventhat PCI is associated with several complications relating to the passage of the coronary wire into the false lumen of the dissected vessel or the loss of coronary flow through the propagation of dissection and displacement of stents, resulting in the propagation of hematoma, PCI may be limited to cases in which there are ongoing symptoms of ischemia [2]. Dissections often involve distal coronary arteries, which are too small to implant stents. Even if the dissected arteries are large, the dissections are often extensive, requiring long stents and thus increasing the risks of restenosis and thrombosis. However, there is no consensus onthe appropriate extent of stent coverage in terms of whether there should be proximal portion coverage only or full coverage of the dissection area. Like wise, adjunctive imaging technologies such as IVUS and OCT should be considered to determine the extent of dissection and to provide real-time guidance about intervention.
Coronary artery fenestration with scoring or cutting balloon has been previously reported. In some cases this technique has been successfully used as the initial invasive treatment of patients with SCAD [41].
CABG is the treatment of choice in multiple vessel disease, especially where there is left main stem involvement with ongoing ischemia refractory to medical or interventional therapy and when there is hemodynamic instability [10]. In one series [2], early results of coronary surgery appeared favorable, but 11 of 15 bypass grafts undergoing late angiographic assessment were occluded.
One recent retrospective study indicates that the risk of emergency CABG is disturbingly high among patients with SCAD treated with PCI, including those with normal flow. In contrast, conservative management was associated with favorable outcomes [42].
A collection of case reports from 1980-2000 showed mortality rates ranging from 0% to 7% [15]. More recent studies reported lower in-hospital mortality rates ranging from 1% to 5% [11,14,17]. One-year mortality rates after discharge were similar, at 1% to 4% [14,17]. One study reported 10-year mortality rates as high as 7.7% [17]. Many also required subsequent hospital visits for recurrent chest pains, and repeat coronary angiograms. Event-free rates at 1 year ranged from 74% to 96% [11,14,20]. In our series, 50% had spontaneous hospital visits for recurrent chest pains, and in one of them we repeated CAG.
Female SCAD patients potentially have a poorer prognosis. An earlier study analyzing 222 patients from several published studies showed that the strongest predictors of death were female sex (odds ratio 4.27) and absence of early treatment (odds ratio 35.5) [23]. In particular, the subgroup patients with postpartum SCAD appear to have the worst prognosis.
The left anterior descending coronary artery (LAD) is the most common site of dissection in female patients, while the right coronary artery (RCA) is often affected in male patients [11].
Possible late complications include progression of the dissection and formation of pseudoaneurysms [10]. Patients should be followed for any symptoms of recurrent ischemia [2]. Stress testing with nuclear perfusion imaging is preferred over CGA as a means of surveillance [2].
Conclusion
SCAD is a fascinating clinical entity. Although our case series report is not the first of its kind, it is a fresh presentation of a very rare disease process. There is as yet no consensus in regard to the management of SCAD, due to the condition’s rarity.
In the clinical setting of ACS, SCAD must be considered as a possibility, particularly in young patients without clear risk factors for coronary artery disease and in women in the peripartum period.
References
-
Saw J (2013) Spontaneous coronary artery dissection. Can J Cardiol 29: 1027-1033.
-
Tweet MS, Hayes SN, Pitta SR, Simari RD, Lerman A, et al. (2012) Clinical features, management, and prognosis of spontaneous coronary artery dissection. Circulation 126: 579-588.
-
Adlam D, Cuculi F, Lim C, Banning A (2010) Management of spontaneous coronary artery dissection in the primary percutaneous coronary intervention era. J Invasive Cardiol 22: 549-553.
-
Gonzalez JI, Hill JA, Conti CR (1989) Spontaneous coronary artery dissection treated with percutaneous transluminal angioplasty. Am J Cardiol 63: 885-886.
-
Pretty HC (1931) Dissecting aneurysm of coronary artery in a woman aged 42: Rupture. Br Med J 1: 667-669.
-
Hering D, Piper C, Hohmann C, Schultheiss HP, Horstkotte D (1998) Prospective study of the incidence, pathogenesis and therapy of spontaneous, by coronary angiography diagnosed coronary artery dissection. Z Kardiol 87: 961-970.
-
Almeda FQ, Barkatullah S, Kavinsky CJ (2004) Spontaneous coronary artery dissection. Clin Cardiol 27: 377-380.
-
Desai S, Sheppard MN (2012) Sudden cardiac death: look closely at the coronaries for spontaneous dissection which can be missed. A study of 9 cases. Am J Forensic Med Pathol 33: 26-29.
-
Saw J (2014) Coronary angiogram classification of spontaneous coronary artery dissection. Catheter Cardiovasc Interv 84: 1115-1122.
-
Sabatine MS, Jaffer FA, Staats PN, Stone JR (2010) Case 28–2010. A 32-year-old woman, 3 weeks post partum, with substernal chest pain. N Engl J Med 363: 1164-1173.
-
Mortensen KH, Thuesen L, Kristensen IB, Christiansen EH (2009) Spontaneous coronary artery dissection: a Western Denmark Heart Registry study. Catheter Cardiovasc Interv 74: 710-717.
-
Hill SF, Sheppard MN (2010) Non-atherosclerotic coronary artery disease associated with sudden cardiac death. Heart 96: 1119-1125.
-
Nishiguchi T, Tanaka A, Ozaki Y, Taruya A, Fukuda S, et al. (2013) Prevalence of spontaneous coronary artery dissection in patients with acute coronary syndrome. Eur Heart J Acute Cardiovasc Care.
-
Vanzetto G, Berger-Coz E, Barone-Rochette G, Chavanon O, Bouvaist H, et al. (2009) Prevalence, therapeutic management and medium-term prognosis of spontaneous coronary artery dissection: results from a database of 11,605 patients. Eur J Cardiothorac Surg 35: 250-254.
-
Yip A, Saw J (2015) Spontaneous coronary artery dissection-A review. Cardiovasc Diagn Ther 5: 37-48.
-
Vijayaraghavan R, Verma S, Gupta N, Saw J (2014) Pregnancy-related spontaneous coronary artery dissection. Circulation 130: 1915-1920.
-
DeMaio SJ Jr, Kinsella SH, Silverman ME (1989) Clinical course and long-term prognosis of spontaneous coronary artery dissection. Am J Cardiol 64: 471-474.
-
Gowda RM, Sacchi TJ, Khan IA (2005) Clinical perspectives of the primary spontaneous coronary artery dissection. Int J Cardiol 105: 334-336.
-
Arnold JR, West NE, van Gaal WJ, Karamitsos TD, Banning AP (2008) The role of intravascular ultrasound in the management of spontaneous coronary artery dissection. Cardiovasc Ultrasound 6: 24.
-
Saw J, Aymong E, Sedlak T, Buller CE, Starovoytov A, et al. (2014) Spontaneous coronary artery dissection: association with predisposing arteriopathies and precipitating stressors and cardiovascular outcomes. Circ Cardiovasc Interv 7: 645-655.
-
Vrints CJ (2010) Spontaneous coronary artery dissection. Heart 96: 801-808.
-
Eleid MF, Guddeti RR, Tweet MS, Lerman A, Singh M, et al. (2014) Coronary artery tortuosity in spontaneous coronary artery dissection: angiographic characteristics and clinical implications. Circ Cardiovasc Interv 7: 656-662.
-
Saw J, Poulter R, Fung A, Wood D, Hamburger J, et al. (2012) Spontaneous coronary artery dissection in patients with fibromuscular dysplasia: a case series. Circ Cardiovasc Interv 5: 134-137.
-
Yan Herry, Alvin Tonang, Victor Herlambang, Novi Anggriyani (2013) Spontaneous coronary artery dissection causing myocardial infarction in an 18-year-old man: A case report. Australas Med J 6: 694-696.
-
Thompson EA, Ferraris S, Gress T, Ferraris V (2005) Gender differences and predictors of mortality in spontaneous coronary artery dissection: a review of reported cases. J Invasive Cardiol 17: 59-61.
-
Tokura M, Taguchi I, Kageyama M, Nasuno T, Nishiyama Y, et al. (2014) Clinical features of spontaneous coronary artery dissection. J Cardiol 63: 119-122.
-
Olin JW, Froehlich J, Gu X, Bacharach JM, Eagle K, et al. (2012) The United States Registry for Fibromuscular Dysplasia: results in the first 447 patients. Circulation 125: 3182-3190.
-
Bergen E, Huffer L, Peele M (2005) Survival after spontaneous coronary artery dissection presenting with ventricular fibrillation arrest. J Invasive Cardiol 17: E4-6.
-
Akyuz A, Alpsoy S, Akkoyun DC (2013) Spontaneous coronary artery dissection and woven coronary artery: three cases and a review of the literature. Korean Circ J 43: 411-415.
-
Lempereur M, Grewal J, Saw J (2014) Spontaneous coronary artery dissection associated with β-HCG injections and fibromuscular dysplasia. Can J Cardiol 30: 464.
-
Choi SW, Nam CW, Bae HJ, Cho YK, Yoon HJ, et al. (2013) Spontaneous coronary artery dissection diagnosed by intravascular ultrasound and followed up by cardiac computed tomography. Korean J Intern Med 28: 370-373.
-
Daoulah AA, Qahtani A, Mazen Malak M, Al Ghamdi S (2012) Role of IVUS in Assessing Spontaneous Coronary Dissection: a Case Report. J Tehran Heart Cent 7: 78-81.
-
O’Gara PT, Kushner FG, Ascheim DD, Casey DE Jr, Chung MK, et al. (2013) 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: executive summary: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation 127: 529-555.
-
Members ATF, Steg PG, James SK, Atar D, Badano LP, et al. (2012) ESC guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur Heart J 33: 2569-2619.
-
Hamm CW, Bassand J-P, Agewall S, Bax J, Boersma E, et al. (2011) ESC guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur Heart J 32: 2999-3054.
-
Lodha A, Mirsakov N, Malik B, Shani J (2009) Spontaneous coronary artery dissection: case report and review of literature. South Med J 102: 315-317.
-
Maeder M, Ammann P, Angehrn W, Rickli H (2002) Spontaneous coronary artery dissection as a rare cause of an acute coronary syndrome. Z Kardiol 91: 951-955.
-
Nienaber CA, Powell JT (2012) Management of acute aortic syndromes. Eur Heart J 33: 26-35b.
-
Anderson JL, Adams CD, Antman EM, Bridges CR, Califf RM, et al. (2007) ACC/AHA 2007 guidelines for the management of patients with unstable angina/non-ST-Elevation myocardial infarction: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines for the Management of Patients With Unstable Angina/Non-ST-Elevation Myocardial Infarction): developed in collaboration with the American College of Emergency Physicians, the Society for Cardiovascular Angiography and Interventions, and the Society of Thoracic Surgeons endorsed by the American Association of Cardiovascular and Pulmonary Rehabilitation and the Society for Academic Emergency Medicine. Circulation 116: e148-304.
-
Shamloo BK, Chintala RS, Nasur A, Ghazvini M, Shariat P, et al. (2010) Spontaneous coronary artery dissection: aggressive vs. conservative therapy. J Invasive Cardiol 22: 222-228.
-
Alkhouli M, Cole M, Ling FS (2015) Coronary artery fenestration prior to stenting in spontaneous coronary artery dissection. Catheter Cardiovasc Interv.
-
Tweet MS, Eleid MF, Best PJ, Lennon RJ, Lerman A, et al. (2014) Spontaneous Coronary Artery Dissection Revascularization Versus Conservative Therapy. Circ Cardiovasc Interv 7: 777-786.





