International Journal of Clinical Cardiology
Ambiguous Left Ventricular Outflow Tract Obstruction: The Key Role of the Cardiac CT-Scan
Marie-Philippe Vergé1*, Géraldine Allain1, Guillaume Lucas2, Luc-Philippe Christians1
1Service de Cardiologie, Centre Hospitalier de Poitiers, France
2Service de Cardiologie, Centre Hospitalier de Niort, France
*Corresponding author: Marie-Philippe Vergé, Service de Cardiologie, Centre Hospitalier de Poitiers, France, Tel: 0630804792, Fax: 05.49.44.38.31, E-mail: scollyfr@yahoo.fr
Int J Clin Cardiol, IJCC-2-064, (Volume 2, Issue 6), Case Report; ISSN: 2378-2951
Received: April 06, 2015 | Accepted: December 05, 2015 | Published: December 09, 2015
Citation: Vergé MP, Allain G, Lucas G, Christians LP (2015) Ambiguous Left Ventricular Outflow Tract Obstruction: The Key Role of the Cardiac CT-Scan. Int J Clin Cardiol 2:064. 10.23937/2378-2951/1410064
Copyright: © 2015 Vergé MP, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Keywords
Subaortic membrane, Aortic valve stenosis, Left ventricular outflow tract, Cardiac CT-scan
Subaortic membranes are uncommon causes of left ventricular outflow tract obstruction. Combined subaortic membrane and aortic valve stenosis is even more rare and can be challenging for echocardiographic diagnosis.
A 58 year old woman, known for symptomatic aortic stenosis was referred to our hospital department for dyspnoea (NYHA III). Her most important medical history was aortic coarctation surgery at the age of 13 without any significant event since that time.
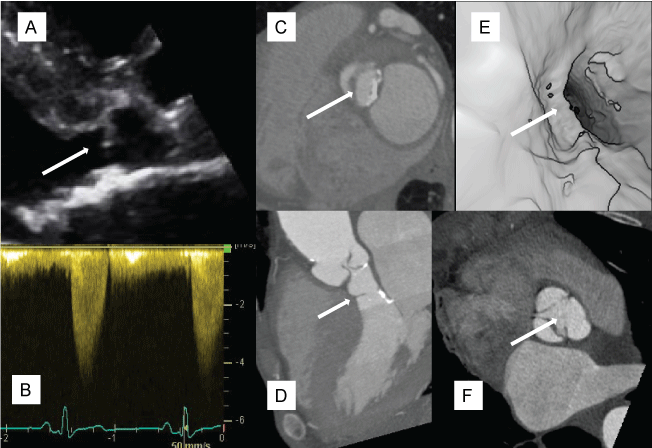
The trans-thoracic echocardiography showed an anatomic obstruction across the left ventricular outflow tract with a maximal gradient of 90 mmHg (Figure 1B), combining an aortic valve stenosis and a subaortic membrane (Figure 1A). However, the accurate location of the main obstruction remained uncertain after this echocardiography in spite of the optimal use of pulse Doppler.
.
(A) Trans-thoracic echocardiography: Aortic valve (star) and subaortic membrane (arrow); (B) Trans-thoracic echocardiography: Left ventricular outflow tract with a maximal gradient of 90 mmHg in continuous Doppler; (C) Cardiac CT: Ring-like subaortic membrane with a residual luminal area of 0.95 cm2 in the left ventricular outflow tract; (D) Cardiac CT: Orthogonal vue of ring-like subaortic membrane; (E) Virtual endoscopic view from the ascending aorta to the left ventricle across the aortic valve and the subaortic stenosis; (F) Cardiac CT: Axial vue: Tricuspid aortic valve, with an area of 1.65 cm2;
View Figure 1
The cardiac-CT with retrospective acquisition had a key role in our diagnostic and therapeutic strategy. This exam enabled us to complete the pre-operative assessment by ruling out any significant coronary artery disease, aneurysm of the ascending aorta and aorta recoarctation. It also enabled us to improve the assessment of the subaortic membrane, which was ring-like and mainly developed from the septal wall, with a width of 7 mm and a residual luminal area of 0.95 cm2 in the left ventricular outflow tract (Figure 1C: orthogonal vue of Figure 1D).
Using 3D reconstruction, we were able to accurately demonstrate this membrane's anatomy and its obstructive nature (Figure 1E), (Video: virtual endoscopic view from the ascending aorta to the left ventricle across the aortic valve and the subaortic stenosis). As for the aortic valve, it was tricuspid, with an area of 1.1 cm2/m2 (Figure 1F).
After debating with the heart-team, surgery was indicated due to the symptomatic nature of the subaortic membrane's obstruction. This surgery consisted of a resection of the fibrous subaortic membrane with septal myectomy and mechanic aortic valve replacement. The follow-up was uneventful and postoperative echocardiography did not show any remaining left ventricular outflow tract flow acceleration.
Membrane subaortic stenosis is a rare congenital abnormality representing 1%-2% of congenital heart diseases and about 10% of cases of left ventricular outflow tract obstruction [1]. It is often associated with other congenital malformations. Usually, the patient remains asymptomatic, which explains the late diagnosis, most of the time in adulthood [2]. Initial presentations of this pathology are very variable, from asymptomatic cases to symptoms such as dyspnea, syncopes, or chest pain [3].
Its physiopathology is still debated but it seems that the combination of factors including an underlying genetic predisposition, various geometric and anatomical variations of the left ventricular outflow tract leading to flow turbulence at this level are involved. As a matter of fact, the resulting turbulent flow may damage the endothelium and promote fibrin deposition which could ultimately progress to fibroelastic or fibromuscular obstruction [4]. As previously described in literature, familial occurrence could suggest a possible genetic cause [5].
Trans-thoracic echocardiography remains the first line exam for the diagnosis, the assessment of the severity of the obstruction and illustration of the mechanism of the left ventricular outflow tract obstruction. This assessment is indeed very important in terms of impact on the clinical outcome. Trans-oesophageal echocardiography is also widely used and 3D reconstructions have a real ad-value in preoperative assessment [6].
Exercise echocardiography may provide additional information in order to better identify the real cardiac origin of dyspnoea. The prognostic impact of this exam has been mainly shown for aortic stenosis [7].
In our case, this exam was not performed due to the symptomatic nature of the obstruction.
Our case-report shows how the widespread use of multimodality imaging enables a more accurate diagnosis and preoperative evaluation.
The CT-scan contributes to the evaluation of the severity of valve disease, directly through the measurement of valve planimetry, the evaluation of the aortic valve anatomy and 3D measurements of the subaortic membrane, and indirectly by quantifying valvular calcification.
CT-scan can also appear very useful to allow a global preoperative assessment of the cardiac structures in particular to rule out coronary artery disease, aneurysm of the ascending aorta and aorta recoarctation. Indeed, due to its high negative predictive value, CT-scan may be useful in excluding CAD in patients who are at low risk of atherosclerosis.
Eventually, thanks to the three-dimensional reconstructions, we were able to provide a real virtual endoscopic view from the ascending aorta to the left ventricle across the aortic valve and the subaortic stenosis, thus guiding the surgeon in preoperative assessment.
In this disease, surgical treatment is generally required and decided according to the symptomatic nature of the membrane and the flow acceleration through the subaortic membrane. A cut-off gradient of 50 mmHg is usually used to lead to surgery [8]. Sub-aortic membrane resection is a possible treatment; however it is associated with a high incidence of recurrence requiring reoperation in 6% to 30% cases [9].
Therefore, the usual surgical approach includes complete subaortic membrane resection and wide septal myectomy in order to improve residual gradients through left ventricular outflow tract and prevent recurrence of subaortic stenosis [10]. This surgical approach leads to a good surgical result and a lower rate of recurrence [9]. The rate of recurrence may be influenced on the one hand by genetic factors [5], on the other hand by higher pre operative left ventricular outflow tract gradient [1].
As a conclusion, our case highlights the usefulness of multimodality imaging and the CT-scan appears to be a very useful tool to assess valve stenosis, subaortic membrane and coronary arteries simultaneously before surgical treatment.
Video
References
-
Uysal F, Bostan OM, Signak IS, Semizel E, Cil E (2013) Evaluation of subvalvular aortic stenosis in children: a 16-year single-center experience. Pediatr Cardiol 34: 1409-1414.
-
Choi JY, Sullivan ID (1991) Fixed subaortic stenosis: anatomical spectrum and nature of progression. Br Heart J 65: 280-286.
-
Weymann A, Schmack B, Rosendal C, Karck M, Szabo G (2013) Surgical management of subaortic stenosis. Ann Thorac Cardiovasc Surg 19: 390-393.
-
Teis A, Sheppard MN, Alpendurada F (2010) Subaortic membrane: correlation of imaging with pathology. Eur Heart J 31: 2822.
-
Fatimi SH, Ahmad U, Javed MA, Shamim S, Ahmad R (2006) Familial membranous subaortic stenosis: review of familial inheritance patterns and a case report. J Thorac Cardiovasc Surg 132: 1484-1486.
-
De Agustin JA, Gomez de Diego JJ, Marcos-Alberca P, Macaya C, Perez de Isla L (2014) Combined subaortic membrane and aortic valve stenosis: additive value of three-dimensional echocardiography. Eur Heart J Cardiovasc Imaging 15: 388.
-
Picano E, Pibarot P, Lancellotti P, Monin JL, Bonow RO (2009) The emerging role of exercise testing and stress echocardiography in valvular heart disease. J Am Coll Cardiol 54: 2251-2260.
-
Arnáiz-García ME, González-Santos JM, Dalmau-Sorlí MJ, Lopez-Rodríguez J, Bueno-Codoñer M, et al. (2014) Aortic stenosis in the background of a subaortic membrane and tunnel-like ventricular outflow tract septal hypertrophy. Arch Cardiol Mex 84: 59-60.
-
Rodríguez-Caulo E, Araji O, de Araujo-Martins D (2013) Septal myectomy in recurrent subaortic membrane. Rev Esp Cardiol (Engl Ed) 66: 585.
-
Valeske K, Huber C, Mueller M, Böning A, Hijjeh N, et al. (2011) The dilemma of subaortic stenosis--a single center experience of 15 years with a review of the literature. Thorac Cardiovasc Surg 59: 293-297.





