International Journal of Clinical Cardiology
Hemolytic Anemia 19 Years after Mitral Valve Replacement with A Porcine Bioprosthesis
Debora Corsi1, Michele Celiento1, Stefano Pratali1, Alberto Balbarini2 and Uberto Bortolotti1*
1Section of Cardiac Surgery, University Hospital, Pisa, Italy
2Section of Angiology, University Hospital, Pisa, Italy
*Corresponding author: U. Bortolotti, MD, Sezione Autonoma di Cardiochirurgia Universitaria, Via Paradisa, 2, 56124 Pisa, Italy, Tel: 39-50-995250, Fax: 39-50-541647, E-mail: uberto.bortolotti@med.unipi.it
Int J Clin Cardiol, IJCC-2-043, (Volume 2, Issue 4), Case Report; ISSN: 2378-2951
Received: March 27, 2015 | Accepted: August 24, 2015 | Published: August 27, 2015
Citation: Corsi D, Celiento M, Pratali S, Balbarini A, Bortolotti U (2015) Hemolytic Anemia 19 Years after Mitral Valve Replacement with A Porcine Bioprosthesis. Int J Clin Cardiol 2:043. 10.23937/2378-2951/1410043
Copyright: © 2015 Corsi D, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
A 58-year-old man required replacement of a porcine mitral prosthesis because of severe hemolysis 19 years after implant. At operation the major pathologic finding was a perforation of one cusp with calcific deposition limited to the commissures. Clinically evident hemolytic anemia due to porcine valve dysfunction is rare particularly occurring very late postoperatively due to cusp perforation as in the present case.
Keywords
Mitral valve replacement, Mitral valve reoperation, Porcine xenograft
Introduction
Clinically evident hemolytic anemia due to porcine bioprosthetic dysfunction and leading to reoperation is a rare event. We report a patient who developed such complication late after mitral valve replacement (MVR) with a porcine valve.
Case Report
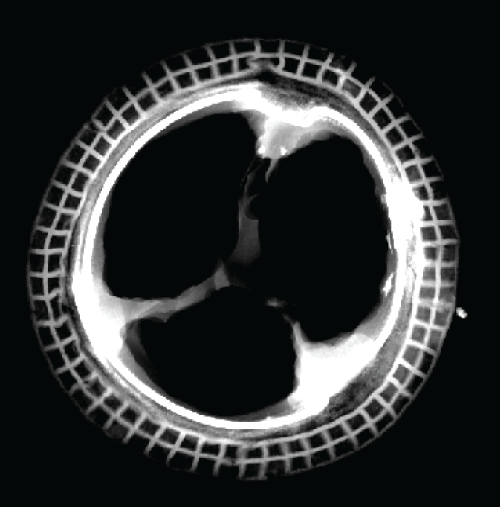
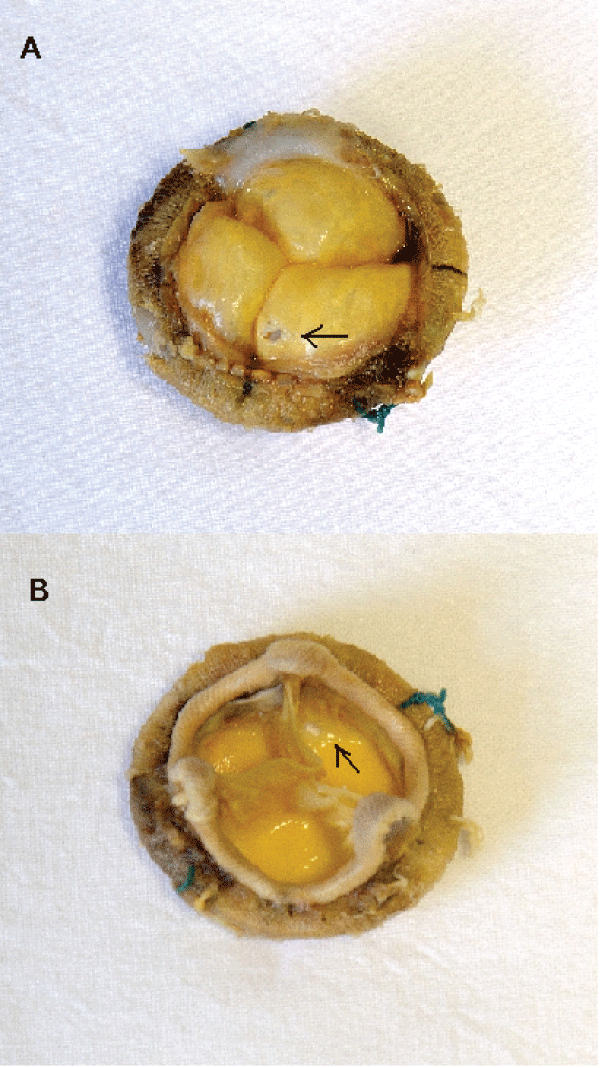
A 58-year-old man had undergone mitral valve replacement with a 27 mm Carpentier-Edwards porcine bioprosthesis (Edwards Lifesciences, Irvine, USA) in 1993. Nineteen years later he presented with dark urine, fatigue and moderate dyspnea denying any history of infection. He was in atrial fibrillation with a systolic mitral regurgitant murmur. Hematologic evaluation was consistent with haemolytic anemia; particularly, reduced hemoglobin, hematocrit and haptoglobin (6.6 mg/dl), and increased total bilirubin and lactic dehydrogenase (752 U/L) with schistocytosis and reticulocytosis were observed. A 2D echocardiogram showed moderate mitral regurgitation and increased pulmonary artery pressure. At reoperation the mitral prosthesis was replaced with a bileaflet mechanical valve. At gross inspection the explant showed pliable leaflets with a small perforation of one cusp; on x-ray calcific spots were detected mainly at one commissure (Figure 1 and Figure 2). The patient recovered uneventfully and was discharged on postoperative day 8 when the haemoglobin hematocrit levels had increased (12.8 g/dl and 37%, respectively), total bilirubin and haptoglobin were normal and lactic dehydrogenase was considerably reduced but still slightly increased (268 U/L).
.
Figure 1: Gross view of the mitral explant from the atrial (A) and ventricular (B) sides showing perforation of one cusp (arrows)
View Figure 1
Comment
Subclinical hemolysis in normally functioning prosthetic valves is a frequent finding [1] but haemolytic anemia secondary to porcine valve dysfunction is rare. A limited number of cases of haemolytic anemia in tissue valve recipients has so far been reported all being related to severe bioprosthetic dysfunction requiring reoperation [2-5]. Conservative treatment of hemolytic anemia occurring in patients after MVR with either a porcine or pericardial bioprosthesis without evidence of hemodynamically significant mitral regurgitation was also reported [6,7]. In 1990 Raymond et al. observed microangiopathic hemolytic anemia in a 59-year-old man who 10 years previously had undergone MVR with a porcine bioprosthesis [8]; at reoperation the cause of anemia was attributed to perforation of one cusp. Similarly, the peculiar finding in our case was the presence of clinically evident haemolytic anemia caused by a cusp perforation in a porcine xenograft who did not exhibit other major pathological changes, particularly in the absence of signs of infection. Interestingly enough in our case this complication occurred very late postoperatively, 19 years after MVR with a Carpentier-Edwards, indicating that first-generation porcine bioprostheses may have an extended durability in the individual patient.
References
-
Mecozzi G, Milano AD, De Carlo M, Sorrentino F, Pratali S, et al. (2002) Intravascular hemolysis in patients with new-generation prosthetic heart valves: a prospective study. J Thorac Cardiovasc Surg 123: 550-556.
-
Magilligan DJ Jr, Fisher E, Alam M (1980) Hemolytic anemia with porcine xenograft aortic and mitral valves. J Thorac Cardiovasc Surg 79: 628-631.
-
Weesner KM, Rocchini AP, Rosenthal A, Behrendt D (1981) Intravascular hemolysis associated with porcine mitral valve calcification in children. Am J Cardiol 47: 1286-1288.
-
Schaer DH, Cheng TO, Aaron BL (1987) Hemolytic anemia and acute mitral regurgitation caused by a torn cusp of a porcine mitral prosthetic valve 7 years after its implantation. Am Heart J 113: 404-406.
-
Brown MR, Hasaniya NW, Dang CR (1995) Hemolytic anemia secondary to a porcine mitral prosthetic valve leaflet dissection. Ann Thorac Surg 59: 1573-1574.
-
Myers TJ, Hild DH, Rinaldi MJ (1978) Hemolytic anemia associated with heterograft replacement of the mitral valve. J Thorac Cardiovasc Surg 76: 214-215.
-
Reddy SB, Pater JL, Pym J, Armstrong PW (1984) Hemolytic anemia following insertion of Ionescu-Shiley mitral valve bioprosthesis. Can Med Assoc J 131: 1469-1470.
-
Enzenauer RJ, Berenberg JL, Cassell PF Jr (1990) Microangiopathic hemolytic anemia as the initial manifestation of porcine valve failure. South Med J 83: 912-917.