International Journal of Clinical Cardiology
A Rare Manifestation of Organophosphorus Poisoning: Hypothermia with Cardiotoxicity
Valliappan Muthu, Sahajal Dhooria and Inderpaul Singh Sehgal*
Department of Pulmonary Medicine, Postgraduate Institute of Medical Education and Research, India
*Corresponding author: Inderpaul Singh Sehgal, Senior Resident, Department of Pulmonary Medicine, Postgraduate Institute of Medical Education and Research, Chandigarh-160012, India, Tel: 91-8437610088; E-mail: ipdoc_2000@hotmail.com
Int J Clin Cardiol, IJCC-1-005, (Volume 1, Issue 1), Case report; ISSN: 2378-2951
Received: September 28, 2014 | Accepted: October 28, 2014 | Published: October 30, 2014
Citation: Muthu V, Dhooria S, Sehgal IS (2014) A Rare Manifestation of Organophosphorus Poisoning: Hypothermia with Cardiotoxicity.Int J Clin Cardiol 1:005. 10.23937/2378-2951/1410005
Copyright: © 2014 Muthu V, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Organophosphorus pesticides related self-harm is afairly common clinical entity responsible for over two thirds of pesticide related deaths in the rural areas of developing countries. Organophosphates by their action at muscarinic and nicotinic receptors can have a myriad of presenting symptoms. Here we describe a fatal case of phorate (organophosphorus) toxicity manifesting with hypothermia and cardiotoxicity.
Introduction
Ingestion of organophosphorus compounds with suicidal intent is a major health issue in rural areas of developing world. According to the World Health Organization (WHO) estimates pesticides account for one to five million cases of poisoning with approximately 200,000 deaths occurring each year [1]. Organophosphorus compounds (OPCs) include a heterogeneous group of chemical compounds designed to control pests, weeds or plant diseases. Their application has led to an enhanced increase in agricultural productivity [1]. The toxicity due to OPCs is due to the avidity of its phosphate radicals for the active sites on cholinesterase enzyme. The inhibition of cholinesterases leads to accumulation of acetylcholine at the synaptic junction, causing overstimulation and subsequent disruption of transmission in central and peripheral nervous systems. The degree of absorption and hence toxicity depends upon the contact period with the skin, lipid solubility of the agent and presence of solvents like xylene and other emulsifiers in the formulations [2]. Phosphorothioates need bio activation to their phosphate analogues (oxon) to become biologically active and hence features of intoxication can be delayed by several hours. Clinical manifestations depend upon their predominant effect on muscarinic receptors, nicotinic receptors, or central nervous system. Diagnosis of organophosphorus poisoning is primarily clinical and estimation of red blood cell (RBC) cholinesterase levels can help in confirming the diagnosis in a given clinical setting [3]. Cardiac manifestations comprise of non-specific ST wave changes, sinus tachycardia, bradyarrhythmias, prolongation of corrected QT (QTc) interval and others [4,5]. By its action at central thermal regulation center OPCs can also cause abnormalities in temperature regulation [6]. Here we describe a rare combination of hypothermia and prolongation of QTc interval in a young female with phorate poisoning.
Case Report
A previously healthy19-year old female was admitted to the emergency department of our hospital eight hours after accidental ingestion of approximately 50 mL of �Thimet� (20% weight by volume phorate, chemically O, O-diethyl S-[ethylthio-methyl] phosphorodithioate) with excessive salivation, bronchospasm, tachycardia (140/minute)and diarrhea along with hypoxemic respiratory failure requiring intubation and mechanical ventilation (tidal volume of 6 mL/kg ideal body weight) at presentation.After intubation she developed hypotension that did not respond to fluid boluses and required vasopressors support (nor adrenaline 0.8�g/kg per minute) to maintain a mean arterial blood pressure of 65 mmHg and a urine output of atleast 0.5mL/kg/min. Gastric lavage and whole body wash were performed to prevent further absorption from gastrointestinal tract and the skin. Atropinization was achieved with 9.6 mg of atropine over the next thirty minutesfollowed by a maintenance infusion of 1.2 mg of atropine per hour. She was also given high dose pralidoxime (2-PAM; 1.5 grams bolus followed by 400 mg per hour infusion).Her initial blood gas analysis revealed combined respiratory and metabolic acidosis, whilerenal and hepatic parameters were within the normal limits at baseline (Table 1). Baseline electrocardiogram showed sinus tachycardia, non-specific ST-T wave changes and a corrected QT interval of 430 msec. At 36 hours of admission, patient developed hypothermia with a core temperature of 32� C. ECG done at that time revealed presence of Osborne wave (a positive deflection at the J point) and a corrected QT interval of 480 milliseconds (Figure 1). The levels of plasma ionized calcium (1.1 mmol/L), serum potassium (4 mmol/L) and serum magnesium (2mg/dL) were within normal limits. Patient was rewarmed withinfusion of warm saline, blankets and to achieve a core temperature of 37�C over the next 7-8 hours. A repeat ECG demonstrated normalization of J point, but corrected QT interval was still prolonged (500 milliseconds). Despite three vasopressors (noradrenaline, adrenaline and vasopressin) shock worsened and the patient developed renal failure. She was started on sustained low efficiency dialysis (SLED) for anuria and refractory metabolic acidosis. Her blood, tracheal and urine culture were sterile with normal leukocyte counts ruling out sepsis as a cause of refractory hypotension. Creatine kinase-MB was elevated (110 U/L) and troponin-I was positive by a qualitative card test. Further, echocardiography showed a left ventricular ejection fraction of 10%. She succumbed to refractory shock 40 hours after consumption of the toxic compound.
![]()
Table 1: Association results of H452Y with memory tests.
View Table 1
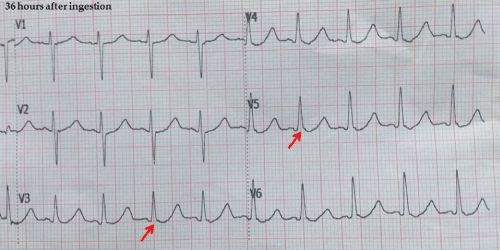
Figure 1: 36 hours after ingestion
View Figure 1
Discussion
Organophosphate poisoning by either intentional or accidental ingestion is still widely prevalent in developing countries and is responsible for roughly 200,000 (two-third of pesticide related deaths) deaths per year [7]. The well-known presentations include:1) cholinergic crisis (overstimulation of muscarinic acetylcholine receptors) manifested by excessive salivation, lacrimation, diarrhea, and bronchorrhea; 2) Nicotinic excess (overstimulation of nicotine acetylcholine receptors) causing tachycardia, mydriasis, hypertension, sweating; 3) neurological (overstimulation of central nervous system muscarinic and nicotinic receptors)manifestations which can be acute (confusion, agitation, and coma), subacute (intermediate syndrome) or delayed (neuropathy due to chronic exposure) [8]. Cardiac manifestations of organophosphate poisoning are often overlooked and anelectrocardiography done at baseline may provide useful information to stratify severity of poisoning and plan further management. Hypothermia with ECG changes occurring along with severe cardiotoxicity as described in the present casehas not been reported with organophosphate poisoning previously.
Thermoregulatory abnormalities have been described previously in animal studies and humans [6,9]. Although fever is common, its occurrence has been attributed primarily to either atropine infusion or the development of sepsis. Hypothermia as a manifestation of organophosphorus poisoning is rare with only few cases described previously. Evidence suggeststhat organophosphorus compounds by their effect on hypothalamus can cause development of hypothermia during the initial 48 hours of ingestion as occurred in the index case [6,9]. However correction of the hypothermia did not correct the electrocardiographic abnormalities and cardiac contractility in our case suggesting primary cardiotoxicity as the cause for the fatal outcome.
Association of OP poisoning with various arrhythmias including ventricular tachycardia has been reported in literature previously [10]. Cardiac complications have been noted in more than half of the patients in various case series, of which prolongation of QTc interval, ST-T changes were the common ones as similar to our patient [4]. Moreover hypotension and prolonged QTc interval have been described to be independent predictors of mortality. Progressive worsening of hypotension in the index patient was associated with a corresponding increase in the QTc interval. Electrocardiographic changes were attributed to organophosphosphate compound due to lack of an alternate explanation for the raised cardiac enzymes and poor LV systolic function. Although the exact cause of cardiac toxicity is still not clear, the postulated mechanisms include parasympathetic/sympathetic imbalance, hypoxemia, electrolyte imbalance, direct cardiotoxicity and others [4,5,11-13]. Higher circulating levels of catecholamines and vaso-active amines (due to increased release by organophosphates) penetrate the myocardial collagen matrix and cause myocardial damage byproducing endothelial erosions and plaque rupture [14]. Additionally, inflammatory mediators like histamine, platelet activating factors, and various cytokines can also cause myocardial damage by causing intense vasospasm and/or coronary artery thrombosis [15]. Myocardium of patients with organophosphate induced cardiotoxicity show patchy interstitial and myocardial inflammation on histopathological examination [11]. Persistence of refractory circulatory failure even after correction of hypothermia ruled out hypothermia as a cause of circulatory failure. Percutaneous cardiopulmonary support has been employed successfully in one such patient of severe organophosphate poisoning previously, but may not be available always [16].
In conclusion, organophosphate compounds account for majority of insecticide related toxicity in humans and can have varied manifestations involving cardiorespiratory and nervous systems. Awareness and early recognition of both common and uncommon manifestations can help in better management of these patients.
References
-
Aktar MW, Sengupta D, Chowdhury A (2009) Impact of pesticides use in agriculture: their benefits and hazards. Interdiscip Toxicol 2: 1-12.
-
Durham WF, Wolfe HR, Elliott JW (1972) Absorption and excretion of parathion by spraymen. Arch Environ Health 24: 381-387.
-
kumar SV, Fareedullah M, Sudhakar Y, Venkateswarlu B, Kumar EA (2010) Current review on organophosphorus poisoning. Arch Appl Sci Res 4: 199-215.
-
Karki P, Ansari JA, Bhandary S, Koirala S (2004) Cardiac and electrocardiographical manifestations of acute organophosphate poisoning. Singapore Med J 45: 385-389.
-
Kose A, Gunay N, Yildirim C, Tarakcioglu M, Sari I, et al. (2009) Cardiac damage in acute organophosphate poisoning in rats: effects of atropine and pralidoxime. Am J Emerg Med 27: 169-175.
-
Moffatt A, Mohammed F, Eddleston M, Azher S, Eyer P, et al. (2010) Hypothermia and Fever after organophosphorus poisoning in humans--a prospective case series. Journal of medical toxicology 6: 379-385.
-
Eddleston M, Phillips MR (2004) Self poisoning with pesticides. BMJ 328: 42-44.
-
Eddleston M, Buckley NA, Eyer P, Dawson AH (2008) Management of acute organophosphorus pesticide poisoning. Lancet 371: 597-607.
-
Gordon CJ (1996) Thermoregulatory aspects of environmental exposure to anticholinesterase agents. Rev Environ Health 11: 101-117.
-
Vijayakumar S, Fareedullah M, Ashok Kumar E, Mohan Rao K (2011) A prospective study on electrocardiographic findings of patients with organophosphorus poisoning. Cardiovasc Toxicol 11: 113-117.
-
Anand S, Singh S, Nahar Saikia U, Bhalla A, Paul Sharma Y, et al. (2009) Cardiac abnormalities in acute organophosphate poisoning. Clin Toxicol (Phila) 47: 230-235.
-
Jayasinghe SS, Pathirana KD (2012) Autonomic function following acute organophosphorus poisoning: a cohort study. PLoS One 7: e37987.
-
Joshi P, Manoria P, Joseph D, Gandhi Z (2013) Acute myocardial infarction: can it be a complication of acute organophosphorus compound poisoning? J Postgrad Med 59: 142-144.
-
Karasu-Minareci E, Gunay N, Minareci K, Sadan G, Ozbey G (2012) What may be happen after an organophosphate exposure: acute myocardial infarction? J Forensic Leg Med 19: 94-96.
-
G�zquez V, Dalmau G, Gaig P, G�mez C, Navarro S, et al. (2010) Kounis syndrome: report of 5 cases. J Investig Allergol Clin Immunol 20: 162-165.
-
Kamijo Y, Soma K, Uchimiya H, Asari Y, Ohwada T (1999) A case of serious organophosphate poisoning treated by percutaneus cardiopulmonary support. Vet Hum Toxicol 41: 326-328.





