International Journal of Anesthetics and Anesthesiology
Systemic Air Embolism during ERCP with Full Recovery
Quy Tran*, Gurpreet Dhaliwal, Christopher Lee and Zohreh Steffens
Department of Anesthesiology, Harbor UCLA Medical Center, USA
*Corresponding author: Quy Tran MD, Department of Anesthesiology, Harbor UCLA Medical Center, 1000 W, Carson St. Box #10, Torrance, CA 90509, California, USA, E-mail: qtran@dhs.lacounty.gov
Int J Anesthetic Anesthesiol, IJAA-3-054, (Volume 3, Issue 4), Case Report; ISSN: 2377-4630
Received: October 17, 2016 | Accepted: November 25, , 2016 | Published: November 28, 2016
Citation: Tran Q, Dhaliwal G, Lee C, Steffens Z (2016) Systemic Air Embolism during ERCP with Full Recovery. Int J Anesthetic Anesthesiol 3:054. 10.23937/2377-4630/3/4/1054
Copyright: © 2016 Tran Q, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Air embolism during gastrointestinal endoscopy procedures is a rare phenomenon but can lead to a catastrophic and fatal event resulting from cardiovascular collapse and neurological injury. Oftentimes, the diagnosis is difficult because the cardiovascular and neurologic symptoms are not specific to systemic air embolism. High index of suspicion with early recognition and treatment is crucial in improving patient outcome. We present a case of cardiac arrest secondary to systemic air embolism during endoscopic retrograde cholangiopancreatography with biliary stent placement which resulted in a good outcome due to early recognition of air embolism and prompt treatment.
Introduction
Endoscopic retrograde cholangiopancreatography (ERCP) is a commonly performed procedure used for both therapeutic and diagnostic purposes which include: assessment of the pancreatobiliary system, endoscopic sphincterotomy for treatment of choledocholithiasis, and placement of biliary duct stents. Visualization of the pancreatobiliary system is achieved with retrograde injection of contrast media through the ampulla of Vater followed by therapeutic intervention. In the United States, approximately 500,000 ERCPs are performed annually with a complication rate between 4% and 10%, and mortality between 0.05% and 1% [1,2]. The most common complications include pancreatitis (3.5%), infection (1.4%), gastrointestinal bleeding (1.3%), and perforation (0.6%). Air embolism following ERCP is rare, with a total of 26 documented cases reported [3]. ERCP involves insufflation of gas into the bowel lumen in order to provide operating space and improve visualization [4]. Air and carbon dioxide are the two most commonly used insufflation agents. Air is not absorbed by bowel and instead is passed via flatus. This can lead to gas retention and contributes to post-ERCP discomfort [4]. In contrast, carbon dioxide (CO2) is easily absorbed into the bloodstream and eliminated via the respiratory system, reducing the risk of embolism [5]. Elevated systemic CO2 in the bloodstream has been associated with physiologic perturbations, including tachycardia and cardiac arrhythmias [4]. Of all the gastrointestinal endoscopic procedures, ERCP has the highest risk of air embolism [6]. Dellon, et al. reported that there were no patients with gas embolism related to CO2 insufflation [5]. We report a case of air embolism during ERCP with full recovery from prompt recognition and adequate resuscitation.
Case Report
A 57-year-old male with history of seizures, hypertension, alcohol abuse and stroke with residual right sided weakness, presented with abdominal pain. Hepatobiliary iminodiacetic acid scan (HIDA) scan showed non-visualization of the gallbladder concerning for cystic duct obstruction. The patient was taken to operating room for laparoscopic cholecystectomy which was converted to an open cholecystectomy, due to a common hepatic duct (CHD) injury. This was repaired over a T-tube. Postoperatively, patient was found to have a bile leak from the CHD injury. On post-operative day 5, the patient underwent an uneventful ERCP procedure with sphincterotomy and common bile duct stent placement. The patient, however, continued to have persistent large volume bilious output from his Jackson-Pratt (JP) drain and a subsequent CT scan revealed a large biloma. A week later, the patient was taken to the GI suite for a second ERCP due to concern for persistent bile leak. The patient was afebrile, with a baseline systolic blood pressure 130-145, heart rate 60-70, and oxygen saturations of 98-100% on room air. The patient was 172 cm in height, and 62 kg in weight. General anesthesia was planned for airway protection. The patient was preoxygenated and premedicated with 2 mg midazolam and 100 mcg fentanyl. Standard ASA monitors were recorded intraoperatively. The patient was then induced with 1 mg/kg of protocol and with 0.7 mg/kg rocuronium for muscle relaxation via a 20 G peripheral IV. Intubation was atraumatic with a MAC 3 blade and a cuffed endotracheal tube #8. End-tidal carbon dioxide (EtCO2) was kept between 29-39 mmHg with mechanical ventilation on volume control mode. Vital signs were stable after induction and intubation. Anesthesia was maintained with 0.3-0.5 MAC of Sevoflurane on 100% FiO2. Patient was turned prone for the procedure. Thirty minutes into the procedure, a precipitous drop in EtCO2 (29 to 17 mmHg), measured via capnography, was noted with concomitant hypertension (BP ~ 170/110 mmHg) and narrow complex tachycardia (HR ~ 120). Within one minute, EtCO2 dropped to 1-2 mmHg associated with severe hypoxia (SpO2 ~ 40%) and central cyanosis, most evident at the neck and face. The carotid pulse was not palpable at this time. Since air has been traditionally used in our GI suite for insufflation, venous air embolism was quickly suspected. The GI endoscopist was immediately notified to abort the procedure. A gurney was brought into the room and the patient was turned supine and positioned with head down position (Trendelenburg). Advanced cardiac life support (ACLS)was initiated as the patient demonstrated pulseless ventricular tachycardia. After 10 minutes of ACLS, with defibrillation × 1 at 200J, 3 mg of epinephrine, and bicarbonate 50 mEq × 4, return of spontaneous circulation was achieved and the patient became normotensive without vasopressor support. A right internal jugular central line was placed during ACLS for fluid resuscitation and 2 L of PlasmaLyte was given over 15 minutes. Transthoracic echocardiography performed intraoperatively showed the presence of diffuse gaseous bubbles in the right cardiac chamber. The patient remained intubated and was transferred to the CT scanner for the total-body computed tomography scan which showed: a few foci of free air adjacent to the T-tube, no evidence of acute infarction or free air in cerebral arteries, no evidence of air in the ventricles or atria. The patient was transferred directly to the intensive care unit for monitoring and initiation of hypothermia protocol. Cardiology was consulted and a formal transesophageal echocardiogram (TEE) showed an ejection fraction of 55-60% with a patent foramen ovale, no wall motion abnormalities, free bubbles noted in left and right atria prior to injection of agitated saline for the PFO study, and in the liver parenchyma, most likely from hepatic veins (Figure 1). After 2 days in the intensive care unit, the patient was successful extubated. The patient remained hemodynamically stable and a neurologic exam did not reveal any neurologic deficits. He was discharged from the hospital 21 days after the incident.
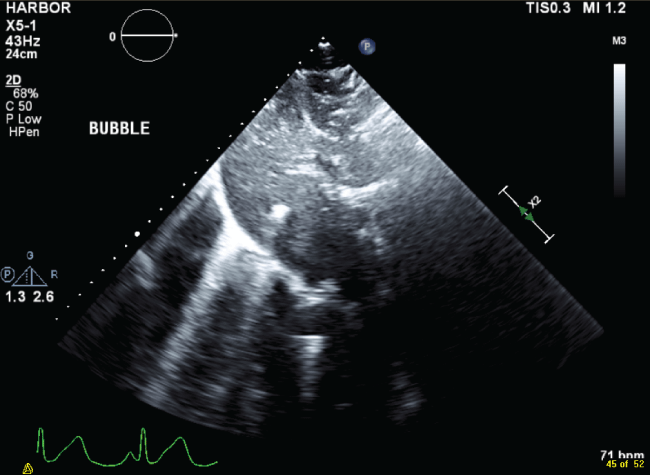
.
Figure 1: Subcostal transesophageal echo view with air (hyperlucent areas) in the liver parenchyma and hepatic veins.
View Figure 1
Discussion
With the increased use of ERCP for both diagnostic and therapeutic modalities, complications such as acute pancreatitis, bleeding, perforation, and infection are common but easily recognizable. Rare complication such as air embolism can be fatal and difficult to detect. Complication rates of ERCP vary widely, up to 5-10% has been reported [7]. The overall mortality rate of ERCP-specific procedures was 0.33%, and the fatality rate associated with cardiopulmonary complications was 0.07% [1]. 41 cases of air embolism following various endoscopic procedures have been published in the literature. Greater than 60% of those cases (26) were related to ERCP and 12 out of those 26 cases had a fatal outcome [3]. A systemic review of case reports by Donepudi et al also found that risk factors for air embolism following ERCP include: previous interventions or surgeries of the bile duct system, transheptaic portosystemic shunts, percutaneous transhepatic biliary drains, blunt or penetrating trauma to the liver, sphincterotomy, metal stent placement, inflammation of the bile duct or surrounding veins, hepatic abscesses or tumors, liver biopsy, and insufflation of air with high pressure, particularly insufflation directly into the bile duct [3]. We presented here a case of non-fatal systemic air embolism during ERCP with sphincterotomy and stent exchange in a male patient with persistent bile leak from previous open cholecystectomy. During ERCP, the high pressure used to distend the duodenum created a pressure gradient allowing the passage of air into the portal venous system during sphincterotomy. Furthermore, bile duct injury from previous cholecystectomy may have facilitated the entrainment of air. In our case, we observed an abrupt and dramatic change in vital signs, most notably the sudden decrease in EtCO2. Our high index of suspicion for air embolism led us to respond quickly. The procedure was immediately stopped; the patient was quickly turned supine on a gurney so the head can be positioned below the level of the feet to prevent air from causing right ventricular outflow obstruction. High flow 100% oxygen was administered to maintain adequate oxygenation, and helps reduce the bubble’s nitrogen content and size. High volume fluid resuscitation was initiated via a central line, and cardiopulmonary resuscitation (CPR) and defibrillation were started in a timely manner. We did not attempt to aspirate the air via the central line because it was a single orifice as opposed to a multi-orifice catheter. We also did not place the patient in a left lateral decubitus position to minimize air lock in the pulmonary artery because the position would interfere with our ability to perform adequate CPR. Return of spontaneous circulation was achieved and the patient remained hemodynamically stable after the arrest so hyperbaric oxygen therapy was not administered. Although the initial intraoperative transesophageal echo did not show a patent foramen ovale, the patient was later found to have a patent foramen ovale on transthoracic echo, which can be present in 25-30% of adults [8]. Despite the presence of a right-to-left shunt predisposing the patient to paradoxical embolism, the patient did not suffer any new neurologic injury as demonstrated on CT imaging and physical exam. Romberg presented a case report where the first sign of a possible air embolism was a decrease in EtCO2 detectable on capnography [9]. Prompt intervention resulted in a good outcome with no morbidity. In his review of nine patients, EtCO2-monitored patients had a 75% survival rate without permanent morbidity, compared to 0% without EtCO2 monitoring [9]. Although ERCP can be done under monitored anesthesia care, at our institution ERCP are preferentially performed under general anesthesia with endotracheal intubation to protect the airway from gastric secretion, and to accurately monitor EtCO2. Additionally, there is evidence to suggest that insufflation with CO2 may be a safer alternative when compared to insufflation with room air. In a systematic review of 9 randomized controlled trials Dellon, et al. reported no adverse pulmonary events related to CO2 insufflation [5]. In conclusion, we present a case of systemic air embolism after ERCP with sphincterotomy under general anesthesia with prompt recognition and intervention for possible air embolism with full recovery. Our case highlights the importance of vigilance in the operating room and maintaining a high index of suspicion for an air embolism in a GI suite that uses air for insufflation. This event led to a practice change in our GI suite with air being replaced with CO2 for insufflation.
References
-
Andriulli A, Loperfido S, Napolitano G, Niro G, Valvano MR, et al. (2007) Incidence rates of post-ERCP complications: a systematic survey of prospective studies. Am J Gastroenterol 102: 1781-1788.
-
Christensen M, Matzen P, Schulze S, Rosenberg J (2004) Complications of ERCP: a prospective study. Gastrointest Endosc 60: 721-731.
-
Donepudi S, Chavalitdhamrong D, Pu L, Draganov PV (2013) Air embolism complicating gastrointestinal endoscopy: a systematic review. World J Gastrointest Endosc 5: 359-365.
-
Cheng Y, Xiong XZ, Wu SJ, Lu J, Lin YX, et al. (2012) Carbon dioxide insufflation for endoscopic retrograde cholangiopancreatography: A meta-analysis and systematic review. World J Gastroenterol 18: 5622-5631.
-
Dellon ES, Hawk JS, Grimm IS, Shaheen NJ (2009) The use of carbon dioxide for insufflation during GI endoscopy: a systematic review. Gastrointest Endosc 69: 843-849.
-
Coelho-Prabhu N, Shah ND, Van Houten H, Kamath PS, Baron TH (2013) Endoscopic retrograde cholangiopancreatography: utilisation and outcomes in a 10-year population-based cohort. BMJ open 3: e002689.
-
Freeman Martin L (2012) Complications of endoscopic retrograde cholangiopancreatography: avoidance and management. Gastrointestinal endoscopy clinics of North America 22: 567-586.
-
Hagen PT, Scholz DG, Edwards WD (1984) Incidence and size of patent foramen ovale during the first 10 decades of life: an autopsy study of 965 normal hearts. Mayo Clin Proc 59: 17-20.
-
Romberg C (2009) Systemic air embolism after ERCP: a case report and review of the literature (with video). Gastrointest Endosc 70: 1043-1045.





