International Journal of Anesthetics and Anesthesiology
Feasibility and Timing of Craniosynostosis Surgery in a Child with Hypoplastic Left Heart Syndrome
C. Hochhold1, P. Paal1, G. Luckner1, M. Rasse2 and C. Velik-Salchner1*
1Department of Anesthesiology and Critical Care Medicine, Medical University of Innsbruck, Austria; Current Address of Peter Paal: Intensive Care Medicine and Cardiac Anaesthesia, Barts Health NHS Trust, London, UK
2Department of Craniomaxillofacial Surgery, Medical University of Innsbruck, Austria
*Corresponding author: Corinna Velik-Salchner, Department of Anaesthesiology and Critical Care Medicine, Medical University of Innsbruck, Anichstrasse 35, A-6020 Innsbruck, Austria, Tel: ++43-512-504-80368, Fax: ++43-512-504-6780368, E-mail: corinna.velik@tirol-kliniken.at
Int J Anesthetic Anesthesiol, IJAA-3-045, (Volume 3, Issue 2), Case Report; ISSN: 2377-4630
Received: January 25, 2016 | Accepted: April 09, 2016 | Published: April 11, 2016
Citation: Hochhold C, Paal P, Luckner G, Rasse M, Velik-Salchner (2016) Feasibility and Timing of Craniosynostosis Surgery in a Child with Hypoplastic Left Heart Syndrome. Int J Anesthetic Anesthesiol 3:045. 10.23937/2377-4630/3/2/1045
Copyright: © 2016 Hochhold C, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Nowadays, children with hypoplastic left heart syndrome (HLHS) survive longer. Thus, the management of such patients undergoing non-cardiac surgery is of increasing relevance. A 12-month-old boy with HLHS following stage II Norwood palliation and Apert syndrome required extended craniosynostosis surgery because of elevated intracerebral pressure. Since extensive blood loss as well as hemodynamic instability were expected, anesthetic and surgical management were carefully planned in a multidisciplinary approach. Advanced invasive hemodynamic monitoring, well-balanced fluid and coagualtion management and an adapted ventilation strategy allowed this complex surgical procedure to be performed safely. Our case illustrates that complex non-cardiac surgery procedures with large volume shifts require a well-thought strategy in children with univentricular repair of congenital heart defects.
Keywords
Anesthesia, Craniosynostosis surgery, Hypoplastic Left Heart Syndrome, Norwood stage II, superior cavopulmonary anastomosis, Apert syndrome
Introduction
Since survival improves in patients with HLHS following each stage of palliation [1], such patients more commonly present for non-cardiac surgery [2,3]. The possibility of extensive blood loss, the risk of sudden cardiovascular instability and the threat of massive air embolism make craniosynostosis surgery challenging even in otherwise healthy children [3,4]. Knowledge of optimal perioperative management of craniosynostosis surgery in children with surgically palliated congenital heart defects is rare. Consequently, our case presentation intends to illustrate that complex non-cardiac surgery procedures with large volume shifts require a well thought out strategy in children with univentricular repair of congenital heart defects. The authors received written permission from the patient's family to report on this case.
Case Description
A 12-month-old boy (69 cm, 7.5 kg) with HLHS and Apert syndrome was scheduled for craniosynostosis surgery. The boy had undergone stage I palliation including a Sano Shunt at the age of 6 days. At the age of 6 months a bilateral bidirectional superior cavopulmonary anastomosis (SCPA, stage II palliation) was performed. At the age of 12 months craniosynostosis surgery was indicated by radiologic signs of increasing intracerebral pressure due to premature closure of the cranial sutures.
Preoperative evaluation showed a cyanotic (SaO2 88%), growth-restricted (69 cm, 7.5 kg) child with developmental deficiency, muscular hypotony and cloni of the lower extremities. Preoperative echocardiography revealed normal ventricular function, minimal insufficiency of the atrioventricular valve and laminar flow profiles in the neo-aorta and Glenn’s anastomoses. Long-term medication included enalapril, furosemide and potassium canrenoate. Acetylsalicylic acid was discontinued ten days prior to surgery.
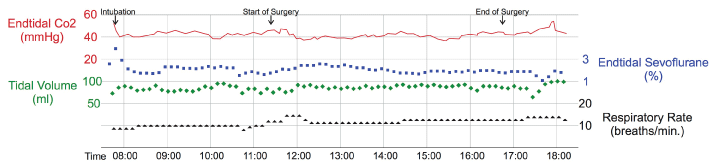
Anesthesia was induced under standard non-invasive monitoring (blood pressure, ECG, pulse oximetry) with midazolam, fentanyl and ketamin-s followed by rocuronium. While bag-valve mask ventilation was unremarkable, endotracheal intubation by direct laryngoscopy was not feasible. Therefore, the airway was immediately secured by fiberoptic intubation. Anesthesia was maintained with sevoflurane and remifentanil. Pressure-controlled ventilation with an inspiratory oxygen fraction of 0.35, a respiratory rate of 16-18/min and an inspiration: expiration ratio of 1:2.5-3.0 was chosen. Positive end-expiratory pressure was limited to 5 mmHg and tidal volume was adjusted to maintain normocapnia (Figure 1).
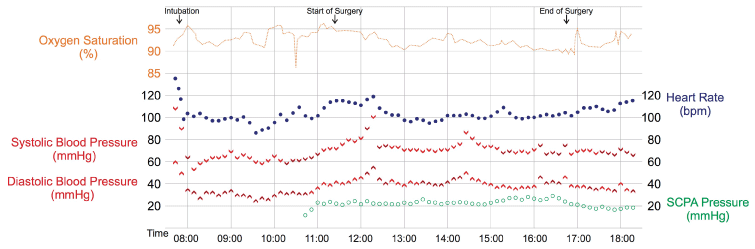
Since the child showed a very poor peripheral vein status and extensive blood loss and inotropic and vasopressor administration were expected, we decided to insert two central venouslines. The central venous catheters (left brachiocephalic vein, left femoral vein) and one arterial line in the right femoral artery were safely established with ultrasound guidance. In order to prevent air embolism, all venous lines were provided with air filters.
Dopamine was administered continuously to maintain mean arterial blood pressure > 50 mmHg. Fluid management was based on balanced isotonic crystalloid solutions with added glucose (Figure 2). Furosemide was given once to avoid volume overload.
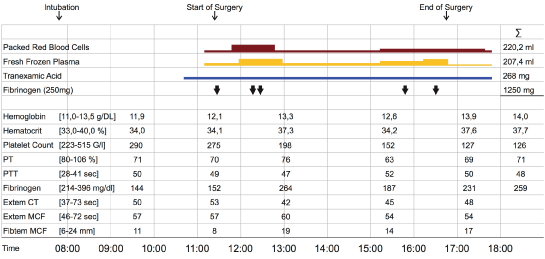
Tranexamic acid was administered during the procedure [5]. Blood loss was replaced with 220 mL of packed red blood cells and 208 mL fresh frozen plasma. Coagulation was supported with fibrinogen concentrate according to coagulation monitoring including rotational thromboelastometry (ROTEM, Figure 3).
For the surgical procedure (Figure 4) a wave line coronal incision and a supraperiosteal dissection to the forehead were performed. A periosteal flap was raised to the orbital rim. The orbital contents were released subperiosteally. The temporalis muscle was reflected from the sphenoid wing. A craniotomy was done with four frontal parts first. Then a supraorbital bandeau was raised. The bandeau was fixed in a position 15mm anteriorly to its former position with a 50 holetitantium plate and the four frontal parts were adjusted to a second plate. The parietal bone was incised and elevated at the anterior end to fit to the frontal bone. An osteosynthesis with resorbable plates (PLLA) was done and the metal plates were removed.
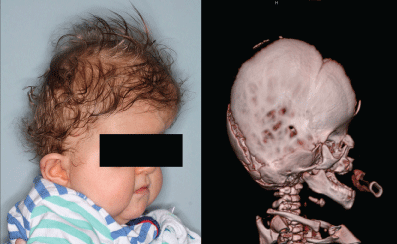
.
Figure 4: Preoperative photo and CT scan of a 12-month-old child with Apert syndrome, which included bilateral synostosis of the coronal sutures and left-sided synostosis of the lambdoid suture, hypoplastic midface with shallow orbits, proptosis.
View Figure 4
After surgery the child was transferred to the pediatric intensive care unit, where he was extubated four hours later. In the further course, cerebrospinal fluid leakage at the surgical site required percutaneous drainage. The boy recovered and was discharged from hospital on day 14 (Figure 5). The patient is doing well six months after surgery.
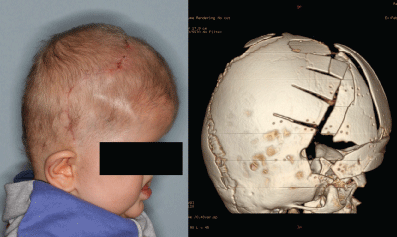
.
Figure 5: Postoperative findings in the same child. The waveline coronal incision and the frontal advancement are clearly visible. In the CT scan the incised parietal bone, and the raised four frontal parts are easily distinguished.
View Figure 5
Discussion
Multidisciplinarily agreed strategy prior to the procedure and surgery performed by an experienced team allowed successful management of craniosynostosis surgery in a child with combined Apert and HLHS following second stage of palliation. Advanced invasive hemodynamic monitoring, well thought out volume management as well as a suitable ventilation strategy allowed this complex surgical procedure to be conducted safely.
Our patient presented after stage II palliation (SCPA) of HLHS. In a child with definitive HLHS the first two steps of the Norwood procedure are of vital importance and should not be delayed. During stage I palliation the neo-aorta is generated from the native pulmonary root. Pulmonary blood flow is ensured by a systemic-to-pulmonary shunt or right ventricle-to-pulmonary conduit. Thereafter, the single ventricle has to supply the pulmonary and the systemic circuit parallel. Thus, complex non-cardiac surgery with expected massive blood loss is not recommended after stage I palliation [6]. Stage II palliation in HLHS is usually performed at the age of 4-6 months and substantially improves hemodynamics by reducing volume load to the single ventricle. We therefore decided to perform craniosynostosis surgery after stage II palliation, even if increased superior venous pressure after SCPA (which is equivalent to the pulmonary arterial pressure) may increase blood loss. Higher venous pressure may be advantageous by reducing the risk of venous air embolism (VAE). In children with cardiac defects right-to-left shunts may lead to fatal paradoxical arterial air emboli in the cerebral or coronary circulation. Following SCPA, arterial air embolism during craniosynostosis surgery is unlikely, since blood flow from the SVC is directly guided into the pulmonary artery [7].
In contrast to our case, Bergmans et al. [8] describe a case of craniosynostosis surgery performed prior to stage II palliation in a child with a complex cyanotic heart defect not suitable for biventricular repair. A definitive HLHS was not present; therefore, stage I palliation (Norwood I) was not necessary. Pulmonary artery banding was performed at the age of 1 month. Since stage II palliation was able to be postponed, the authors decided to perform craniosynostosis surgery prior to SCPA, because bleeding risk was deemed to be lower [8]. On the other hand, this strategy entails the risk of fatal complications of intraoperative arterial air embolism [7].
Hemodynamics and oxygenation are mainly influenced by SCPA flow. For optimal SCPA flow it is crucial to have preserved ventricular function, atrioventricular valve competence and low pulmonary vascular resistance (PVR). Therefore, preoperative echocardiography is mandatory. To maintain cardiac output, hypovolemia, arrhythmia and tachycardia should be aggressively treated. Inserting a central venous line via SVC/SCPA allows pulmonary arterial pressure and central venous oxygen saturation to be measured, thus facilitating hemodynamic and volume management [9].
In patients with SCPA or total cavopulmonary anastomosis (TCPA), positive pressure ventilation limits pulmonary blood flow mainly to the expiration phase. Thus, inspiratory time needs to be minimized and an inspiration: expiration ratio of 1 : 2.5-3.0 is favorable. A respiratory rate < 15-20/min is recommended; higher mean airway pressures resulting from PEEP values > 6 mmHg should be avoided. In our patient, low-frequency mechanical ventilation with limited inspiratory time and low mean airway pressures in combination with controlled administration of fluids enabled stable hemodynamics and adequate oxygenation. Since low PVR is crucial for adequate SCPA flow; conditions increasing PVR (e.g. hypoxemia, acidosis, atelectasis, fluid overload) should be avoided [10,11]. Nevertheless, hyperventilation is deleterious, because hypocapnia causes cerebral vasoconstriction and diminishes blood flow through the SCPA. Even though increasing pCO2 improves oxygenation in SCPA, it should be avoided because effects on cerebral blood flow (vasodilation) may increase surgical bleeding [12]. Spontaneous ventilation is most advantageous for patients with SCPA or TCPA, because pulmonary blood flow preferentially takes place during spontaneous inspiration. Thus, extubation or at least spontaneous breathing should be achieved as soon as possible in such patients under consideration of an individually tailored analgesic and sedation regime [13].
In conclusion, for successful perioperative management of major non-cardiac surgery in children with palliated single ventricle physiology, a previously defined plan for hemodynamic and respiratory management is mandatory. Decision making on timing of surgery requires a multidisciplinary approach. In consideration of the underlying pathophysiology, the patient’s capacity for hemodynamic toleration of expected large volume shifts at each stage of palliation should be estimated and critically discussed.
Quintessence
• Knowing and understanding the pathophysiology of the underlying heart defect is crucial for planning high-risk non-cardiac surgery in patients with complex heart defects.
• Decision making for timing of surgery according to stage of palliation should be based on estimation of hemodynamic tolerance for expected large volume shifts.
• Multidisciplinary approach by pediatricians, pediatric cardiologists, pediatric anesthesiologists experienced in congenital heart disease and surgeons for planning the procedure in a timely manner is absolutely required.
Funding
Funded by department funds.
References
-
Ravishankar C, Gerstenberger E, Sleeper LA, Atz AM, Affolter JT, et al. (2015) Factors affecting Fontan length of stay: Results from the Single Ventricle Reconstruction trial. J Thorac Cardiovasc Surg 151: 669-675.
-
Barron DJ, Kilby MD, Davies B, Wright JG, Jones TJ, et al. (2009) Hypoplastic left heart syndrome. Lancet 374: 551-564.
-
Shuhaiber J, Morgan B, Gottliebson W (2015) Survival outcomes following norwood procedure for hypoplastic left heart. Pediatr Cardiol 36: 57-63.
-
Hughes C, Thomas K, Johnson D, Das S (2013) Anesthesia for surgery related to craniosynostosis: a review. Part 2. Paediatr Anaesth 23: 22-27.
-
Goobie SM, Meier PM, Pereira LM, McGowan FX, Prescilla RP, et al. (2011) Efficacy of tranexamic acid in pediatric craniosynostosis surgery: a double-blind, placebo-controlled trial. Anesthesiology 114: 862-871.
-
Christensen RE, Gholami AS, Reynolds PI, Malviya S (2012) Anaesthetic management and outcomes after noncardiac surgery in patients with hypoplastic left heart syndrome: a retrospective review. Eur J Anaesthesiol 29: 425-430.
-
Adetayo OA, Haider S, Adamo MA, Ehlers MA (2015) Craniosynostosis reconstruction in patients with cyanotic heart defects-risk factors for venous air embolism and overview of preventative strategies. Paediatr Anaesth 25: 754-755.
-
Bergmans B, Kammeraad JA, van Adrichem LN, Staals LM (2014) Craniosynostosis surgery in an infant with a complex cyanotic cardiac defect. Paediatr Anaesth 24: 788-790.
-
Andropoulos DB (2009) Anesthesia for the Patient with a Single Ventricle, in Anesthesia for Congenital Heart Disease. Wiley-Blackwell: 456-479.
-
Lake CM (2004) Hypoplastic Left Heart Syndrome, in Pediatric Cardiac Anesthesia, C.M. Lake and P.D. Booker, Lippincott Williams Wilkins: Philadelphia: 445-466.
-
Yuki K, Casta A, Uezono S (2011) Anesthetic management of noncardiac surgery for patients with single ventricle physiology. J Anesth 25: 247-256.
-
Fogel MA, Durning S, Wernovsky G, Pollock AN, Gaynor JW. Et al. (2004) Brain versus lung: hierarchy of feedback loops in single-ventricle patients with superior cavopulmonary connection. Circulation 110: II147-152.
-
Leyvi G, Wasnick JD (2010) Single-ventricle patient: pathophysiology and anesthetic management. J Cardiothorac Vasc Anesth 24: 121-130.