International Journal of Anesthetics and Anesthesiology
Early Signs and Perinatal Diagnosis of Tracheal Agenesis
Mayke E. van der Putten1*, Viola Christmann1, Lucas E. Matthyssens2, Arthur R.T. Scheffer3 and Chris Bleeker4
1Department of Pediatrics, Radboud University Medical Centre, The Netherlands
2Department of Pediatric surgery, University Hospital of Ghent, Belgium
3Department of Otorhinolaryngology/ENT, Radboud University Medical Centre, The Netherlands
4Department of Anesthesiology, Radboud University Medical Centre, The Netherlands
*Corresponding author: Mayke van der Putten, Department of Pediatrics, Radboud University Medical Centre, P.O. Box 9101, 6500 HB Nijmegen, The Netherlands, Tel: +31 24 3614430, E-mail: Mayke.vanderPutten@radboudumc.nl
Int J Anesthetic Anesthesiol, IJAA-1-029, (Volume 2, Issue 2), Case Report; ISSN: 2377-4630
Received: April 17, 2015 | Accepted: May 09, 2015 | Published: May 11, 2015
Citation: Putten ME, Christmann V, Matthyssens LE, Scheffer ART, Bleeker C (2015) Early Signs and Perinatal Diagnosis of Tracheal Agenesis. Int J Anesthetic Anesthesiol 2:029. 10.23937/2377-4630/2/2/1029
Copyright: © 2015 Putten ME, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
A preterm boy of 30 weeks and five days did not breathe spontaneously at birth and despite visualization of the vocal cords, intubation attempts did not succeed, until a tube 2.0 was placed using an Eschmann stylette, mistakenly considered endotracheal. A few hours later, suspicion of tube dislocation arose and renewed intubation attempts failed. The ENT specialist performed a tracheoscopy and found a blind ending of the trachea in the subglottic area. The trachea could not be felt below this area. Retrospectively the ventilation went via the esophagus to the lungs.
The clinical presentation, the tracheoscopic and CT-scan findings indicated a tracheal agenesis Floyd type 3. This is a rare congenital and usually lethal malformation. Specific antenatal and clinical findings may indicate and facilitate early diagnosis. We hope that this case presentation will raise the awareness of a tracheal agenesis.
Keywords
Airway management, Intubation, Trachea, Tracheal agenesis
Abbreviations
TA: Tracheal Agenesis, TEF: Tracheoesophageal Fistula, GA: Gestational Age, CT: Computed Tomography, V(A)CTE(R)(L) syndrome: Vertebral, Anal, Cardiovascular, Tracheal, Esophageal, Renal/urogenital and/or Limb Anomalies Syndrome, CHAOS: Congenital High Airway Obstruction Syndrome, TACRD: Tracheal/Laryngeal Agenesis/Atresia, Complex Congenital Heart Anomalies, Radial Ray Defects and Duodenal Atresia
Introduction
Tracheal agenesis (TA) is a rare congenital malformation with an incidence of 1 per 50,000-100,000 newborns and approximately 150 cases reported since its first description by Payne in 1900 [1-3]. It comprises a complete or partial absence of the trachea below the larynx, with or without a concomitant tracheoesophageal fistula (TEF) [1,4].
We report the fourth TA patient at our institution with special emphasis on early recognition and awareness for this diagnosis [2].
Case Description
A healthy 41-year-old mother of two healthy boys was evaluated at 19 weeks and five days gestational age (GA) by ultrasound, showing a polyhydramnion and a perimembranous ventricle septum defect. At 30 weeks and four days GA, she was admitted with preterm labour. Ultrasound revealed polyhydramnion with little stomach filling, suspect for esophageal atresia. Despite drainage of five litres of amniotic fluid and inhibition of contractions, she gave birth one day later to a boy weighing 1750 grams, and without any dysmorphic signs. The neonate did not breathe spontaneously, was pale and bradycardic. Apgar scores were one, five and six at 1, 5 and 10 minutes, respectively. After five insufflation breaths and mask ventilation his heart rate and oxygen saturation normalized. Because of insufficient spontaneous breathing we decided to intubate, which initially failed despite visualization of the vocal cords. The anesthesiologist assisted with the intubation and after several attempts, a tube 2.0 was placed using an Eschmann stylette assumed endotracheal, leading to adequate oxygenation and ventilation.
A few hours later, a large air leak with increasing oxygen requirement and hypercapnia was seen. Adjustment of the ventilator settings gave no improvement, and tube dislocation was suspected. Renewed attempts to intubate failed. Nevertheless, mask ventilation remained possible and saturation and hypercapnia were managed. Notable was that the stomach area and thorax moved up and down synchronously.
At tracheoscopy, the ENT specialist found a blind ending trachea in the subglottic area. The trachea could not be felt below this area.
Computed Tomography (CT)-scan showed a short trachea with a 5mm subglottic trajectory below of which no trachea is present. The left and right main bronchi were in open connection with the distal esophagus to the stomach. (Figure 1,2)
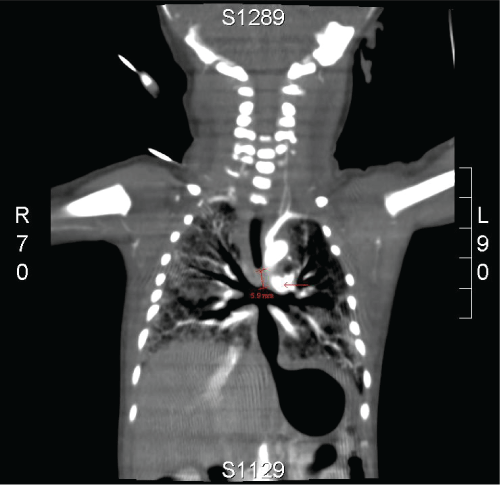
.
Figure 1: CT images show a short trachea with a subglottic trajectory of 5 mm below of which no trachea is present. The left and right main bronchi are in open connection with the distal esophagus.
View Figure 1
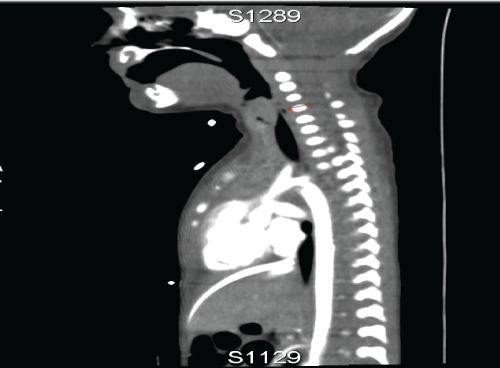
.
Figure 2: CT images show a short trachea with a subglottic trajectory of 5 mm below of which no trachea is present. The left and right main bronchi are in open connection with the distal esophagus.
View Figure 2
Additional findings were an atrial septum defect, ventricular septum defect and rib malformations, compatible with a V(A)CTE(R)(L) syndrome (Vertebral, Anal, Cardiovascular, Tracheal, Esophageal, Renal/urogenital and/or Limb anomalies).
The clinical presentation, tracheoscopic and CT-scan findings led to the diagnosis of TA with TEF, Floyd type 3. For this type of TA, treatment is not available. Further care was considered futile and the patient died a few hours later. The parents refused an autopsy.
Discussion
This case report of the fourth TA patient at our institution demonstrates that clinicians can be confronted with unexpected rare congenital upper airway anomalies [2]. Knowledge of perinatal signs and symptoms may help in diagnosis and management.
The clinical presentation was deceptive since the patient initially responded to normal resuscitation therapy, and had apparently normal vocal cords. Therefore, endotracheal intubation and ventilation seemed a viable option and we erroneously assumed that the tube was in the trachea during his first hours of life. Retrospectively the ventilation was achieved via the distal oesophagus to the lungs. The movement of the stomach in sync with the in- and deflation of the lungs/chest is highly suggestive of a large unobstructed connection between the stomach and the airways.
Prenatal diagnosis
Antenatal diagnosis of TA is difficult, especially in case of a TEF. The combination of polyhydramnion and other anomalies could serve as a clue: 72% of the patients with TA present with polyhydramnion and over 80% have associated anomalies [1,2,5].
TA without TEF is detectable antenatally, presenting with ultrasonographic signs of congenital high airway obstruction syndrome (CHAOS): large echogenic lungs, flattened or inverted diaphragm, dilated airways distal to the obstruction, polyhydramnion, fetal ascites or hydrops, and vigorous, large amplitude fetal breathing movements [6,7].
Fetal MRI may confirm the diagnosis and guide management [4,[8].
Postnatal diagnosis
TA can be detected at birth by the inability to cry in combination with severe respiratory distress and intubation failure [9,10]. Even if associated with TEF, the TA patient cannot breathe spontaneously, because the esophagus collapses during negative intrathoracic pressure [11]. After esophageal intubation the respiratory status may temporarily improve, and may provide time for further evaluation.
Preterm labour (mean gestational age 34 weeks), male predominance (2M:1F), low birth weight (mean 2,048 kg) and low Apgar score (mean 3,5 after 1 and 5 minutes) are aspecific common characteristics [1,2].
Laryngotracheoscopy can diagnose TA, and at esophagoscopy the TEF can be seen [1,2].
CT-scan is the procedure of choice to quickly confirm the diagnosis, delineating the complete anatomy of the distal airway, bronchi and lungs [5].
Pathophysiology
Over a hundred years after its first description by Payne, the etiology of TA remains unclear. In gestational week 5 the ventral lung buds appear from the foregut and thereafter the foregut normally divides into trachea and esophagus [2,12]. Deformation of the foregut on the ventral side may lead to TA, with or without the development of a TEF [12]. External/environmental factors may have a role, as no causal genetic pattern has been identified [1].
Clinical classification and associated anomalies
TA can be classified according to Floyd or Faro, based on the anatomical types [1] (Figure 3). Classification system of tracheal agenesis according to Faro consists of 7 types.
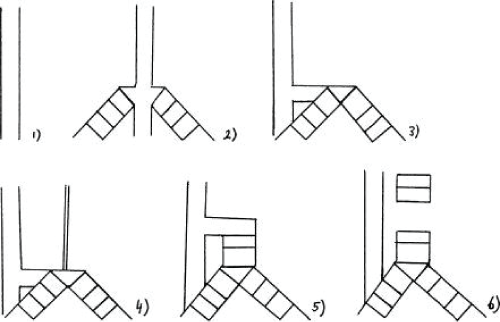
.
Figure 3: Classification system of tracheal agenesis (TA). (1) Faro type A: total pulmonary agenesis. (2) Faro type B/Floyd type III: complete TA with a separate origin of the main bronchi from the esophagus. (3) Faro type C: total TA with normal main bronchi fusing in the midline at the carine; a trachea-esophageal fistula (TEF) is present. Floyd type II: total TA with normal main bronchi fusing in the midline at the carine; a TEF may or may not (not shown) be present. (4) Faro type D: larynx joined by atresic strand to distal trachea with a TEF present. (5) Faro type E/Floyd type I: agenesis of the proximal trachea with a normal caudal segment of the distal trachea and a TEF. Faro type F: agenesis of the proximal trachea with a normal caudal segment of the distal trachea and no TEF is present (not shown). (6) Faro type G: TA in a short segment [1].
View Figure 3
Faro type A is total pulmonary agenesis. Faro type B (Floyd type III, 23-30%) is a complete TA with a separate origin of the main bronchi from the esophagus. Faro type C (Floyd type II, 50-61%) is a total TA with normal main bronchi fusing in the midline at the carine; a trachea-esophageal fistula (TEF) is present. Faro type D: the larynx joined by atresic strand to distal trachea with a TEF present. Faro type E (Floyd type I, 11-15% ) is an agenesis of the proximal trachea with a normal caudal segment of the distal trachea and a TEF. Faro type F is an agenesis of the proximal trachea with a normal caudal segment of the distal trachea and no TEF is present. Faro type G is TA in a short segment [1,2,10,13].
80% or more cases are associated with other congenital malformations, mostly VA(C)TER(L) or TACRD (tracheal/laryngeal agenesis/atresia, complex congenital heart anomalies, radial ray defects and duodenal atresia) [1,14].
Therapy
The ultimate treatment of TA would be surgical. Current surgical treatments are often experimental and unsatisfactory. Multi-stage procedures have been described, using the esophagus or synthetic (tissue-engineered) material as an airway substitute [2,15]. Nevertheless most patients have died within a few hours or days (85% in the first two days) with so far only four long-term survivors reported [1,2,15,16]. The longest survivor in the literature was a girl with Floyd type 1, who became 6 years and 10 months. She was well ventilated with an endotracheal tube inserted through the cervical esophagostomy after thoracic esophageal banding. At 6 years and 10 months she died of a massive bleeding because of perforated esophagus [16].
Conclusion
Tracheal agenesis is a rare congenital and usually lethal malformation. Recognition of specific antenatal and clinical findings may facilitate an early diagnosis.
References
-
de Groot-van der Mooren MD, Haak MC, Lakeman P, Cohen-Overbeek TE, van der Voorn JP, et al. (2012) Tracheal agenesis: approach towards this severe diagnosis. Case report and review of the literature. Eur J Pediatr 171: 425-431.
-
van Veenendaal MB, Liem KD, Marres HA (2000) Congenital absence of the trachea. Eur J Pediatr 159: 8-13.
-
Shankar Raman V, Sugandhi N, Bhatnagar V (2011) Role of the esophagus in surgery for tracheal agenesis. Eur J Pediatr Surg 21: 198-200.
-
Lange P, Fishman JM, Elliott MJ, De Coppi P, Birchall MA (2011) What can regenerative medicine offer for infants with laryngotracheal agenesis? Otolaryngol Head Neck Surg 145: 544-550.
-
Strouse PJ, Newman B, Hernandez RJ, Afshani E, Bommaraju M (2006) CT of tracheal agenesis. Pediatr Radiol 36: 920-926.
-
Hedrick MH, Ferro MM, Filly RA, Flake AW, Harrison MR, et al. (1994) Congenital high airway obstruction syndrome (CHAOS): a potential for perinatal intervention. J Pediatr Surg 29: 271-274.
-
Saadai P, Jelin EB, Nijagal A, Schecter SC, Hirose S, et al. (2012) Long-term outcomes after fetal therapy for congenital high airway obstructive syndrome. J Pediatr Surg 47: 1095-1100.
-
Yamamura J, Schnackenburg B, Kooijmann H, Frisch M, Hecher K, et al. (2010) Magnetic resonance angiography of fetal vessels: feasibility study in the sheep fetus. Jpn J Radiol 28: 720-726.
-
De Jos� Mar�a B, Drudis R, Moncl�s E, Silva A, Santander S, et al. (2000) Management of tracheal agenesis. Paediatr Anaesth 10: 441-444.
-
Iszuari M, Mazita A, Tan GC, Hayati AR, Shareena I, et al. (2010) Tracheal agenesis: a rare cause of unsuccessful tracheal intubation during resuscitation. Med J Malaysia 65: 317-318.
-
Krause U, R�del RM, Paul T (2011) Isolated congenital tracheal stenosis in a preterm newborn. Eur J Pediatr 170: 1217-1221.
-
Kluth D, Steding G, Seidl W (1987) The embryology of foregut malformations. J Pediatr Surg 22: 389-393.
-
Floyd J, Campbell DC Jr, Dominy DE (1962) Agenesis of the trachea. Am Rev Respir Dis 86: 557-560.
-
Evans JA, Greenberg CR, Erdile L (1999) Tracheal agenesis revisited: analysis of associated anomalies. Am J Med Genet 82: 415-422.
-
Fuchimoto Y, Mori M, Takasato F, Tomita H, Yamamoto Y, et al. (2011) A long-term survival case of tracheal agenesis: management for tracheoesophageal fistula and esophageal reconstruction. Pediatr Surg Int 27: 103-106.
-
Soh H, Kawahawa H, Imura K, Yagi M, Yoneda A, et al. (1999) Tracheal agenesis in a child who survived for 6 years. J Pediatr Surg 34: 1541-1543.





