International Archives of Urology and Complications
New Insights into the Chemical and Isotopic Composition of Human-Body Biominerals. II: COM Kidney Stones from Greece
Dimitra Athanasiadou1,2, Athanasios Godelitsas1*, Dimosthenis Sokaras3,4, Andreas-Germanos Karydas3,5, Elisavet Dotsika6, Stelios Xanthos7 and Maria N Gamaletsou8
1School of Science, National and Kapodistrian University of Athens, Greece; aFaculty of Dentistry, McGill University, Canada
2Faculty of Dentistry, McGill University, Montreal, QC, Canada
3Institute of Nuclear and Particle Physics, National Center for Scientific Research "Demokritos", Greece
4Stanford Synchrotron Radiation Lightsource, SLAC National Accelerator Laboratory, USA
5International Atomic Energy Agency (IAEA), Nuclear Spectrometry and Applications Laboratory, Seibersdorf Laboratories, Austria
6Institute of Nanoscience & Nanotechnology, National Center for Scientific Research "Demokritos", Greece
7Department of Automation Engineering, Alexander Technological Educational Institute of Thessaloniki, Greece
8Department of Pathophysiology, Laikon General Hospital and Medical School, National and Kapodistrian University
of Athens, Greece
*Corresponding author: Athanasios Godelitsas, School of Science, National and Kapodistrian University of Athens, Zografou Campus, 15784, Athens, Greece, E-mail: agodel@geol.uoa.gr
Int Arch Urol Complic, IAUC-3-020, (Volume 3, Issue 1), Research Article; ISSN: 2469-5742
Received: October 17, 2016 | Accepted: February 07, 2017 | Published: February 10, 2017
Citation: Athanasiadou D, Godelitsas A, Sokaras D, Karydas AG, Dotsika E, et al. (2017) New Insights into the Chemical and Isotopic Composition of Human-Body Biominerals. II: COM Kidney Stones from Greece. Int Arch Urol Complic 3:020. 10.23937/2469-5742/1510020
Copyright: © 2017 Athanasiadou D, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
We have analyzed kidney stones from patients from Greece in which the mineral phase was calcium oxalate monohydrate (COM) as identified by powder-XRD and FTIR spectra. SEM-EDS analysis revealed microscale-COM crystal aggregates mixed with organic/biological matter. XRF revealed that in addition to Ca (max. ca. 21.4 wt.%; av. ca. 21 wt.%) and Sr (max. 35 ppm; av. 34 ppm), there were significant levels of heavy metal impurities, namely Zn (max. 214 ppm; av. 204 ppm), Pb (max. 149 ppm; av. 136 ppm), Fe (max. 136 ppm; av. 132 ppm) and Cu (max. 17 ppm; av. 15 ppm), as well as minor amounts of Br. As identified by IRMS, all three examined kidney stones presented a very light δ13C signature (average δ13C ca. -25.4‰ PDB) as compared to previously reported data on kidney stones from humans from different geographical locations. The δ18O values averaged ca. -7.31‰ PDB. With regard to radioactive isotopes, HR γ-ray spectrometry demonstrated the existence of the natural radionuclides 214Pb and 214Bi due to 238U-series, and also an additional amount of 40Κ. We conclude that these kidney stones from southeastern Europe are enriched in essential biometals (Zn and Fe), and also contain a high content of harmful heavy metals such as Pb, and traces of U. This elemental composition may be related to a toxic diet and/or environmental pollution.
Keywords
Biominerals, Kidney stones, Calcium oxalate monohydrate (COM), Metals, Isotopes
Abbreviations
COM: Calcium Oxalate Monohydrate; SEM: Scanning Electron Microscopy; EDS: Energy Dispersive Spectrometry; XRD: X-ray Diffraction; FTIR: Fourier-Transform Infrared Spectroscopy; XRF: X-ray Fluorescence; IRMS: Isotope Ratio Mass Spectrometry; HR: High Resolution; γ-ray: Gamma Ray
Introduction
Kidney stone disease (urolithiasis) is a serious health problem, involving more than 5% of the population, especially in developing countries [1,2]. Urolithiasis, the formation of urinary calculi in the kidney, bladder and/or urethra, can be attributed to several risk factors related to geographical region, gender and dietary habits [e.g. 3-5]. Urine ion supersaturation, and the effects of various mineralization promoters and inhibitors, in combination with genetic risk factors, plays a significant role in the crystallization process within kidney stones [5]. Understanding the elemental and isotopic composition of kidney stones is central to understanding their initiation and growth, and to developing treatments that prevent their formation.
Kidney stones vary in composition and contain a wide variety of different organic and inorganic compounds. They can be broadly classified into five types of stones depending on their major chemical composition: (a) calcium oxalate, (b) calcium phosphate, (c) uric acid, (d) struvite and (e) cystine [e.g. 3,6]. Calcium oxalate-containing stones are the most common type, where the mineral phase can exist as calcium oxalate monohydrate (COM), calcium oxalate dihydrate (COD) and rarely calcium oxalate trihydrate (COT), the latter being the most thermodynamically unstable form of calcium oxalate. COM is the most stable form, and it predominates in more than 80% of total stones. Struvite, uric acid and cystine stones are found in a smaller percentage of cases, at 15%, 5-10% and 1% respectively of total kidney stones [3,5-7].
In previous studies by others, analytical techniques such as X-ray Fluorescence/XRF and Proton Induced X-ray Emission/PIXE were used to verify the concentrations of trace elements in COM stones [8-14]. Further analytical techniques, such as Inductively Coupled Plasma -Optical Emission & -Mass Spectrometry/ICP-OES & ICP-MS, Atomic Absorption Spectrometry & Emission Spectrometry/AAS & AES, Instrumental Neutron Activation Analysis/INAA and Synchrotron-based methods were also used [9,11,15-25].
The main purpose of the present work was to contribute to information on the chemical composition of European kidney stones, specifically those from southeastern Europe (Greece). Here, XRF was also used for the quantitative analysis of COM kidney stones which were previously characterized using multiple techniques (Scanning Electron Microscopy/SEM, X-ray Diffraction/XRD, and Fourier-Transform Infrared Spectrometry/FTIR). Also, carbon and oxygen isotopic ratios (δ13C and δ18O) were measured using Isotope Ratio Mass Spectrometry/IRMS whereas high-resolution (HR) Gamma (γ)-ray spectrometry measurements provided with data of natural radionuclides (238U-series, 232Th-series, 40K) in COM kidney stones from the human body.
Materials and Methods
Kidney stones of different sizes were examined after surgical removal from patients at the Laikon General Hospital in Athens (Greece). Three kidney stones were subjected to further detailed analysis after optical examination.
Microscopic analysis was performed using a Jeol JSM-5600 microscope equipped with an Oxford EDS microanalytical system. XRD patterns were acquired by a Siemens D5005 (Bruker AXS) X-ray diffractometer using Cu Kα-radiation. FTIR spectra were collected using a Perkin Elmer Spectrum One spectrometer operating in the frequency range 4000-450 cm-1 with a 2 cm-1 nominal energy resolution. KBr discs were prepared for each measurement from 1.3 mg of powdered sample. An in-house developed transferable milli-beam XRF spectrometer was used for the XRF analysis. This XRF spectrometer involves a Rh-anode side-window low power X-ray tube (50 W, 50 kV maximum high voltage, 75 μm Be window) encompasses a Si-PiN diode X-ray detector (500 μm nominal crystal thickness, energy resolution of about 165 eV (FWHM) at Mn-Kα). For this study, the X-ray tube was functioned at 40 kV with hard filtering (i.e., Ni: 42.5 mg/cm2, V: 33.0 mg/cm2) to improve peak-to-background ratio for the determination of metallic trace elements. Quantitative analysis was based on elemental calibration factors obtained experimentally using a set of thin mono-elemental targets with endorsed areal density. Thin pellets of a 1.3 cm diameter each were prepared by compressing the homogenized sample powder using a 5 tonnes hydraulic press.
Stable carbon and oxygen isotopes were measured by Isotope Ratio Mass Spectrometry/IRMS (Thermo Scientific Delta V Plus equipped with a FlashEA 1112 elemental analyser). For evaluating the δ13C and δ18O values of the studied kidney stones, 150-200 mg of each sample was used. Finally, the radionuclides contained in the COM kidney stones were determined by high-resolution γ-ray spectrometry using a Canberra high-purity Ge detector (HPGe).
Results and Discussion
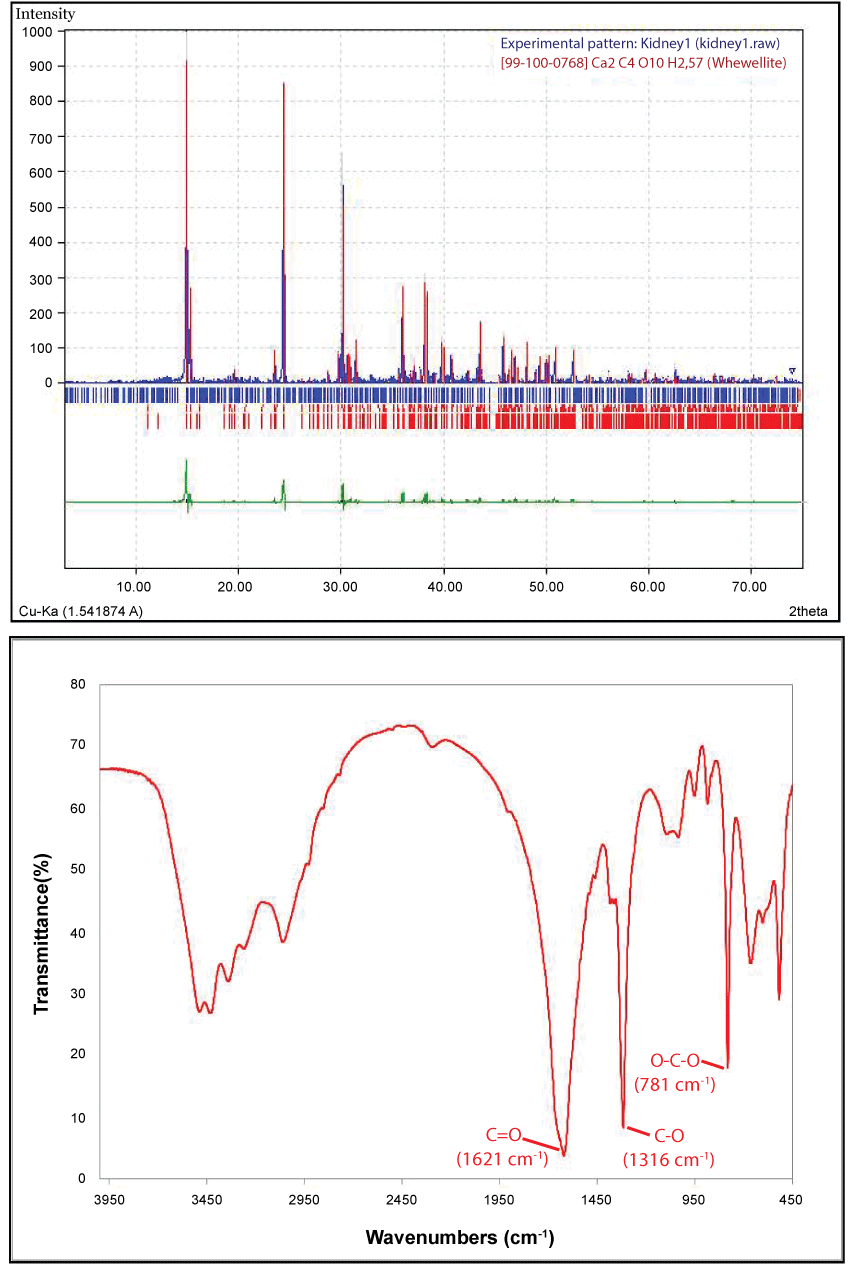
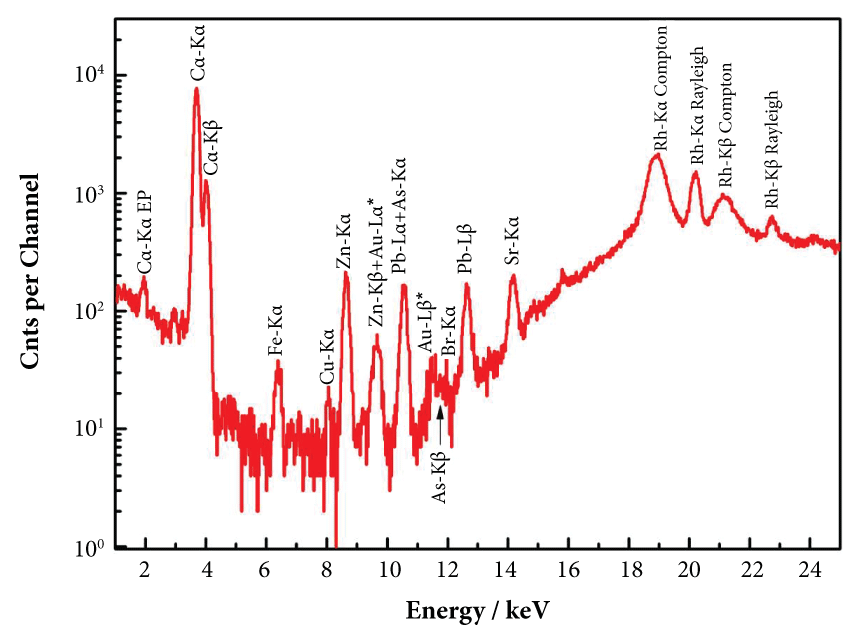
Representative morphology and aggregations of COM microcrystals comprising human kidney stones from Greece, as recorded by SEM, are shown for one sample in Figure 1. In vitro studies have shown that carboxylate-rich urinary macromolecules, as well as anionic molecules, can serve as adhesives that stimulate the aggregation of COM crystals [26]. The powder XRD patterns of all three samples disclosed characteristic peaks corresponding to COM [27,28] whereas the FTIR spectra included characteristic bands in the region between 4000-450 cm-1 attributable to the vibrations of O-C-O, C-O and C=O, at 781, 1316 and 1621 cm-1, arising from oxalates of COM [29,30] (Figure 2). All the above bulk data confirmed that the examined biominerals belong to the COM type of kidney stones. XRF analysis (Figure 3) revealed the presence of essential biometals (Ca, Fe, Cu, Zn) within the stones, as well as potentially toxic metals, alkali earth metals and halogens (Pb, Sr, Br). The presence of Au L-characteristic X-rays in the XRF spectrum is attributable to contamination by the XRF spectrometer component materials, namely the X-ray detector (contact electrode). Calcium was found to be the major nonorganic constituent in all kidney stones (max. ca. 21.4 wt.%; av. ca. 21 wt.%).

.
Figure 1: SEM images of kidney stones from the present study (upper: COM crystal aggregates; lower: post-colored image indicating COM crystals partly covered by organic/biological matter).
View Figure 1
.
Figure 2: Powder-XRD pattern (upper) and FTIR spectrum (lower) of COM kidney stones from the present study.
View Figure 2
.
Figure 3: Representative XRF spectrum concerning the COM kidney stones from Greece. The presence of Au L-characteristic X-rays is due to interference by the XRF spectrometer materials, namely the X-ray detector (contact electrode).
View Figure 3
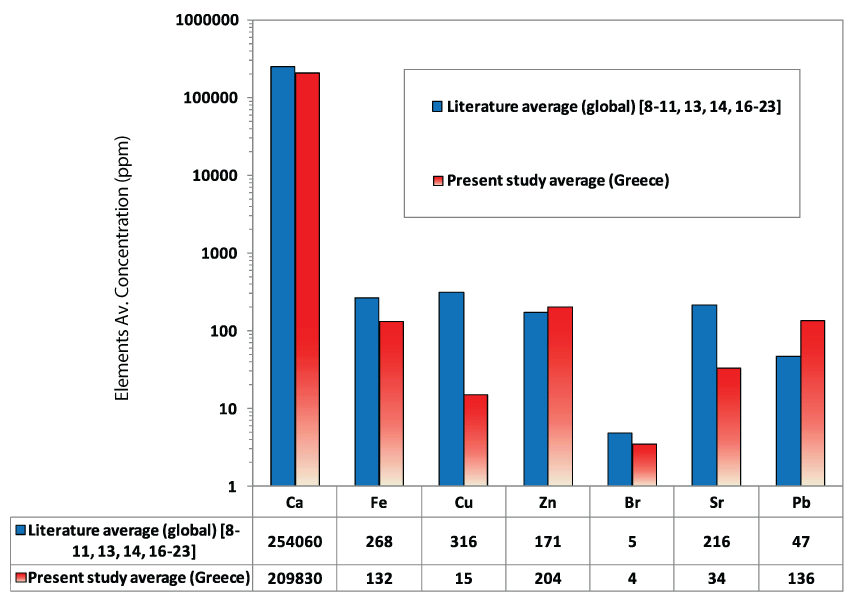
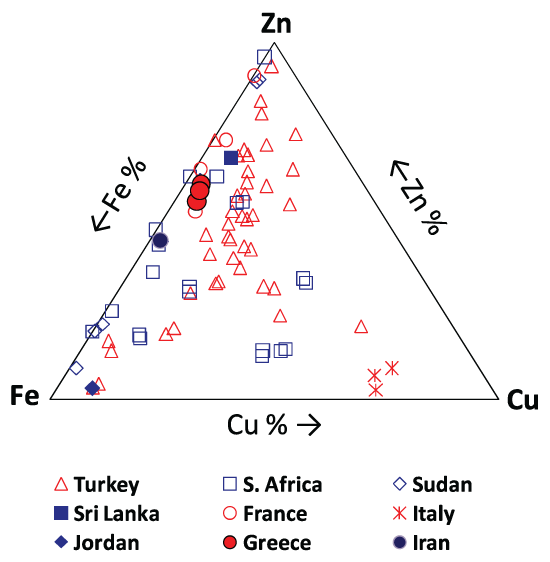
Dairy and milk products, eggs, water and tea, amongst others, influences Ca content in humans [11]. Strontium (max. 35 ppm; av. 34 ppm) might substitute for Ca due to its valence and similar ionic radius [31]. The precipitation of bromide salts could explain the existence of Br (av. 4 ppm) in the studied kidney stones, but the role of Br in calcium oxalate stone formation needs further investigation [16]. Iron and copper were present in all examined samples, with a maximum concentration of 136 ppm and 17 ppm, respectively (av. 132 and 15 ppm). Beans, meats and water are the main source of Fe, whereas green foods, meats, flour and milk products are rich in Cu [11]. The role of Fe in stone formation has not been elucidated completely. According to Muñoz, et al. [32], ferric ions affect the development of calcium oxalate stones by forming stable chemical interactions with the oxalate ions on their crystal surface. Nevertheless, Meyer, et al. [33] reported that Fe does not influence calcium oxalate crystal growth. The relative low concentration of Cu compared to the global average could be related to its low excretion in urine [14]. In addition, Zn (max. 214 ppm; av. 204 ppm) was found to occur in concentrations slightly higher than the global average (Figure 4). Similar to Sr, Zn can substitute for Ca in the crystal lattice of calcium oxalate stones [14,34]. The presence of Zn could also be associated with levels of atmospheric pollution [35]. In comparison to other geographic areas around the world, these Greek kidney stones contained comparatively high amounts of Zn and Fe, and less Cu (max. 17 ppm; av. 15 ppm), as illustrated in Figure 5.
It is evident that the kidney stones examined here are highly enriched in potentially toxic metals such as Pb, as compared to the global average (Figure 4). The presence of Pb (max. 149 ppm; av. 136 ppm) may be natural and/or industrial, as well as unexpected (e.g., home-made wine [36]), whereas high concentrations of Pb are found to be related to Ca-containing stones rather than organic phases [14]. It is notable that no concentrations of As were observed, even though high As concentrations have been observed in other biominerals such as cholesterol gallstones from England and Greece [37].
The carbon and oxygen stable isotopic composition of the kidney stones was found to be in the same range. The average δ13C and δ18O values of the samples were calculated at -25.4‰ PDB and ca. -7.31‰ PDB, respectively. The δ13C values are low compared with findings from other studies [38,39]. These low values could be attributable to remaining organic material trapped between the calcium oxalate crystals (Figure 1). The isotopic composition of carbon reflects the dietary habits of the patients. From the δ13C values the kidney stones are enriched in the light isotope 12C. In a previous study, it was showed that biogenic calcium oxalate was enriched in the light isotope 12C compared to geological calcium oxalate which was enriched in the heavy isotope 13C [40]. DeNiro and Epstein [41] and Jim, et al. [42] were able to determine the influence of various food animals (pigs and rats) in the isotopic composition of carbon. However, the accurate determination of food influence in the isotopic composition of humans is difficult to establish because of the variety of dietary habits. Likewise, the isotopic composition of oxygen reflects the living geographical location of the patients. Dotsika, et al. [43] showed that δ18O values around -7.5‰ PDB are typical values corresponding to areas near the sea.
Here we reveal for the first time using high-resolution γ-ray spectrometry that the kidney stones of urolithiasis patients may contain radioactive isotopes. Natural radionuclides such as 214Pb (475 Bq/Kg) and 214Bi (375 Bq/Kg) due to 238U- series and 40K (4 Bq/Kg) were determined (Figure 6). This also reveals that traces of U are present in the kidney stones. Finally, it is noticeable that artificial radionuclides such as 137Cs (arising from nuclear accidents including Chernobyl) were not detected.
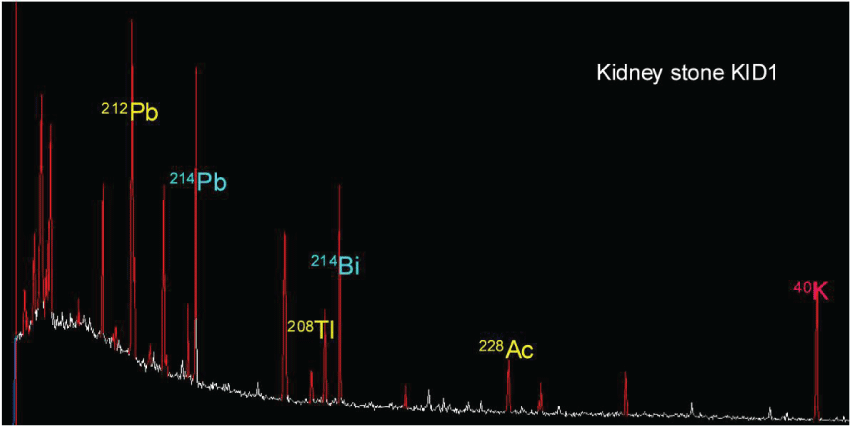
.
Figure 6: Representative HR γ-ray spectrum, indicating natural radioactivity, concerning the studied COM kidney stones from Greece.
View Figure 6
Conclusions
The present study characterized calcium oxalate monohydrate human kidney stones from Greece by means of microscopic, diffraction and spectroscopic techniques (SEM-EDS, XRD, FTIR), as well as by XRF, IRMS and high-resolution γ-ray spectrometry techniques, to elucidate their chemical and isotopic composition. XRF analysis indicated that Ca is the principal metal in the COM aggregates, along with Zn, Pb and Fe. According to IRMS results, all kidney stones showed a similar δ13C signature (average δ13C ca. -25.4‰ PDB) which is rather low compared to values reported in the literature, whereas the δ18O values were found to have an average of ca. -7.31‰ PDB. Concerning radioactive isotopes, γ-ray spectrometry revealed the existence of 214Pb and 214Bi, due to 238U- series, and also a detectable amount of 40K. From these results, we can conclude that the examined Greek kidney stones are enriched in metals essential for the normal functioning of the body such as Zn (max. 214 ppm; av. 204 ppm), but they also contain excess harmful elements such as Pb (max. 149 ppm; av. 136 ppm) and traces of U. This might be related to a potentially toxic diet and/or environmental pollution.
Acknowledgements
We would like to thank Prof. M. D. McKee (Faculty of Dentistry, McGill University, Canada) for comments and reading of the manuscript, as well as Prof. E. Iatrou (Dept. of Chemistry, University of Athens, Greece) for collaboration during FTIR measurements.
References
-
Parmar MS (2004) Kidney stones. BMJ 328: 1420-1424.
-
Stamatelou KK, Francis ME, Jones CA, Nyberg LM, Curhan GC (2003) Time trends in reported prevalence of kidney stones in the United States: 1976-1994. Kidney Int 63: 1817-1823.
-
John Fitzpatrick (2007) Urinary stone disease: The practical guide to medical and surgical management. New Jersey: Humana Press Inc.
-
Goldfarb S (1994) Diet and nephrolithiasis. Annu Rev Med 45: 235-243.
-
Moe OW (2006) Kidney stones: pathophysiology and medical management. Lancet 367: 333-344.
-
Wesson JA, Ward MD (2007) Pathological biomineralization of kidney stones. Elements 3: 415-421.
-
Singh VK, Rai AK, Rai PK, Jindal PK (2009) Cross-sectional study of kidney stones by laser-induced breakdown spectroscopy. Lasers Med Sci 24: 749-759.
-
Pineda CA, Peisach M (1994) Micro-analysis of kidney stones sequentially excreted from a single patient. Nucl Instrum Methods Phys Res B 85: 896-900.
-
Paluszkiewicz C, Galka M, Kwiatek W, Parczewski A, Walas S (1997) Renal stone studies using vibrational spectroscopy and trace element analysis. Biospectroscopy 3: 403-407.
-
Pineda-Vargas CA, Rodgers AL, Eisa ME (2004) Nuclear microscopy of human kidney stones, comparison between two population groups. Radiat Phys Chem 71: 947-950.
-
Abboud IA (2008) Concentration effect of trace metals in Jordanian patients of urinary calculi. Environ Geochem Health 30: 11-20.
-
Moroz TN, Palchik NA, Dar'in AV (2009) Microelemental and mineral compositions of pathogenic biomineral concrements: SRXFA, X-ray powder diffraction and vibrational spectroscopy data. Nucl Instrum Methods Phys Res A 603: 141-143.
-
Zarasvandi A, Heidari M, Sadeghi M, Mousapoor E (2013) Major and trace element composition of urinary stones, Khuzestan province, southwest, Iran. J Geochem Explor 131: 52-58.
-
Bazin D, Chevallier P, Matzen G, Jungers P, Daudon M (2007) Heavy elements in urinary stones. Urol Res 35: 179-184.
-
Donev I, Mashkarov S, Maritchkova L, Gotsev G (1977) Quantitative investigation of some trace elements in renal stones by neutron activation analysis. J Radioanal Nucl Chem 37: 441-449.
-
Sarmani S, Kuan LL, Bakar MA (1990) Instrumental neutron activation analysis of kidney stones. Biol Trace Elem Res 26: 497-502.
-
Koeberl C, Bayer PM, Höbarth K (1993) Determination of rare earth and other trace elements abundances in human kidney stones and brain tissue by instrumental neutron activation analysis. J Radioanal Nucl Chem 169: 269-276.
-
Durak I, Kilic Z, Sahin A, Akpoyraz M (1992) Analysis of calcium, iron, copper and zinc contents of nucleus and crust parts of urinary calculi. Urol Res 20: 23-26.
-
Turgut M, Unal I, Berber A, Demir TA, Mutlu F, et al. (2008) The concentration of Zn, Mg and Mn in calcium oxalate monohydrate stones appears to interfere with their fragility in ESWL therapy. Urol Res 36: 31-38.
-
Jhaumeer-Laulloo S, Subratty AH (1999) Analysis of urinary calculi in Mauritius. Sci Technol 3: 87-93.
-
Giannossi ML, Summa V, Mongelli G (2013) Trace element investigations in urinary stones: A preliminary pilot case in Basilicata (Southern Italy). J Trace Elem Med Biol 27: 91-97.
-
Chandrajith R, Wijewardana G, Dissanayake CB, Abeygunasekara A (2006) Biomineralogy of human urinary calculi (kidney stones) from some geographic regions of Sri Lanka. Environ Geochem Health 28: 393-399.
-
Keshavarzi B, Yavarashayeri N, Irani D, Moore F, Zarasvandi A, Salari M (2015) Trace elements in urinary stones: a preliminary investigation in Fars province, Iran. Environ Geochem Health 37: 377-389.
-
Siritapetawee J, Pattanasiriwisawa W (2008) An attempt at kidney stone analysis with the application of synchrotron radiation. J Synchrotron Radiat 15: 158-161.
-
Blaschko SD, Miller J, Chi T, Flechner L, Fakra S, et al. (2013) Microcomposition of human urinary calculi using advanced imaging techniques. J Urol 189: 726-734.
-
Sheng X, Ward MD, Wesson JA (2003) Adhesion between molecules and calcium oxalate crystals: critical interactions in kidney stone formation. J Am Chem Soc 125: 2854-2855.
-
Orlando MTD, Kuplich L, de Souza DO, Belich H, Depianti JB, et al. (2008) Study of calcium oxalate monohydrate of kidney stones by X-ray diffraction. Powder Diffr 23: S59-S64.
-
Abboud IA (2008) Mineralogy and chemistry of urinary stones: patients from North Jordan. Environ Geochem Health 30: 445-463.
-
Shippey TA (1980) Vibrational studies of calcium oxalate monohydrate (whewellite) and an anhydous phase of calcium oxalate. J Mol Struct 63: 157-166.
-
Estepa L, Daudon M (1997) Contribution of Fourier transform infrared spectroscopy to the identification of urinary stones and kidney crystal deposits. Biospectroscopy 3: 347-369.
-
Zhou XS, Shen GR, Wu JG, Li WH, Xu YZ, et al. (1997) A spectroscopic study of pigment gallstones in China. Biospectroscopy 3: 371-380.
-
Muñoz JA, Valiente M (2005) Effects of trace metals on the inhibition of calcium oxalate crystallization. Urol Res 33: 267-272.
-
Meyer JL, Angino EE (1977) The role of trace metals in calcium urolithiasis. Invest Urol 14: 347-350.
-
Blaschko SD, Chi T, Miller J, Flechner L, Fakra S, et al. (2013) Strontium substitution for calcium in lithogenesis. J Urol 189: 735-739.
-
Godelitsas A, Nastos P, Mertzimekis TJ, Toli K, Simon R, et al. (2011) A microscopic and Synchrotron-based characterization of urban particulate matter (PM10-PM2.5 and PM2.5) from Athens atmosphere, Greece. Nucl Instrum Methods Phys Res B 269: 3077-3081.
-
Sipos P, Szentmihályi K, Fehér E, Abaza M, Szilágyi M, et al. (2003) Some effects of lead contamination on liver and gallbladder bile. Acta Biol Szeged 47: 139-142.
-
Athanasiadou D, Godelitsas A, Sokaras D, Karydas AG, Dotsika E, et al. (2013) New insights into the chemical and isotopic composition of human-body biominerals. I: Cholesterol gallstones from England and Greece. J Trace Elem Med Biol instead of Trace Elem Med Biol
27: 79-84.
-
Krouse HR, Levinson AA (1984) Geographical trends of carbon and sulphur isotope abundances in human kidney stones. Geochim Cosmochim Acta 48: 187-191.
-
Manning DA, Lopez-Capel E, White ML, Barker S (2008) Carbon isotope determination for separate components of heterogeneous materials using coupled thermogravimetric analysis/isotope ratio mass spectrometry. Rapid Commun Mass Spectrom 22: 1187-1195.
-
Hoefs J, Armbruster Th (1978) 13C/12C-Verhaltnisse in menschlichen Harnkonkrementen. Naturwissenschaften 65: 586-589.
-
DeNiro MJ, Epstein S (1978) Influence of diet on the distribution of carbon isotopes in animals. Geochim Cosmochim Acta 42: 495-506.
-
Jim S, Ambrose SH, Evershed RP (2004) Stable carbon isotopic evidence for differences in the dietary origin of bone cholesterol, collagen and apatite: implications for their use in palaeodietary reconstruction. Geochim Cosmochim Acta 68: 61-72.
-
Dotsika E, Lykoudis S, Poutoukis D (2010) Spatial distribution of isotopic composition of precipitation and spring water in Greece. Glob Planet Change 71: 141-149.