International Archives of Urology and Complications
Mesothelioma of the Tunica Vaginalis Testis: Case Report and Review
Ruiz Hernández M1*, Fabuel Alcañiz JJ1, Gutiérrez-Pecharromán AM2, Romio de las Heras E2, Rodríguez-Patrón Rodríguez R1, Varona Crespo C2 and Burgos Revilla FJ1
1Department of Urology, Hospital Universitario Ramón y Cajal, IRYCIS, Madrid, Spain
2Department of Pathology, Hospital Universitario Ramón y Cajal, IRYCIS, Madrid, Spain
*Corresponding author: Mercedes Ruiz Hernández, Department of Urology, Hospital Universitario Ramón y Cajal, IRYCIS, Madrid, Spain, E-mail: merh06@gmail.com
Int Arch Urol Complic, IAUC-2-015, (Volume 2, Issue 1), Case Report; ISSN: 2469-5742
Received: March 06, 2016 | Accepted: May 17, 2016 | Published: May 20, 2016
Citation: Ruiz HM, Fabuel AJJ, Gutiérrez-Pecharromán AM, Romio de las HE, Rodríguez-Patrón RR, et al. (2016) Mesothelioma of the Tunica Vaginalis Testis: Case Report and Review. Int Arch Urol Complic 2:015. 10.23937/2469-5742/1510015
Copyright: © 2016 Ruiz HM, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
A 93-year-old man with chronic hydrocele and no history of asbestos exposure underwent a hydrocelectomy, which revealed several nodules in the tunica vaginalis. The histopathological diagnosis was malignant mesothelioma, requiring a second procedure. A radical inguinal orchiectomy with hemiscrotectomy was performed, reporting tumor-free surgical margins; therefore, no adjuvant treatments were given. Three months postoperative, the patient continued asymptomatic, scrotal examination was normal, and control CT scan showed no evidence of lymphatic disease or metastases.
Keywords
Malignant mesothelioma, Tunica vaginalis, Hydrocele, Scrotal pain
Introduction
Malignant mesothelioma of the tunica vaginalis (MMTV) is a rare neoplasm which represents approximately 5% of mesotheliomas [1]. Its clinical presentation is unspecific (the most common is chronic hydrocele) [1], making preoperative suspicion unusual. This causes a negative impact on prognosis; for example if a hydrocelectomy is intervened via scrotum, the neoplasm lymphatic drainage is altered and may facilitate metastases [2].
We report our case to create awareness amongst urologists regarding the incidence and clinical presentation of this tumor. Preoperative ultrasound exam or an intraoperative biopsy may help in identifying this pathology and provide a better opportunity to manage these cases.
Case Report
We report a case of a 93-year-old man with a chronic history of hydrocele on his left testicle. During the first evaluation, surgery was ruled out due to the age and comorbidities of the patient, however, in a second consultation, his primary complaint was significant scrotum growth. A sonograph of the scrotum was performed, reporting numerous solid nodules on left hemiscrotum, varying from 3-27 mm, some of which showed hypervascularity in Doppler ultrasound (Figure 1).
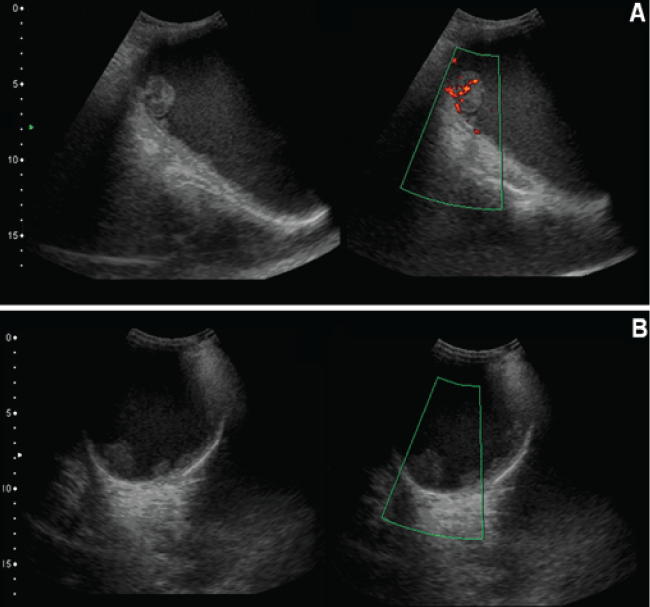
.
Figure 1: Doppler Ultrasound Imaging: a Thick tunica vaginalis with abundant nodules. A) Nodules with hypervascularity; B) Nodules with hypervascularity.
View Figure 1
Due to the low incidence of malignant tumors of the tunica testis, malignancy was not suspected. Instead, the findings were associated with pachivaginalitis which is often related with chronic hydrocele. A hydrocelectomy was indicated, however, transoperative findings revealed exudative yellow hydrocele fluid, a thick wall, and abundant nodules ranging from 3 mm to 3 cm in diameter.
Histopathology reported a membranous and thickened structure with whitish nodules in gross examination. The microscopic description revealed a neoplastic proliferation formed by cells, mostly arranged in a papillary or nodular pattern, although diffused sheets of cells without any recognisable distribution was observed with inflammatory infiltrates focally dispersed, alternating with desmoplastic areas. The tumor cells were medium size with rounded and polygonal shapes. The cytoplasm was homogeneous eosinophilic with tiny translucent vacuoles. The nucleus-cytoplasm ratio was high and the nuclei were rounded with an irregular contour, coarse chromatin and prominent nucleoli. The mitotic count was high. Immunohistochemistry evidenced a strong Calretinin and D2-40 expression in cells. The pathological diagnosis was epithelioid MMTV.
A thoracic-abdominal CT scan was performed for staging. Neither lymphatic nor organ metastases were detected. Inguinal orchiectomy with left hemiscrotectomy was performed reporting tumor-free surgical margins and tumoral cell infiltrate of the testicular parenchyma, equivalent to the hydrocelectomy specimen.
There was no indication for adjuvant chemo or radiotherapy. Three months after surgery, the patient was reported asymptomatic and there was no evidence of local recurrence or metastases in control CT scan.
Discussion
Mesotheliomas are low incidence tumors with MMTV representing around 5% of all cases [1]. Only 250 cases have been reported in medical literature. Onset occurs in males between 55 and 75 years old, with up to 10% affecting men younger than 25 [1]. They are locally aggressive tumors with as many as 34% of cases evidencing infiltration of the subvaginal connective tissue and testis at the time of diagnosis, as was the case of our patient [1,3].
MMTV develops in conjunction to asbestos exposure and is considered its main risk factor increasing incidence by 30 to 67%. It has been associated with direct contact as well as family and occupational history [1,4].
Asbestos fibres in the scrotum can cause chronic inflammation and lead to malignant transformation [5]. It is thought that asbestos could reach the scrotum via lymphatic or hematological routes [4]. The latency period is long with a mean time of 46.2 years (range 16.2-59.7) [4]. It can also be associated with chronic inflammation processes such as: epididymitis, (similarly to tumorigenesis mechanism that occurs in pleural peritoneal mesothelioma) [6], with chronic hydrocele [7,8], radiation exposure, herniorrhaphy/hernioplasty, and chromosomal abnormalities such as the losses on 1p, 3p, 6q, 9q and monosomy 22 [9]. Currently, these associated risk factors have not been proven to cause MMTV, only asbestos exposure has been confirmed to be associated with MMTV. In this case, our patient denies exposure to asbestos.
Clinical presentation is unspecific [1,5,10]. The majority of patients suffer from: hydrocele (49.5%), testicular tumor (36.6%), scrotal hernia (5.9%), spermatocele (2%), testicular torsion (2%), or posttraumatic testicular lesion (1.6%) [11]. Bilateral presentation is extremely rare, present in only 2% of cases, with similar incidence between right and left scrotum [1].
The diagnosis is frequently made in the postoperative stage (97%), as was our case [1]. During surgery, MMTV should be suspected when hemorrhagic hydroceles, nodules or fibrotic thickening of the tunica vaginalis is present [1,3,10,11]. In some cases, ultrasonography may suggest MMTV findings, although these are often unspecific (irregular thickening of the tunica vaginalis, presence of heterogeneous nodules that vary in size, and associated hydrocele) [8,11-15]. Color Doppler ultrasound may report either hypervascularity [16] or hypervascularity [15]. Preoperative diagnosis is seldom determined by fine-needle aspiration (FNA) of the hydrocele fluid; however, FNA can diagnose 75-80% of cases when puncturing suspicious nodules [16]. The use of FNA generates a hypothetical risk of tumor seeding during the procedure, reason why some authors do not recommend its use when there is a suspicion of mesothelioma infiltration into the testis layers [5,17].
The gross characteristics of MMTV are a thick tunica vaginalis with white-to-yellow nodules (Figure 2). The tumor has an invasive growth, which commonly infiltrates the testicular parenchyma, epididymis and spermatic cord, and from there it can reach the peritoneum [12]. Microscopically, cells tend to be cuboid with an eosinophilic cytoplasm. Immunohistochemistry (Figure 3) is necessary for diagnosis [12,18-20]. The tumor shows strong positivity for Calretinin, WT1 (Willm's Tumor antibody), EMA (epithelial membrane antigen), Thrombomodulin, Cytokeratin 7, variable positivity to Cytokeratin 5/6 and negativity to Cytokeratin 20 and Carcinoembryonic antigen. MMTV has been sub-classified into four groups: epithelial (65%), sarcomatous (15%), desmoplastic (2%) and biphasic (18%) [21].
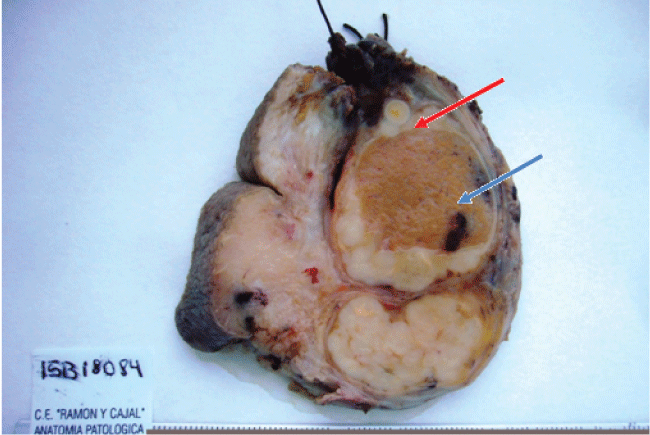
.
Figure 2: Surgical specimen of orchiectomy with hemiscrotectomy after hydrocelectomy. Thick tunica vaginalis and nodules. Evidence of neoplasm infiltration to the testicular parenchyma (red arrow) with growth towards the previous hydrocelectomy area (blue marrow).
View Figure 2
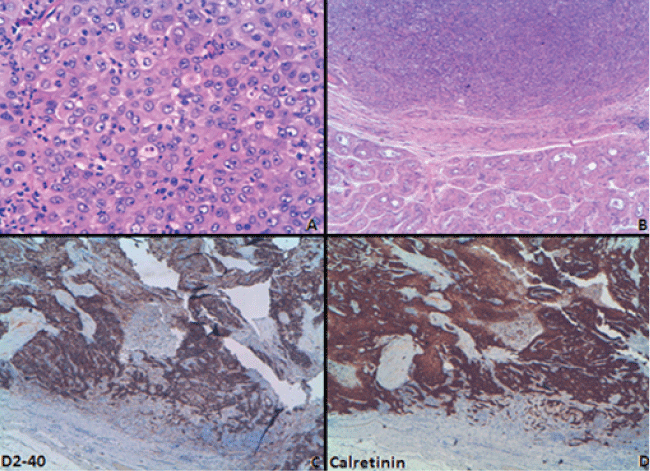
.
Figure 3: Microscopic description. Cuboid cells with eosinophilic cytoplasm and translucent vacuoles (A); Inflamatory infiltration (B); D2-40 positive expression cells (C); Presence of calretinin (D).
View Figure 3
The differential diagnosis must be done with tumors originating from Mullerian vestiges, reactive mesothelial hyperplasia (which has been associated with inguinal hernia and is limited to the serosal layer without stromal invasion), adenomatoid tumor (benign testicular tumor), etc [10].
The standard treatment for non-metastatic MMTV is inguinal radical orchiectomy with hemiscrotectomy [5,10]. Inguinal or iliac lymphadenectomy is not necessary in the majority of the cases, since less than 15% of the patients have lymph node disease [1,22]. If there is suspicion of metastases, then a staging lymphadenectomy must be performed. Evidence of adjuvant therapy such as chemo- or radiotherapy is limited. A report of gross residual tumor after surgery that exhibited adequate response to chemo-radiotherapy [10] and a case involving lymph node dissemination treated with chemotherapy after lymphadenectomy, with a 3 year tumor-free outcome, have been documented [23].
There are some prognostic factors such as: age, primary disseminated disease, type of surgery (tunica vaginalis resection or orchiectomy), and recurrence [1]. Patients older than 60 years of age have a worse prognosis than younger patients. The most common metastases is lymphatic, mainly to retroperitoneal nodes, followed by inguinal and iliac nodes. Organ metastases are rare at the time of diagnosis (lungs, bones, omentum, etc.) Depending on the type of surgery, there are different recurrence rates: 35.7% with resection of the hydrocele wall, 10.5% after scrotal orchiectomy and 11.5% after inguinal orchiectomy [1]. Tumor recurrence after surgery is observed in 52.5% of the cases, most often local recurrence, followed by lymph node metastases, and finally by organ dissemination [1] (lung, liver, omentum, bone, pleura [24], and in other isolated cases, colon, spleen, mesentery or mediastinum metastases). A thoracic and abdominal CT scan should be performed for staging or to dismiss suspicion of metastases.
60% of recurrences occur within the first two years. The mean survival ranges from 18 to 23 months [1]. Although, this prognosis seems to be better than in patients with mesotheliomas in other locations (ex. Mean survival range of 4-18 months in pleural mesotheliomas) [11]. This is maybe because the location allows for a more radical surgery.
Clinical examinations and CT scan or retroperitoneal ultrasound should be performed every 3 months for the first 2 years. Afterwards, patients should be monitored on a yearly basis until the fifth year of follow-up [1,3].
Urologists should suspect MMTV in patients with chronic hydrocele, predominantly when there are associated symptoms such as rapid growth or palpable inguinal nodules. Ultrasound can be helpful in the diagnosis, when nodules are detected. If nodules are detected during surgery a frozen section can help to distinguish a benign from a malign neoplasm; however, immunohistochemistry is mandatory for a definitive diagnosis.
In conclusion, although nodular thickening of the tunica vaginalis is unspecific and can appear in benign neoplasms, a malignant neoplasm should be suspected.
Acknowledgment
We would like to acknowledge our Department of Pathology for the images.
References
-
Plas E, Riedl CR, Pfl üger H (1998) Malignant mesothelioma of the tunica vaginalis testis: review of the literature and assessment of prognostic parameters. Cancer 83: 2437-2446.
-
Stephenson AJ, Gilligan TD (2011) Neoplasms of the testis. In: Wein AJ, (ed). Campbell-Walsh Urology. 10th ed. Philadelphia, PA: Elsevier Saunders, 845-882.
-
Guney N, Basaran M, Karayigit E, M üsl ümanoglu A, Guney S, et al. (2007) Malignant mesothelioma of the tunica vaginalis testis: a case report and review of the literature. Med Oncol 24: 449-452.
-
Mensi C, Pellegatta M, Sieno C, Consonni D, Riboldi L, et al. (2012) Mesothelioma of tunica vaginalis testis and asbestos exposure. BJU Int 110: 533-537.
-
Yen CH, Lee CT, Su CJ, Lo HC (2012) Malignant mesothelioma of the tunica vaginalis testis: a malignancy associated with recurrent epididymitis? World J Surg Oncol 10: 238.
-
Peterson JT Jr, Greenberg SD, Buffler PA (1984) Non-asbestos-related malignant mesothelioma. A review. Cancer 54: 951-960.
-
Albino G, Nenna R, Inchingolo CD, Marucco EC (2010) Hydrocele with surprise. Case report and rewiew of literature. Arch Ital Urol Androl 82: 287-290.
-
G ürdal M, Erol A (2001) Malignant mesothelioma of tunica vaginalis testis associated with long-lasting hydrocele: could hydrocele be an etiological factor? Int Urol Nephrol 32: 687-689.
-
Taguchi T, Jhanwar SC, Siegfried JM, Keller SM, Testa JR (1993) Recurrent deletions of specific chromosomal sites in 1p, 3p, 6q, and 9p in human malignant mesothelioma. Cancer Res 53: 4349-4355.
-
Segura-González M, Urias-Rocha J, Castelán-Pedraza J (2015) Malignant Mesothelioma of the Tunica Vaginalis: A Rare Neoplasm--Case Report and Literature Review. Clin Genitourin Cancer 13: e401-405.
-
Schure PJ, van Dalen KC, Ruitenberg HM, van Dalen T (2006) Mesothelioma of the tunica vaginalis testis: a rare malignancy mimicking more common inguino-scrotal masses. J Surg Oncol 94: 162-164.
-
Chekol SS, Sun CC (2012) Malignant mesothelioma of the tunica vaginalis testis: diagnostic studies and differential diagnosis. Arch Pathol Lab Med 136: 113-117.
-
Fields JM, Russell SA, Andrew SM (1992) Case report: ultrasound appearances of a malignant mesothelioma of the tunica vaginalis testis. Clin Radiol 46: 128-130.
-
Aggarwal P, Sidana A, Mustafa S, Rodriguez R (2010) Preoperative diagnosis of malignant mesothelioma of the tunica vaginalis using Doppler ultrasound. Urology 75: 251-252.
-
Wolanske K, Nino-Murcia M (2001) Malignant mesothelioma of the tunica vaginalis testis: atypical sonographic appearance. J Ultrasound Med 20: 69-72.
-
Boyum J, Wasserman NF (2008) Malignant mesothelioma of the tunica vaginalis testis: a case illustrating Doppler color flow imaging and its potential for preoperative diagnosis. J Ultrasound Med 27: 1249-1255.
-
Bruno C, Minniti S, Procacci C (2002) Diagnosis of malignant mesothelioma of the tunica vaginalis testis by ultrasound-guided fine-needle aspiration. J Clin Ultrasound 30: 181-183.
-
Berti E, Schiaffino E, Minervini MS, Longo G, Schmid C (1997) Primary malignant mesothelioma of the tunica vaginalis of the testis. Immunohistochemistry and electron microscopy. Pathology 29:96-99.
-
Churg A (2003) Paratesticular mesothelial proliferations. Semin Diagn Pathol 20: 272-278.
-
Bisceglia M, Dor DB, Carosi I, Vairo M, Pasquinelli G (2010) Paratesticular mesothelioma. Report of a case with comprehensive review of literature. Adv Anat Pathol 17: 53-70.
-
Attanoos RL, Gibbs AR (2000) Primary malignant gonadal mesotheliomas and asbestos. Histopathology 37: 150-159.
-
Abe K, Kato N, Miki K, Nimura S, Suzuki M, et al. (2002) Malignant mesothelioma of testicular tunica vaginalis. Int J Urol 9: 602-603.
-
Lopez JI, Angulo JC, Ibañez T (1995) Combined therapy in a case of malignant mesothelioma of the tunica vaginalis testis. Scand J Urol Nephrol 29: 361-364.
-
Pannier D, Caty A, Hysi I, Bouchindhomme B, Copin MC, et al. (2011) [Pleuro-pulmonary metastases from a malignant mesothelioma of the tunica vaginalis]. Rev Mal Respir 28: 1155-1157.





