International Archives of Urology and Complications
Importance of Prostate-Specific Antigen Screening before and after Bacillus Calmette-Guérin Intravesical Bladder Instillation Treatment to Avoid Unnecessary Prostate Biopsy
Takehiko Okamura1*, Hidetoshi Akita1, Kenji Yamada1, Takahiro Kobayashi1, Ryosuke Ando2, Noriyasu Kawai2, Keiichi Tozawa2 and Takahiro Yasui2
1Department of Urology, J.A. Aichi Anjo Kosei Hospital, Japan
2Department of Nephro-Urology, Nagoya City University Graduate School of Medical Sciences, Japan
*Corresponding author: Takehiko Okamura, Department of Urology, J.A. Aichi Anjo Kosei Hospital, 28 Higashihirokute, Anjo-cho, Anjo 446-8602, Japan, Tel: +81-566-75-2111, Fax: +81-566-76-4335, E-mail: hiko2546@sf.commufa.jp
Int Arch Urol Complic, IAUC-1-006, (Volume 1, Issue 1), Research Article; ISSN: 2469-5742
Received: May 29, 2015 | Accepted: June 26, 2015 | Published: June 29, 2015
Citation: Okamura T, Akita H, Yamada K, Kobayashi T, Ando R, et al. (2015) Importance of Prostate-Specific Antigen Screening before and after Bacillus Calmette-Guérin Intravesical Bladder Instillation Treatment to Avoid Unnecessary Prostate Biopsy. Int Arch Urol Complic 1:006. 10.23937/2469-5742/1510006
Copyright: © 2015 Okamura T, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objective: The adverse effects of Bacillus Calmette-Guérin (BCG) intravesical instillation therapy for non-muscle invasive bladder cancer (NMIBC) vary widely, and can include BCG-induced prostatitis. In contrast, prostate cancer may occasionally develop along with bladder cancer. Therefore, if prostate-specific antigen (PSA) levels increase after BCG therapy, a prostate biopsy should be performed to exclude prostate cancer.
Methods: PSA values were determined in 114 men who had undergone intravesical BCG instillation at our institution between 2000 and 2012. The mean age of the patients was 69.2 years (range, 40-87 years). All cases had NMIBC; the histological classification was G1 in 9 patients, G2 in 74 patients, G3 in 17 patients, and carcinoma in situ in 14 patients. PSA was measured more than once in 37 out of 100 cases, excluding the cases wherein prostate cancer had already been diagnosed.
Results: Patients showed an increase in PSA levels in 7 cases, and 3 patients showed an increase in PSA levels after BCG treatment. Two cases developed granulomatous prostatitis after BCG treatment, and a definitive diagnosis was made via biopsy.
Conclusions: Our results confirm that PSA examination is essential both before and after BCG treatment. PSA level elevation in patients treated with intravesical BCG is self-limiting; prostate biopsies are not essential in these patients and could be delayed while the PSA levels are monitored.
Keywords
Bacille Calmette-Gu�rin, Non-muscle-invasive bladder cancer, PSA, Granulomatous prostatitis
Introduction and Objective
Several side effects have been documented following the use of Bacillus Calmette-Gu�rin (BCG) intravesical instillation therapy for the treatment of non-muscle invasive bladder cancer (NMIBC), including BCG-induced prostatitis. Lamm et al. [1] reported that 17 of 1,278 patients (1.3%) developed symptomatic prostatitis during BCG treatment [1], whereas 17 of 3,377 patients (0.2%) were found to have developed symptomatic prostatitis during BCG treatment in a post-marketing surveillance study in Japan [2]. A number of similar findings have also been reported [3-5]. On the other hand, prostate cancer occasionally develops along with bladder cancer, and it can be difficult to make a clinical diagnosis in such cases. Therefore, if a patient showed an elevated PSA level and induration on digital rectal examination, there was previously no option but to perform a prostate biopsy after BCG intravesical instillation therapy, to exclude prostate cancer. We have encountered 2 cases of granulomatous prostatitis after BCG treatment in our hospital, wherein a definitive diagnosis was made via biopsy.
This study was designed to assess whether a significant association exists between intravesical BCG immunotherapy and an increase in serum PSA levels by analyzing cases in which PSA levels were measured before, during, and after BCG treatment. A better understanding of the relationship between BCG immunotherapy and serum PSA levels could help avoid unnecessary prostate biopsies.
Materials and Methods
Of the patients who underwent BCG intravesical instillation treatment (Tokyo 172 strain purchased from Nihon BCG Laboratory Co., Tokyo) for NMIBC, including carcinoma in situ (CIS), in our hospital between January 2000 and February 2012, 114 cases underwent at least one PSA level measurement. BCG (40-80mg) was administered 6-8 times, once every week, for prophylaxis, with or without the addition of the same dose of maintenance BCG therapy 6-8 times, once every month [6]. Follow-up was performed once a week during the weekly treatment period, and then every 1 to 3 months after the cessation of treatment, depending on the patients' condition. The mean patient age was 69.2 years (range, 40-87 years). All the cases had NMIBC, and the disease grade was found to be G1, G2, G3, and CIS in 9, 74, 17, and14 cases, respectively. Transurethral resections (TUR-Bt) were performed in all non-CIS cases before BCG treatment. The patient characteristics are summarized in table 1.
![]()
Table 1: Characteristics of patients
View Table 1
MRI was performed using a 1.5-T MR unit (MAGNETOM Avanto, Avanto SQ, and Symphony; Siemens). A body coil was used for signal excitation, and a multichannel phased-array coil for signal reception. The MRI examination for all patients included transverse T1- and T2-weighted fast spin-echo imaging, and transverse diffusion weighted imaging (DWI). Transverse T2-weighted fast spin-echo imaging was performed under specific conditions of Repetition Time/Echo Time [TR/TE]=3620/116.0 msec, signal average of 1, echo train length of 15, 170� flip angle, 230Hz/Px bandwidth, 24cm � 24cm field of view, 5-mm section thickness, 1-mm intersection gap, and a 256 � 256 matrix, and transverse T1-weighted fast spin-echo imaging was performed in conditions of TR/TE=503/7.7msec, signal average of 1, echo train length of 3, 170� flip angle, 230Hz/Px bandwidth, 24cm � 24cm field of view, 5-mm section thickness, 1-mm intersection gap, and a 256 � 256 matrix through the prostate gland and seminal vesicles.
Transverse DWI was performed using an Echo Planar Imaging (TR/TE=3300/77 msec, signal average of 7, 90� flip angle, 1698Hz/Px bandwidth, Echo Planar Imaging, 35cm � 35cm field of view, 5-mm section thickness, 1-mm intersection gap, and a 128 � 102 matrix). Images were acquired with motion-probing gradient pulses that were applied sequentially along three orthogonal orientations with two b factors (0 and 1000sec/mm2).
Results
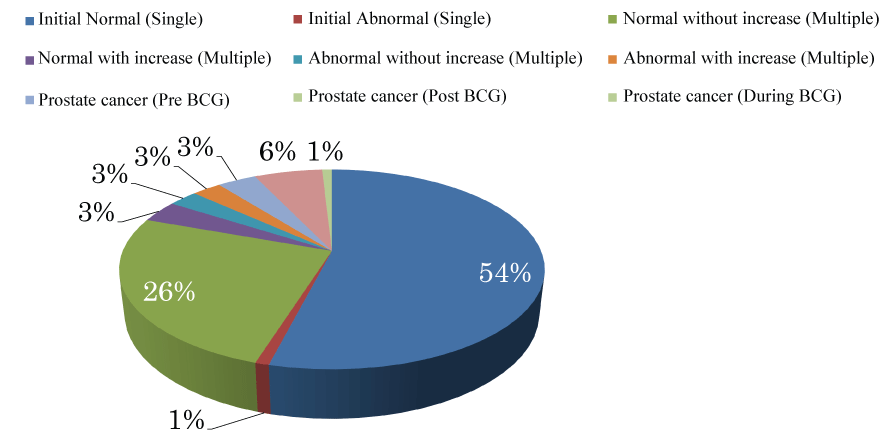
In at least 40% of cases, the serum PSA level was only measured before BCG treatment; pre- and post- BCG treatment PSA measurements were only performed in 26% of cases (Figure 1). The frequency of single or multiple PSA measurements, increases in PSA levels, and the timing of PSA measurements are summarized in figure 2. In more than 50% of cases, PSA levels were only assessed once, and the levels were within the normal range. Half of the remaining cases did not show elevated PSA levels, despite BCG treatment.
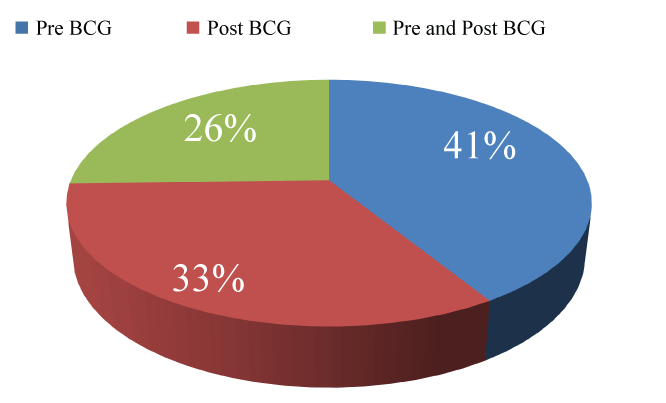
.
Figure 1: Timing of prostate-specific antigen (PSA) level measurements. In at least 40% of cases, PSA levels were only measured before Bacillus Calmette-Gu�rin (BCG) treatment, whereas PSA levels were measured both pre- and post-BCG treatment in only 26% of cases.
View Figure 1
.
Figure 2: Prostate-specific antigen (PSA) values. More than 50% of cases received only a single PSA measurement, and the levels were within the normal range. Half of the remaining cases did not have an elevated PSA level, despite undergoing Bacillus Calmette-Gu�rin treatment.
View Figure 2
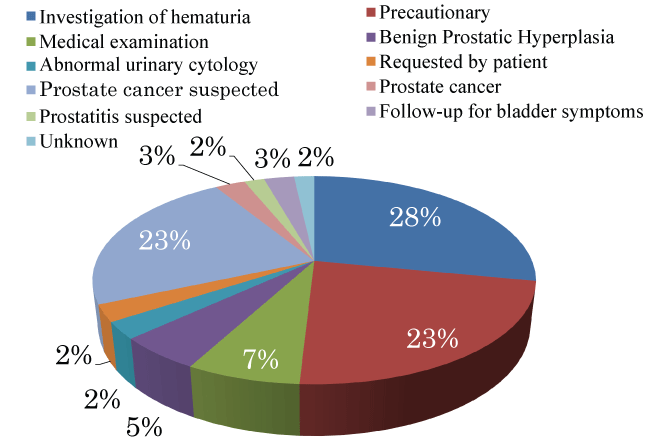
We also studied the reasons for measuring PSA levels (Figure 3). PSA levels were measured several times in 37 of 100 cases, excluding the patients who had already been diagnosed with prostate cancer. Fourteen patients were diagnosed with prostate cancer before or after BCG treatment due to suspected prostate cancer in 8 cases, hematuria in 2 cases, and screening in one case; the remaining 3 patients were receiving treatment for a previously diagnosed prostate cancer. According to the International Union Against Cancer (UICC) TNM staging system classification, the prostate cancer was graded as T1c in 7 cases, T1a in 4 cases, T2b in 2 cases (one case diagnosed before bladder cancer was detected and one case diagnosed at total cystectomy after being refractory to BCG treatment), and T4 in 1 case (diagnosed before bladder cancer). Initially, many patients had micro hematuria due to bladder cancer, and the PSA levels were measured to screen for prostate cancer in more than one quarter of these patients. Thus, prostate cancer was suspected in 37 patients, 7 of whom had elevated PSA levels; 3 of these had elevated PSA levels after BCG treatment. Of these 3 patients, 2 underwent prostate biopsy, and were consequently diagnosed with granulomatous prostatitis. These two cases are further detailed below:
.
Figure 3: Reasons for prostate-specific antigen (PSA) level measurement. Many patients had micro hematuria due to bladder cancer, and PSA levels were assessed for prostate cancer screening in more than a quarter of these patients. In almost one quarter of the cases, PSA levels were measured because prostate cancer was suspected, or simply as a precaution.
View Figure 3
Case 1
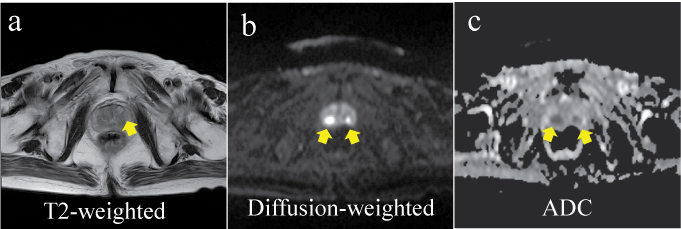
A 75 year old man presented to our hospital for further examination of left hydronephrosis. His PSA level was slightly elevated (4.6ng/ml). He was diagnosed as having left ureteral cancer and a left nephrouretelectomy was performed. He subsequently experienced repeated NMIBC recurrence, and prophylactic BCG intravesical instillation therapy was performed after TUR-Bt. His PSA level subsequently increased to 17.5ng/ml, and prostate biopsy was performed. The MRI findings just before biopsy are shown in figure 4. T2-weighted imaging showed a low signal (Figure 4a) and DWI showed a high signal (Figure 4b); a low apparent diffusion coefficient (ADC) was noted in the marginal area (Figure 4c). A diagnosis of granulomatous prostatitis was made based on the histopathological findings.
.
Figure 4: Case 1; Magnetic resonance imaging findings just before the biopsy. The T2-weighted image shows a low signal (a), the diffusion-weighted image shows a high signal (b), and the apparent diffusion coefficient (ADC) appears to be low in the marginal area (c).
View Figure 4
Case 2
A 73 year old man had previously undergone a prostate biopsy because he had an elevated PSA level of 5.6ng/ml, and revisited our hospital for further examination after a further increase in his PSA levels, to 7.0ng/ml. He was also found to have bladder cancer. TUR-Bt and a second prostate biopsy were performed, and no sign of malignancy was found in the biopsy specimen. Six months after BCG treatment, he developed left epididymitis; after an additional 6 months, his PSA level had increased to 15.4ng/ml. On DWI, a high signal was observed, along with a low ADC in the marginal area (Figure 5a-5c). On histopathological examination, multifocal epithelioid granulomas were observed, and there was no evidence of malignancy. Hence, based on the histopathological findings, the patient was diagnosed with granulomatous prostatitis (Figure 5d,5e).
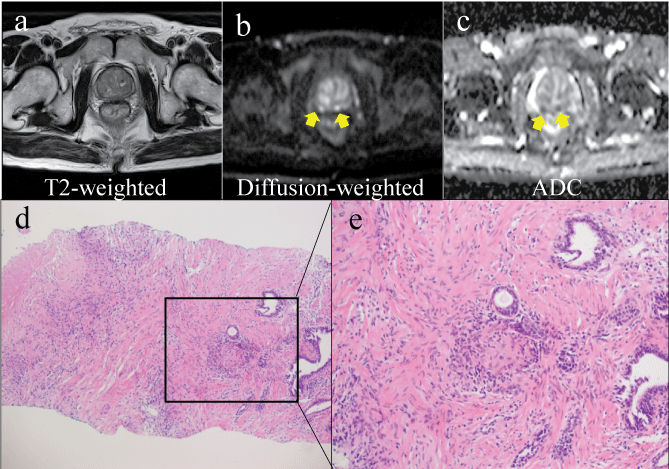
.
Figure 5: Case 2; Magnetic resonance imaging findings just before the biopsy and histopathological findings. The T2-weighted image shows a low signal (a), the diffusion-weighted image shows a high signal (b), the apparent diffusion coefficient (ADC) appears to be low in the marginal area (c). These findings are similar to those in case 1. On histopathological examination, multifocal epithelioid granulomas are observed, and no evidence of malignancy is noted (hematoxylin and eosin staining, d: �40, e: �100).
View Figure 5
MRI findings could not distinguish between granulomatous prostatitis and prostate cancer in either of these cases.
Discussion
Our data indicated the importance of PSA level measurement before and after BCG treatment for NMIBC. The relevance of PSA levels in BCG therapy was addressed by Leibovici and Beltrami [7,8], who found that the serum PSA level was at least 4.0ng/ml in 41-46% of these patients, but that the PSA values returned to within the normal range over the following 3-12 months. These findings are inconsistent with those observed in cases receiving mitomicin C and epirubicin instillation, wherein no increase in PSA level was observed. Based on these data, it may be possible to avoid an unnecessary prostate biopsy in some cases [8].
Positron-emission tomography-computed tomography (PET-CT) has recently been introduced as a diagnostic tool in many areas of medicine, including urology, and the reports on 2 cases have described its use in BCG-induced granulomatous prostatitis [9,10]; in both these cases, a prostate biopsy was performed because cancer was suspected. The MRI features of granulomatous prostatitis caused by BCG infection have recently been reported in several studies [11-13]. Although major advances have been made in diagnostic imaging, including MRI and PET-CT, this diagnostic imaging problem remains unresolved.
Furthermore, our data (including the 2 cases described above) demonstrated the difficulty of diagnosing granulomatous prostatitis after BCG intravesical instillation therapy. The urethra is surrounded by the prostate, which is located adjacent to the urinary bladder. BCG is administered through the urethra, thus increasing the risk of prostate infection. Therefore, the incidence of non-symptomatic prostate infection would be expected to be relatively high after this procedure.
A further complication is the difficulty of distinguishing between granulomatous prostatitis and prostate cancer, which was demonstrated in a report of 26 cases in Japan [2], as well as in reports from Europe and the United States [14,15]. In 1990, Mukamel et al. [15] reported that granulomatous prostatitis was found in 8 of 20 cases (40%) on prostate biopsy following abnormal digital rectal examination and/or ultrasonography after BCG treatment [15]. A high incidence of granulomatous prostatitis, including non-symptomatic cases, was also reported in a study in the 1980s (37.5%: 12/32 cases) [14]. Naik et al. [16] evaluated 10 histopathologically confirmed granulomatous prostatitis cases using transrectal ultrasound and MRI, and compared the findings to those of prostate cancer patients; no apparent difference was noted between these groups [16]. Hence, further assessments are needed for the development of a sufficient and easy PSA level monitoring protocol in the near future.
Conclusion
Our results confirm that PSA level examination is essential before and after BCG treatment. PSA level elevation in patients treated with intravesical BCG is self-limiting, and hence, prostate biopsies may not be necessary in these patients while PSA levels are being monitored. However, prostate biopsies are essential for patients with elevated PSA levels, as prostate cancer can occur metachronously.
References
-
Lamm DL, Stogdill VD, Stogdill BJ, Crispen RG (1986) Complications of bacillus Calmette-Guerin immunotherapy in 1,278 patients with bladder cancer. J Urol 135: 272-274.
-
Oshima K, Okabe H, Tamura H (2006) Results of Post-marketing Surveilance of Immunobladder? Intravesical [Freeze-dried BCG intravesical (Japan)] -Drug Use Results Survey-. Jpn J Urol Surg 19: 1409-1420.
-
LaFontaine PD, Middleman BR, Graham SD Jr, Sanders WH (1997) Incidence of granulomatous prostatitis and acid-fast bacilli after intravesical BCG therapy. Urology 49: 363-366.
-
Rifkin MD, Tessler FN, Tublin ME, Ross JS (1998) US case of the day. Granulomatous prostatitis resulting from BCG therapy. Radiographics 18: 1605-1607.
-
Joaquim A, Cust�dio S, Pimentel FL, Matos JF, Peixoto V, et al. (2012) Bacillary prostatitis after intravesical immunotherapy: a rare adverse effect. Case Rep Oncol 5: 80-83.
-
Okamura T, Akita H, Ando R, Ikegami Y, Taku Naiki T, et al. (2012) Single monthly bacillus Calmette-Gue�rin intravesical instillation is effective maintenance therapy to prevent recurrence in Japanese patients with non-muscle-invasive bladder cancer. Int J Clin Oncol 17: 477-481.
-
Leibovici D, Zisman A, Chen-Levyi Z, Cypele H, Siegel YI, et al. (2000) Elevated prostate specific antigen serum levels after intravesical instillation of bacillus Calmette-Guerin. J Urol 164: 1546-1549.
-
Beltrami P, Ruggera L, Cazzoletti L, Schiavone D, Zattoni F (2008) Are prostate biopsies mandatory in patients with prostate-specific antigen increase during intravesical immuno- or chemotherapy for superficial bladder cancer? Prostate 68: 1241-1247.
-
Wilkinson C, Chowdhury F, Scarsbrook A, Smith J (2012) BCG-induced granulomatous prostatitis-an incidental finding on FDG PET-CT. Clin Imaging 36: 413-415.
-
Bour L, Schull A, Delongchamps NB, Beuvon F, Muradyan N, et al. (2013) Multiparametric MRI features of granulomatous prostatitis and tubercular prostate abscess. Diagn Interv Imaging 94: 84-90.
-
Suzuki T, Takeuchi M, Naiki T, Kawai N, Kohri K, et al. (2013) MRI findings of granulomatous prostatitis developing after intravesical Bacillus Calmette-Gu�rin therapy. Clin Radiol 68: 595-599.
-
Dianat SS, Matoso A, Carter BH, Macura KJ (2014) Multiparametric MRI findings of granulomatous prostatitis after intravesical bacillus Calmette-Gu�rin therapy in a patient undergoing active surveillance. Clin Genitourin Cancer 12: 215-219.
-
Logan JK, Walton-Diaz A, Rais-Bahrami S, Merino MJ, Turkbey B, et al. (2014) Changes observed in multiparametric prostate magnetic resonance imaging characteristics correlate with histopathological development of chronic granulomatous prostatitis after intravesical Bacillus Calmette-Guerin therapy. J Comput Assist Tomogr 38: 274-276.
-
Oates RD, Stilmant MM, Freedlund MC, Siroky MB (1988) Granulomatous prostatitis following bacillus Calmette-Guerin immunotherapy of bladder cancer. J Urol 140: 751-754.
-
Mukamel E, Konichezky M, Engelstein D, Cytron S, Abramovici A, et al. (1990) Clinical and pathological findings in prostates following intravesical bacillus Calmette-Guerin instillations. J Urol 144: 1399-1400.
-
Naik KS, Carey BM (1999) The transrectal ultrasound and MRI appearances of granulomatous prostatitis and its differentiation from carcinoma. Clin Radiol 54: 173-175.





