International Archives of Nursing and Health Care
Novelties in the Treatment of Diabetic Foot: Hyperbaric Oxygen and Rich Platelet Plasma Therapy
Ana Maria Arnaiz-Garcia*
University Hospital Marques de Valdecilla, Spain
*Corresponding author: Dr. Ana Maria Arnaiz-Garcia, Specialist Consultant in Internal Medicine, Infectious Diseases Unit, University Hospital Marques de Valdecilla, Spain, Tel: +34 659708320, E-mail: anarnaiz@humv.es
Int Arch Nurs Health Care, IANHC-1-019, (Volume 1, Issue 1), Case Report; ISSN: 2469-5823
Received: September 27, 2015 | Accepted: November 25, 2015 | Published: November 30, 2015
Citation: Arnaiz-Garcia AM (2015) Novelties in the Treatment of Diabetic Foot: Hyperbaric Oxygen and Rich Platelet Plasma Therapy. Int Arch Nurs Health Care 1:019. 10.23937/2469-5823/1510019
Copyright: © 2015 Arnaiz-Garcia AM. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and
reproduction in any medium, provided the original author and source are credited.
Hyperbaric Oxigen Treatment
Introduction
Hyperbaric oxygen therapy (HBOT) is useful as an adjunct or primary therapy of multiple processes, such as gas embolism, carbon monoxide poisoning, decompression treatments, myonecrosis, ischemic traumatic injuries, compartment syndrome, severe anemia, brain abscesses dued to Actinomyces spp., necrotizing infections, refractory osteomyelitis, radiation necrosis, burns and it is also useful in situations in which the evolution of a graft or skin flap is unfavorable despite other treatments [1].
Mechanisms of action
Most of the benefits of HBOT are explained by the relationship established between the oxygen concentration, volume and pressure that is administered. HBOT is used primarily in situations of tissue hypoxia, treatment of gas embolism and decompression.
Increasing the flow of oxygen to the tissues:According to Henry's Law, the amount of an ideal gas dissolved in a solution is directly proportional to its partial pressure. Therefore, if the oxygen plasma concentration at sea level (1 atmosphere pressure) is 0.3 ml/dl, after the administration of oxygen at a pressure of 100%, it is raised to 1.5 ml/dl. With HBOT (3 atmospheres pressure), partial blood oxygen concentration becomes to 6 ml/dl [1,2].
The ability of HBOT to increase the content of oxygen and meet the needs of oxygen in tissues, has made it to be one of the mainstays of treatment of all situations in which the flow of oxygen to the cells and tissues is reduced (anemia, carbon monoxide poisoning and acute and chronic ischemia).
Wound healing:In vitro, HBOT modulates the local and systemic effects found in acute and chronic situations of ischemia and inflammation. Many of these effects are mediated by reactive oxygen species (free radicals of oxygen, hydrogen peroxide, hypochlorous acid) and reactive nitrogen species (nitric oxide) that are generated by the tissues. Hyperoxia induces local vasoconstriction and reduced vasogenic edema secondary to acute trauma, facilitates the proliferation of fibroblasts, angiogenesis and thus tissue healing processes. HBOT increases the bactericidal activity of neutrophils, limits the production of exotoxins and spores of Clostridium spp., has anaerobicidal effect on Clostridium perfringens and inhibits the growth of many other bacteria [1-3].
Reducing the size of the nitrogen gas bubble:Based on Boyle's Law, HBOT can be used as treatment of decompression, whereby a constant temperature, the volume of a fixed mass of gas is inversely proportional to the pressure it exerts. So, at 3 atm, a nitrogen gas bubble reduces its volume nearly 75%. Furthermore, in the case of HBOT, the reduction of the bubble size is accompanied by the replacement of those nitrogen bubbles for oxygen, which is rapidly metabolized by tissues [3].
Antagonism with carbon monoxide (CO):CO binds to hemoglobin with a avidity that is up to 250 times higher than oxygen. Therefore, the presence of carboxyhemoglobin, reduces the transport and release of oxygen to the tissues. The half-life of carboxyhemoglobin is 40-80 minutes at 1 atmosphere of pressure, but is reduced to 15-30 minutes after the administration of HBOT [3].
Technique
Generally, the spacious chambers in which HBOT is provided, allow treatment of various patients in each session and allow health workers to come inside to carefully monitor critically ill patients. HBOT is a kind of treatment in which the patient breathes 100% oxygen at a pressure > 1 atm. The pressures that are used range between 2 and 3 atmospheres [1-3].
A complete HBOT is usually administered in 15-30 sessions, each of them lasting between 60 and 120 minutes (depending on the indication for which the treatment is given).
The treatment of acute situations (carbon monoxide poisoning, decompressive treatments, etc.) typically needs only 1-2 sessions, while treatment of chronic diseases (diabetic foot, myonecrosis, etc.) requires longer treatment (15-30 sessions). When oxygen pressures exceeds 2.8-3 atm, and especially after prolonged periods of exposure to HBOT (Figure 1), the risk of neurological damage and pulmonary toxicity is increased [1-3].
Contraindications and complications
The only absolute contraindication to HBOT is pneumothorax. Relative contraindications include chronic obstructive (COPD), pulmonary disease, presence of lung bullae, respiratory tract infections, recent ear surgery or lung area, uncontrolled fever and claustrophobia. However, all these relative contraindications should never preclude treatment with HBOT of patients in serious situations [3,4].
As regards, patients diagnosed with epilepsy have an increased risk of neurological complications dued to the toxicity of central nervous system by oxygen overpressure. However, the incidence of complications of the central nervous system in the population without this antecedent, is very low, so the potential risk of developing this neurological toxicity should not deter the doctor from the use of HBOT in circumstances where it is needed [2,4].
It has been theorized possibility that HBOT could exacerbate the adverse effects of some drugs (bleomycin and doxorubicin). However, this hypothesis has no scientific basis. Given the lack of clinical studies, the decision to treat with HBOT to patients who have been treated with these drugs (bleomycin and/or doxorubicin) should be individualized [4].
HBOT is a safe and well tolerated therapy in most cases. Generally, side effects are usually mild and reversible, but serious complications can occur in very rare situations [1-5].
Reversible myopia:Because the direct exposure of the eye to oxygen. It is the most common side effect, occurring in up to 20% of patients. Completely reversed within weeks or months of treatment.
Otic barotrauma:It happens in 3-20% of patients, being more frequent in patients receiving multiple treatments.
Otorrhagia dued to ruptured eardrum and damage of the middle ear:It happens in < 1% of patients, occurring mainly in patients receiving several rounds of sessions TOH.
Pulmonary barotrauma:Very rare complication, dued to the improving of current diagnostic techniques for the identification and treatment of pneumothorax before the start of TOH.
Pulmonary toxicity dued to oxygen:It usually manifests by increased work of breathing, shortness of breath, coughing and a reversible decrease in lung function. It usually occurs in patients receiving multiple treatments or who have been exposed to high oxygen pressures previously.
Seizures:The incidence of seizures in patients receiving HBOT is estimated at 2.4 cases/100,000 treatments. The risk is increased if the sessions of HBOT exceed 90-120 minutes and 2.8 to 3 atmospheres of pressure. The risk is increased in patients who are also receiving treatments with corticosteroids, insulin and levothyroxine. Since HBOT has been associated with an increased risk of hypoglycemia in diabetic patients, this possibility should always be considered in any patient in whom a compatible neurological symptoms (seizures) appears after HBOT. Seizures resulting from oxygen toxicity, rarely cause permanent structural damage to the brain.
Seizures must be treated promptly by reducing the inspired oxygen concentration, that is breathing air in the room with a FiO2 = 0.21, and administering anticonvulsant therapy.
The neurotoxicty dued to oxygen can be prevented interspersing periods of 5 minutes in which ambient air is administered. Thus, the formation of free oxygen radicals is reduced and the potential neurotoxicty derived of HBOT too.
Gas embolism and decompression gaseous:Usually divers and pilots employed in patients using compressed air.
Clinical Utility
In this section we will focus on its applicability in situations of tissue ischemia and infection, often found together in the diabetic foot [3-5].
Diabetic foot:Diabetes mellitus has become a global epidemic. Because the prevalence of diabetes has increased, more and more therapeutic approaches to its complications are needed. These complications are often very difficult to treat and includes from the development of a simple foot ulcer, to the development of infection and limb amputation in extreme cases.
Each year, 1.9% of people with diabetes develop foot ulcers. These wounds are often refractory to conventional treatments, requiring limb amputation in 15-20% of diabetic patients in the first 5 years after the development of the ulcer [3-5].
In diabetic patients, peripheral neuropathy seems to be the most important risk factor for developing foot ulcers, being the main cause in 80% of cases. Peripheral neuropathy causes the loss of sweat glands, increasing the dryness of the skin, and therefore the probability of small wounds, that could be infected, causing further tissue damage. The changes in the biomechanic of foot is also a negative effects of peripheral neuropathy, and also another important factor in the development of wounds and chronic ulcers that result from disruption of normal pressure points on the foot, with inappropriate distribution of the weight, exerting high pressure in these areas that are not physiologically prepared for that purpose. The treatments of ulcers of diabetic foot usually are: debridement of nonviable tissue, relieving pressure on the ulcerated area (splints, proper footwear), wound care, optimizing glycemic control and assess the possible need for antibiotic treatment and limb revascularization [1-3].
Diabetic patients are at high risk of developing ischemic ulcers. Hypoxia happens in diabetic foot ulcer, directly caused by ischemia due to the breakdown of the local microcirculation. The inflammatory response increases the demand of oxygen in the wound bed. Fibrin stimulates the release of cytokines which, in turn, recruit macrophages and fibroblasts in the affected area. Leukocytes also migrate into the damaged tissue, increasing its activity and therefore, the oxygen demand. This situation of initial hypoxia, however persists in chronic ulcers. In normal conditions, the partial pressure of oxygen in the wound usually is < 3 mmHg, however the pressure in the periphery of the wound is generally close to 20 mmHg. This oxygen gradient, stimulates the wound healing. However, wounds that are surrounded by hypoxic tissue, have less oxygen gradient from the periphery to the center, being harder healing. Therefore, dued to the healing of diabetic foot wounds depends on oxygen from the peripheria, HBOT has its place in the treatment of ischaemic wound. Peripherial hyperoxygenation of the area due to HBOT reduces edema (vasoconstrictor effect due to oxygen occurs), increases bactericidal capacity of leukocytes and macrophages, has anaerobicidal effect, increases the gradient of oxygen from the periphery to the wound bed and stimulates angiogenesis [1-5].
Acute and chronic ischemia:TOH has been recommended as part of the general treatment of ischemic pathology, including compartment syndrome and situations where there is vascular compromise. Animal models of ischemia and compartment syndrome suggest that the benefit of TOH in these situations may be due to a combination of increased tissue oxygenation, reducing edema secondary to vasospasm hyperoxia, tissue protection from ischemia and reperfusion injury secondary and antibacterial effects.
Infection:The main treatment of severe infections of the skin and soft tissues (myonecrosis, necrotizing fasciitis and Fournier gangrene) is the antibiotherapy and, in acases of severe infections, also the surgical debridement of the wound. The use of HBOT reduces mortality and limit the area to debridement in Fournier's gangrene and necritizante fasciitis. A retrospective study using the national database of Singapore, identified 45913 cases of necrotizing infections of skin and soft tissue, of which 405 received HBOT. It was found that the mortality in the group of patients who had used HBOT was 4.5%, compared with mortality in the group of patients whom not received HBOT that was 9.4%, with a statistically significant difference. Although it is also common to use HBOT in other infectious diseases such as refractory osteomyelitis, brain abscess zygomycosis and Actinomyces spp, there are insufficient data in the literature to support its use, so more studies are needed to confirm the benefits of HBOT in these situations and justify its cost and potential risks in this group of patients.
Clinical cases
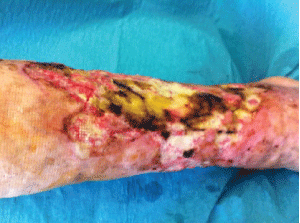
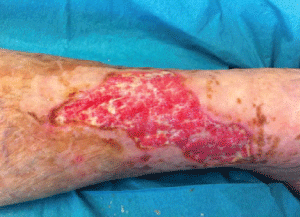
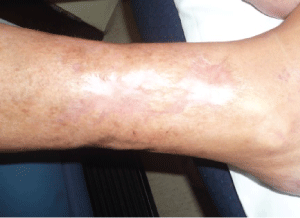
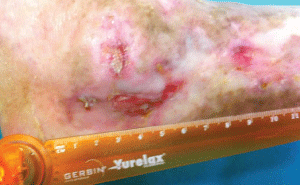
Case 1:Woman, 72 year old, with insullin-dependant diabetes mellitus type 2 and chronic bad glycemic control. She suffered traumatic injury in her right leg with development of a superficial necrosis and cellulitis. She had data of microangiopathy in her legs, not being possible treatment of revascularization, so antibiotic, wound debridement and early treatment with HBOT (receiving a total of 15 sessions) were prescribed. The wound healing was achieved within 1.5 months.
1. - Initial wound: Surgical debridement, antibiotics and starting HBOT were indicated (Figure 2).
2. - Wound after 15 sessions of HBOT (Figure 3).
3. - Healing wound (1.5 months) (Figure 4).
Rich Platelet Plasma Therapy
Introduction
Since 2000, they have appeared in the literature articles that reflect the experience of the application of Rich platelet plasma therapy (RPPT) especially in the field of maxillofacial surgery, ophthalmology (corneal ulcers), orthopedic surgery (osteosynthesis and joint replacement) and cosmetic surgery (skin rejuvenation techniques) [6-9]. However, it is in these last two years, when it has begun to implement as adjunctive therapy in the field of diabetic foot, focusing especially on chronic ulcers.
Function of platelets in the wound
The primary role of platelets is the regeneration of injured tissue. Once the activation of the platelet occurs, the degranulation occurs, releasing alpha granules which contain multiple tissue growth factors. Growth factors act attracting undifferentiated cells to the site of injury (chemoattraction and cell migration) and stimulating the processes of cellular mitosis (proliferation) [6,7].
Any wound has a number of physiological processes to promote tissue healing. For this reason, processes are triggered:
Coagulation/sealing of the wound:(formation of a hemostatic barrier): Fibrinogen, the fibrin strands and platelet activation.
Inflammation:(leukocyte to eliminate the infection): Through signal proteins of platelet and white blood cells.
Tissue regeneration:(Cellular attraction and differentiation in the wound bed): Platelet growth factors, endothelial growth factors and tissue adhesion molecules.
Growth factors in PRPT
There is a lot of growth factors in the RPPT, most still unidentified [6-8]. The main growth factors in the RPPT are:
PDGF (Growth factor platelet derived):Promotes the cellular chemoattraction of mesenchymal and endothelial cells to the site of injury. It promotes differentiation of fibroblasts and osteoblasts and regulates the effects of other growth factors on cells.
TGF-β (Transforming growth factor beta):Promotes cellular mitosis and differentiation of connective tissue.
IGF (Growth factor insulin):Stimulates the bone formation from existing osteoblasts.
VEGF (Vascular endothelial growth factor):Stimulates the angiogenesis and chemoatracttion of osteoblasts.
EGF (Epithelial growth factor):Induces the epithelial development and promotes angiogenesis.
Properties of growth factors
The properties of the growth factors present in RPPT are [6-9]:
• Induction of cell mitosis: Increasing the number of cells involved in processes of tissue reparation.
• Angiogenesis: Generating new capillaries in the injured area.
• Release of growth factors from other cells that promote synthesis of fibroblasts and osteoblasts.
Platelet rich plasma (RPPT) is an autologous concentrated, non-toxic, non-allergenic therapy that contains a high concentration of growth factors and cytokines that have been released by the process of degranulation of platelets. These substances stimulate the bone and soft tissue regeneration [7-9].
It shortens the physiological processes of wound healing by stimulating revascularization and stimulating the synthesis and differentiation of precursor cells [7,8].
Preparation of RPPT
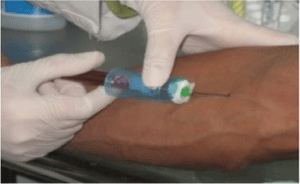
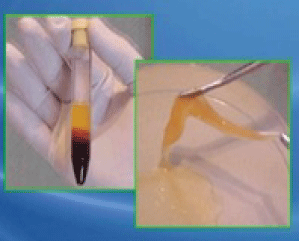
The first step is the extraction of venous blood of the patient (range 5-30 ml) (Figure 5).
Second step is to centrifuge the blood sample for selecting the RPPT and discard cells (red blood cells and white blood cells) that they will not have place in the healing process (Figure 6).
Third step is the aspiration of the plasma and to place it in a container. After that, we will activate it with calcium chloride, and administering topically to the wound (Figure 7 and Figure 8).
Clinical cases
Case 1: Man, 86 year old, diabetic and with non-revascularizable arterial disease in both lower limbs. He has a chronic ulcer in his right leg of 20 years of evolution. Treatment with PPRT was started, with a total of 4 sessions separated by an interval of three weeks administrations. The wound healed in two months (Figure 9 and Figure 10).
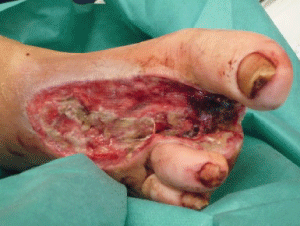
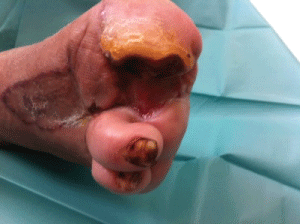
Case 2: Man, 56 year old, diabetic with very poor metabolic control. Signs of necrosis of second and third fingers of his right foot, needing amputation of fingers and extensive debridement of the wound. Topical treatment with RPPT was started (3 sessions spaced 3 weeks). Wound was totally healed in the second month, not needed to perform any graft or procedure to cover the wound (Figure 11 and Figure 12).
References
-
Soh CR, Pietrobon R, Freiberger JJ, Chew ST, Rajgor D, et al. (2012) Hyperbaric oxygen therapy in necrotising soft tissue infections: a study of patients in the United States Nationwide Inpatient Sample. Intensive Care Med 38: 1143-1151.
-
Wilkinson D, Doolette D (2004) Hyperbaric oxygen treatment and survival from necrotizing soft tissue infection. Arch Surg 139: 1339-1345.
-
Abidia A, Laden G, Kuhan G, Johnson BF, Wilkinson AR, et al. (2003) The role of hyperbaric oxygen therapy in ischaemic diabetic lower extremity ulcers: a double-blind randomised-controlled trial. Eur J Vasc Endovasc Surg 25: 513-518.
-
Tibbles PM, Edelsberg JS (1996) Hyperbaric-oxygen therapy. N Engl J Med 334: 1642-1648.
-
Riseman JA, Zamboni WA, Curtis A, Graham DR, Konrad HR, et al. (1990) Hyperbaric oxygen therapy for necrotizing fasciitis reduces mortality and the need for debridements. Surgery 108: 847-850.
-
Slesaczeck T, Paetzold H, Nanning T, Reichel A, Barthel A, et al. (2012) Autologous-derived, platelet rich plasma gel in the treatment of non-healing diabetic foot ulcer: A case report. Ther Adv Endocrinol Metab 3: 75-78.
-
Scimeca CL, Bharara M, Fisher TK, Kimbriel H, Armstrong DG (2010) Novel use of platelet-rich plasma to augment curative diabetic foot surgery. J Diabetes Sci Technol 4: 1121-1126.
-
Villela DL, Santos VL (2010) Evidence on the use of platelet-rich plasma for diabetic ulcer: a systematic review. Growth Factors 28: 111-116.
-
Lacci KM, Dardik A (2010) Platelet-rich plasma: support for its use in wound healing. Yale J Biol Med 83: 1-9.