International Archives of Addiction Research and Medicine
Pneumomediastinum and Pneumothorax: A Rare Presentation of Synthetic Cannabinoids Toxicity
Marvin Louis Roy Lu*, Sana Mazher, Andres Mora Carpio and Anita Govil
Department of Medicine, Einstein Medical Center, Philadelphia, USA
*Corresponding author:
Marvin Louis Roy Lu, Department of Medicine, Albert Einstein Medical Center, 5501 Old York Road, Philadelphia PA, USA, Tel: 267-648-8126, Email: lumarvin@einstein.edu
Int Arch Addict Res Med, IAARM-2-017, (Volume 2, Issue 1), Case Report; ISSN: 2474-3631
Received: February 01, 2016 | Accepted: April 04, 2016 | Published: April 07, 2016
Citation: Lu MLR, Mazher S, Carpio AM, Govil A (2016) Pneumomediastinum and Pneumothorax: A Rare Presentation of Synthetic Cannabinoids Toxicity. Int Arch Addict Res Med 2:017. 10.23937/2474-3631/1510017
Copyright: © 2016 Lu MLR, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Keywords
Synthetic cannabinoids, Pneumomediastinum, Pneumothorax
Introduction
Synthetic cannabinoids (SCs) are chemically produced analogs of natural cannabinoids and are blended with herbs to appear as a natural product. They are typically marketed as "Cloud 9", "k2", "spice", "chill X", and "crazy monkey" among many others depending on their herbal blend [1]. They are an emerging recreational drug with significant adverse effects in the entire western hemisphere including Europe and the United States use and abuse have been steadily increasing since its emergence in the country in 2008, particularly in the younger population [2]. Clinical effects are similar to marijuana but with more severe psychomotor agitation and paranoia. Pneumothorax and pneumomediastinum are rare but potentially life-threatening and should be ruled-out in these patients who present with chest pain.
Case
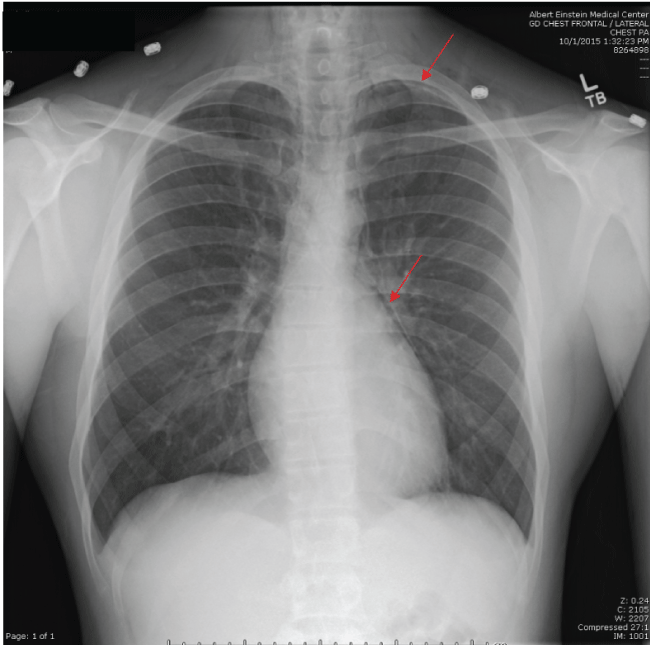
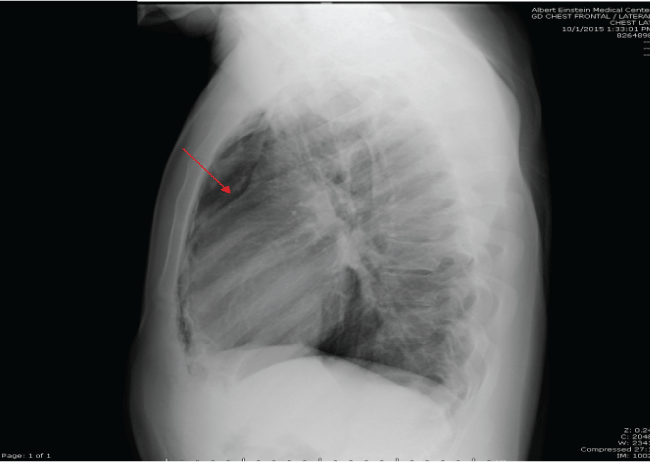
A 23 year-old male with no significant past medical history presented to the emergency department with generalized body pain. He had been smoking 1-2 blunts of "K2" everyday for a year but has recently been smoking twice more after having a fight with his girlfriend. He is otherwise healthy, does not smoke cigarettes and takes no medications. He had bilateral upper and lower extremity pain described as "muscle cramps" and sharp pleuritic chest pain associated with nausea and vomiting. On physical exam, he is a well-looking male with lean body mass and appropriate arm span. His BMI was 23. He was tachycardic to 110 beats/minute but all other vital signs were normal. On exam, he was in no acute distress but was very agitated. Crepitus was noted on neck palpation. He had substernal chest tenderness, clear lungs and normal cardiovascular exam. He also had diffuse muscle tenderness on all extremities. Neurologic exam was normal. Laboratory exam showed a creatine phosphokinase of 18,944 IU/L, creatinine 3.8 mg/dl, BUN of 55 mg/dl and a troponin of < 0.01 ng/ml. Urine drug screen was negative for tetrahydrocannabinol, opiates, cocaine, phencyclidine, benzodiazepines, barbiturates, and amphetamines. Electrocardiogram (ECG) showed sinus tachycardia at 115 beats/minute with no ST or T-wave changes. Chest x-ray (CXR) revealed a small left apical pneumothorax, pneumomediastinum and subcutaneous emphysema in the neck bilaterally (Figure 1 and Figure 2, red arrow). He was given aggressive intravenous fluid hydration, and given oxygen through nasal cannula. His urine output and electrolytes were monitored closely. His symptoms eventually improved on day three of admission and his serum creatinine and BUN normalized. Serial CXR revealed that the pneumomediastinum and pneumothorax was stable. He was seen by cardiothoracic surgery who agreed with our plan to just observe him closely. He was referred to psychiatry and has agreed to undergo rehabilitation and detoxification. At that time, he was less agitated and the chest pain was improving. He was seen later at the primary care clinic and has been drug-free for 1 month and feeling well.
.
Figure 2: CXR (Lateral View) showing left sided pneumothorax and pneumomediastinum.
View Figure 2
Discussion
Synthetic cannabinoids have the same clinical effects as regular cannabis which includes conjunctival erythema, palpitations and tachycardia, increased appetite (munchies), slurred speech and gait imbalance. However, they have a significantly greater potential for psychosis, hallucinations and psychomotor agitation [3-6]. The exact mechanism behind SC's greater potential for neuropsychiatric toxicity and life-threatening toxicity from severe agitation and seizures remain unknown but is thought to be secondary to their increased potency [7]. SCs act on similar receptors (cannabinoid 1 and 2, serotonin and N-methyl-D-aspartate receptors) but are 2-800 times more potent than delta-9 tetrahydrocannabinol [8]. SCs act as full agonists in Cannabinoid receptor 1 and 2 (CB1R and CB2R) in both cellular assays and animal studies [7]. Moreover, the herbs and other products which serve as its carrier may also contain other contaminants such as O-desmethyltramadol that can contribute to its harmful effect [8].
Air leaks such as pneumomediastinum and pneumothorax are rare and may be secondary to inhalation and breathholding during SC use. Breathholding and its relation to these air-leak syndromes have been described in other inhalational drug abuse such as tobacco, marijuana, and cocaine [1]. Literature regarding these air leak syndromes remain scarce and have only been described as a potential adverse effect of synthetic cannabinoids. No population based studies have been made to measure its prevalence. Patients typically present with chest pain associated with use of the drug. Management varies depending on the size of the air leak and vital signs. Small pneumothorax and pneumomediastinum is managed conservatively with supportive oxygen but these patients should be monitored for hemodynamic compromise. Large pneumothorax with the pleural line extending more than 2 cm. from the chest wall and significant respiratory distress should be managed with prompt evacuation with chest tube placement [8].
Other illicit drugs known to cause pneumomediastinum and pneumothorax are tobacco, amphetamines, cocaine, ecstasy and heroin [1].
Using the search term "synthetic cannabinoids", "pneumomediastinum" and "pneumothorax", we performed searches for MEDLINE and PUBMED databases to look for case reports or series associating synthetic cannabinoids with these complications. As far as the authors are concerned, this may be the first reported case of pneumomediastinum and pneumothorax from synthetic cannabinoid use in the young. Physicians should be aware of this rare but potentially life-threatening condition and should rule it out in all patients who present with chest pain who have a history of long-standing synthetic cannabinoid use.
References
-
Camus P, Rosenow EC (2010) Drug-induced and Iatrogenic Respiratory Disease. In: Taylor and Francis Group, 294-295.
-
European Monitoring Centre for Drugs and Drug Addiction (2009) Understanding the ‘Spice' phenomenon. Luxembourg: 37.
-
Hermanns-Clausen M, Kneisel S, Szabo B, Auwärter V (2013) Acute toxicity due to the confirmed consumption of synthetic cannabinoids: clinical and laboratory findings. Addiction 108: 534-544.
-
Harris CR, Brown A (2013) Synthetic cannabinoid intoxication: a case series and review. J Emerg Med 44: 360-366.
-
Cohen J, Morrison S, Greenberg J, Saidinejad M (2012) Clinical Presentation of intoxication due to synthetic cannabinoids. Pediatrics 129: e 1064-1067.
-
Winstock AR, Barratt MJ (2013) The 12-month prevalence and nature of adverse experiences resulting in emergency medical presentations associated with the use of synthetic cannabinoid products. Hum Psychopharmacol 28: 390-393.
-
Musselman ME, Hampton JP (2014) "Not for human consumption": a review of emerging designer drugs. Pharmacotherapy 34: 745-757.
-
Fantegrossi WE, Moran JH, Radominska-Pandya A, Prather PL (2014) Distinct pharmacology and metabolism of K2 synthetic cannabinoids compared to Δ(9)-THC: mechanism underlying greater toxicity? Life Sci 97: 45-54.