SARS-CoV-2 causes placental changes that can lead to embryonic/foetal death. However, the pregnant woman who in other diseases is considered (e.g., H1N1) a risk group for the development of severe forms of the disease, in COVID-19 rarely evolves with severity, except when childbirth occurs, and the woman loses the bond with the placenta. The answer for the pregnant woman is in the immunosuppression promoted by the placenta, in the metabolism of tryptophan and phenylalanine (Try) and functions of furins and cathepsins. This article discusses at crucial points the main pathophysiological mechanisms that can explain the events observed during pregnancy and the peri-and postpartum phenomena. It also shows the vital role of the placenta in this period of gestation, which is one of the most significant evolutionary adaptations that humans have in the face of SARS-CoV-2 infection. The studies for the elaboration of this article could also bring to light theories about the formation of the syncytial lung in COVID-19, about the physiology of the Faget's Sign and justify new therapeutic approaches that should be carried out seeking a better outcome for patients in this case aimed at pregnant and postpartum women. This article names the main clinical manifestations of COVID-19 as the thrombometabolic and immune tolerant syndrome induced by SARS-CoV-2 infection (TMITSy-CoV-2). Finally, this article aims to bring to light a host-parasite interaction that, if well understood, can help in the management of the pregnant woman and the first postpartum moments, because even without specific medications, we can perform better clinical management, resulting in a good outcome for the pregnant woman and the foetus. Reading the Appendix is essential as it complements the general pathophysiology of COVID-19.
Coronaviruses (CoVs) are a family of positive-stranded RNA viruses with envelopes widely distributed among mammals and birds that cause respiratory and intestinal infections in animals and humans. Brazil reported a disproportionately large number of maternal deaths attributed to COVID-19, particularly during the postpartum period. There were 160 notified deaths associated with COVID-19 among pregnant and 106 deaths among postpartum hospitalized women considering data from the Brazilian Ministry of Health's Influenza Epidemiological Surveillance Information System. The high maternal death rates in Brazil are accompanied by large racial, geographic, and socioeconomic disparities. The COVID-19 mortality rate among pregnant and postpartum women is 7.2% in Brazil (data published in 2021/June). This percentage is 2.5 times higher than the national rate of 2.8%. According to the Covid-19 Observatory of the Oswaldo Cruz Foundation (Fiocruz) [1].
Pregnant women are not part of the risk group for hospital admission, but they present risks due to the pregnant woman's status and because there are cases of placental changes that may lead to the embryo or foetus death. Maternal mortality rate by COVID-19 increased and Brazil is responsible for an important portion of cases. This rate may be even higher due to underreporting, and it is necessary to consider "The Long COVID-19" as the greatest causes of mortality that is underreported. A recent study published COVID-19 is diagnosed in the third trimester. Thus, women with COVID-19 diagnosis or whose pregnancy ended earlier in pregnancy are underrepresented either because our study was exclusively hospital based or earlier infection may manifest with mild symptoms, which are either ignored or managed in primary care [2] (Tables 2, Table 3, Table 4 and Table 5 in the Appendix).
Table 1: The peptidases involved in the infection and replication of CoVs. View Table 1
This article based on praxis aims to bring Hypotheses that allows physicians to have more possibilities to perform better management of the pregnant women facing a reality that show us no active antiviral to avoid COVID-19. Understanding the pathophysiology of COVID-19 is the way that we have to improve medical conduct and offer more safety to pregnant women and foetus.
Performing a search for articles in PubMed and SciELO databases - Scientific Electronic Library Online, Articles published in the last 20 years were considered valid. The search for articles was based on the following descriptors: (mj :( "Coronavirus Infections" OR "Pregnant Women") AND mha ("Infectious Complications in Pregnancy/MO" OR "Infectious Complications in Pregnancy/PA" OR "Infectious Complications in Pregnancy/PP")), considering all types of articles published on COVID-19 and pregnancy, as only 71 articles were found to be considered to perform the idea inside this manuscript. Only articles published between 2001 and 2021 and in English were considered, totalling 67 articles. Official sites of the Brazilian government and sites of universities were accepted to complement information and research. More information about mechanisms that may happen during the gestational period were complemented by search on Google Scholar. Articles in Russian were also included, mainly because they addressed the shock in COVID-19, as many articles present interesting and unique views that help in understanding the pathophysiological mechanisms. Overall, considering all the subjects covered, 202 articles were selected based on the quality of publication and relevance to the subject, but 101 are in the main text. Many other articles were used to support the pathophysiology of SARS-CoV-2 and host hospitalization. The figures used from sources other than those drawn by the author himself had their origins duly identified. Figures modified or constructed by the author were performed using the platform www.biorrender.com (Created with BioRender).
amh: document subject descriptors mh: The search result was N records. N = number of found records. mj: primary descriptors of the document - example: mj: The search result was N records.The maternal organism only tolerates the foetus because the placenta develops an immune tolerance mechanism in the pregnant woman's body, so the embryo is not recognized as not proper. About four days after fertilization, the morula reaches the uterus and fluid from the uterine cavity passes through the pellucid zone to form - the blastocystic cavity. Blastomeres are separated into two parts with increasing fluid, and two structures arise due to this separation 1) Trophoblast: External cell layer that will form the embryonic part of the placenta and 2) Embryoblast: A group of centrally located blastomeres that will give rise to the embryo. After six days of fertilization, the embryo base is adhered to the uterine wall and physiologically by the embryoblast structure by the action of proteolytic metalloproteinases of the endometrium. On the opposite side, the trophoblast starts differentiation to 1) Cytotrophoblast: Inner cell layer and 2) Syncytiotrophoblast: Outer cell layer [3-5]. The Syncytiotrophoblast is highly invasive and responsible for producing the hormone hCG that maintains hormonal activity in the corpus luteum during pregnancy and forms the basis for pregnancy tests. More about placental development, immune response and vasculogenesis, about changes in placental physiology in the face of gestational or Sars-CoV-2 induced diabetes are more detailed in the Appendix. Figures numbered 1 through 22 remain in the main text as they are self-explanatory, but more details are presented in the Appendix in supplementary material [6-8] (Figure 1, Figure 2, Figure 3 and Figure 4).
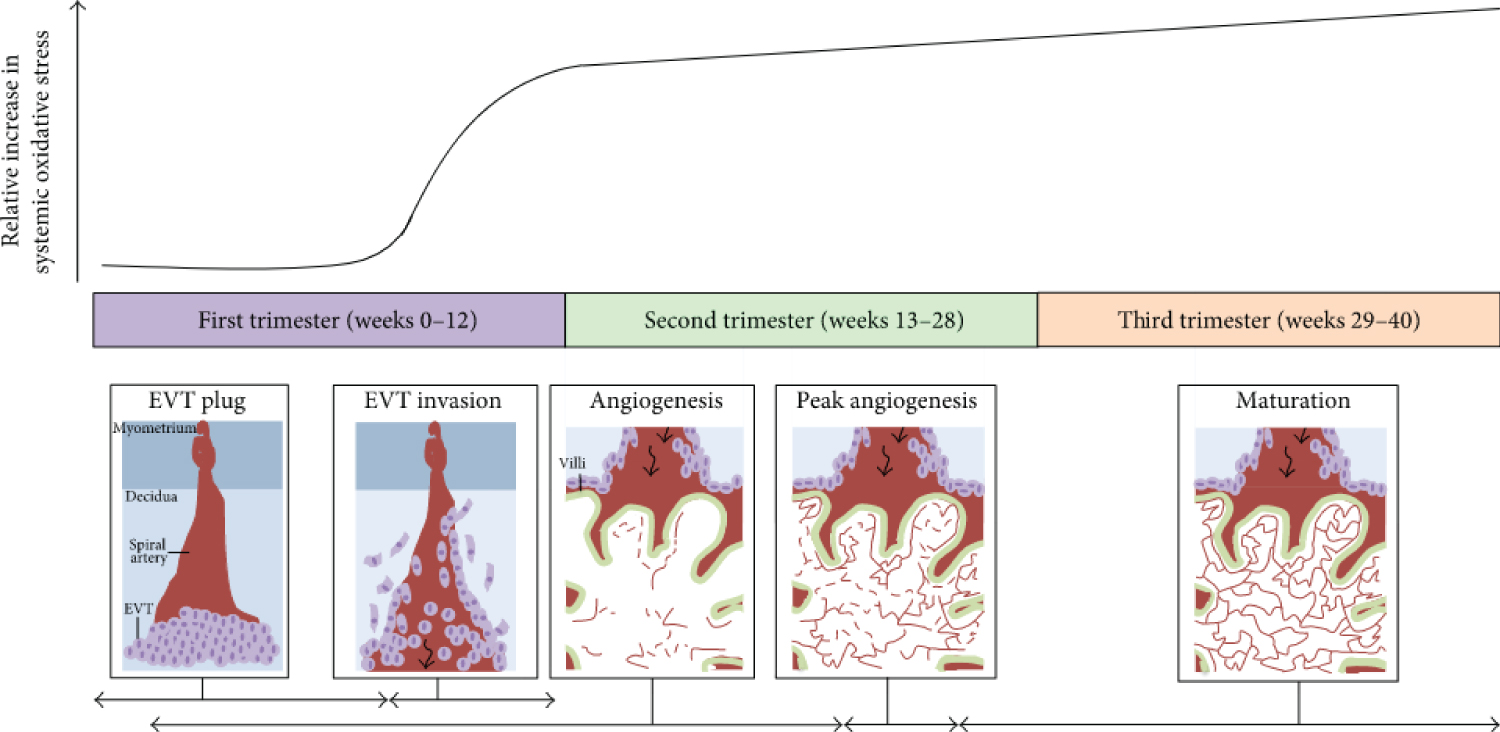 Figure 1: Oxidative stress throughout human pregnancy and its relation to placental angiogenesis. At the beginning of the first trimester of pregnancy, there are low levels of systemic oxidative stress and no blood flow into the placenta because extra villous trophoblasts (EVTs) (depicted as light purple circles) plug the maternal spiral arteries (depicted in red) in the decidua as shown in the first panel. Between 8 and 12 weeks of gestation, the EVT plug dissipates and the EVTs invade maternal spiral arteries to allow blood to enter the placenta (black arrow), as illustrated in the second panel. This coincides with a sharp increase in maternal oxidative stress. Furthermore, the state of oxidative stress increases with gestational age as depicted by the black curve. The first signs of placental angiogenesis occur at 3 weeks of gestation. However, from about 12 weeks onwards, blood vessels (red lines) protrude towards the trophoblastic layers of the villi (outlined in green), where blood exchange between maternal and foetal circulation is optimal (shown in panel three). From about 9-23 weeks of gestation, there is an expansion of the foetal capillary bed by branching and nonbranching angiogenesis (dashed red lines in angiogenesis panel). From 23-24 weeks of gestation, the greatest changes in blood vessel development and villous composition are observed (peak angiogenesis panel). Angiogenesis continues until term with the maturation of blood vessels and development of a more complex vascular network to facilitate exponential foetal growth. The horizontal black arrows indicate the approximate time each process depicted in the panels occurs. Note: this graph has been constructed by interpretations of multiple studies reporting findings of systemic oxidative stress markers present in women during normal pregnancy as studies on placental/uterine oxidative stress are limited. Source: [101].
View Figure 1
Figure 1: Oxidative stress throughout human pregnancy and its relation to placental angiogenesis. At the beginning of the first trimester of pregnancy, there are low levels of systemic oxidative stress and no blood flow into the placenta because extra villous trophoblasts (EVTs) (depicted as light purple circles) plug the maternal spiral arteries (depicted in red) in the decidua as shown in the first panel. Between 8 and 12 weeks of gestation, the EVT plug dissipates and the EVTs invade maternal spiral arteries to allow blood to enter the placenta (black arrow), as illustrated in the second panel. This coincides with a sharp increase in maternal oxidative stress. Furthermore, the state of oxidative stress increases with gestational age as depicted by the black curve. The first signs of placental angiogenesis occur at 3 weeks of gestation. However, from about 12 weeks onwards, blood vessels (red lines) protrude towards the trophoblastic layers of the villi (outlined in green), where blood exchange between maternal and foetal circulation is optimal (shown in panel three). From about 9-23 weeks of gestation, there is an expansion of the foetal capillary bed by branching and nonbranching angiogenesis (dashed red lines in angiogenesis panel). From 23-24 weeks of gestation, the greatest changes in blood vessel development and villous composition are observed (peak angiogenesis panel). Angiogenesis continues until term with the maturation of blood vessels and development of a more complex vascular network to facilitate exponential foetal growth. The horizontal black arrows indicate the approximate time each process depicted in the panels occurs. Note: this graph has been constructed by interpretations of multiple studies reporting findings of systemic oxidative stress markers present in women during normal pregnancy as studies on placental/uterine oxidative stress are limited. Source: [101].
View Figure 1
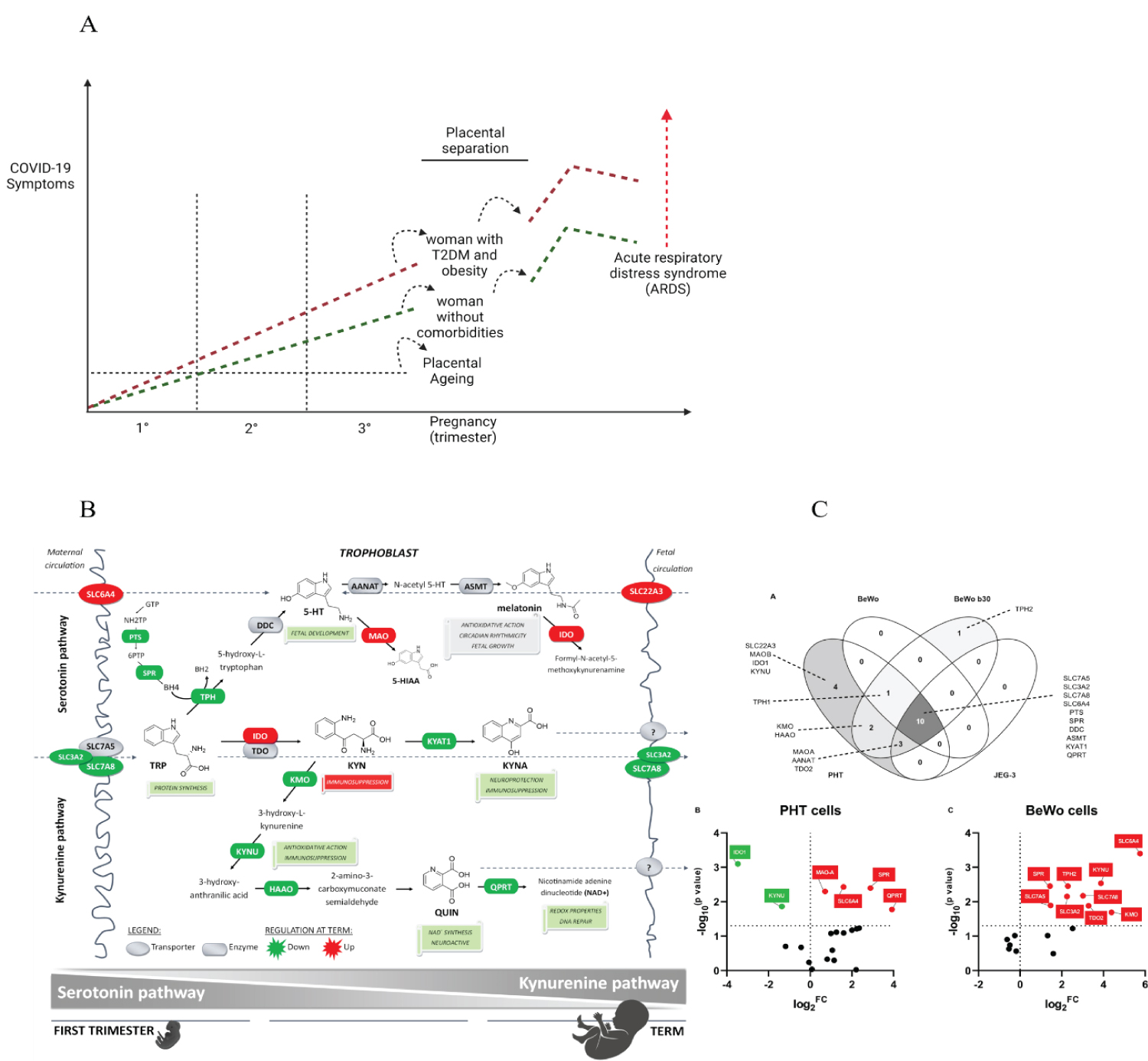 Figure 2: Symptomatic COVID-19 during pregnancy and immediately post-delivery. A mosaic built from figures adapted from Rona Karahoda, et al. A) The scheme theorizes why pregnant women progress to symptomatic forms of COVID-19 hours after childbirth has taken place. The immunosuppressive effect of KYN abruptly ceases to exist with the separation of the placenta, making the puerperal woman regain her immunocompetent status, re-establishing her capacity for inflammation against the SARS-CoV-2 virus. As aggravating factors, the puerperal woman may have presented significant gastrointestinal involvement by the virus, leading to a deficiency of Tryptophan (Try) and Phenylalanine (Phe). These metabolic changes are even more pronounced in women who have inflammatory comorbidities such as obesity and type 2 diabetes mellitus (T2DM). It is recurrent that women after childbirth evolve to orotracheal intubation or need a high demand for supplemental oxygen. SARS-CoV-2 infection can be a little symptomatic during the entire pregnancy, as the placenta provides substrates to the pregnant woman absent in the general population in COVID-19; furthermore, natural immunosuppression exerts a unique protective power of adaptation. Of human placentals concerning SARS-CoV-2. In the first's trimesters, pregnant women are not totally immune to viral aggressions, as multiple mechanisms can cause thrombotic events in the placenta, embryo/foetus and pregnant women, in addition to autoimmune events triggered by viral infection. B) Proposed gestational age-dependent changes in placental Try metabolic pathways. The schematic illustration is based on gene expression studies and rate-limiting enzymes/key transporters, protein, or functional activity. The first-trimester placenta preferentially uses Try to generate 5-HT involved in foetal development and synthesis of KYN metabolites implied with neuroprotection, antioxidative and redox effects, and DNA repair. On the other hand, the term placenta synthesizes significant amounts of KYN necessary for immune regulation activities. Collectively, our findings strongly indicate gestational age-related changes in placental handling of Try and its main metabolites, 5-HT and KYN. All the presented changes may reflect foetal demands for metabolites of either pathway to ensure proper embryonic and foetal development throughout pregnancy-Gene expression of Try pathway-related enzymes and transporters in placental-derived cells. Placental-derived cells were obtained from three different passages of BeWo, BeWo b30 (choriocarcinoma cell lines), and JEG cells and five primary human trophoblast cells (PHT) isolations. (A) Venn diagram of the overlap in gene expression among four cell lines. PHT and BeWo b30 cells share the highest homology, with the PHT cells additionally expressing four important genes, including the rate-limiting enzyme, IDO1, and the polyspecific organic transporter, SLC22A3. Subsequently, effect of differentiation in these two cell lines was tested, revealing dissimilarity between spontaneous syncytialization "(B)" and forskolin-induced differentiation "(C)". Data are visualized as volcano plots of fold changed expression in the differentiated cells, compared to the undifferentiated counterpart. Colour indicates statistically significant changes: red - up-regulation, green - down-regulation. Statistical significance was evaluated using the Mann-Whitney test. Choriocarcinoma-derived cell lines (BeWo, BeWob30, JEG-3). Source: [5].
View Figure 2
Figure 2: Symptomatic COVID-19 during pregnancy and immediately post-delivery. A mosaic built from figures adapted from Rona Karahoda, et al. A) The scheme theorizes why pregnant women progress to symptomatic forms of COVID-19 hours after childbirth has taken place. The immunosuppressive effect of KYN abruptly ceases to exist with the separation of the placenta, making the puerperal woman regain her immunocompetent status, re-establishing her capacity for inflammation against the SARS-CoV-2 virus. As aggravating factors, the puerperal woman may have presented significant gastrointestinal involvement by the virus, leading to a deficiency of Tryptophan (Try) and Phenylalanine (Phe). These metabolic changes are even more pronounced in women who have inflammatory comorbidities such as obesity and type 2 diabetes mellitus (T2DM). It is recurrent that women after childbirth evolve to orotracheal intubation or need a high demand for supplemental oxygen. SARS-CoV-2 infection can be a little symptomatic during the entire pregnancy, as the placenta provides substrates to the pregnant woman absent in the general population in COVID-19; furthermore, natural immunosuppression exerts a unique protective power of adaptation. Of human placentals concerning SARS-CoV-2. In the first's trimesters, pregnant women are not totally immune to viral aggressions, as multiple mechanisms can cause thrombotic events in the placenta, embryo/foetus and pregnant women, in addition to autoimmune events triggered by viral infection. B) Proposed gestational age-dependent changes in placental Try metabolic pathways. The schematic illustration is based on gene expression studies and rate-limiting enzymes/key transporters, protein, or functional activity. The first-trimester placenta preferentially uses Try to generate 5-HT involved in foetal development and synthesis of KYN metabolites implied with neuroprotection, antioxidative and redox effects, and DNA repair. On the other hand, the term placenta synthesizes significant amounts of KYN necessary for immune regulation activities. Collectively, our findings strongly indicate gestational age-related changes in placental handling of Try and its main metabolites, 5-HT and KYN. All the presented changes may reflect foetal demands for metabolites of either pathway to ensure proper embryonic and foetal development throughout pregnancy-Gene expression of Try pathway-related enzymes and transporters in placental-derived cells. Placental-derived cells were obtained from three different passages of BeWo, BeWo b30 (choriocarcinoma cell lines), and JEG cells and five primary human trophoblast cells (PHT) isolations. (A) Venn diagram of the overlap in gene expression among four cell lines. PHT and BeWo b30 cells share the highest homology, with the PHT cells additionally expressing four important genes, including the rate-limiting enzyme, IDO1, and the polyspecific organic transporter, SLC22A3. Subsequently, effect of differentiation in these two cell lines was tested, revealing dissimilarity between spontaneous syncytialization "(B)" and forskolin-induced differentiation "(C)". Data are visualized as volcano plots of fold changed expression in the differentiated cells, compared to the undifferentiated counterpart. Colour indicates statistically significant changes: red - up-regulation, green - down-regulation. Statistical significance was evaluated using the Mann-Whitney test. Choriocarcinoma-derived cell lines (BeWo, BeWob30, JEG-3). Source: [5].
View Figure 2
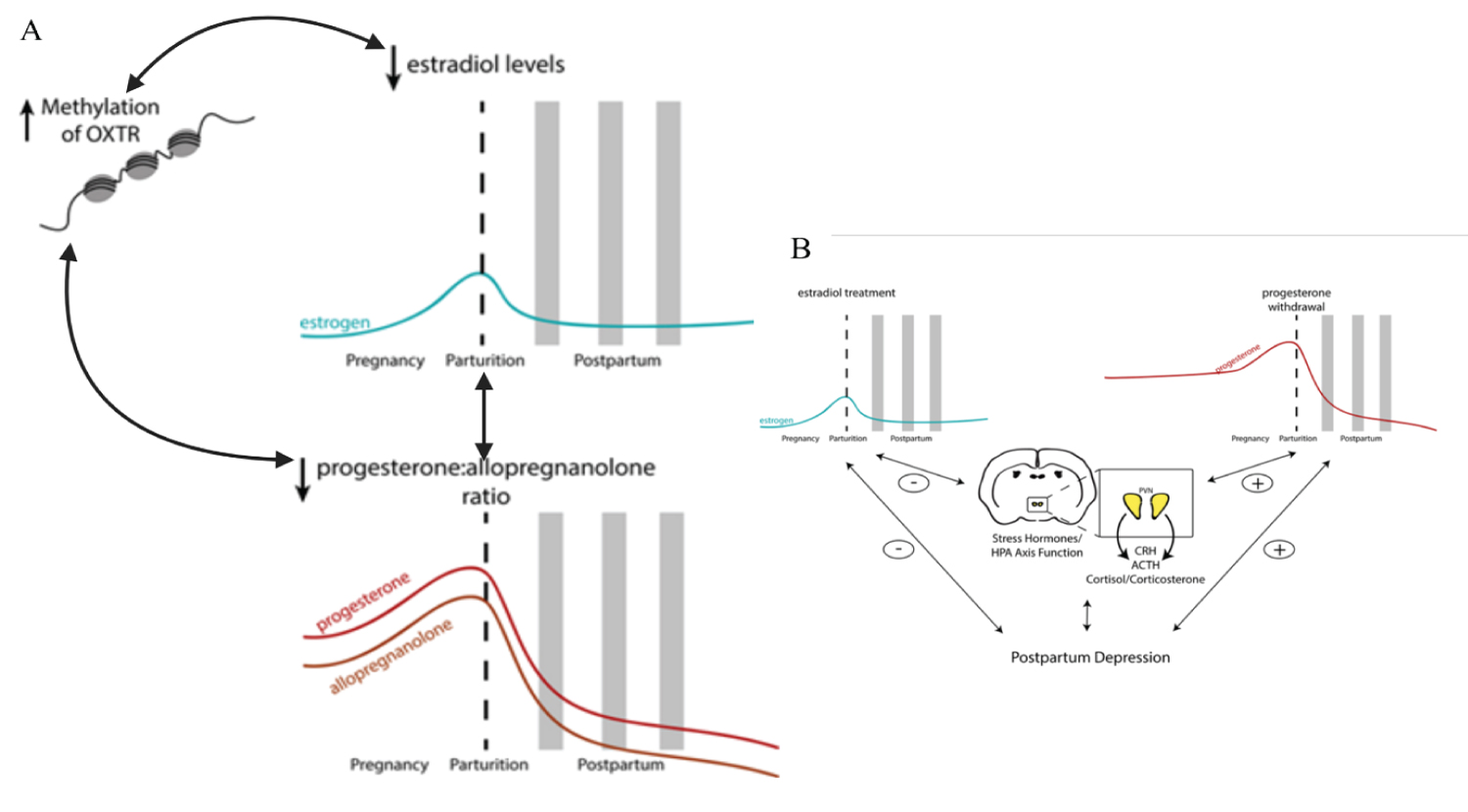 Figure 3: Postpartum depression (PPD). Figure adapted. PPD is associated with higher β-endorphin levels, reduced platelet serotonin levels, increased monoamine oxidase-A density, low omega-3 levels, and lower vitamin D levels. Stress hormones during the peripartum period have also been implicated in the underlying neurobiology of postpartum mood disorders. Neuroendocrine abnormalities PPD are associated with ovarian and lactogenic hormones (oestrogen, progesterone, oxytocin, prolactin) and stress hormones (cortisol, ACTH, CRH) and their neurosteroid metabolites; reproductive hormones impact the function of the hypothalamic-pituitary-adrenal axis (HPA axis) and vice versa. Strogen and progesterone play an ambiguous role in developing PPD - higher levels of progesterone are correlated with worse depression scores in postpartum women childbirth. Placental hormones are also implicated in the development of PPD: corticotropin-releasing hormone (CRH) has been shown to be a strong predictor of postpartum depression. Oxytocin (OXTR) and oxytocin (OXT) receptor: A single nucleotide polymorphism (SNP) in OXT was predictive of variation in breastfeeding duration and postpartum depression scores. At the same time, the interaction between SNP in OXTR and adverse life events did not correlate with maternal behaviours but was predictive of prepartum depression scores. Variations in DNA methylation of the OXTR gene in women with postpartum depression are negatively correlated with serum oestradiol levels. A significant interaction between oestradiol, OXTR DNA methylation and the ratio of allopregnanolone to progesterone was also saw in women with postpartum depression. Thus, epigenetic changes can influence the biochemical pathways associated with postpartum depression. It is important to consider that the inflammatory response in COVID-19 is intense and mediated by IL-6 (perhaps the most important cytokine in the Central Nervous System) and the immunosuppressive role of IL-10. In addition, tryptophan has its metabolism altered by kynurenine products due to IDO-1, and Kynurenine by-products are GABAergic and glutaminergic receptors ligands. Severe hypoxemia due to lung injury by SARS-CoV-2 and systemic injury causes adenosine overproduction that can mimic depressive and anxiety symptoms. Source: [111].
View Figure 3
Figure 3: Postpartum depression (PPD). Figure adapted. PPD is associated with higher β-endorphin levels, reduced platelet serotonin levels, increased monoamine oxidase-A density, low omega-3 levels, and lower vitamin D levels. Stress hormones during the peripartum period have also been implicated in the underlying neurobiology of postpartum mood disorders. Neuroendocrine abnormalities PPD are associated with ovarian and lactogenic hormones (oestrogen, progesterone, oxytocin, prolactin) and stress hormones (cortisol, ACTH, CRH) and their neurosteroid metabolites; reproductive hormones impact the function of the hypothalamic-pituitary-adrenal axis (HPA axis) and vice versa. Strogen and progesterone play an ambiguous role in developing PPD - higher levels of progesterone are correlated with worse depression scores in postpartum women childbirth. Placental hormones are also implicated in the development of PPD: corticotropin-releasing hormone (CRH) has been shown to be a strong predictor of postpartum depression. Oxytocin (OXTR) and oxytocin (OXT) receptor: A single nucleotide polymorphism (SNP) in OXT was predictive of variation in breastfeeding duration and postpartum depression scores. At the same time, the interaction between SNP in OXTR and adverse life events did not correlate with maternal behaviours but was predictive of prepartum depression scores. Variations in DNA methylation of the OXTR gene in women with postpartum depression are negatively correlated with serum oestradiol levels. A significant interaction between oestradiol, OXTR DNA methylation and the ratio of allopregnanolone to progesterone was also saw in women with postpartum depression. Thus, epigenetic changes can influence the biochemical pathways associated with postpartum depression. It is important to consider that the inflammatory response in COVID-19 is intense and mediated by IL-6 (perhaps the most important cytokine in the Central Nervous System) and the immunosuppressive role of IL-10. In addition, tryptophan has its metabolism altered by kynurenine products due to IDO-1, and Kynurenine by-products are GABAergic and glutaminergic receptors ligands. Severe hypoxemia due to lung injury by SARS-CoV-2 and systemic injury causes adenosine overproduction that can mimic depressive and anxiety symptoms. Source: [111].
View Figure 3
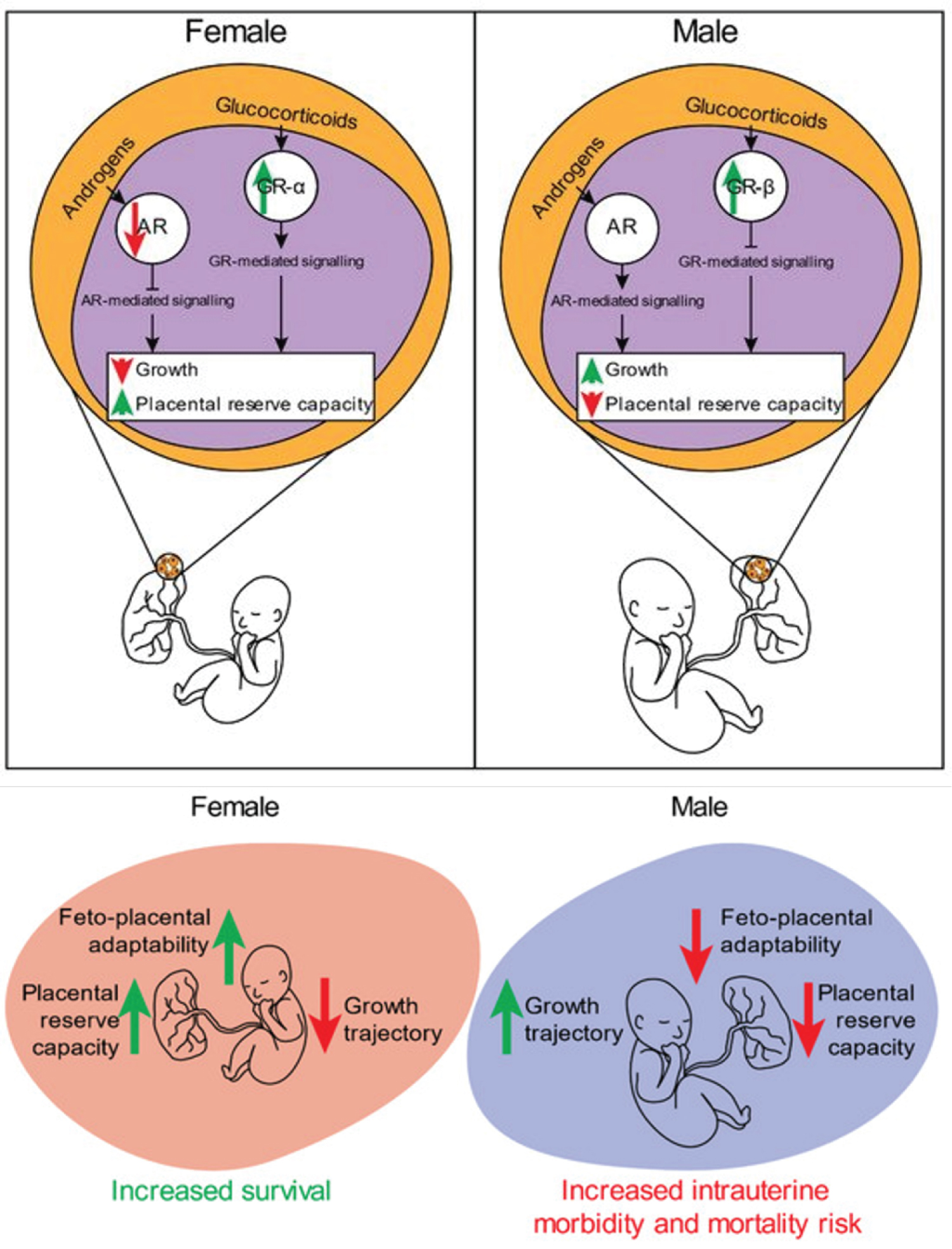 Figure 4: Overview of sex-specific in utero adaptations that drive differences in growth and survival outcomes. In females, increased foeto-placental adaptability, and placental reserve capacity result in increased survival rates at the expense of a reduced growth trajectory, whereas the opposite is observed in males. Changes in GR expression and localisation may contribute to sex-specific differences in growth outcomes. Placentae from IUGR females have been shown to increase nuclear GRα expression in both extra villous trophoblast (EVT) and syncytiotrophoblast cells (as measured by immunohistochemistry in humans) and increased nuclear expression of GRαD1-3 isoforms (as measured by Western blot in mice). Unlike in females, growth restriction in males was associated with increased GRβ expression in humans, and a decrease in the activity of GrαA in mice, as determined by its cytoplasmic sequestration. These sex-specific differences in the placental responsivity to changes in glucocorticoids favour the work conducted in human population studies of maternal asthma associated with sex-specific intrauterine growth outcomes. Throughout gestation, the levels of maternal circulating androgens including testosterone, dihydrotestosterone (DHT), dehydroepiandrosterone (DHEA), and androstenedione (A4) increase three-fold by the third trimester when compared to non-pregnant levels. There are sex differences in the concentrations of circulating fetal and maternal androgens, with levels higher in male fetuses and in the maternal circulation of individuals carrying a male fetus. Fetal derived androgens are readily aromatised to estrogens by the placental cytochrome P450 (CYP) enzyme, CYP19A1 (aromatase). These fetal-derived androgens are required for placental-biosb synthesised estrogens, an important physiological process that modulates uteroplacental vasculature. Although the placenta is known to be involved in the aromatisation of androgens, other studies have reported placental-specific androgen biosynthesis. Escobar, et al. 2011 reported third trimester human placentae were able to synthesise androgens de novo. More recently, levels of tissue-specific androgens were measurable in first trimester human male placentae; the levels of DHEA, A4, testosterone, and DHT were all detectable, with some of these androgens having higher levels when compared with liver-, adrenal-, or testis-specific levels. In line with these findings, the same study reported expression of genes involved in androgen biosynthesis in the male placenta. Source: [112].
View Figure 4
Figure 4: Overview of sex-specific in utero adaptations that drive differences in growth and survival outcomes. In females, increased foeto-placental adaptability, and placental reserve capacity result in increased survival rates at the expense of a reduced growth trajectory, whereas the opposite is observed in males. Changes in GR expression and localisation may contribute to sex-specific differences in growth outcomes. Placentae from IUGR females have been shown to increase nuclear GRα expression in both extra villous trophoblast (EVT) and syncytiotrophoblast cells (as measured by immunohistochemistry in humans) and increased nuclear expression of GRαD1-3 isoforms (as measured by Western blot in mice). Unlike in females, growth restriction in males was associated with increased GRβ expression in humans, and a decrease in the activity of GrαA in mice, as determined by its cytoplasmic sequestration. These sex-specific differences in the placental responsivity to changes in glucocorticoids favour the work conducted in human population studies of maternal asthma associated with sex-specific intrauterine growth outcomes. Throughout gestation, the levels of maternal circulating androgens including testosterone, dihydrotestosterone (DHT), dehydroepiandrosterone (DHEA), and androstenedione (A4) increase three-fold by the third trimester when compared to non-pregnant levels. There are sex differences in the concentrations of circulating fetal and maternal androgens, with levels higher in male fetuses and in the maternal circulation of individuals carrying a male fetus. Fetal derived androgens are readily aromatised to estrogens by the placental cytochrome P450 (CYP) enzyme, CYP19A1 (aromatase). These fetal-derived androgens are required for placental-biosb synthesised estrogens, an important physiological process that modulates uteroplacental vasculature. Although the placenta is known to be involved in the aromatisation of androgens, other studies have reported placental-specific androgen biosynthesis. Escobar, et al. 2011 reported third trimester human placentae were able to synthesise androgens de novo. More recently, levels of tissue-specific androgens were measurable in first trimester human male placentae; the levels of DHEA, A4, testosterone, and DHT were all detectable, with some of these androgens having higher levels when compared with liver-, adrenal-, or testis-specific levels. In line with these findings, the same study reported expression of genes involved in androgen biosynthesis in the male placenta. Source: [112].
View Figure 4
Cathepsins [9-13] identified as a lysosomal protease that degrades substrates in acidic compartments, Furins [14-16] ubiquitous proprotein convertase cleaves substrates at the consensus sequence -Arg-X-Lys/Arg-Arg↓. For these two proteins to have their actions guaranteed, they need the microenvironment where they act to be acidic. Cathepsin and Furins are induced to be expressed mainly in environments with low oxygen tension, that is, in a hypoxemic environment. Both proteins are related to mental diseases, cardiovascular diseases and the development of neoplasms and metastasis. However, they also participate essentially and fundamentally in protein processing when patients are in homeostasis. They also play an essential role in placental implantation and the process of invasive waves related to placental development and implantation, and these reasons justify their presence, in high concentrations, in syncytiotrophoblast. Cathepsin L [16-18] and the rasHa oncogene are expressed in two coincident waves corresponding to periods during which the placenta is invasive and just before parturition. Large amounts of cathepsin L in the placenta suggest that the proteinase has a significant function there. Expression of cathepsin L in the placenta is potentially under the control of the ras product gene p21; both are under developmental control [19-21] (Table 1 and Figure 5).
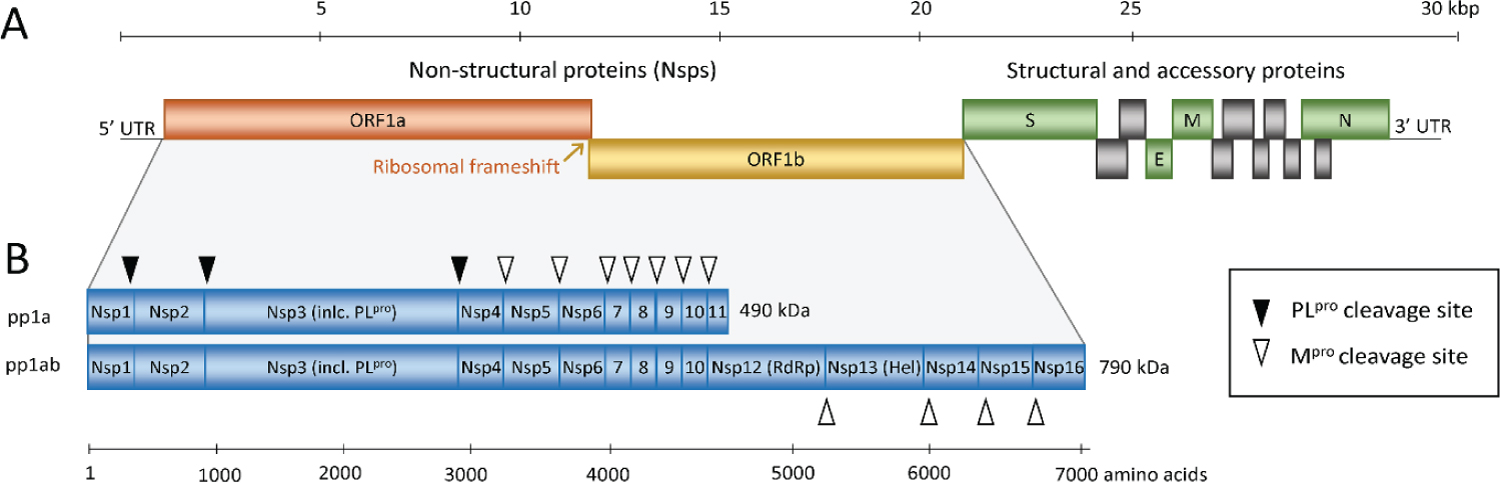 Figure 5: Schematic representation of the SARS-CoV and SARS-CoV-2 genome (A) and the replicase polyproteins (B). The genomic RNA comprises 2 parts, of which the first part (ORF1a and ORF1b) directly translates into 2 polyproteins pp1a and pp1ab due to a -1 frameshift between the 2 ORFs (orange arrow). They are composed of 16 Nsps (Nsp1-Nsp16) that form the replication-transcription complex. The polyproteins are processed into mature Nsps by the 2 peptidases as indicated in panel B by black and white triangles, namely Mpro, which is Nsp5, cleaves the polyprotein at 11 sites and PLpro, which is part of the Nsp3, performs cleavage at 3 sites. Among the released Nsps are RdRp (Nsp12) and RNA helicase (Nsp13). The 3' third of the genome encodes the structural and accessory proteins, including spike (S), envelope (E), membrane (M), and nucleocapsid (N) shown in green and gray. Nsp, nonstructural protein; ORF, open reading frame; RdRp, RNA-dependent RNA polymerase; SARS-CoV, Severe Acute Respiratory Syndrome Coronavirus; SARS-CoV-2, Severe Acute Respiratory Syndrome Coronavirus 2. Source: [31].
View Figure 5
Figure 5: Schematic representation of the SARS-CoV and SARS-CoV-2 genome (A) and the replicase polyproteins (B). The genomic RNA comprises 2 parts, of which the first part (ORF1a and ORF1b) directly translates into 2 polyproteins pp1a and pp1ab due to a -1 frameshift between the 2 ORFs (orange arrow). They are composed of 16 Nsps (Nsp1-Nsp16) that form the replication-transcription complex. The polyproteins are processed into mature Nsps by the 2 peptidases as indicated in panel B by black and white triangles, namely Mpro, which is Nsp5, cleaves the polyprotein at 11 sites and PLpro, which is part of the Nsp3, performs cleavage at 3 sites. Among the released Nsps are RdRp (Nsp12) and RNA helicase (Nsp13). The 3' third of the genome encodes the structural and accessory proteins, including spike (S), envelope (E), membrane (M), and nucleocapsid (N) shown in green and gray. Nsp, nonstructural protein; ORF, open reading frame; RdRp, RNA-dependent RNA polymerase; SARS-CoV, Severe Acute Respiratory Syndrome Coronavirus; SARS-CoV-2, Severe Acute Respiratory Syndrome Coronavirus 2. Source: [31].
View Figure 5
The performance of autopsies has shown that some lungs that progress with severity from COVID-19 have pneumocytes II modified to syncytium formation, and this phenomenon occurs mainly in cases where the disease has evolved into severe or critical forms [22-24].
This article shows that lesion caused by SARS-CoV-2 promotes a highly hypoxemic environment that induces the expression of cathepsins and furins which, in addition to their usual functions, also help in the viral replication of the coronavirus [16,25-27].
There is, in this process, a tendency to acidification of cell environment so that the functions of cathepsins and furins occur optimally. So, this justifies the difficulty in maintaining, in critically ill patients, an adequate pH in the blood gases and what happens is that, in the usual management of the severely ill patient COVID-19, the hypoxemic environment is allowed to be allowed, conduct called permissive hypercapnia [28].
This permissive hypercapnia has been allowed. Nevertheless, this article shows that this conduct must be proscribed. The critically ill patient should not remain with arterial blood gases showing acidosis due to elevated PCO2, allowing the cellular environment to remain acidified means allowing more cathepsins and Furins to be expressed, facilitating viral replication and dissemination, and enabling syncytium formation in lung cells. In other words, the syncytium formation process in lung cells shows that the hypoxemia of the disease intensified by the tendency of the cell medium to try to acidify so that cathepsin and furin act efficiently is how COVID-19 perpetuates viral replication forms syncytia that also help SARS-COV-2 in its infection and is the phenomenon that is presented to us to manage in critically ill patients. The acidosis presented by many intubated patients is very difficult to reverse, justifying the medical management of permissive hypercapnia. The problem with this approach is that we are permissive with something that keeps the virus in replication and dissemination, in addition to allowing a severe tendency to immunosuppression and tolerance in these critically ill patients, justifying opportunistic infections and providing a highly tumorigenic environment [29-31].
Permissive hypercapnia must be proscribed in patients COVID-19, and the patient should be managed with improved mechanical ventilation and the use of 8.4% sodium bicarbonate in order to adjust the pH to the most physiological possible so that expression induction of Cathepsins and Furins, as well as the maintenance of a medium with optimal pH to carry out their functions, are blocked [32,33].
This article shows the vital role of Methylprednisolone in attenuating the inflammatory manifestations promoted by SARS-CoV-2. More than this drug acting on classical inflammation pathways, corticosteroids seem to act on proteins that directly favour COVID-19 severity, that is, on furins and cathepsins [34].
The effect of Methylprednisolone (MTP), an anti-inflammatory drug, upon purified calpain and the Ca2+-mediated degradation of endogenous proteins of spinal cord homogenate in vitro has been examined. The activity of calpain purified from rabbit muscle was greatly inhibited in a dose-dependent fashion by MTP. Calpain-mediated degradation of myelin basic protein (MBP) was also inhibited by MTP and cathepsin B-mediated MBP breakdown. MTP acts as a proteinase (calpain) inhibitor and defines a new mechanism for its actions.
Cathepsin B-like activity was measured in lung secretions. The enzyme had a pH optimum of approximately 5.5 and had the characteristics of an alkaline-stable cysteine proteinase. Enzyme activity in the sputum from ten subjects with chronic bronchitis was significantly reduced after 5 days of treatment with prednisolone. A pharmacologic dose of Methylprednisolone (50 mg/kg) significantly delayed the subcellular redistribution of cathepsin D and the other hydrolases in the ischemic heart. Unlike untreated hearts, no evidence of enzyme diffusion from lysosomes could be demonstrated immunohistochemically in corticosteroid-treated ischemic hearts for over 45 min. This study demonstrates that corticosteroid pre-treatment does not prevent alterations in cardiac lysosomes during severe ischemia indefinitely, but it does significantly delay their development.
Cathepsins increased in cystic fibrosis and have a role in airway remodelling during fibrogenesis in pulmonary fibrosis. An association has led to the hypothesis that emphysema in subjects with normal serum alpha-antitrypsin concentrations arises as a result of an imbalance between the enzymes and inhibitors within the lung such that enzyme activity persists. The main serum and alveolar inhibitor of proteolytic enzymes are alpha-antitrypsin3, and severe deficiency of this protein is associated with the development of pulmonary emphysema. The sputum-to-serum concentration ratios of alpha-antichymotrypsin rose during corticosteroid treatment, and steroid treatment may increase protection against these proteolytic enzymes, which could benefit some patients, particularly those with recurrent bacterial infections [35,36].
Furin inhibits inflammatory responses in the myeloid cells by reducing the production of pro-inflammatory cytokines. The function of Furin in immunoregulation is not entirely clear, but its expression in immune cells is critical for the functional maturation of anti-inflammatory pro-TGF-β1 cytokine and the suppressive function of regulatory T cells.
Furin levels were significantly elevated in Peripheral Blood (PB) and Synovial Fluid in some Rheumatic Disorders. Mononuclear cells, T cells, and monocytes from Rheumatoid Arthritis patients compared to healthy controls had Furin levels significantly associated with the prevailing prednisolone treatment, higher prednisolone doses, and increased C-reactive protein levels and Health and with functional maturation of anti-inflammatory protransforming growth factor-β1 (pro-TGF-β1) cytokine and the suppressive function of regulatory T cell. Some cytokines as IL-1β, IL-2, IL-4, IL-6, IL-7, IL-10, IFN-γ, and TNF-α levels in plasma were correlated with Furin.
Recently a study showed differences in ACE2, TMPRSS2, and furin epithelial and airway gene expression are unlikely to confer enhanced COVID-19 pneumonia risk in patients with asthma across all treatment intensities and severity. ACE2 expression was inversely correlated with markers of type 2 immunity, with no influence of sex or use of nasal corticosteroids. The same article showed that segmental bronchial allergen challenge in adults with mild asthma led to decreases in ACE2 expression and that IL-13 reduces ACE2 expression on cultured bronchial epithelial cells. The data shows the importance of understanding the effects of TH2 and IL-17-driven inflammation, inhaled corticosteroids on airway epithelial cell ACE2 expression, and the susceptibility of these cells to infection and replication by SARS-CoV-2 [37,38].
This article highlights the importance of using corticosteroids in COVID-19, including pulse therapy. The use of corticosteroids deserves further clinical studies. However, in a situation in which we do not have specific medications, it is essential to show that corticosteroids have a fundamental role because they have nonspecific anti-inflammatory actions and can act in numerous ways, contributing to the improvement of the patient COVID-19 in moderate, severe and critical illness [39-41].
Mitochondria are multifunctional organelles, and their primary role is in ATP generation by oxidative phosphorylation (OXPHOS) using substrates derived from β-oxidation and the tricarboxylic acid cycle, and are also involved in cell signalling via the production of reactive oxygen species (ROS). ROS are a normal by-product of OXPHOS but, when produced in excess, e.g., during disrupted oxygen (O2) or substrate supply, they can cause oxidative stress and damage DNA, lipids, and proteins [42-45].
Hypoxia is the main cause of foetal growth restriction at high altitudes and is a common feature of sea-level pregnancy complications. Some studies in rodents show that inhalation hypoxia adapts placental morphology and nutrient transport to the foetus depends upon the degree, timing, and length of O2 restriction by the low oxygen environment and that, even as oxygen becomes available, glycolysis would be preferred to oxidative phosphorylation. Since oxidative phosphorylation has a more significant potential to produce free radicals, supporting glycolysis may act as a protective mechanism in preventing teratogenesis in a tissue that is undergoing rapidly (Figure 6 and Figure 7).
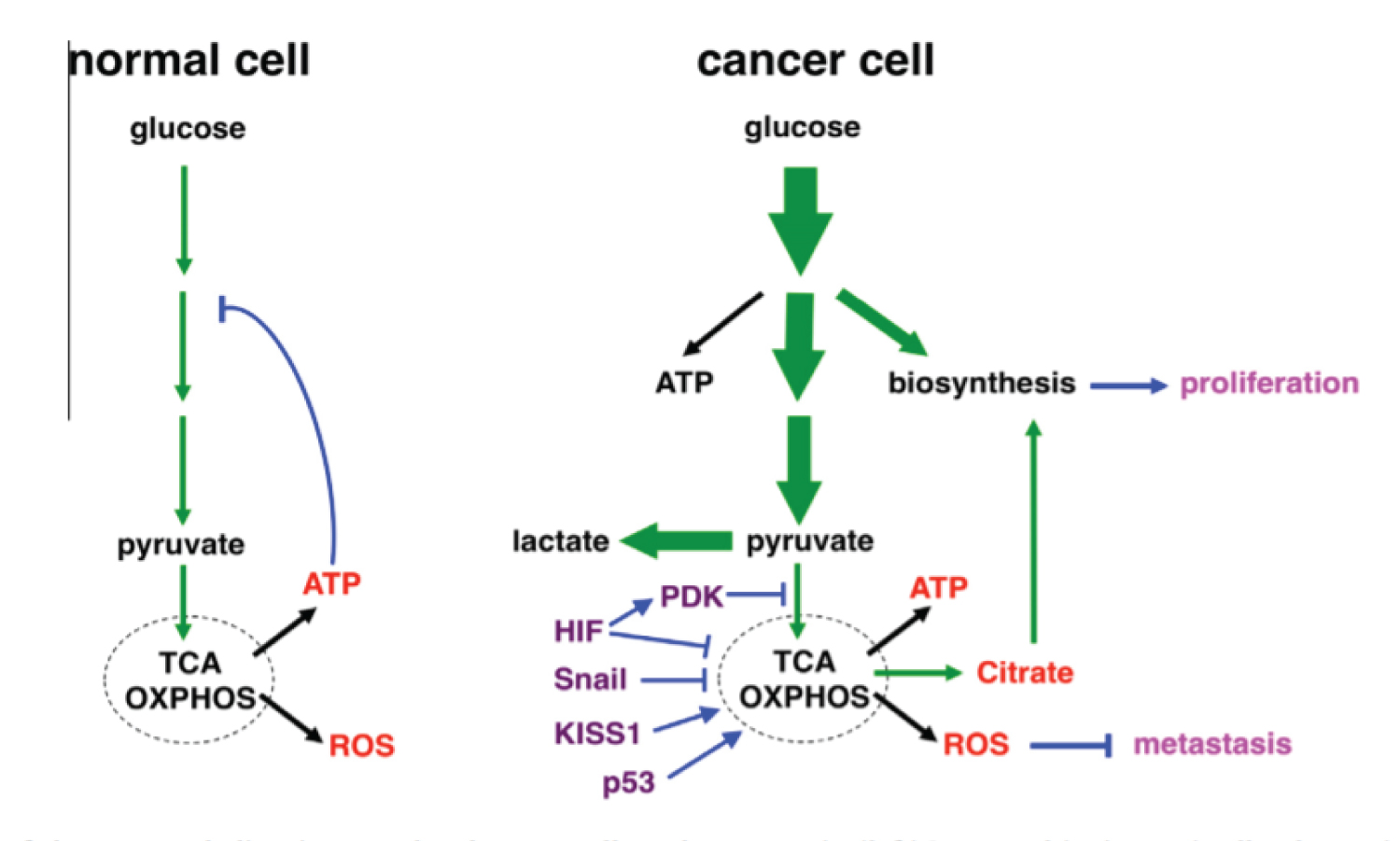 Figure 6: Text was modified by the author of this article. Schematic illustration of glucose metabolism in normal and cancer cells under normoxia. There is a idera that placenta uses the same via to generate ATP. (left) In normal (quiescent) cells, glucose is converted to pyruvate through glycolysis, and most pyruvate enters mitochondrial oxidative metabolism for efficient energy generation (in the form of ATP). Glucose is predominantly used for energy production. High levels of ATP attenuate glycolysis via feedback inhibition. (right) Cancer cells dramatically increase glucose uptake and glycolysis (indicated by bold arrows). A significant portion of glucose carbon is diverted to biosynthetic pathways to fuel cell proliferation. Pyruvate is preferentially shunted to lactate, resulting in increased lactate production. Oxidative metabolism persists, but is uncoupled from increased glycolysis. The respiration byproducts ROS exhibit anti-metastasis activity, which may explain why cancer cells keep glucose oxidation in check. The flux of glucose carbon is indicated by green arrows (The thickness of arrows reflects the relative amount of the flow). Major mitochondrial products are depicted in red; metastatic regulators are in purple; regulatory steps are in blue; and the metabolic effects on cancer are in pink. Source: [42].
View Figure 6
Figure 6: Text was modified by the author of this article. Schematic illustration of glucose metabolism in normal and cancer cells under normoxia. There is a idera that placenta uses the same via to generate ATP. (left) In normal (quiescent) cells, glucose is converted to pyruvate through glycolysis, and most pyruvate enters mitochondrial oxidative metabolism for efficient energy generation (in the form of ATP). Glucose is predominantly used for energy production. High levels of ATP attenuate glycolysis via feedback inhibition. (right) Cancer cells dramatically increase glucose uptake and glycolysis (indicated by bold arrows). A significant portion of glucose carbon is diverted to biosynthetic pathways to fuel cell proliferation. Pyruvate is preferentially shunted to lactate, resulting in increased lactate production. Oxidative metabolism persists, but is uncoupled from increased glycolysis. The respiration byproducts ROS exhibit anti-metastasis activity, which may explain why cancer cells keep glucose oxidation in check. The flux of glucose carbon is indicated by green arrows (The thickness of arrows reflects the relative amount of the flow). Major mitochondrial products are depicted in red; metastatic regulators are in purple; regulatory steps are in blue; and the metabolic effects on cancer are in pink. Source: [42].
View Figure 6
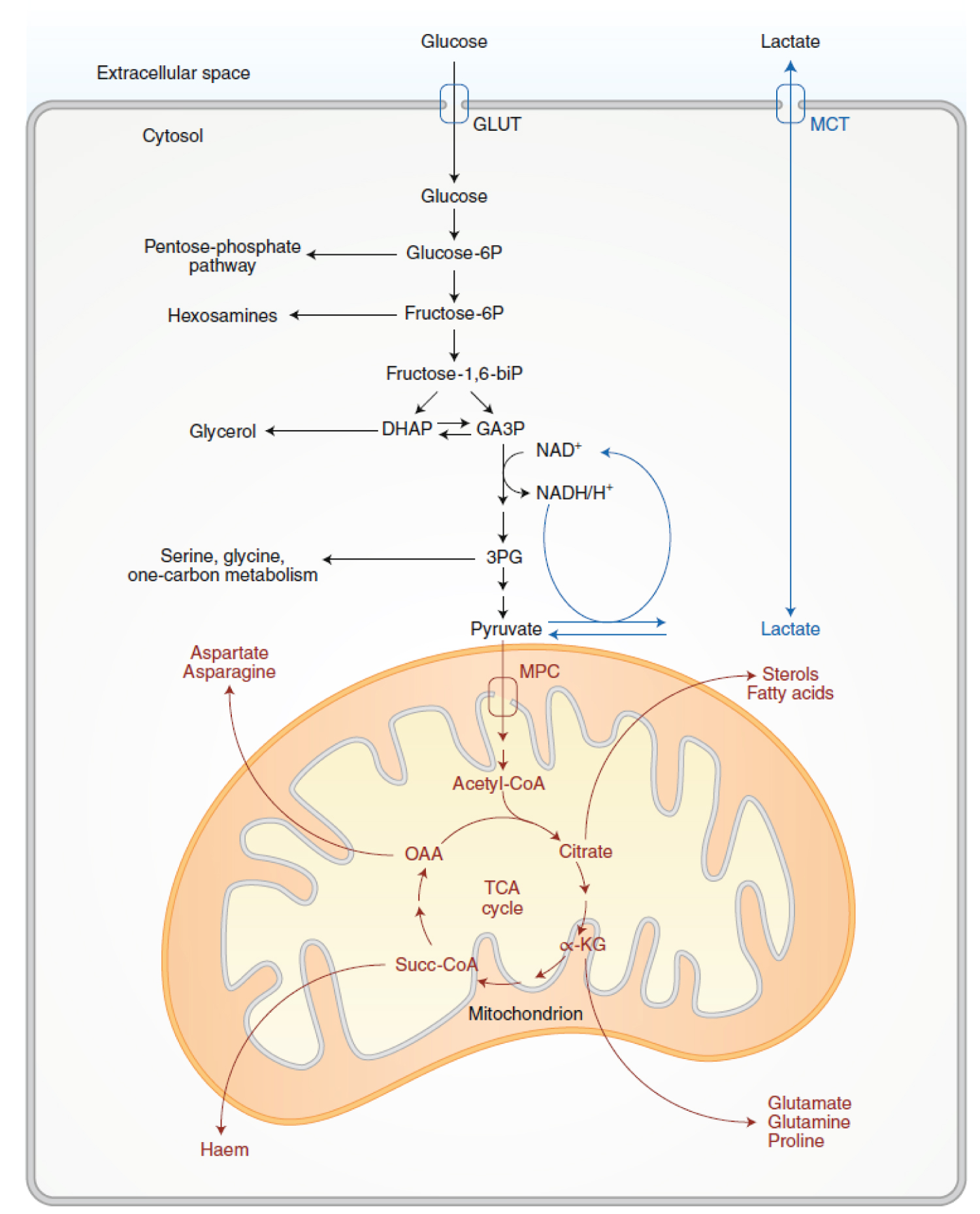 Figure 7: Glycolysis and TCA-cycle metabolism supply anabolic pathways. Activities related to glycolysis are in black, and activities related to pyruvate oxidation and the TCA cycle are in red. Both processes support cell growth by feeding branch pathways required for anabolism. In the Warburg effect, glycolysis terminates with lactate production and secretion despite the presence of oxygen. These latter steps (blue) provide a means of recycling NADH to NAD+ but result in loss of carbon from the cell upon lactate release. GLUT, glucose transporter; MCT, monocarboxylate transporter; MPC, mitochondrial pyruvate carrier; glucose-6P, glucose-6-phosphate; fructose-6P, fructose-6-phosphate; fructose-1,6-biP, fructose-1,6-bisphosphate; DHAP, dihydroxyacetone-phosphate; GA3P, glyceraldehyde-3-phosphate; 3PG, 3-phosphoglycerate; acetyl-CoA, acetyl coenzyme-A; α-KG, α-ketoglutarate; Succ-CoA, succinylCoA; OAA, oxaloacetate; NAD+, oxidized nicotinamide adenine dinucleotide; NADH, H+, reduced nicotinamide adenine dinucleotide. Source: [113].
View Figure 7
Figure 7: Glycolysis and TCA-cycle metabolism supply anabolic pathways. Activities related to glycolysis are in black, and activities related to pyruvate oxidation and the TCA cycle are in red. Both processes support cell growth by feeding branch pathways required for anabolism. In the Warburg effect, glycolysis terminates with lactate production and secretion despite the presence of oxygen. These latter steps (blue) provide a means of recycling NADH to NAD+ but result in loss of carbon from the cell upon lactate release. GLUT, glucose transporter; MCT, monocarboxylate transporter; MPC, mitochondrial pyruvate carrier; glucose-6P, glucose-6-phosphate; fructose-6P, fructose-6-phosphate; fructose-1,6-biP, fructose-1,6-bisphosphate; DHAP, dihydroxyacetone-phosphate; GA3P, glyceraldehyde-3-phosphate; 3PG, 3-phosphoglycerate; acetyl-CoA, acetyl coenzyme-A; α-KG, α-ketoglutarate; Succ-CoA, succinylCoA; OAA, oxaloacetate; NAD+, oxidized nicotinamide adenine dinucleotide; NADH, H+, reduced nicotinamide adenine dinucleotide. Source: [113].
View Figure 7
The placenta is compared with tumours and other proliferating or developing cells, the rate of glucose uptake dramatically increases and lactate is produced, even in the presence of oxygen and fully functioning mitochondria [46,47]. This pathway, known as The Warburg Effect (Figure 5 and Figure 6), is the regeneration of NAD+ from NADH in the pyruvate to lactate step that completes aerobic glycolysis. In this scenario, NADH produced by glyceraldehyde phosphate dehydrogenase (GAPDH) must be consumed to regenerate NAD+ to keep glycolysis active. This high rate of glycolysis allows supply lines to remain open that can, for example, siphon 3-phosphoglycerate (3PG) to serine for one-carbon metabolism-mediated production of NADPH nucleotides. The Warburg Effect might support a metabolic environment that allows for rapid biosynthesis to support growth and proliferation [17,48,49].
A first publication based on observations of COVID-19 patients already indicated to us a possibility that the infection by SARS-CoV-2 is responsible for autoimmune and immunosuppressive events and for causing a tolerant and tumorigenic environment. Time has come to say that this idea is, unfortunately, something real and that COVID-19 also causes dysmetabolism due to the need for oxygen that is almost always, in severe cases, not overcome due to intense hypoxemia and lack of substrates for aerobic respiration to be appropriately performed. Furins, cathepsins, Tryptophan metabolism shifted from the hepatic enzyme TDO to the inflammatory IDO-1 are examples of the multiple pathways that affect inflammation and immunosuppression in COVID-19, pathways that go far beyond the internalisation of ACE-2 and its impact on the Renin-Angiotensin-Aldosterone pathway [30,50].
In general, we are beginning to understand why even asymptomatic or mildly symptomatic patients may present some persistent inflammatory aftermath or symptoms of hypertension and dysglycemia, in addition to psychiatric conditions, after the infection. The virus passed through the human body at some point (with high or low inoculum), and some modification, no matter how small, was caused. Multiple pathways are responsible for controlling inflammation, so there are many possibilities of sequelae after infection by SARS-CoV-2 because it acts on these vias.
The inflammation in COVID-19 is multicausal: Direct tissue injury or metabolic and molecular changes, the parasite-host relationship, viral inoculum, patient comorbidities, time of exposure to hypoxemia (for the most symptomatic cases) and autoimmune events induced by autoantibodies and immune complex formation.
TMITSy-CoV-2 is a viral aetiology syndrome caused by SARS-CoV-2, with variable spectrum due to the multiple pathways influencing its clinical presentation. These factors depend on 1) The specific points of intersection of each patient concerning their genetic factors and their comorbidities, especially those considered to have an inflammatory profile, 2) On viral mutations that can direct the different variants to one or the other route, preferentially and 3) The hypoxemic environment, whose intensity and time of exposure to this environment have a directly proportional impact on the magnitude of the Syndrome.
It is clinically characterised by:
a) Thrombosis due to multiple causes: cathepsins impacting factor VIII, adenosine and platelet activation, changes in the endothelial response due to changes in tryptophan metabolism and the action of IDO-1, vasospasm due to angiotensin and aldosterone, or autoimmune events (Takotsubo Syndrome and Takayasu's Arteritis). Catastrophic events usually occur at multiple sites [51-54].
b) Generalised lymphocytic apoptosis: generalised apoptosis by cathepsins, by the action of hypoxemia and Kynurenin products, inducing cell death of CD4+ cells, CD8+ cells, B cells and mainly natural killer cells (NK cells). This fact justifies the existence of cases with reactivation of latent infectious diseases [55-58].
- Rapid growth of neoplasms: the magnitude of the disease can cause intense lymphopenia; the SARS-CoV-2 infection causes a highly tumorigenic environment.
- Immunoparalysis with the sustained presence of recurrent bacteremias, most of them by dermatological and mainly intestinal translocation (which, as an inflammatory site, is the leading site of bacterial dissemination due to barrier break).
c) Variable eosinophilia dependent on adenosine and tryptophan metabolites, in addition to inflammatory stimulation by IL-6 [30,59,60].
d) Basophilia [61].
e) Neuropsychiatric manifestations due to alterations in tryptophan metabolism, due to the action of Kynurenine by-products and adenosine excess. It includes peripheral pain and difficulty in sedation [13,62,63].
f) Anemia due to the use of pyrrolic rings to build enzymes in the inflammatory pathways, to a decrease in serotonin in the bone marrow and general consumption of iron used for the metabolism of the intracellular airways [63-66].
Inflammation "de novo": Every time there is an increased need for oxygen supply, there is a stimulus for the inflammatory pathways of oxidative stress to be reactivated. This fact is real and reflects the high prevalence of the event "failure to extubate". Patients have worsening neurological and respiratory status, in addition to systemic repercussions with neutrophilia and generalized inflammation, causing bacterial translocation and refractory vasoplegic shock.
The extubation guidelines do not consider COVID-19 as a disease that presents different demands and as long as this disease is seen as an already known disease, we will be doomed to new inflammation and failures.
The relationship between SARS-CoV-2 and the pregnant woman deserves essential considerations.
Had it been thought that pregnant and postpartum women should be a risk group for severity, plenty of hospital admissions would happen. However, there were few admissions concerning pregnant women, as they usually present with no symptoms or oligosymptomatic, especially in early pregnancy; however, the embryo/foetus is not necessarily saved from suffering from SARS-CoV-2 infection.
Intriguingly, the closer to term, the more symptomatic the pregnant woman becomes due to coronavirus infection. Perhaps the common thinking has considered that, because the placenta naturally immunosuppresses pregnant women, the usual thing would be to be more susceptible to infections, which can reactivate latent viral diseases. However, COVID-19 has three distinct phases similar to other biphasic diseases such as yellow fever. COVID-19 has a viremic phase, followed by a period of defervescence, which may be the resolution of the disease or proceed to an immune-mediated inflammatory phase which, in the case of COVID-19, starts around 8 days after the patient perceives the first symptoms [2,67].
So far, there is no specific antiviral drug that is effective. Even in the face of many controversies, Corticosteroid is an essential [2,68] option in alleviating the symptoms caused by the cytokine storm, responsible for the period in which many orotracheal intubations occur (OTI), especially in people with predictors of severity (mainly obese, diabetic, and elderly) because they already have an inflammatory profile related to their comorbidities [40,69-71].
This article shows that the placenta plays a similar role to the Corticosteroids immunosuppressing the patient in COVID-19, reducing the magnitude of the inflammatory storm and its consequences. Thus, the treatment for a disease that progresses with inflammation - and being biphasic (second phase without viral presence) - is to perform immunosuppression. The placenta naturally immunosuppresses pregnant women.
The structural and molecular changes that trigger specific diseases in pregnancy are based on changes in the formation of blood vessels, changes in invasive waves and changes that promote premature placental ageing mediated by multiple pathways, but with oxidative stress being one of the most critical factors and determinants of molecular and biochemical changes leading to structural ones.
SARS-CoV-2 infection promotes alterations based on the magnification of hypoxia and, more directly, aggression to the vascular endothelium. These changes simulate or are superimposed on pre-existing conditions such as diseases such as HELLP Syndrome, preeclampsia and eclampsia [72]. Furthermore, oxidative stress promotes [46,73] an environment of insulin resistance and the internalisation of ACE-2, an environment that allows pregnant women to develop a hypertensive period (Figure 8, Figure 9, Figure 10, Figure 11, Figure 12 and Figure 13) [74-76].
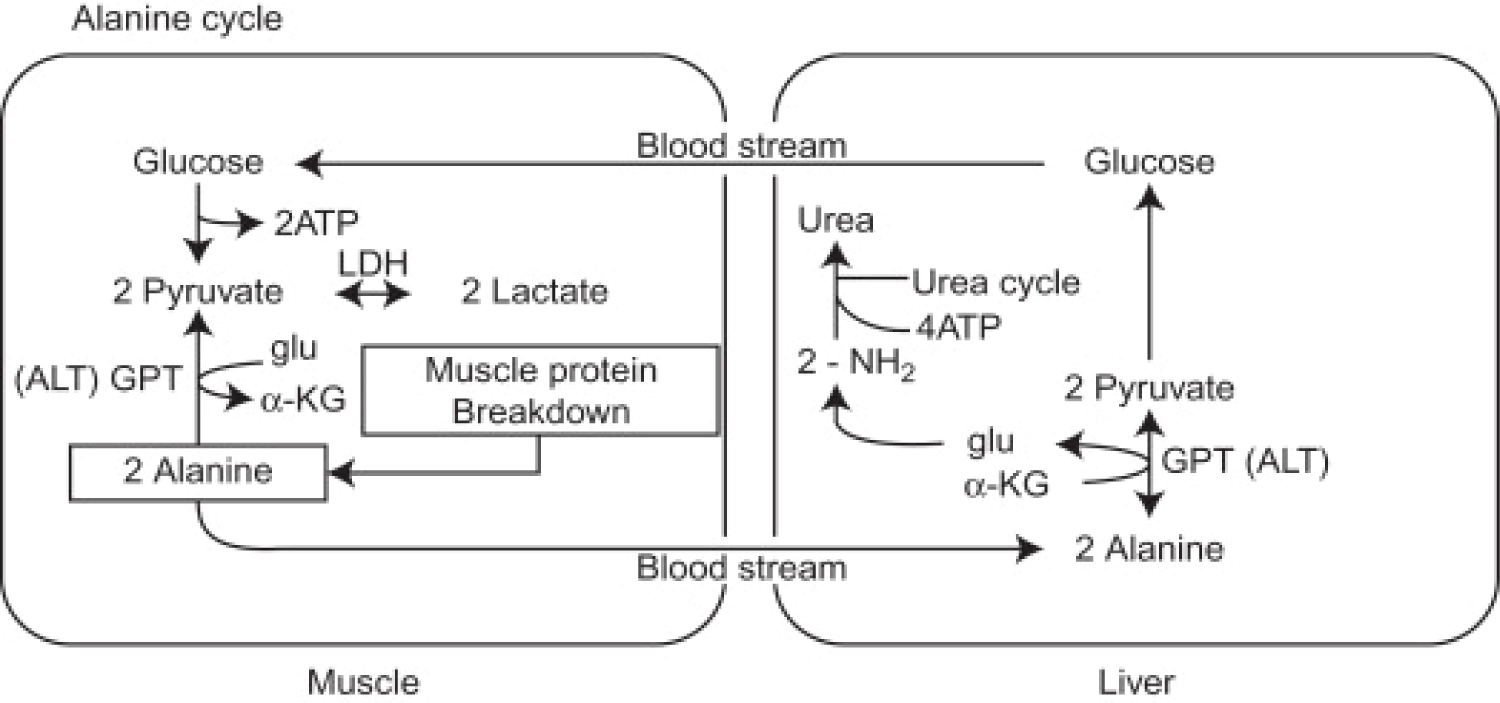 Figure 8: The Alanine Cycle. On the left are the reactions that take place in skeletal muscle. Alanine is a major product of the breakdown of skeletal muscle during fasting or other stresses. Also some other amino acid products from this breakdown can be converted to alanine. Alanine then accesses the bloodstream and is carried to the liver where, through the action of glutamate-pyruvate aminotransferase (ALT), it is converted to pyruvate. Pyruvate can then be converted to glucose through the gluconeogenic pathway. The amino group of alanine is converted to urea, by the urea cycle, and excreted. The glucose formed in the liver from Alanine May then enters the skeletal muscle again through the bloodstream and serves as an energy supply. Glu, glutamate; α-KG, alpha-ketoglutarate; GPT, glutamate-pyruvate aminotransferase (or ALT). Source: [114].
View Figure 8
Figure 8: The Alanine Cycle. On the left are the reactions that take place in skeletal muscle. Alanine is a major product of the breakdown of skeletal muscle during fasting or other stresses. Also some other amino acid products from this breakdown can be converted to alanine. Alanine then accesses the bloodstream and is carried to the liver where, through the action of glutamate-pyruvate aminotransferase (ALT), it is converted to pyruvate. Pyruvate can then be converted to glucose through the gluconeogenic pathway. The amino group of alanine is converted to urea, by the urea cycle, and excreted. The glucose formed in the liver from Alanine May then enters the skeletal muscle again through the bloodstream and serves as an energy supply. Glu, glutamate; α-KG, alpha-ketoglutarate; GPT, glutamate-pyruvate aminotransferase (or ALT). Source: [114].
View Figure 8
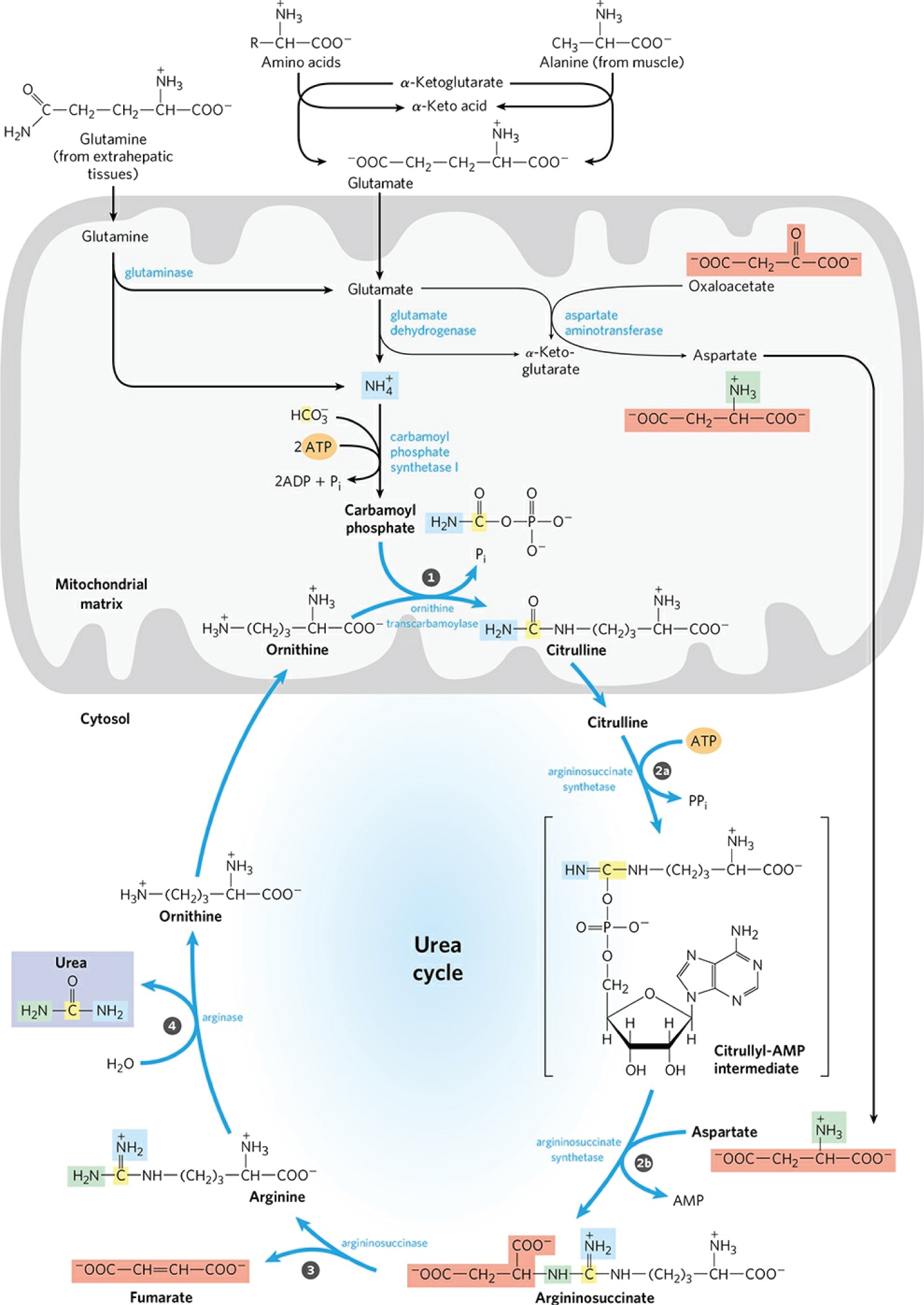 Figure 9: Urea synthesis is a five-step cyclic process with five distinct enzymes. The first two enzymes are present in mitochondria, while the others are located in the cytosol. Consequently, the urea cycle begins within the liver's mitochondria, but three of the subsequent steps take place in the cytosol; the cycle therefore encompasses two cell compartments. Urea has two amino groups (-NH2), one derived from NH3 in the mitochondrial matrix and the other from aspartate (generated in the mitochondria by transamination and transported to the cytosol). The carbon atom in urea is obtained from CO2 (as HCO3-) produced by mitochondrial respiration. A) Synthesis of carbamoyl phosphate: NH4+ generated in liver mitochondria is used, along with CO2 (as HCO3-) produced by cellular respiration, to form carbamoyl phosphate in the matrix. This ATP-dependent condensation reaction consumes two ATP and is irreversible and rate-limiting. It is catalyzed by a regulatory enzyme, carbamoyl phosphate synthetase I (CPS I), which requires N-acetyl glutamate for its activity. The mitochondrial form of CPS I is distinct from the cytosolic form (CPS II), which has a separate function in pyrimidine biosynthesis. CPS II accepts the amino group of glutamine and does not require N-acetyl glutamate for its activity. Carbamoyl phosphate, which functions as an activated carbamoyl group donor, now enters the urea cycle. B) Citrulline Formation: Carbamoyl phosphate donates its carbamoyl group to ornithine to form citrulline catalyzed by ornithine transcarbamoylase. Ornithine plays a role similar to that of oxaloacetate in the citric acid cycle. Ornithine and citrulline are basic amino acids. They are never found in the protein structure due to the lack of codons. The citrulline produced in this reaction is transported from the mitochondrial matrix to the cytosol by a transport system. C) Synthesis of Argininosuccinate: The second amino group of urea is incorporated in this reaction. Argininosuccinate synthetase condenses citrulline with aspartate to produce argininosuccinate. The condensation reaction takes place between the amino group of aspartate and the ureido (carbonyl) group of citrulline. D) A citrulyl-AMP intermediate is formed. This step requires ATP and it is cleaved into AMP and pyrophosphate (PPi). The latter is immediately decomposed to inorganic phosphate (Pi). E) Argininosuccinate Cleavage: Argininosuccinate is cleaved by argininosuccinase to give free Arginine and Fumarate, which is the only reversible step in the urea cycle. Arginine is the immediate precursor to urea. The fumarate thus formed enters the mitochondria to join the pool of TCA cycle intermediates that connect the TCA cycle, gluconeogenesis. Urea formation: In the last reaction of the urea cycle, the cytosolic enzyme arginase cleaves arginine to produce urea and ornithine. Arginase is activated by Co2+ and Mn2+. Ornithine, thus regenerated, is transported to the mitochondria to initiate another round of the urea cycle. The urea thus formed passes into the bloodstream and kidneys and is excreted in the urine. Ornithine and lysine compete with arginine (competitive inhibition). Arginase is found mainly in the liver, while the rest of the urea cycle enzymes are also present in other tissues. For this reason, arginine synthesis can occur to varying degrees in many tissues, but only the liver can produce urea. Source: [115].
View Figure 9
Figure 9: Urea synthesis is a five-step cyclic process with five distinct enzymes. The first two enzymes are present in mitochondria, while the others are located in the cytosol. Consequently, the urea cycle begins within the liver's mitochondria, but three of the subsequent steps take place in the cytosol; the cycle therefore encompasses two cell compartments. Urea has two amino groups (-NH2), one derived from NH3 in the mitochondrial matrix and the other from aspartate (generated in the mitochondria by transamination and transported to the cytosol). The carbon atom in urea is obtained from CO2 (as HCO3-) produced by mitochondrial respiration. A) Synthesis of carbamoyl phosphate: NH4+ generated in liver mitochondria is used, along with CO2 (as HCO3-) produced by cellular respiration, to form carbamoyl phosphate in the matrix. This ATP-dependent condensation reaction consumes two ATP and is irreversible and rate-limiting. It is catalyzed by a regulatory enzyme, carbamoyl phosphate synthetase I (CPS I), which requires N-acetyl glutamate for its activity. The mitochondrial form of CPS I is distinct from the cytosolic form (CPS II), which has a separate function in pyrimidine biosynthesis. CPS II accepts the amino group of glutamine and does not require N-acetyl glutamate for its activity. Carbamoyl phosphate, which functions as an activated carbamoyl group donor, now enters the urea cycle. B) Citrulline Formation: Carbamoyl phosphate donates its carbamoyl group to ornithine to form citrulline catalyzed by ornithine transcarbamoylase. Ornithine plays a role similar to that of oxaloacetate in the citric acid cycle. Ornithine and citrulline are basic amino acids. They are never found in the protein structure due to the lack of codons. The citrulline produced in this reaction is transported from the mitochondrial matrix to the cytosol by a transport system. C) Synthesis of Argininosuccinate: The second amino group of urea is incorporated in this reaction. Argininosuccinate synthetase condenses citrulline with aspartate to produce argininosuccinate. The condensation reaction takes place between the amino group of aspartate and the ureido (carbonyl) group of citrulline. D) A citrulyl-AMP intermediate is formed. This step requires ATP and it is cleaved into AMP and pyrophosphate (PPi). The latter is immediately decomposed to inorganic phosphate (Pi). E) Argininosuccinate Cleavage: Argininosuccinate is cleaved by argininosuccinase to give free Arginine and Fumarate, which is the only reversible step in the urea cycle. Arginine is the immediate precursor to urea. The fumarate thus formed enters the mitochondria to join the pool of TCA cycle intermediates that connect the TCA cycle, gluconeogenesis. Urea formation: In the last reaction of the urea cycle, the cytosolic enzyme arginase cleaves arginine to produce urea and ornithine. Arginase is activated by Co2+ and Mn2+. Ornithine, thus regenerated, is transported to the mitochondria to initiate another round of the urea cycle. The urea thus formed passes into the bloodstream and kidneys and is excreted in the urine. Ornithine and lysine compete with arginine (competitive inhibition). Arginase is found mainly in the liver, while the rest of the urea cycle enzymes are also present in other tissues. For this reason, arginine synthesis can occur to varying degrees in many tissues, but only the liver can produce urea. Source: [115].
View Figure 9
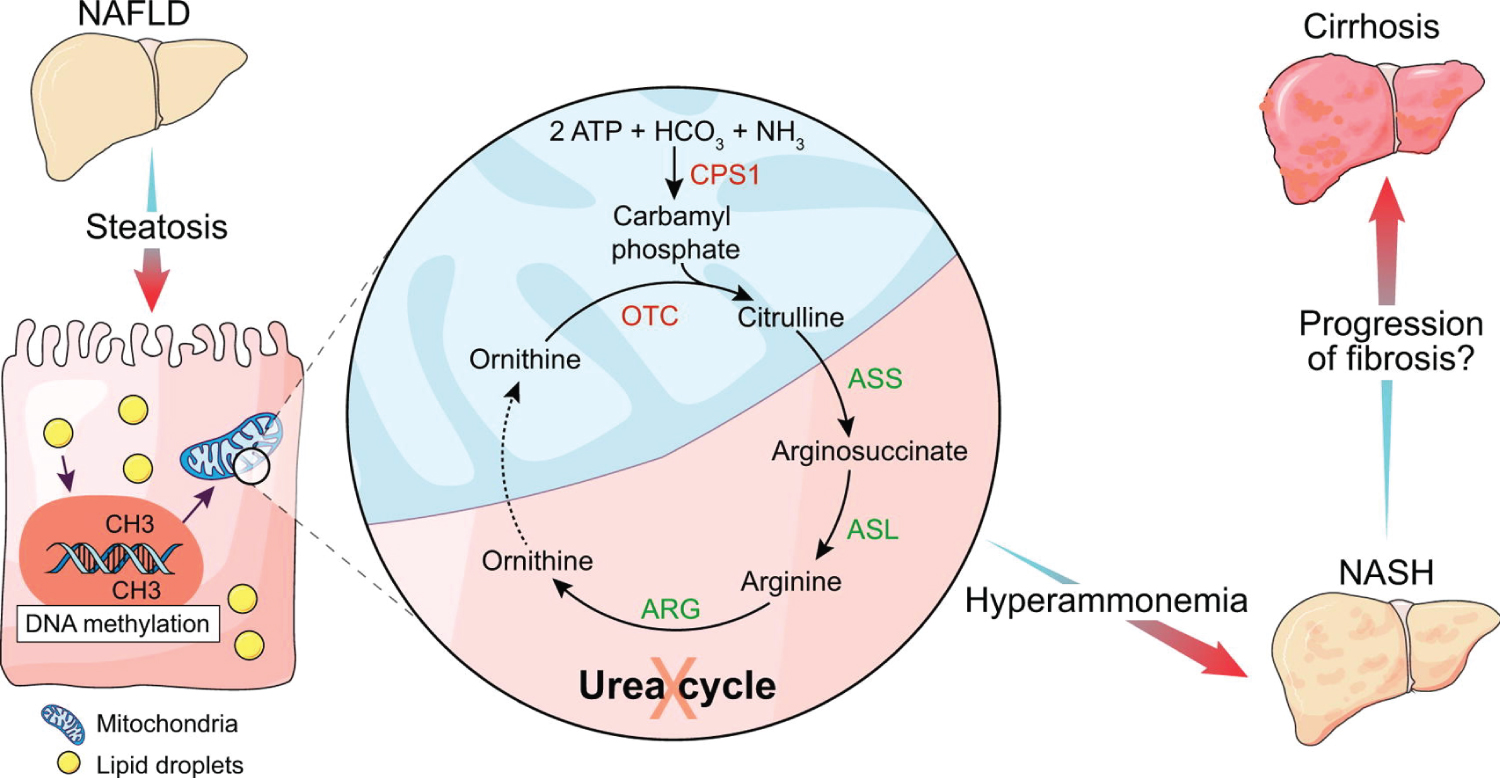 Figure 10: Experimental and human NASH is associated with reduced urea cycle enzyme expression. Hypermethylation of urea cycle enzymes is a potential underlying mechanism. The functional changes of urea synthesis in NASH are associated with hyperammonemia. Hyperammonemia can cause progression of liver injury and fibrosis. Ammonia is a potential novel target for prevention of progression of NASH. Severe disease in COVID-19 patients is prevalente in T2DM and obese ones. They show high azotemia due to Urea. It is important assessment of the seric ammonium facing neurologic phenomena. Source: [116].
View Figure 10
Figure 10: Experimental and human NASH is associated with reduced urea cycle enzyme expression. Hypermethylation of urea cycle enzymes is a potential underlying mechanism. The functional changes of urea synthesis in NASH are associated with hyperammonemia. Hyperammonemia can cause progression of liver injury and fibrosis. Ammonia is a potential novel target for prevention of progression of NASH. Severe disease in COVID-19 patients is prevalente in T2DM and obese ones. They show high azotemia due to Urea. It is important assessment of the seric ammonium facing neurologic phenomena. Source: [116].
View Figure 10
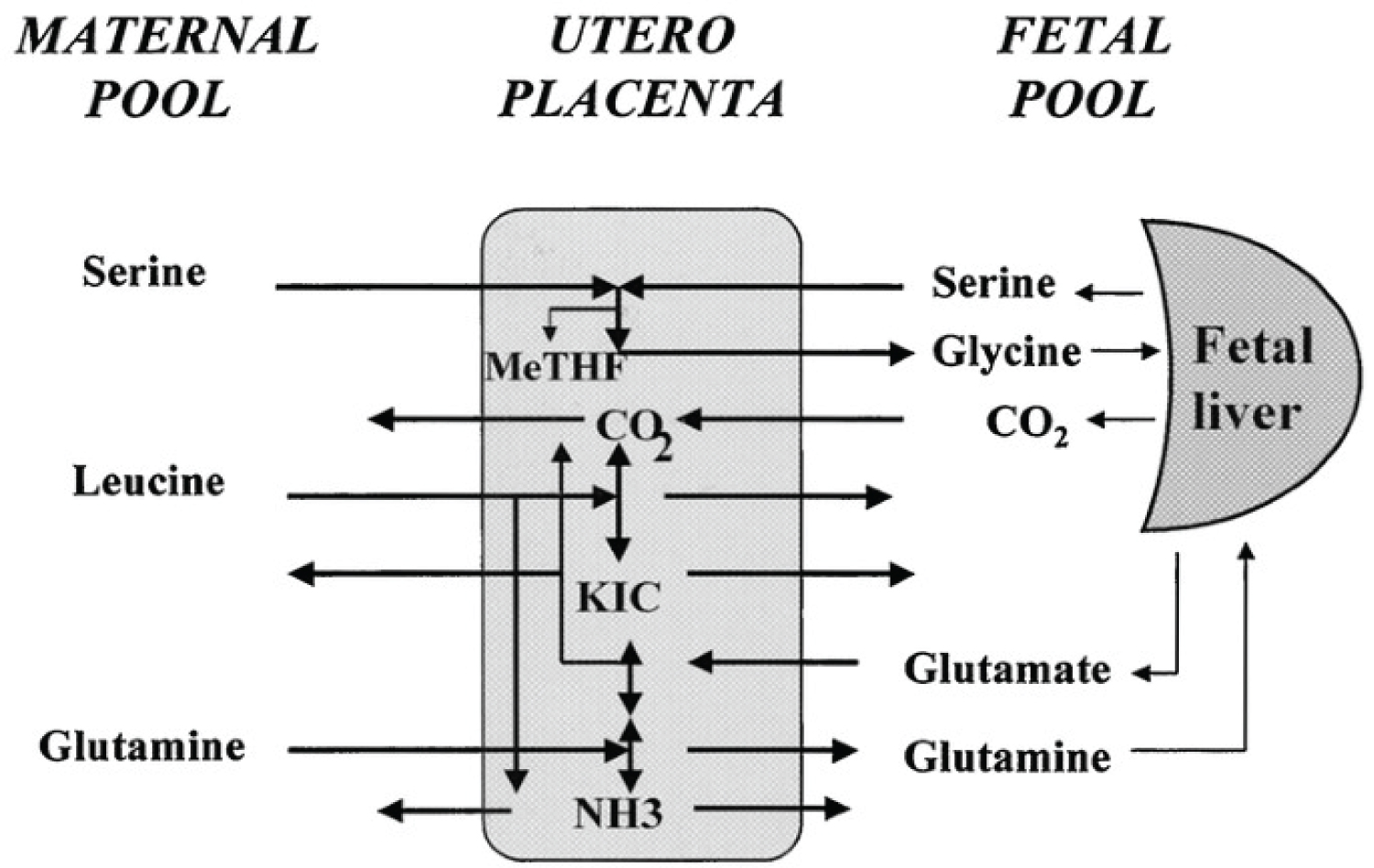 Figure 11: Diagram of amino acid placental exchanges between maternal and fetal plasma pools in chronically catheterized pregnant sheep. Serine is taken up by the placenta from both maternal and fetal circulations: Glycine is produced and released in the fetal circulation, and then taken up by fetal liver and partly converted to serine and CO2. Glutamine is taken up by the maternal circulation but also produced from glutamate within the placenta, released into the fetal circulation, and significantly converted to glutamate within the fetal liver. Leucine is also taken up by the maternal circulation, deaminated to KIC within the placenta, and then released into the fetal circulation together with KIC. Placental amino acid utilization leads to ammonia production.
View Figure 11
Figure 11: Diagram of amino acid placental exchanges between maternal and fetal plasma pools in chronically catheterized pregnant sheep. Serine is taken up by the placenta from both maternal and fetal circulations: Glycine is produced and released in the fetal circulation, and then taken up by fetal liver and partly converted to serine and CO2. Glutamine is taken up by the maternal circulation but also produced from glutamate within the placenta, released into the fetal circulation, and significantly converted to glutamate within the fetal liver. Leucine is also taken up by the maternal circulation, deaminated to KIC within the placenta, and then released into the fetal circulation together with KIC. Placental amino acid utilization leads to ammonia production.
View Figure 11
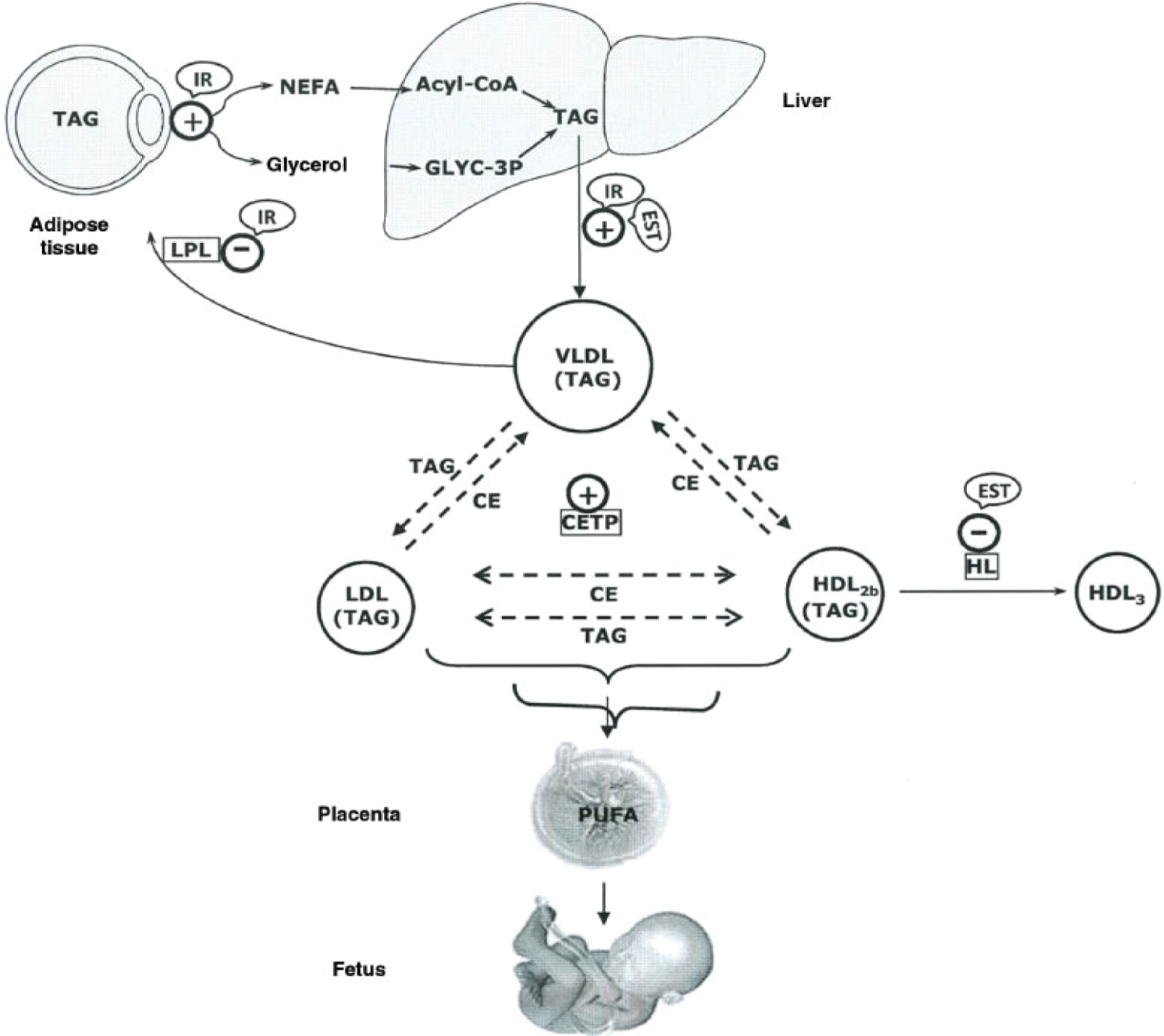 Figure 12: Major lipid metabolic interactions during late pregnancy, showing control by the insulin-resistant condition (IR) and enhanced estrogen levels (EST) in specific steps: +, activated steps; -, inhibited steps; NEFA, non-esterified fatty acid; Glyc-3P, glycerol-3P; TAG, triacylglyceride; CE, cholesterol ester; LPL, lipoprotein lipase; HL, hepatic lipase; PUFA, polyunsaturated fatty acid; CETP, cholesterol ester transfer protein. During late pregnancy, the net catabolic condition of maternal adipose tissue is associated with hyperlipidemia, which mainly corresponds to increases in TAG levels with smaller rises in phospholipids and cholesterol. The greatest increase in plasma TAGs corresponds to VLDLs as result of their increased production by the liver and decreased removal from the circulation as a result of decreased LPL activity in adipose tissue. Plasma lipoproteins of higher density than VLDLs, such as LDLs and HDLs that normally do not carry TAGs, also become enriched in TAGs during late pregnancy as result of an increase in cholesteryl ester transfer protein (CETP) activity and a decrease in hepatic lipase activity. This latter change also seems to be responsible for the proportional accumulation of TAGs in buoyant TAG-rich HDL2b subfractions at the expense of cholesterol-rich and TAG-poor HDL3. The increase in plasma estrogen levels during pregnancy probably contributes to these changes, because estrogens are known to stimulate hepatic VLDL production in pregnancy and to decrease hepatic lipase activity. It is proposed that the two main hormonal factors responsible for the metabolic interactions that result in the development of maternal hypertriglyceridemia during late pregnancy are the high lipolytic activity and low LPL activity in adipose tissue caused by the insulin-resistant condition and the increased liver secretion of VLDLs and decreased hepatic lipase activity caused by oestrogen. Source: [117].
View Figure 12
Figure 12: Major lipid metabolic interactions during late pregnancy, showing control by the insulin-resistant condition (IR) and enhanced estrogen levels (EST) in specific steps: +, activated steps; -, inhibited steps; NEFA, non-esterified fatty acid; Glyc-3P, glycerol-3P; TAG, triacylglyceride; CE, cholesterol ester; LPL, lipoprotein lipase; HL, hepatic lipase; PUFA, polyunsaturated fatty acid; CETP, cholesterol ester transfer protein. During late pregnancy, the net catabolic condition of maternal adipose tissue is associated with hyperlipidemia, which mainly corresponds to increases in TAG levels with smaller rises in phospholipids and cholesterol. The greatest increase in plasma TAGs corresponds to VLDLs as result of their increased production by the liver and decreased removal from the circulation as a result of decreased LPL activity in adipose tissue. Plasma lipoproteins of higher density than VLDLs, such as LDLs and HDLs that normally do not carry TAGs, also become enriched in TAGs during late pregnancy as result of an increase in cholesteryl ester transfer protein (CETP) activity and a decrease in hepatic lipase activity. This latter change also seems to be responsible for the proportional accumulation of TAGs in buoyant TAG-rich HDL2b subfractions at the expense of cholesterol-rich and TAG-poor HDL3. The increase in plasma estrogen levels during pregnancy probably contributes to these changes, because estrogens are known to stimulate hepatic VLDL production in pregnancy and to decrease hepatic lipase activity. It is proposed that the two main hormonal factors responsible for the metabolic interactions that result in the development of maternal hypertriglyceridemia during late pregnancy are the high lipolytic activity and low LPL activity in adipose tissue caused by the insulin-resistant condition and the increased liver secretion of VLDLs and decreased hepatic lipase activity caused by oestrogen. Source: [117].
View Figure 12
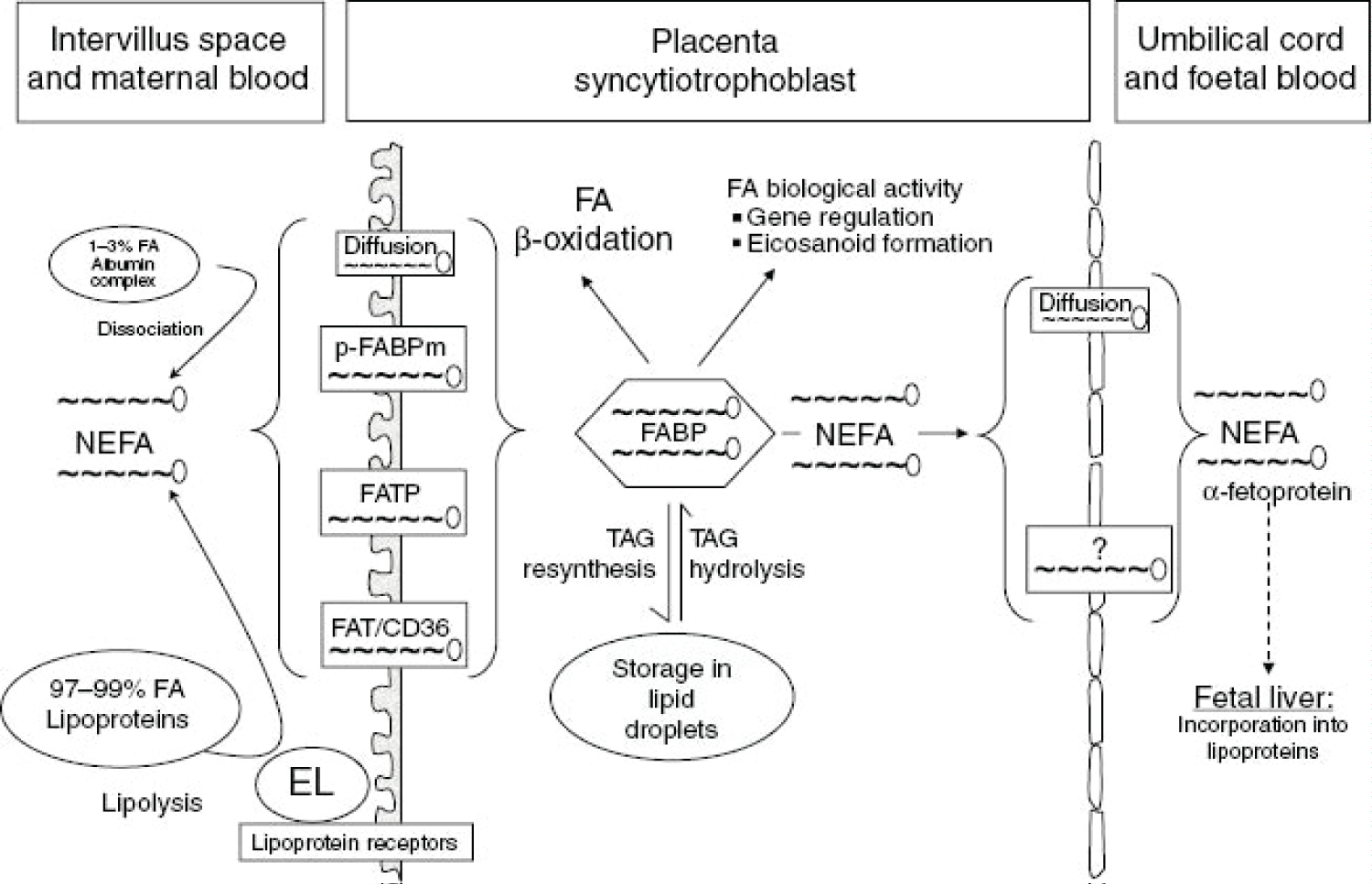 Figure 13: Current concept of maternal-to-fetal transfer of fatty acids (FA).NEFA, non-esterified fatty acid; TAG, triacylglyceride, p-FABPm, plasma membrane fatty acid binding protein; FATP, fatty acid transport protein; FAT/CD36, fatty acid translocase; EL, endothelial lipase; FABP, fatty acid binding protein. depicts current understanding of maternal-to-fetal transfer of fatty acids. About 1%-3% of the fatty acids in the maternal circulation are non-esterified (i.e. the vast majority are in lipoproteins). LDL is the major lipoprotein in the maternal circulation and can be taken up into the syncytiotrophoblast (the placental tissue interfacing with maternal blood) by receptor-mediated endocytosis. After uptake, the LDL-TAG and LDL-cholesterol esters can become hydrolyzed by intracellular lipases and cholesterol-ester hydrolases and contribute to the intracellular fatty acid pool. Other lipoproteins such as VLDLs and HDLs bind to receptors on the syncytiotrophoblast surface and become hydrolyzed extracellularly. Endothelial lipase has been identified as the major extracellular placental lipase, but this is not a uniform finding and there are claims about the presence of LPL. Regardless of the molecular identity of the TAG hydrolase activity, the resulting NEFAs must be taken up by the syncytiotrophoblast to eventually contribute to the intracellular fatty acid pool. Uptake is accomplished by fatty acid transporters such as fatty acid transport proteins (FATB)-1, FATB-4, fatty acid translocase (FAT/CD36), and plasma membrane fatty acid binding protein (p-FABPm). These transporters have overlapping specificities for the various fatty acids, and their relative contribution to the uptake of distinct fatty acids is unknown. Once inside the syncytiotrophoblast, the fatty acids must be bound to fatty acid binding proteins (FABP), of which the heart-type isoform appears to be the major FABP in the human placenta. From there, fatty acids can enter the various metabolic routes, including β-oxidation, conversion into eicosanoids, and re-esterification to form phospholipids and triglycerides. The latter may then be stored in lipid droplets in the syncytiotrophoblast. NEFAs can traverse the cytoplasm of the syncytiotrophoblast and are then released into the fetal circulation, where they bind to transport proteins such as α-fetoprotein. They are taken up in the fetal liver, esterified and metabolized or used to form lipoproteins, mostly HDL, which is the main cholesterol-carrying fetal lipoprotein in humans. The various fatty acids contribute differentially to fetal lipid pools (i.e. NEFA, TAG, phospholipids - mainly phosphatidylcholine, and cholesterol-esters). Interestingly, the human placenta has a preference for the transport of docosahexaenoic acid (DHA) over arachidonic acid (AA), α-linolenic acid (ALA), and linoleic acid (LA). This could explain the enrichment of LCPUFAs in fetal plasma and in particular of DHA and also reflect the high demand of the growing fetus for DHA, particularly to sustain brain and retinal development. Source: [117].
View Figure 13
Figure 13: Current concept of maternal-to-fetal transfer of fatty acids (FA).NEFA, non-esterified fatty acid; TAG, triacylglyceride, p-FABPm, plasma membrane fatty acid binding protein; FATP, fatty acid transport protein; FAT/CD36, fatty acid translocase; EL, endothelial lipase; FABP, fatty acid binding protein. depicts current understanding of maternal-to-fetal transfer of fatty acids. About 1%-3% of the fatty acids in the maternal circulation are non-esterified (i.e. the vast majority are in lipoproteins). LDL is the major lipoprotein in the maternal circulation and can be taken up into the syncytiotrophoblast (the placental tissue interfacing with maternal blood) by receptor-mediated endocytosis. After uptake, the LDL-TAG and LDL-cholesterol esters can become hydrolyzed by intracellular lipases and cholesterol-ester hydrolases and contribute to the intracellular fatty acid pool. Other lipoproteins such as VLDLs and HDLs bind to receptors on the syncytiotrophoblast surface and become hydrolyzed extracellularly. Endothelial lipase has been identified as the major extracellular placental lipase, but this is not a uniform finding and there are claims about the presence of LPL. Regardless of the molecular identity of the TAG hydrolase activity, the resulting NEFAs must be taken up by the syncytiotrophoblast to eventually contribute to the intracellular fatty acid pool. Uptake is accomplished by fatty acid transporters such as fatty acid transport proteins (FATB)-1, FATB-4, fatty acid translocase (FAT/CD36), and plasma membrane fatty acid binding protein (p-FABPm). These transporters have overlapping specificities for the various fatty acids, and their relative contribution to the uptake of distinct fatty acids is unknown. Once inside the syncytiotrophoblast, the fatty acids must be bound to fatty acid binding proteins (FABP), of which the heart-type isoform appears to be the major FABP in the human placenta. From there, fatty acids can enter the various metabolic routes, including β-oxidation, conversion into eicosanoids, and re-esterification to form phospholipids and triglycerides. The latter may then be stored in lipid droplets in the syncytiotrophoblast. NEFAs can traverse the cytoplasm of the syncytiotrophoblast and are then released into the fetal circulation, where they bind to transport proteins such as α-fetoprotein. They are taken up in the fetal liver, esterified and metabolized or used to form lipoproteins, mostly HDL, which is the main cholesterol-carrying fetal lipoprotein in humans. The various fatty acids contribute differentially to fetal lipid pools (i.e. NEFA, TAG, phospholipids - mainly phosphatidylcholine, and cholesterol-esters). Interestingly, the human placenta has a preference for the transport of docosahexaenoic acid (DHA) over arachidonic acid (AA), α-linolenic acid (ALA), and linoleic acid (LA). This could explain the enrichment of LCPUFAs in fetal plasma and in particular of DHA and also reflect the high demand of the growing fetus for DHA, particularly to sustain brain and retinal development. Source: [117].
View Figure 13
Once the virus infects the human cell, there is a trace of the internalisation of ACE-2 and for each person. The time for this protein to be expressed again is variable and very individual.
The materno-foetal interface presents syncytial cells with many Furins and Cathepsins, molecules that act under acidic pH. SARS-CoV-2 needs these proteins to be able to perform better during the infection. Extreme hypoxia can lead to syncytium formation in pneumocytes II, as cathepsins and furins are induced by hypoxia. Perhaps this fact justifies the formation of syncytium in the lungs. This hypothesis raises an essential issue in the management of the COVID-19 patient, as there is permissiveness in the face of higher pCO2, conduct called permissive hypercapnia, however allowing the acidic and hypoxemic environment causes more cathepsins and furins to be expressed in a way that facilitates viral replication and protein processing. Based on this idea, the behaviour of permissive hypercapnia should be proscribed, and mechanical ventilation should be better managed, including the intravenous administration of 8.4% sodium bicarbonate, despite the current protocols.
COVID-19 provides a deficient cellular environment for aerobiosis. The internalisation of ACE-2 in enterocytes causes fewer amino acids to be absorbed, and among the most important for the proposed pathophysiology, we have Tryptophane (Try) and Phenylalanine (Phe). The Try deficit will cause a deficit of NAD/NADH+, of serotonin (5-HT) presenting a shift in metabolism for the production of Kynurenine and its derivatives, many of them toxic to the central nervous system, which explains the difficulty in sedating patients [77-79]. Try during inflammatory processes has been metabolised by IDO-1 [80-82], produced mainly by dendritic cells, macrophages and monocytes, mainly when stimulated by IFN-gamma. In physiological normality, Try is metabolised by the hepatic enzyme TDO. With the switch to IDO-1, the Kynurenine pathway is prioritised (Figure 14, Figure 15, Figure 16 and Figure 17).
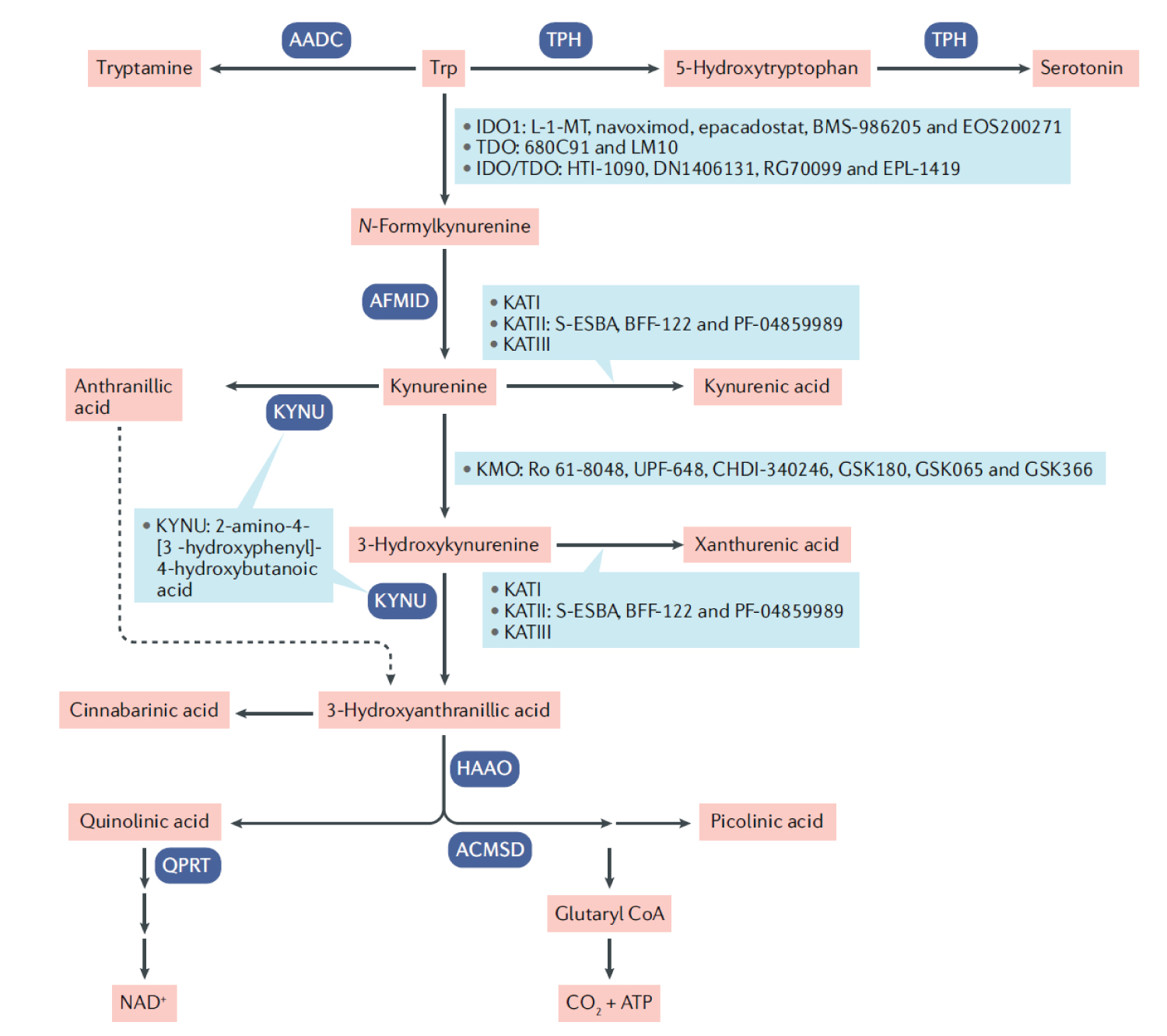 Figure 14: Tryptophan catabolism, key therapeutic targets and drugs in development. A small fraction of free l-tryptophan (Try) is used for protein synthesis and the production of neurotransmitters such as serotonin and neuromodulators such as tryptamine. However, over 95% of free Try is a substrate for the kynurenine (Kyn) pathway (KP) of Try degradation, which generates several metabolites. Drugs in development and their therapeutic targets within the KP are shown in blue boxes. The tissue-specific expression of enzymes in the kynurenine (Kyn) pathway (KP) is best studied for tryptophan-2,3-dioxygenase (TDO) and indoleamine-2,3-dioxygenase 1 (IDO1). In humans, TDO and IDO1 are localized in different cells and tissues and are used in different physiological processes. TDO. The TDO enzyme is expressed in liver, bone marrow, the immune system, muscle, gastrointestinal tract, kidney and urinary bladder and brain258, and expression levels are regulated by systemic levels of l-tryptophan (Try) and corticosteroids, which change in response to stress or food; for example, in mice, starvation results in a dramatic decrease in TDO expression. In addition, TDO is induced in glioma cells and in neurons by prostaglandins. TDO activity balances the total amount of Trp in the body and is thought to regulate a behavioural response to food availability by initiating the production of bioactive KP metabolites. Such behavioural regulation by the KP has been demonstrated in Caenorhabditis elegans, in which the availability of food, as measured by trp, increases the levels of kynurenic acid (Ka). Ka then increases the foraging behaviour via evolutionary conserved neuronal N-methyl-d-aspartate (NMDa) receptors until the trp balance is restored. Fluctuations in levels of KP metabolites in the human brain may similarly regulate behavioural responses, which may be impaired under pathological conditions and lead to behavioural alterations. IDO1. The IDO1 enzyme is expressed in most tissues at low levels, including cells of the central nervous system (CNs) and macrophages, but not in the liver. The expression and activity of IDO1 in the immune compartment is tightly regulated. It is highly expressed in inflamed tissues, with its expression induced (mainly in myeloid cells) by the pro-inflammatory cytokine iFNγ, interleukin-6 (iL-6) and toll-like receptor (tLr) ligands. IDO1 expression can also be induced through interaction with T cells. The immune checkpoint cytotoxic t lymphocyte-associated protein 4 (CtLa4), which is expressed on regulatory T cell (Treg) cells, stimulates IDO1 expression in dendritic cells (DCs) through outside-in signalling of the co-stimulatory molecules CD80 and CD86. This activation is tightly controlled by suppressor of cytokine signalling 3 (SOCS3), which targets IDO1 for proteasomal degradation. Thus, the degradation of IDO1 appears to represent a default programme to ensure full stimulation of an antigen specific T cell response197. IDO1 is thus an integral part of an immunoregulatory network controlling T cell activation. Here, Treg cells play an important role as both inducers and targets of IDO1-mediated Trp metabolism134. IDO1 is also constitutively expressed in the placenta and epididymis, where it has been shown to maintain immune privilege by suppressing T cell responses. Particularly in the lung, elevated levels of IDO1 are thought to prevent the growth of Try-dependent intracellular pathogens by depleting local Try. Endothelial IDO1 expression in response to pro-inflammatory stimuli is thought to be involved in the regulation of vascular tone. IDO2. the IDO2 gene is located immediately downstream of the IDO1 gene and is believed to possess a more ancestral function. IDO2 mRNA expression is more restricted than IDO1; it is constitutively expressed in human liver, brain, thyroid, placenta, endometrium and testis but can be induced in antigen-presenting cells (aPCs) and B cells14. IDO2 expression is regulated in APCs and B cells by Kyn binding to the aryl hydrocarbon receptor (AHR). This observation suggests that IDO2 may be part of a cellular restricted feed forward loop of Trp metabolism induced by IDO1. Downstream enzymes. The other enzymes in the KP have been mostly studied in the CNS. Kynurenine monooxygenase (KMO) and kynurenine aminotransferases (KATI-KATIII) are expressed in the brain and induced in several pathological conditions, including traumatic injury267, viral infection268 and neurodegeneration2 L-1-MT, 1-methyl-l-Trp; AADC, aromatic-l-amino acid decarboxylase; ACMSD, α-amino-β-carboxymuconate-ε-semialdehyde decarboxylase; AFMID, kynurenine formamidase; HAAO, 3-hydroxyanthranilate 3,4-dioxygenase; IDO, indoleamine-2,3-dioxygenase; KATs, kynurenine amino transferases I-III; KMO, kynurenine 3-monooxygenase; KYNU, kynureninase; QPRT, quinolinic acid phosphoribosyl transferase; TDO, tryptophan-2,3-dioxygenase; TPH, tryptophan hydroxylase. Source: [118].
View Figure 14
Figure 14: Tryptophan catabolism, key therapeutic targets and drugs in development. A small fraction of free l-tryptophan (Try) is used for protein synthesis and the production of neurotransmitters such as serotonin and neuromodulators such as tryptamine. However, over 95% of free Try is a substrate for the kynurenine (Kyn) pathway (KP) of Try degradation, which generates several metabolites. Drugs in development and their therapeutic targets within the KP are shown in blue boxes. The tissue-specific expression of enzymes in the kynurenine (Kyn) pathway (KP) is best studied for tryptophan-2,3-dioxygenase (TDO) and indoleamine-2,3-dioxygenase 1 (IDO1). In humans, TDO and IDO1 are localized in different cells and tissues and are used in different physiological processes. TDO. The TDO enzyme is expressed in liver, bone marrow, the immune system, muscle, gastrointestinal tract, kidney and urinary bladder and brain258, and expression levels are regulated by systemic levels of l-tryptophan (Try) and corticosteroids, which change in response to stress or food; for example, in mice, starvation results in a dramatic decrease in TDO expression. In addition, TDO is induced in glioma cells and in neurons by prostaglandins. TDO activity balances the total amount of Trp in the body and is thought to regulate a behavioural response to food availability by initiating the production of bioactive KP metabolites. Such behavioural regulation by the KP has been demonstrated in Caenorhabditis elegans, in which the availability of food, as measured by trp, increases the levels of kynurenic acid (Ka). Ka then increases the foraging behaviour via evolutionary conserved neuronal N-methyl-d-aspartate (NMDa) receptors until the trp balance is restored. Fluctuations in levels of KP metabolites in the human brain may similarly regulate behavioural responses, which may be impaired under pathological conditions and lead to behavioural alterations. IDO1. The IDO1 enzyme is expressed in most tissues at low levels, including cells of the central nervous system (CNs) and macrophages, but not in the liver. The expression and activity of IDO1 in the immune compartment is tightly regulated. It is highly expressed in inflamed tissues, with its expression induced (mainly in myeloid cells) by the pro-inflammatory cytokine iFNγ, interleukin-6 (iL-6) and toll-like receptor (tLr) ligands. IDO1 expression can also be induced through interaction with T cells. The immune checkpoint cytotoxic t lymphocyte-associated protein 4 (CtLa4), which is expressed on regulatory T cell (Treg) cells, stimulates IDO1 expression in dendritic cells (DCs) through outside-in signalling of the co-stimulatory molecules CD80 and CD86. This activation is tightly controlled by suppressor of cytokine signalling 3 (SOCS3), which targets IDO1 for proteasomal degradation. Thus, the degradation of IDO1 appears to represent a default programme to ensure full stimulation of an antigen specific T cell response197. IDO1 is thus an integral part of an immunoregulatory network controlling T cell activation. Here, Treg cells play an important role as both inducers and targets of IDO1-mediated Trp metabolism134. IDO1 is also constitutively expressed in the placenta and epididymis, where it has been shown to maintain immune privilege by suppressing T cell responses. Particularly in the lung, elevated levels of IDO1 are thought to prevent the growth of Try-dependent intracellular pathogens by depleting local Try. Endothelial IDO1 expression in response to pro-inflammatory stimuli is thought to be involved in the regulation of vascular tone. IDO2. the IDO2 gene is located immediately downstream of the IDO1 gene and is believed to possess a more ancestral function. IDO2 mRNA expression is more restricted than IDO1; it is constitutively expressed in human liver, brain, thyroid, placenta, endometrium and testis but can be induced in antigen-presenting cells (aPCs) and B cells14. IDO2 expression is regulated in APCs and B cells by Kyn binding to the aryl hydrocarbon receptor (AHR). This observation suggests that IDO2 may be part of a cellular restricted feed forward loop of Trp metabolism induced by IDO1. Downstream enzymes. The other enzymes in the KP have been mostly studied in the CNS. Kynurenine monooxygenase (KMO) and kynurenine aminotransferases (KATI-KATIII) are expressed in the brain and induced in several pathological conditions, including traumatic injury267, viral infection268 and neurodegeneration2 L-1-MT, 1-methyl-l-Trp; AADC, aromatic-l-amino acid decarboxylase; ACMSD, α-amino-β-carboxymuconate-ε-semialdehyde decarboxylase; AFMID, kynurenine formamidase; HAAO, 3-hydroxyanthranilate 3,4-dioxygenase; IDO, indoleamine-2,3-dioxygenase; KATs, kynurenine amino transferases I-III; KMO, kynurenine 3-monooxygenase; KYNU, kynureninase; QPRT, quinolinic acid phosphoribosyl transferase; TDO, tryptophan-2,3-dioxygenase; TPH, tryptophan hydroxylase. Source: [118].
View Figure 14
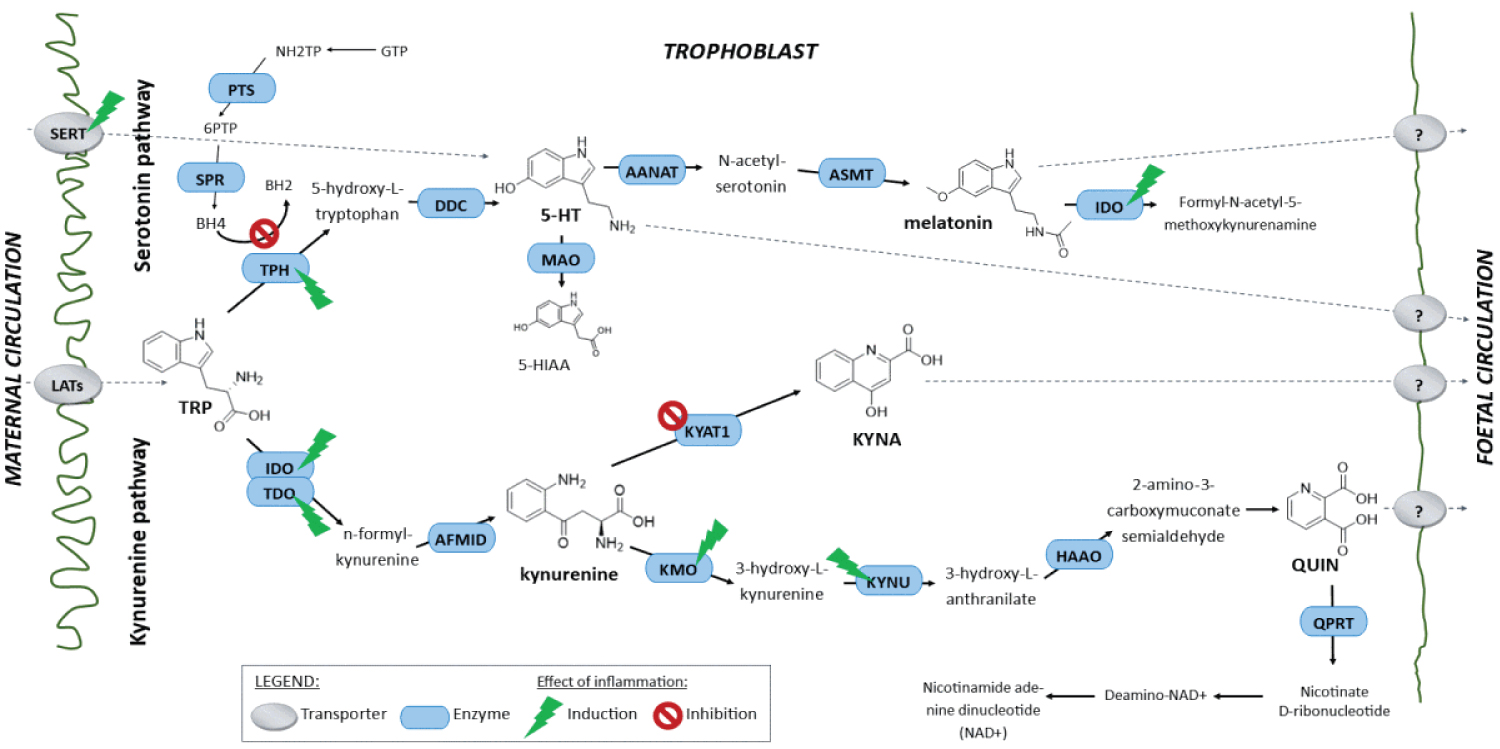 Figure 15: Schematic depiction of serotonin and kynurenine pathways of tryptophan metabolism in the placenta and effects of inflammation on induction or inhibition of rate-limiting transporters and enzymes. Important metabolites are also shown. Although these metabolites are known to be produced in the adult brain, liver, and monocytes, their production in the syncytiotrophoblast has not been fully described and their transport from the placenta to foetal circulation has not been elucidated. The serotonin pathway: Serotonin (5-HT), an essential trophic factor for foetal brain development, was previously believed to cross the placenta from mother to foetus. However, recent work suggests that maternal 5-HT is largely degraded by placental monoamine oxidases, and the placenta serves as a transient source of 5-HT for the foetus, synthesizing it from maternal TRP. The rate-limiting enzyme of the 5-HT pathway is TRP hydroxylase (TPH), identified in both human and murine placenta. Placenta-derived 5‑HT is then released into the foetal circulation and plays a crucial role in foetal brain development. 5-HT in the placenta may be further transformed into melatonin, which is associated with foetal circadian rhythmicity. Since the placenta is an essential source of 5-HT for the foetal brain, perturbations in placental TRP metabolism may disrupt 5-HT signalling in specific regions of the developing foetal brain and lead to abnormal programming of the major axonal pathways. The kynurenine pathway: The kynurenine pathway in the placenta has received great attention in recent years following the discovery that its products play important roles in health and disease, particularly conditions associated with immune dysfunction and central nervous system disorders. In placenta, the pathway also putatively plays crucial roles in preventing foetal rejection by the maternal immune system. However, intermediates in this pathway include several potentially neuroprotective (kynurenic acid) or neurotoxic metabolites (quinolinic acid). Both 5-HT and KYN pathways are modulated by interferon gamma (IFN-γ) and pro-inflammatory cytokines, which influence the expression and activities (inter alia) of rate-limiting transporters and enzymes such as serotonin transporter (SERT), tryptophan hydroxylase (TPH), indoleamine 2,3-dioxygenase (IDO), and tryptophan 2,3-dioxygenase (TDO). Therefore, both metabolic pathways of TRP are modulated during inflammatory insults.KYN is further metabolized by either KYN amino transferase (KYAT1) or KYN monooxygenase (KMO), which respectively generate neuroprotective kynurenic acid (KYNA) or neurotoxic quinolinic acid (QUIN). Inflammation shifts the KYNA/QUIN balance in favour of neurotoxic QUIN, with potentially deleterious effects on the developing foetus. Accordingly, increased levels of QUIN in umbilical blood have been detected in pregnant women with intrauterine infection. Several other studies have demonstrated increases in TRP metabolism via the KYN pathway in the placenta of women with intrauterine infections and related them to increases in risks of autism spectrum disorders associated with prenatal infection. However, further tests of these hypotheses with various experimental approaches are required, particularly confirmatory tests of the transport of KYNA and QUIN from the placenta to the foetal circulation, which has not been elucidated. Source: [119].
View Figure 15
Figure 15: Schematic depiction of serotonin and kynurenine pathways of tryptophan metabolism in the placenta and effects of inflammation on induction or inhibition of rate-limiting transporters and enzymes. Important metabolites are also shown. Although these metabolites are known to be produced in the adult brain, liver, and monocytes, their production in the syncytiotrophoblast has not been fully described and their transport from the placenta to foetal circulation has not been elucidated. The serotonin pathway: Serotonin (5-HT), an essential trophic factor for foetal brain development, was previously believed to cross the placenta from mother to foetus. However, recent work suggests that maternal 5-HT is largely degraded by placental monoamine oxidases, and the placenta serves as a transient source of 5-HT for the foetus, synthesizing it from maternal TRP. The rate-limiting enzyme of the 5-HT pathway is TRP hydroxylase (TPH), identified in both human and murine placenta. Placenta-derived 5‑HT is then released into the foetal circulation and plays a crucial role in foetal brain development. 5-HT in the placenta may be further transformed into melatonin, which is associated with foetal circadian rhythmicity. Since the placenta is an essential source of 5-HT for the foetal brain, perturbations in placental TRP metabolism may disrupt 5-HT signalling in specific regions of the developing foetal brain and lead to abnormal programming of the major axonal pathways. The kynurenine pathway: The kynurenine pathway in the placenta has received great attention in recent years following the discovery that its products play important roles in health and disease, particularly conditions associated with immune dysfunction and central nervous system disorders. In placenta, the pathway also putatively plays crucial roles in preventing foetal rejection by the maternal immune system. However, intermediates in this pathway include several potentially neuroprotective (kynurenic acid) or neurotoxic metabolites (quinolinic acid). Both 5-HT and KYN pathways are modulated by interferon gamma (IFN-γ) and pro-inflammatory cytokines, which influence the expression and activities (inter alia) of rate-limiting transporters and enzymes such as serotonin transporter (SERT), tryptophan hydroxylase (TPH), indoleamine 2,3-dioxygenase (IDO), and tryptophan 2,3-dioxygenase (TDO). Therefore, both metabolic pathways of TRP are modulated during inflammatory insults.KYN is further metabolized by either KYN amino transferase (KYAT1) or KYN monooxygenase (KMO), which respectively generate neuroprotective kynurenic acid (KYNA) or neurotoxic quinolinic acid (QUIN). Inflammation shifts the KYNA/QUIN balance in favour of neurotoxic QUIN, with potentially deleterious effects on the developing foetus. Accordingly, increased levels of QUIN in umbilical blood have been detected in pregnant women with intrauterine infection. Several other studies have demonstrated increases in TRP metabolism via the KYN pathway in the placenta of women with intrauterine infections and related them to increases in risks of autism spectrum disorders associated with prenatal infection. However, further tests of these hypotheses with various experimental approaches are required, particularly confirmatory tests of the transport of KYNA and QUIN from the placenta to the foetal circulation, which has not been elucidated. Source: [119].
View Figure 15
 Figure 16: Placental nutrient transport/handling and foetal protection against xenobiotics. A) STB expresses many transporters in the MVM and BM for vertical transport of nutrients across the placenta. It is also equipped with biotransformation enzymes capable of metabolizing and using nutrients for placental needs. Active metabolites can also be produced in the STB and delivered to the foetus across BM. AAs and glucose are the primary stimuli for secretion of foetal insulin; therefore, any dysregulation of AAs or glucose transport across the placenta appears to result in IUGR or LGA babies. Optimal maternal-fetal disposition of nutrients is thus critical through the whole period of pregnancy. B) Efflux transporters, mainly P-gp and BCRP, are functionally expressed on MVM. They can recognize hundreds of substrates such as toxins or drugs and direct them back to maternal circulation, thus providing protection to the fetus. Although the expression of several biotransformation enzymes (both phase I and phase II) has been detected at protein and/or transcript level in STB, placental metabolism of xenobiotics is relatively insignificant and does not present a barrier for their passage across the placenta. C) Placental nutrient transport is regulated by signals of maternal (blue), placental (red) and foetal (yellow) origin, acting in a direct (straight lines) or indirect (dotted lines) manner. To date, three main signalling pathways within the STB have been described to integrate various factors and regulate placental nutrient transport: while mTOR has been well confirmed to constitute a positive AA transport regulator, placental AA and fatty acid transport has also been shown to be regulated through a STAT3- and PPARγ-dependent mechanisms. Abbreviations: AAs - amino acids; BCRP - breast cancer resistance protein; BM - basal membrane; FABPs - fatty acid binding proteins; FATPs - fatty acid transport proteins; FFAs - free fatty acids; GLUTs - glucose transporters; LAT - L-type amino acid transporter; LIP - lipase; mTOR - mammalian target of rapamycin; MVM - microvillus membrane; NTs - nucleoside transporters; P-gp - p-glycoprotein; PPAR - peroxisome proliferator-activated receptor; SNAT - A-type amino acid transporter; STAT - signal transducer and activator of transcription protein; STB - syncytiotrophoblast; TAG - triacylglycerol. Source: [3].
View Figure 16
Figure 16: Placental nutrient transport/handling and foetal protection against xenobiotics. A) STB expresses many transporters in the MVM and BM for vertical transport of nutrients across the placenta. It is also equipped with biotransformation enzymes capable of metabolizing and using nutrients for placental needs. Active metabolites can also be produced in the STB and delivered to the foetus across BM. AAs and glucose are the primary stimuli for secretion of foetal insulin; therefore, any dysregulation of AAs or glucose transport across the placenta appears to result in IUGR or LGA babies. Optimal maternal-fetal disposition of nutrients is thus critical through the whole period of pregnancy. B) Efflux transporters, mainly P-gp and BCRP, are functionally expressed on MVM. They can recognize hundreds of substrates such as toxins or drugs and direct them back to maternal circulation, thus providing protection to the fetus. Although the expression of several biotransformation enzymes (both phase I and phase II) has been detected at protein and/or transcript level in STB, placental metabolism of xenobiotics is relatively insignificant and does not present a barrier for their passage across the placenta. C) Placental nutrient transport is regulated by signals of maternal (blue), placental (red) and foetal (yellow) origin, acting in a direct (straight lines) or indirect (dotted lines) manner. To date, three main signalling pathways within the STB have been described to integrate various factors and regulate placental nutrient transport: while mTOR has been well confirmed to constitute a positive AA transport regulator, placental AA and fatty acid transport has also been shown to be regulated through a STAT3- and PPARγ-dependent mechanisms. Abbreviations: AAs - amino acids; BCRP - breast cancer resistance protein; BM - basal membrane; FABPs - fatty acid binding proteins; FATPs - fatty acid transport proteins; FFAs - free fatty acids; GLUTs - glucose transporters; LAT - L-type amino acid transporter; LIP - lipase; mTOR - mammalian target of rapamycin; MVM - microvillus membrane; NTs - nucleoside transporters; P-gp - p-glycoprotein; PPAR - peroxisome proliferator-activated receptor; SNAT - A-type amino acid transporter; STAT - signal transducer and activator of transcription protein; STB - syncytiotrophoblast; TAG - triacylglycerol. Source: [3].
View Figure 16
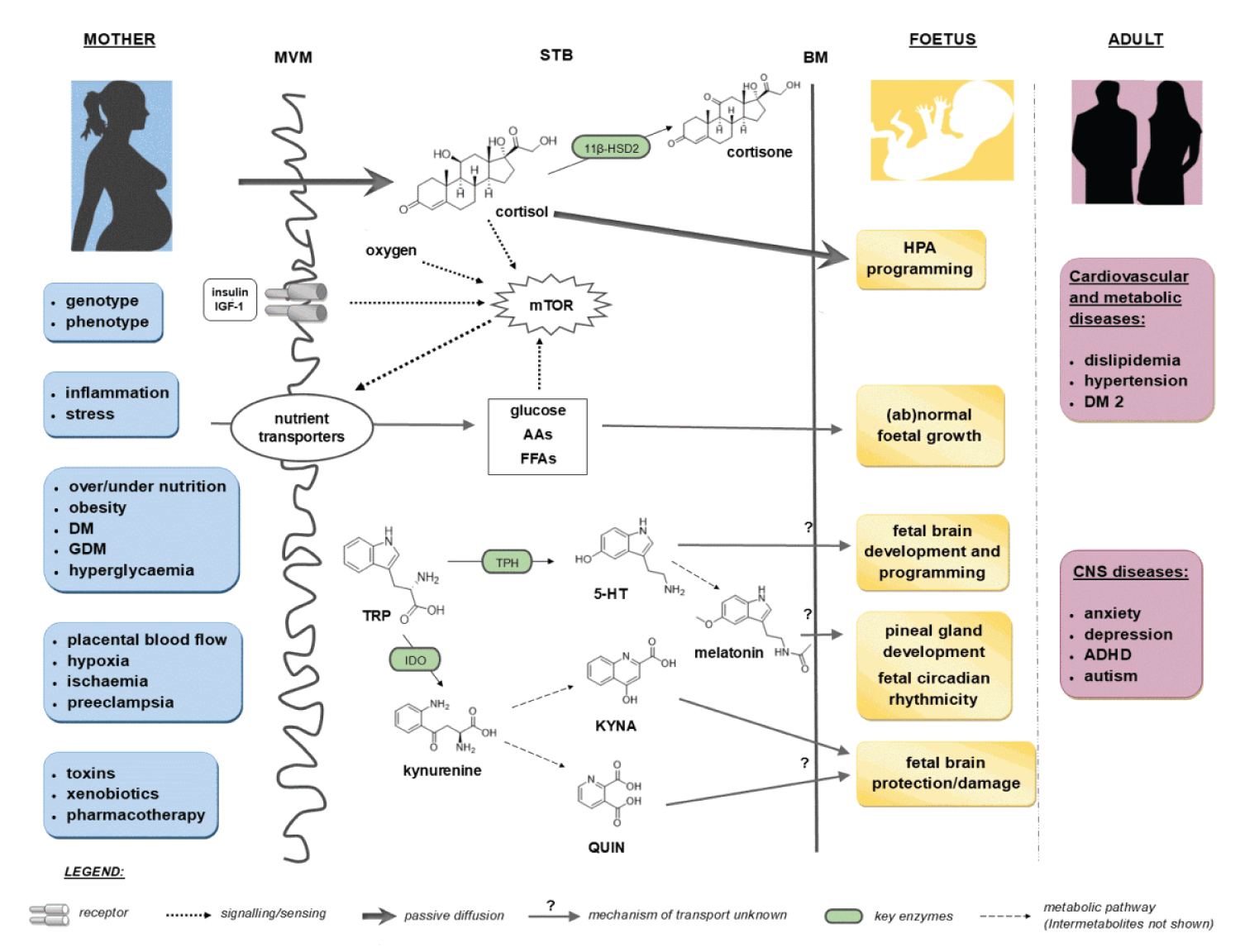 Figure 17: Proposed mechanism of mother-to-foetus communication and foetal programming occurring in the STB. Adverse prenatal conditions on the maternal side (blue boxes) may alter enzyme/transporter expression and function or mTOR signalling within STB, and subsequently nutrient transporter expression/function. Defects in nutrient delivery as well as glucocorticoid and tryptophan systems may lead to sub-optimal in utero conditions and faulty programming of the foetus (yellow boxes), giving rise to diseases in adult life (pink boxes). Specifically, placental transport and mother-to-foetus ratio of cortisol is modulated by the biotransformation activity of 11β-HSD2 in the STB; mTOR functions as a placental sensing mechanism for nutrients, regulating their transport from mother to foetus. Alterations in these regulatory mechanisms affects foetal growth, provoking cardiovascular and metabolic disease later in life (see text). Both serotonin and kynurenine pathways of TRP metabolism have been detected within STB. While serotonin and melatonin are important for foetal neurodevelopment, several products of kynurenine pathway may be of neuroprotective (KYNA) or neurotoxic (QUIN) nature. The key enzymes of both pathways, TPH and IDO, may be affected by external insults including maternal and placental pathologies and thus result in inappropriate formation and/or transport of neuroactive metabolites to foetus. These events have been associated with occurrence of CNS diseases in adulthood. Abbreviations: 11β-HSD2 - 11beta-hydroxysteroid dehydrogenase; 5-HT - serotonin; AAs - amino acids; ADHD - attention-deficit/hyperactivity disorder; BM - basal membrane; DM - diabetes mellitus; DM2 - DM type 2; FFAs- free fatty acids; GDM - gestational DM; HPA - hypothalamic-pituitary-adrenal axis; IDO - indoleamine 2, 3-dioxygenase; IGF - insulin-like growth factor; KYNA - kynurenic acid; mTOR - mammalian target of rapamycin; MVM - microvillus membrane; QUIN - quinolinic acid; STB - syncytiotrophoblast; TDO - tryptophan 2,3-dioxygenase; TPH - tryptophan hydroxylase; TRP - tryptophan. Source: [3].
View Figure 17
Figure 17: Proposed mechanism of mother-to-foetus communication and foetal programming occurring in the STB. Adverse prenatal conditions on the maternal side (blue boxes) may alter enzyme/transporter expression and function or mTOR signalling within STB, and subsequently nutrient transporter expression/function. Defects in nutrient delivery as well as glucocorticoid and tryptophan systems may lead to sub-optimal in utero conditions and faulty programming of the foetus (yellow boxes), giving rise to diseases in adult life (pink boxes). Specifically, placental transport and mother-to-foetus ratio of cortisol is modulated by the biotransformation activity of 11β-HSD2 in the STB; mTOR functions as a placental sensing mechanism for nutrients, regulating their transport from mother to foetus. Alterations in these regulatory mechanisms affects foetal growth, provoking cardiovascular and metabolic disease later in life (see text). Both serotonin and kynurenine pathways of TRP metabolism have been detected within STB. While serotonin and melatonin are important for foetal neurodevelopment, several products of kynurenine pathway may be of neuroprotective (KYNA) or neurotoxic (QUIN) nature. The key enzymes of both pathways, TPH and IDO, may be affected by external insults including maternal and placental pathologies and thus result in inappropriate formation and/or transport of neuroactive metabolites to foetus. These events have been associated with occurrence of CNS diseases in adulthood. Abbreviations: 11β-HSD2 - 11beta-hydroxysteroid dehydrogenase; 5-HT - serotonin; AAs - amino acids; ADHD - attention-deficit/hyperactivity disorder; BM - basal membrane; DM - diabetes mellitus; DM2 - DM type 2; FFAs- free fatty acids; GDM - gestational DM; HPA - hypothalamic-pituitary-adrenal axis; IDO - indoleamine 2, 3-dioxygenase; IGF - insulin-like growth factor; KYNA - kynurenic acid; mTOR - mammalian target of rapamycin; MVM - microvillus membrane; QUIN - quinolinic acid; STB - syncytiotrophoblast; TDO - tryptophan 2,3-dioxygenase; TPH - tryptophan hydroxylase; TRP - tryptophan. Source: [3].
View Figure 17
Kynurenine has an immunosuppressive [83-85] role by acting on AhR receptors, promoting a tendency for lymphocytes to differentiate into Treg. Cathepsins and Furins also induce the tolerance pathway as well as promote apoptosis of lymphocytes. These facts explain much of what we see in patients who exhibit intense lymphopenia associated with the severity of hypoxia (Figure 18 and Figure 19).
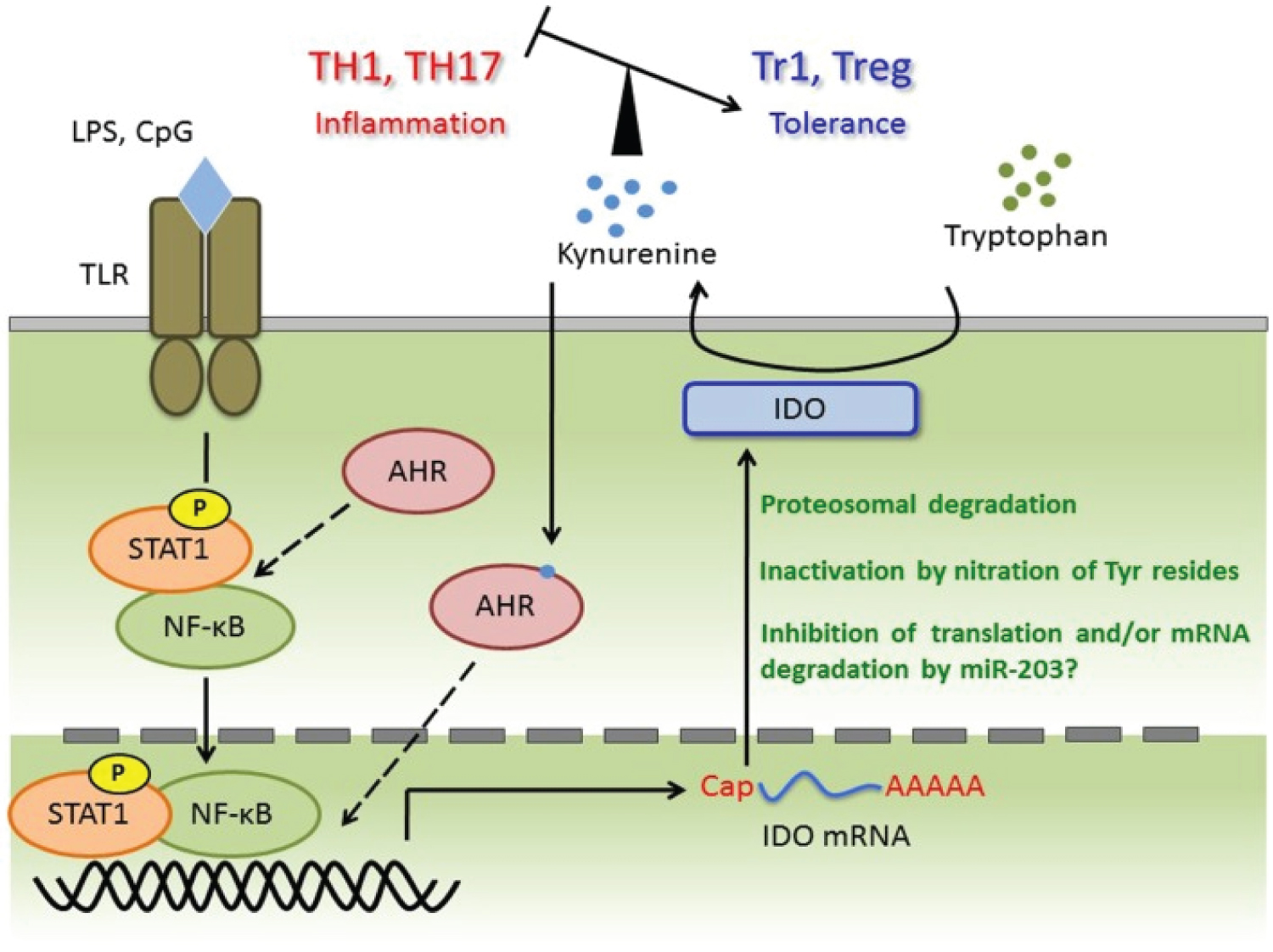 Figure 18: TLR ligands trigger transcriptional activation of STAT-1 and NF-κB and then induce IDO mRNA. Although AhR forms complex with STAT-1 and NF-κB in macrophages under pro-inflammatory cytokines production, this complex appears in DC or required for IDO expression is not known. Induced IDO mRNA may be controlled by miR-203 (not investigated), and the activity or amount of IDO protein is regulated at post-translational modification, such as nitration of Tyr and ubiquitin ligation. Kynurenine catalysed by IDO induces tolerances via regulating TH1, TH17, Tr1, and Treg balance. Kynurenine may activate the AhR for IDO induction in an autocrine manner and form AHR/Kynurenine positive-feedback loop. Source: [78].
View Figure 18
Figure 18: TLR ligands trigger transcriptional activation of STAT-1 and NF-κB and then induce IDO mRNA. Although AhR forms complex with STAT-1 and NF-κB in macrophages under pro-inflammatory cytokines production, this complex appears in DC or required for IDO expression is not known. Induced IDO mRNA may be controlled by miR-203 (not investigated), and the activity or amount of IDO protein is regulated at post-translational modification, such as nitration of Tyr and ubiquitin ligation. Kynurenine catalysed by IDO induces tolerances via regulating TH1, TH17, Tr1, and Treg balance. Kynurenine may activate the AhR for IDO induction in an autocrine manner and form AHR/Kynurenine positive-feedback loop. Source: [78].
View Figure 18
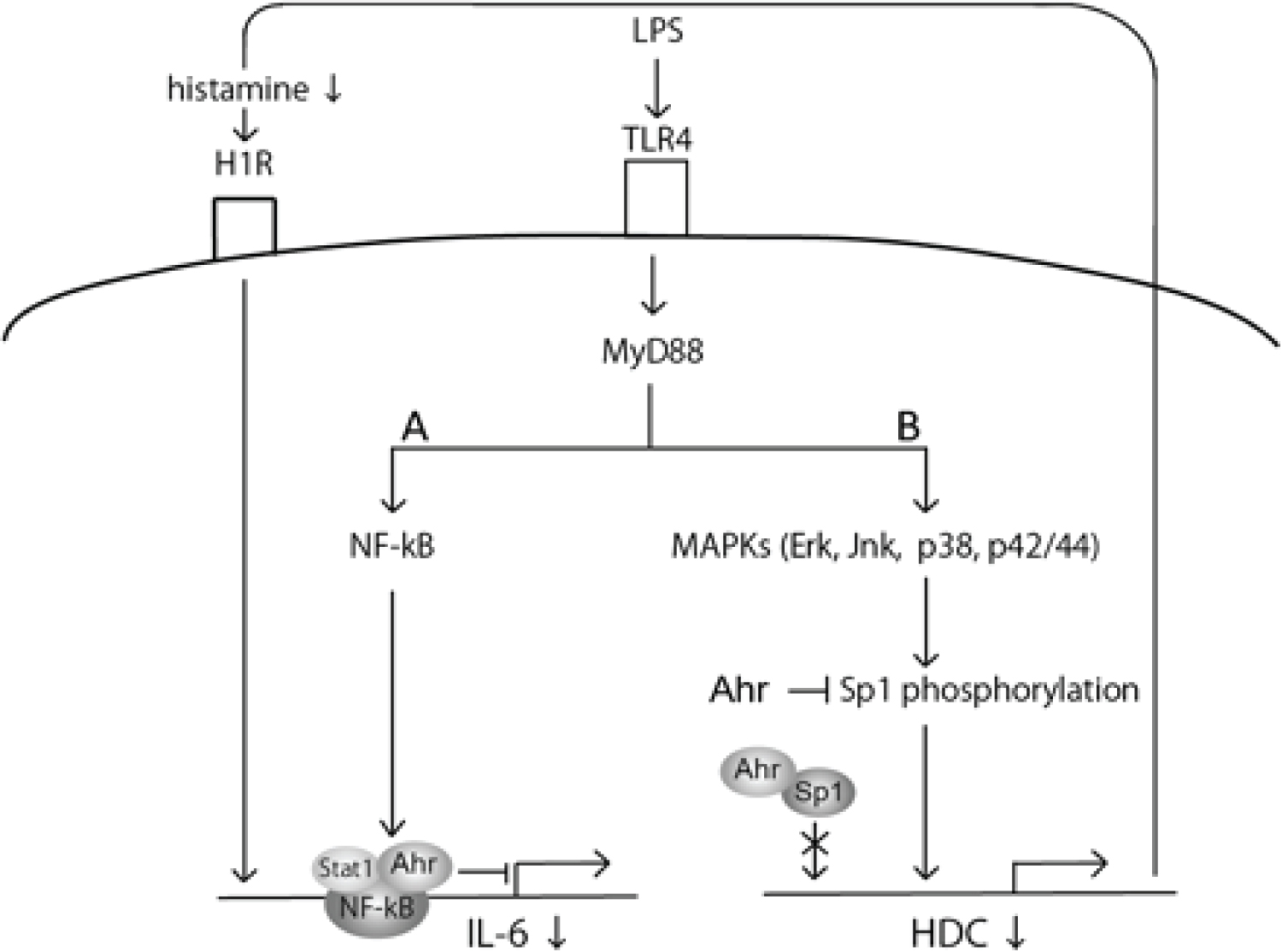 Figure 19: AhR negatively regulates IL-6 production by inhibiting two pathways of MyD88-dependent LPS signalling. (A)AhR in combination with Stat1 forms a complex with NF-κB, which negatively regulates LPS-induced IL-6 production through inhibition of MyD88-dependent NF-κB activation. Ahr-Sp1 complex attenuates the ability of Sp1 DNA binding in the HDC promoter by impairing Sp1 phosphorylation at Ser residues, which leads to suppression of histamine production. Finally, the decrease of histamine production contributes to the reduction of H1R-mediated IL-6 production. Source: [120].
View Figure 19
Figure 19: AhR negatively regulates IL-6 production by inhibiting two pathways of MyD88-dependent LPS signalling. (A)AhR in combination with Stat1 forms a complex with NF-κB, which negatively regulates LPS-induced IL-6 production through inhibition of MyD88-dependent NF-κB activation. Ahr-Sp1 complex attenuates the ability of Sp1 DNA binding in the HDC promoter by impairing Sp1 phosphorylation at Ser residues, which leads to suppression of histamine production. Finally, the decrease of histamine production contributes to the reduction of H1R-mediated IL-6 production. Source: [120].
View Figure 19
Phe is a substrate for the formation of catecholamines; when Phe is in deficit, the chronic stress of COVID-19 can deplete catecholamine reserves and justify the difficulty in suspending the intravenous administration of norepinephrine in patients.
The production of Kynurenine and IDO-1 is altered so that in the first trimester, there is a tendency towards the formation of 5-HT involved in fetal development and blood vessels. By the action of IDO-1, the Try metabolism tends towards the KYN pathway, generating both protective and toxic metabolites to the Central Nervous System. During inflammatory processes, the preference for the KYN pathway is orchestrated to decrease inflammation, as KYN binds to AhR receptors, down-regulating IL-6 production by inhibiting two MyD88-dependent LPS signalling pathways (Figure 19 and Figure 20) [86-88].
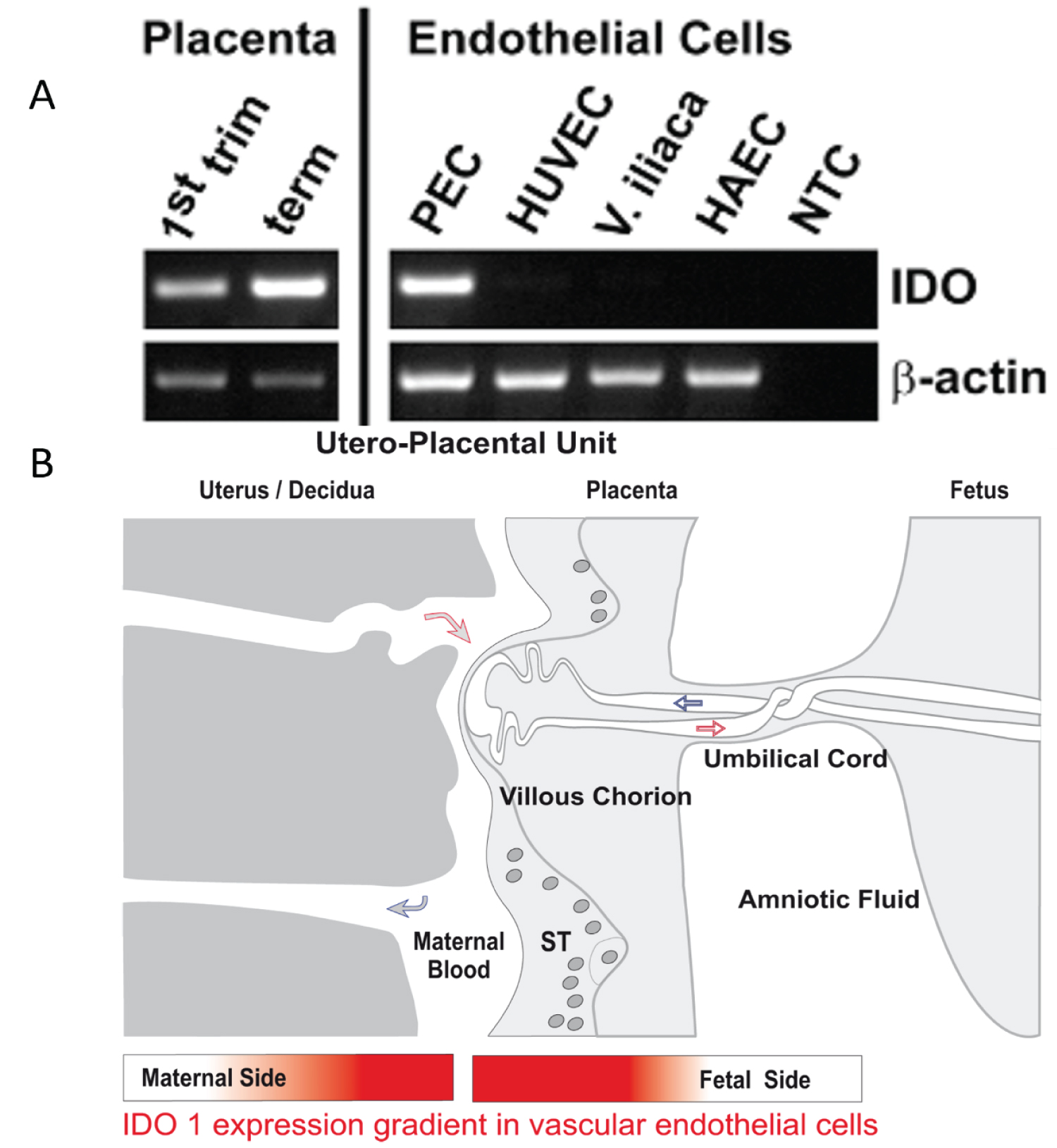 Figure 20: A. Semi-quantitative RT-PCR comparing IDO1 mRNA expression in first trimester and term placenta tissues with endothelial cells isolated from term placenta chorionic plate (PEC), a human umbilical vein endothelial cell line (HUVEC), endothelial cells isolated from the iliac vein (V. iliaca) and a cell line from human aortic endothelium (HAEC). The expression of beta-actin was used as internal reference and water instead of total RNA served as non template control (NTC). The upregulation of IDO1 is confirmed from first trimester to term placenta by measuring the levels of tryptophan and kynurenine and the Kyn/trp ratio in lysed tissues. In parallel to immunohistochemical and RT-PCR results, tryptophan concentration measured in the tissue decreased (p < 0.001) from first trimester villi (57.4 + 31.13 µmol/mg, mean + standard deviation) to term villi (7.2 + 8.14 µmol/mg), the concentration of kynurenine increased (p < 0.001) from first trimester (5.3 + 8.94 µmol/mg) to term villi (38.0 + 11.97 µmol/mg). So the mean kynurenine/tryptophan ratio, allowing for an estimation of antecedent IDO activity of placental tissue increases about thousand-fold (p < 0.001) from first trimester (0.076 + 0.072) to term placenta (74.419 + 126.291). B. Illustration of the IDO1 expression gradient in vascular endothelial cells of fetal and maternal blood circulations at the utero-placental maternofoetal interface. Source: [121].
View Figure 20
Figure 20: A. Semi-quantitative RT-PCR comparing IDO1 mRNA expression in first trimester and term placenta tissues with endothelial cells isolated from term placenta chorionic plate (PEC), a human umbilical vein endothelial cell line (HUVEC), endothelial cells isolated from the iliac vein (V. iliaca) and a cell line from human aortic endothelium (HAEC). The expression of beta-actin was used as internal reference and water instead of total RNA served as non template control (NTC). The upregulation of IDO1 is confirmed from first trimester to term placenta by measuring the levels of tryptophan and kynurenine and the Kyn/trp ratio in lysed tissues. In parallel to immunohistochemical and RT-PCR results, tryptophan concentration measured in the tissue decreased (p < 0.001) from first trimester villi (57.4 + 31.13 µmol/mg, mean + standard deviation) to term villi (7.2 + 8.14 µmol/mg), the concentration of kynurenine increased (p < 0.001) from first trimester (5.3 + 8.94 µmol/mg) to term villi (38.0 + 11.97 µmol/mg). So the mean kynurenine/tryptophan ratio, allowing for an estimation of antecedent IDO activity of placental tissue increases about thousand-fold (p < 0.001) from first trimester (0.076 + 0.072) to term placenta (74.419 + 126.291). B. Illustration of the IDO1 expression gradient in vascular endothelial cells of fetal and maternal blood circulations at the utero-placental maternofoetal interface. Source: [121].
View Figure 20
As pregnancy progresses, the placenta loses its immunosuppressive strength and perhaps the increase in oxygen tension is an explanation for this phenomenon since hypoxia makes the environment more tolerant. Furthermore, the ageing process of the placenta tends to a natural increase in oxidative stress indicating that the pathways that activate inflammation are gradually increased as the placenta ages. Thus, in the third trimester and after 34 weeks, the pregnant woman is more symptomatic when the SARS-CoV-2 infection occurs during this period [88,89].
There are many cases of acute COVID-19 that start with psychiatric manifestations, especially depression. The deficit of 5-HT and dopamine can explain these changes due to low intestinal absorption, which can have repercussions in more intense puerperal melancholia [87,90,91].
Added to the 5-HT and dopamine deficits, cell damage due to intense hypoxia generates Adenosine as a product whose function is initially cellular protection, acting in an immunosuppressive way.
Hypoxia induces ATP release via ATP-binding cassette (ABC) transporters, pannexin 1 or connexins. Accumulated ATP can stimulate purinergic P2 receptors (P2XRs and P2YRs) or be further degraded to Adenosine by the sequential action of CD39 and CD73 ectonucleotidases. In situations of great metabolic stress, in this case, the magnitude of hypoxia produced in SARS-CoV-2 infection in a cellular medium depleted of NAD/NADH+ [58,92-94], requiring tissue lysis to guarantee amino acids for gluconeogenesis, Adenosine is produced in large quantities. It may reflect metabolic exhaustion in the face of cell damage. This article theorises that Adenosine [95-97] promotes bradycardia even in a febrile state, a fact known as the Faget's Sign (Figure 21 and Appendix). This phenomenon is also a known sign of other illnesses, for example, yellow fever. The tendency towards bradycardia is related to tissue hypoxia with implications for aerobic metabolism [98-101]. An electrocardiogram must be performed whenever a pregnant woman (or any patient) is admitted to the hospital. A baseline comparison is necessary to have a better assessment of the possible causes of bradycardia. Another essential role of Adenosine is to cause peripheral vasodilation, adding points for refractory shock COVID-19.
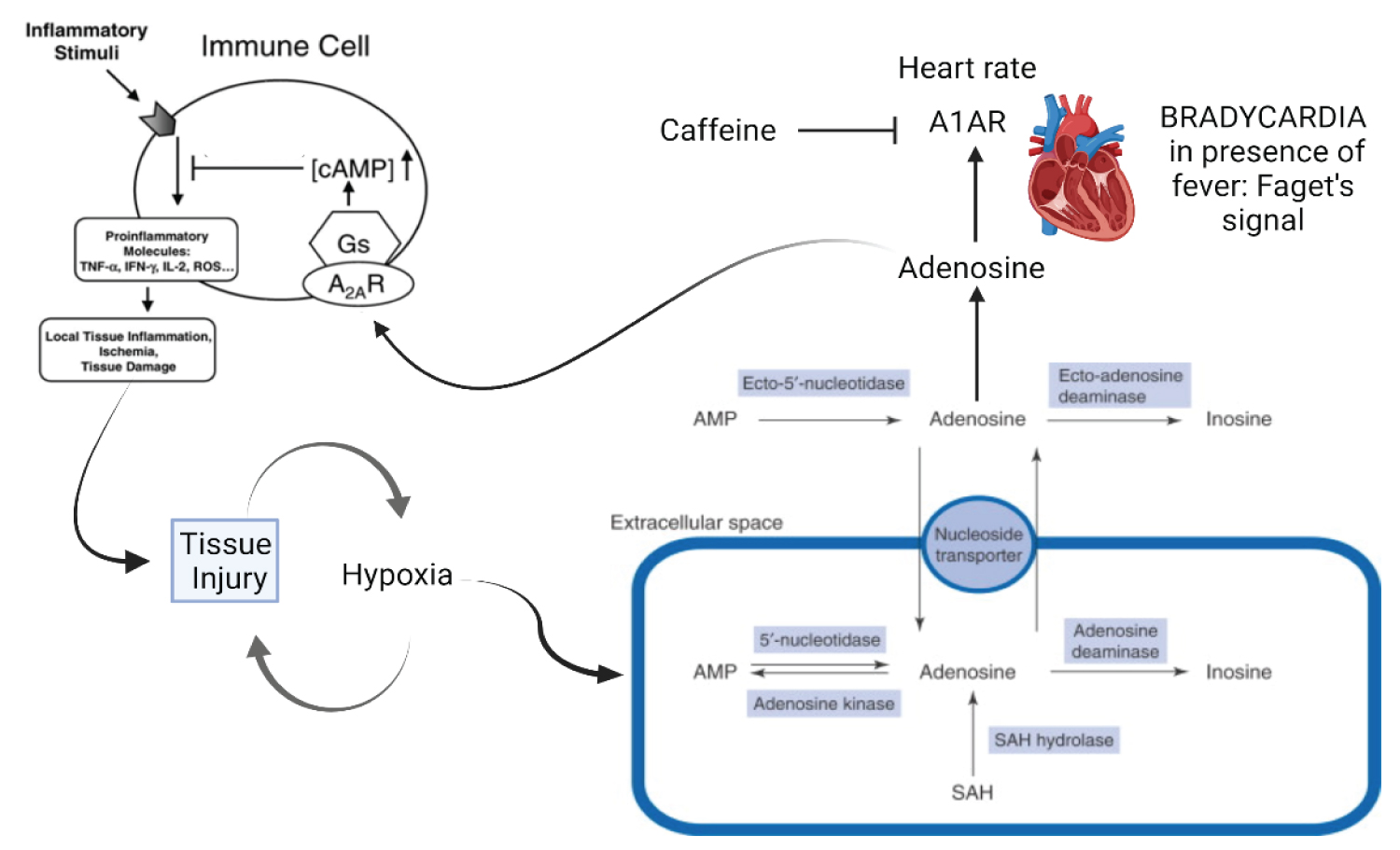 Figure 21: Created with BioRender.com. Faget's sign. Faget's sign is bradycardia concerning fever when the usual thing is tachycardia due to temperature elevations. It so happens that the pathophysiology behind this phenomenon is still controversial. Given the studies in COVID-19, the possibility arises that this phenomenon is related to the production of Adenosine (ADA) in the face of severe hypoxia that occurs in SARS-COV-2 infection. Adenosine plays an essential role against cell damage, has an anti-inflammatory/immunosuppressive role in addition to causing psychiatric phenomena that are very prevalent in COVID-19. However, this adaptation to cellular damage can become a sign of physical and metabolic exhaustion in the face of such intense inflammation resulting in Bradycardia with a risk of cardiorespiratory arrest due to the amount of serum adenosine and hypoxemia resulting from COVID-19. This situation also appears in other diseases that progress with severity and tissue hypoxia due to disseminated intravascular coagulation, causing cell damage due to lack of tissue oxygen.
View Figure 21
Figure 21: Created with BioRender.com. Faget's sign. Faget's sign is bradycardia concerning fever when the usual thing is tachycardia due to temperature elevations. It so happens that the pathophysiology behind this phenomenon is still controversial. Given the studies in COVID-19, the possibility arises that this phenomenon is related to the production of Adenosine (ADA) in the face of severe hypoxia that occurs in SARS-COV-2 infection. Adenosine plays an essential role against cell damage, has an anti-inflammatory/immunosuppressive role in addition to causing psychiatric phenomena that are very prevalent in COVID-19. However, this adaptation to cellular damage can become a sign of physical and metabolic exhaustion in the face of such intense inflammation resulting in Bradycardia with a risk of cardiorespiratory arrest due to the amount of serum adenosine and hypoxemia resulting from COVID-19. This situation also appears in other diseases that progress with severity and tissue hypoxia due to disseminated intravascular coagulation, causing cell damage due to lack of tissue oxygen.
View Figure 21
This article shows that exposure to hypoxia is linked to the magnitude of the patients' phenomena since cell responses tend towards tolerance. Patients with predictors of severity (T2DM, obese, elderly, heart diseases) gets immunosuppressed by SARS-CoV-2 and can rapidly induce tumour growth due to apoptosis of all lineages of lymphocytes. The activation of latent diseases of any infectious etiologies [102-104], which can, in pregnant women, cause secondary damage to the foetus, is a fact. Publications showing activation of the virus of the Herpesviridae family [105-107] in the face of lymphopenia in COVID-19 has not been rare [108-110]. Considering the role of neutrophils and the formation of immune complexes in COVID-19, tolerance also allows for autoimmune diseases.
In pregnant women, the placenta acts as a buffer by immunosuppressing the pregnant woman, preventing her from progressing to an inflammatory phase with intense symptoms. At the same time, pregnant women with T2DM and obesity are more symptomatic because they present placental changes that prevent them from performing adequate immunosuppression.
Soon after placental separation, postpartum women rapidly progress to respiratory failure (depending on comorbidities and the extent of pulmonary involvement). This phenomenon may occur because, without the placenta, the postpartum woman's homeostasis returns to the basal level. Inflammation occurs quickly and generates the cytokine storm that drives them to Acute Respiratory Distress Syndrome (ARDS). For this reason, it is necessary to perform a chest tomography immediately after placental separation (see guide in the Appendix).
Due to COVID-19 has a viral phase and another one almost (most cases) inflammatory exclusively, immunosuppression by the placenta is the natural treatment against cytokine storm.
T2DM or obese patients have a high chance of progressing to severe or critical forms of COVID-19, as they already have a comorbidity profile that stimulates inflammation. Even in early pregnancy, pregnant women with diabetes or obesity will be more symptomatic, as the metabolic pathways of the placenta are already altered, tending to premature ageing and more significant oxidative stress (Figure 22).
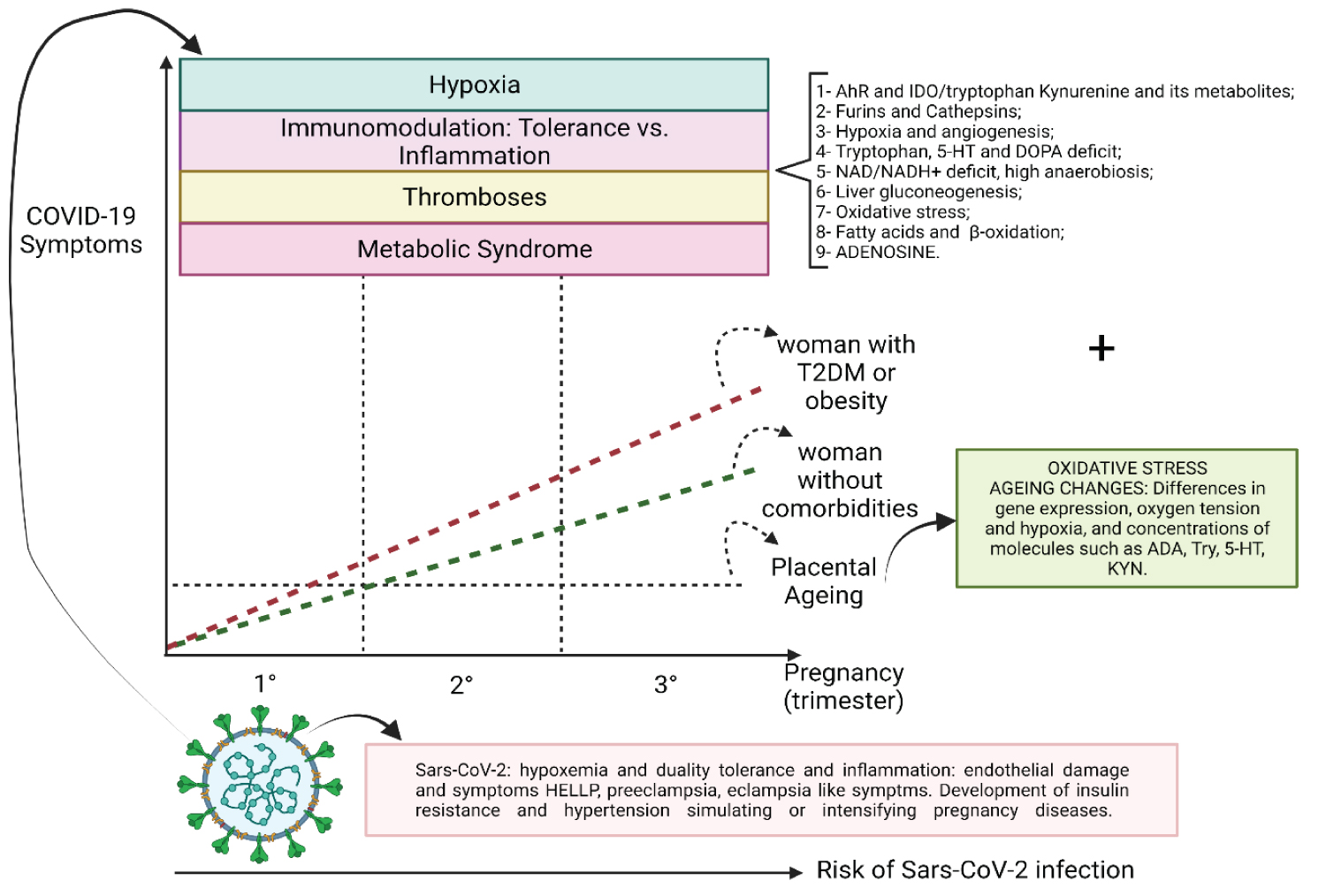 Figure 22: Created with BioRender.com. Phenomena presented at COVID-19. Thrombotic phenomena, dysmetabolism, immunotolerance magnified by sustained hypoxia are facts that promote the maintenance of viral replication by maintaining an acidic environment. Clinical manifestations during SARS-COV-2 infection are more intense as the placenta ages, both naturally and early, in cases of gestational diseases that accentuate oxidative stress. The pregnant woman can become infected at any time of pregnancy and the fetus is not exempt from suffering from SARS-Cov-2 infection. Numbers from 1 to 9 show altered metabolic pathways in COVID-19 or induced or magnified by hypoxia.
View Figure 22
Figure 22: Created with BioRender.com. Phenomena presented at COVID-19. Thrombotic phenomena, dysmetabolism, immunotolerance magnified by sustained hypoxia are facts that promote the maintenance of viral replication by maintaining an acidic environment. Clinical manifestations during SARS-COV-2 infection are more intense as the placenta ages, both naturally and early, in cases of gestational diseases that accentuate oxidative stress. The pregnant woman can become infected at any time of pregnancy and the fetus is not exempt from suffering from SARS-Cov-2 infection. Numbers from 1 to 9 show altered metabolic pathways in COVID-19 or induced or magnified by hypoxia.
View Figure 22
The placenta acts as a buffer against SARS-CoV-2 infection: 1) Immunosuppression prevents the pregnant woman from presenting symptoms resulting from an inflammatory storm, 2) The placenta is initially supported in an oxygen-poor environment, and its energy metabolism prioritises via an anaerobic route, a fact that can mitigate the effects of hypoxia caused by SARS-CoV-2, 3) The materno-foetal interface is a syncytium, a site that can be tropic for the coronavirus since this environment has high expression of cathepsins and furins; however, it is the place where maternal macrophages and neutrophils are stimulated to contain the infection. In this case, syncytium would have a protective role against SARS-CoV-2 infection, functioning as a trap.
Given the pandemic and, in the case of something that I had never seen before, a virus for which we do not have adequate treatment, the only solution was to study the virus-host relationships to establish better medical practices on an understanding of the pathophysiology involved in infection.
I am aware that all of these are hypotheses, but hypotheses based on observation of almost two years dealing with the clinical management of COVID-19 patients. I am fully convinced that tomorrow all this can change, as we are talking about science. The important thing is that we are not static in the face of the new. Our knowledge is limited, and we need to expand it to understand acute SARS-CoV-2 infection and the consequences of chronic inflammation that has a terrible impact on people's quality of life, a situation called "Long COVID-19". So far, the main approaches have a more palliative than curative bias. We are acting on the symptoms and not on the essence that causes the presented clinical phenomena. With that, we have a polymedication that often causes more problems in the patient's life than benefits.
Understanding the disease is the best way to know how to treat it.
-To Syomara Regina de Almeida, an Obstetrician with who I have the pleasure of sharing plenty of doubts facing the pregnant women that Syomara trusted me to follow-up considering each patient as unique in their individualities.
I had the obligation (with a grateful satisfaction) of studying deeply each pregnant woman to find options to offer facing the real life, considering my professional and human ethics.
-To Maria Luiza Benício. An Obstetrician and a friend of mine who believed in my opinion when I explained by phone, what I was thinking about COVID-19 and pregnancy relationship. It happened about 1 year ago.
-To All patients that allow me to grow up as a human being, as a physician, as a researcher. All this work has been developed to offer better choices when no other option has already been available.
-To the hospital do servidor público (public servants) in the state of São Paulo (HSPE). My eternal gratitude.
-To my friends, parents and family who supported me to continue, even in the face of storms: Be they cytokine storms or human storms.
-And to Andrea Lúcia Silva Ladeira de Almeida, clinical manager of the HSPE, for understanding that these studies have value not only for me, who grows with the doubts and with the search for solutions in the face of doubts, but that the most significant value is the outcome we can achieve for the COVID-19 patients when we understand that SARS-CoV-2 does not behave like a known virus.
Luiz Zanella: I wrote this article based on my nearly two years of clinical experience and studies focused on the management of the patient COVID-19. The experience and existing doubts in the management of pregnant patients originated both from consultations carried out at the HSPE and from outpatients for which I was appointed a specialist in infectious diseases to perform care in conjunction with the Obstetricians.
Miriane Marques: My friend and an Obstetrician who helped me searching for articles about pregnant women's pathophysiology.
This work does not require authorization and there is no identification of patients. There was no need for a consent form to this review according to the National Ethics Committee.
All data are available in the electronic medical record system of Hospital do Servidor Estadual (Sao Paulo-Brazil) and in the authors' research files.
The authors report on conflict of interest.
Luiz Gonzaga Francisco de Assis 'Barros D'Elia Zanella is a physician specialised in Infectious Diseases. I work fully in Public Health (SUS-Sistema Único de Saúde) in Brazil, providing private care in my own clinic. Also, I conduct clinical research at the "Hospital do Servidor Público do Estado de São Paulo (HSPE)". I have been dealing with COVID-19 patients in COVID-19 wards, Intensive Care Units, and I work with the training of medical residents. I have no conficts of interest and I do not have founds that.
Miriane Marques Borges is a Gynecologist and Obstetrician. She provides private care and to the Public Health System. She does not receive any funding that conflicts with the article's proposal.
This article did not receive funding from any kind of sources to be written.
The recent publication by Nóbrega-Cruz, et al. [74], favouring the idea that COVID-19 needs a different understanding of its pathophysiology, addresses the molecular relationships that lead pregnant women to be more symptomatic in the final stages of pregnancy. Adding to the ideas proposed in Nóbrega-Cruz, et al. this article shows changes in metabolic pathways by the action of SARS-Cov-2. Changes related to ACE-2 internalization associated with the cellular injury caused by SARS-CoV-2 triggering intense hypoxia indicate that the pathophysiology of COVID-19 needs reconsiderations every moment. COVID-19 is a new disease, and hypoxia has differences that make it something serious, whose management requires a different view concerning other diseases that course with respiratory severity. Together, the two articles highlight the need for clinical approaches to be modified for all COVID-19 patients. However, this article focuses on pregnant and postpartum women.
It is important to emphasize that the conduct of pregnant and postpartum women is necessarily Obstetric, a speciality focused on improvements in this area. My role, as an Infectious Diseases specialist, is to expose situations where I was involved by jointly conducting obstetric cases, which supplied me moments of learning and study.
It is essential to consider that pregnant and postpartum women need improvement in Clinical, Obstetrical, and Intensive Care management, which a joint inter-clinical specialities discussion is essential, adding ideas and knowledge facing a new disease makes possible to achieve better and targeted approaches for each patient.
From the contact with specialists in obstetrics, conducting patients at the Hospital do Servidor Público do Estado de São Paulo (HSPE), where I work as an Infectious disease specialist/emergency physician and intensive care physician helping in the care in COVID-19, I expose the result after studies triggered by the necessity of better management of COVID-19 pregnant women.
Due to this confidence in joint management, I nurtured an essential desire for a study to improve conduct based on understanding the virus-pregnant woman relationship.
The following key points summarize what I consider essential given the conditions that make the placenta the exceptional protection for pregnancy to come close to term (about 280 days or 40 weeks) during SARS-CoV-2 infection.
1. The placenta is an organ that provides the woman with immunosuppression due to vasculogenesis, which tends the microenvironment to tolerance so that the embryo is not recognised as not proper by the maternal immune system (Figure 1, Figure 2, Figure 3, Figure 4 and Appendix).
2. The tolerant microenvironment occurred by the action of cytokines that act systemically, providing the pregnant woman with a status of physiological immunosuppression. However, SARS-CoV-2 infection has handled saving women from symptoms in the early stages of pregnancy (until the end of the second trimester).
3. When the placenta is still a highly active organ, from the third trimester onwards, the placenta shows more intense and rapid ageing, the mechanisms that maintain immunosuppression are less and less intense, allowing the standard tools of inflammation to come back. The pregnant woman becomes more symptomatic in diseases that progress with inflammatory processes such as COVID-19. For this reason, the placenta is an organ that protects the pregnant woman during COVID-19, allowing a normal pregnancy when thrombosis in placental vessels does not occur during SARS-CoV-2 infection in the first trimester of pregnancy.
4. The placenta allows the woman to be a safe environment for the embryo and foetus, but the woman is more symptomatic in the third trimester and manifests symptoms linked to endothelial damage by SARS-CoV-2.
5. Symptoms related to endothelial damage caused by the coronavirus simulate classic diseases of pregnant women such as HELLP syndrome, preeclampsia, and eclampsia. The internalization of ACE-2 by the virus causes an increase in systemic blood pressure, simulating Gestational Diabetes mellitus (GDM). At the same time, the general metabolism has a hyperglycaemic environment, affecting GDM, in oxidative stress (Figure 1, Figure 2 and Figure 3) and aldosterone elevation. Placenta has mechanisms that can compensate the oxidative stress under normal hypoxia (Figure 8, Figure 9, Figure 10, Figure 11, Figure 12, Figure 13 and Figure 18) via The Warburg effect (Appendix).
6. Deficient tryptophan can generate significant symptoms of puerperal blues. Try replacement associated with Bupropion or Citalopram can bring good results to be studied in clinical protocols of specific and necessary clinical studies. So far, I only present hypotheses and experiences (Figure 2 and Figure 3).
7. The placenta produces tryptophan, an amino acid deficient in patients with SARS-CoV-2 infection of the gastrointestinal system, as ACE-2 absorbs it in enterocytes. Phenylalanine is also absorbed by ACE-2 in the intestines and becomes deficient in COVID-19. The lack of tryptophan promotes a more tolerant environment due to immunosuppression by lymphocyte apoptosis (via caspase, CD4+, CD8+, NK and B cells). Some latent infections can be reactivated due to lymphopenia, such as Herpes zoster, other viral diseases, in addition to mycobacteriosis, fungal diseases.
8. Lack of tryptophan (Try) and phenylalanine (Phe) has caused acute psychiatric symptoms and catecholamine deficiency.
9. The lack of vitamin B3 due to the shift to Kynurenine (via IDO1) promotes deficient cellular respiration, generating hypoxemia and adenosine in response to this environment of cellular aggression. However, all this pathophysiology is alleviated in pregnant women with the intraplacental production of Try and a hypoxemic buffering (Figure 5, Figure 6, Figure 7, Figure 8, Figure 13, Figure 14, Figure 15, Figure 16, Figure 17, Figure 18 and Figure 19).
10. When it is necessary to induce the delivery due to signs and symptoms of gestational diseases mimicked by SARS-CoV-2, the woman loses the organ that supported her as a marvellous shield to the foetus. There is, at this time, a considerable acute hormonal variation, in addition to the temporary organ loss that allows an infection in the third trimester to be sustained until the symptoms of gestational diseases reach the criteria for resolution of childbirth.
11. With the loss of the placenta, the woman progressively becomes symptomatic, and, in about 10 to 12 hours after the delivery, she can progress to respiratory failure with orotracheal intubation (in cases where infection occurred near this period).
12. Symptoms related to endothelial damage caused by the coronavirus mimic classical diseases of pregnant women such as HELLP syndrome, preeclampsia, and eclampsia. ACE-2 internalization by the virus causes an increase in systemic blood pressure, simulating GHD, and metabolic syndrome.
13. Furins and Cathepsins are present in large quantities in syncytiotrophoblast and act in the process of placental implantation (Figure 5). Usually, the expressions of these proteins are related to hypoxia in the environment. Thus, the hypoxia developed in COVID-19 can promote important changes in the invasive waves of the placenta, impacting the development of diseased pregnancies and foetal/embryo death. Perhaps the high expression of furins and cathepsins contribute to the process of "symptom buffering" of COVID-19 in pregnant women, associated with the fact that the placenta can meet metabolic needs due to the lack of tryptophan.
14. The uteroplacental location of Ang-(1-7) and ACE2 in pregnancy suggests an autocrine function of Ang-(1-7) in the vasoactive regulation that characterizes placentation and established pregnancy. ACE2 at the maternal–foetal interface a prerequisite for transplacental transmission. Strong and diffuse membranous staining of cytotrophoblast and syncytiotrophoblast (ST) cells of placental villi, as well as a membranous expression in extra villous trophoblast. ACE2 expression was detected in villous stroma, Hofbauer cells, or endothelial cells. TMPRSS2 expression was only present weakly in the villous endothelium and rarely in the ST.
15. In COVID-19 patients, endothelial activation has been shown to be associated with elevated circulating markers of coagulation (e.g., D-dimers) and inflammation (e.g., TNF-a, IL-6, IL-2, and MCP-1). COVID-19 patients usually present with elevated platelet numbers, highly increased fibrinogen and slightly prolonged prothrombin and activated partial thromboplastin time.
16. Placentas from pregnancies complicated with chorioamnionitis (ChA), exhibited increased expression of ACE2 mRNA. Placental ACE2 protein localized to syncytiotrophoblast, in foetal blood vessels and M1/M2 macrophage and neutrophils within the villous stroma. Increased numbers of M1 macrophage and neutrophils were present in the placenta of ChA pregnancies. Placentas from pregnant women with COVID-19 and uninfected neonates show significant variability in the spectrum of pathology findings. Placentas from infected maternal-neonatal dyads are characterized by the finding of mononuclear cell inflammation of the intervillous space, termed chronic histiocytic intervillositis, together with syncytiotrophoblast necrosis.
17. Perhaps because the infection induces greater expression of ACE-2, attenuating the effects of internalization of ACE-2 itself. The virus finds an environment rich in cathepsins and furins, and with syncytial cells ready, the placenta acts as a buffer for the adverse effects of SARS-CoV-2 infection. However, both the pregnant woman and the placenta are not exempt from suffering thrombotic events, or even the foetus is exempt from suffering an infection.
18. The fact that M1 macrophages express more ACE-2 and mediate an inflammatory response offers a break in the immunosuppression offered by the placenta, which can be harmful to the foetus, but which can be located in the ST region, allowing viral containment to this site.
19. Never belittle the unknown opponent. We don't know of his strength. For outpatient management, consider that any symptom with pain in the hypochondria and epigastrium should be considered an urgency, and obstetric ultrasonography is necessary. We had a patient with acute loss of amniotic fluid, which could be signs of low (lung base) pneumonia with referred ipsilateral abdominal pain. The guidance to go to the hospital in the face of signs and symptoms caused by a virus, whose pathophysiology is unknown, was crucial for the excellent evolution of the pregnant woman and the foetus (Figure 19, Figure 20, Figure 21 and Figure 22).
20. In summary, SARS-CoV-2 has a high performance to infect individuals of Homo sapiens, of any population, due to the presence of receptors that allow the infection to occur in different tissues. At the same time, the placenta offers specific mechanisms that favour protection for most pregnancies to generate viable foetuses.
The placenta buffers the effects of SARS-CoV-2 infection by:
a) Immunosuppression - acting as corticosteroids, inhibiting the exacerbated inflammatory phase.
b) Syncytiotrophoblast and trophoblast that "trap" the virus in an optimal environment, but that allows the action of macrophages and neutrophils that will act to eliminate the virus.
c) The anaerobic glycolytic pathway very preserved in the first months of pregnancy.
d) Perhaps due to the exchange of deficient substances resulting from the internalization of ACE-2, but which the placenta produces, attenuating the nutritional demands related to Tryptophan and Phenylalanine.