Background: This article is a small review with a hypothesis to be considered, focusing on neuropsychiatric symptoms (NPSs) in COVID-19 patients. Almost 2 years passed by of dealing with COVID-19 patients it is undeniable how patients suffer from continuous pain and how it lasts a long time to return to the previous neurologic status they had before infection; they have been admitted at the hospital with psychiatric symptoms or neurological symptoms without a historical morbid past these before, and soon they are present COVID-19 symptoms some days ahead. This footage can be related to ACE-2 protein internalization, which absorbs Tryptophan and Phenylalanine in the intestine. Neuropsychiatric symptoms have been treated like classical disorders rather than treat these patients properly covered by a new context in a new disease.
Hypothesis: Might physician be iatrogenic in COVID-19 by using inhibitors of the dopaminergic and serotoninergic pathways, since which are already inhibited by the action of SARS-CoV-2.
Objectives: This article aims to 1) Show the importance of knowing the real pathophysiology of a new disease and 2) Avoid patients being treated mistakenly by prescribed drugs that are proper to diseases that are already known, but harmful to COVID-19, generating iatrogenesis.
Methods: Articles searching occurred by PubMed and Google Scholar platforms respecting the previous limit of 60 articles, imposed by the author. After selected sixty articles there was a second sieve based on author's view endorsed by scientific and objective clarified in the Appendix of this manuscript. Forty-five articles composed this work all cited in the "references" at the end of this manuscript.
Results: The review found a link between ACE-2 protein and Try and Phe amino acids. ACE-2, expressed on enterocytes in some sites in the gut, is responsible for absorbing amino acids. The ACE-2 internalisation resulted from Sars-CoV-2 infection triggers a plethora of metabolic changes in the dopaminergic and serotoninergic pathways resulting from the lack of Phe and Try due to low intestinal absorption, responsible for acute neuropsychiatric (NPSs) phenomena. Inflammation is responsible for shifting the Try metabolism towards the formation of Kynurenine, mediated by the enzyme IDO1 (indoleamine 2,3-dioxygenase 1). The products derived from Kynurenine are, for the most part, toxic to the Central Nervous System, causing symptoms like Alzheimer's and Parkinson's diseases, in addition to covering the central GABAergic and glutaminergic receptors. Hypoxemia amplifies the inflammatory reaction and blocks enzymatic reactions, inhibiting the formation of other Try products, for example, Vitamin B3 (Niacin or N/NADH+), drawing in deficient aerobic respiration with systemic repercussions. Lack of Try in a hypoxemic and inflamed environment also implies a decrease in Try directed to the CNS for serotonin formation. Lack of Phe implies less formation of Tyrosine, resulting in an overall decrease in catecholamines, including DOPA.
Conclusion: Physicians must be aware that the NPSs phenomena presented can have a different pathophysiological essence, and treat patients based on already known diseases might be a fallacy, generating iatrogenesis.
COVID-19, Neurological symptoms, Psychiatric symptoms, ACE-2, SARS-CoV-2 infection
The SARS-CoV-2 infection has been a challenge to physicians, among other reasons, because the ACE-2 protein is distributed throughout many human body tissues, allowing COVID-19 disease to be systemic and not just pulmonary [1-4].
Among the systemic manifestations, NPSs have been a severe problem in both the acute and chronic phases of COVID-19 and knowing how to differentiate them from primary psychiatric or neurological diseases is the best way to provide adequate treatment [5-10].
The ACE-2 protein, with wide tissue distribution, is present in all parts of the small intestinal epithelium cells, including the duodenum, ileum, but not in enterocytes of the colon, acts on enterocytes by absorbing some amino acids, including Tryptophan. ACE2 interacts with the neutral amino acid (AA) transporter B0AT1 via its collectrin-like domain, and studies have been shown decreased Try plasma levels in ACE2-deficient mice may result in reduced 5-HT levels in the brain. ACE2 binds to the B0AT1 amino acid transporter and contributes to the absorption of neutral amino acids, for example, Tryptophan (Try) and Phenylalanine (Phe). ACE-2 plays an essential role in the kidneys but will not be covered in this article [11-15]. A few weeks after submitting this article for analysis, there was an understanding of another critical pathway that can be one of the leading causes of the maintenance of inflammation and generate and maintain neuropsychiatric symptoms in COVID-19. This critical pathway is related to adenosine and will be covered in the appendix to this article.
The MEDLINE database search was performed using PubMed. The following 'Tryptophan' Medical Subject Heading (MeSH) terms have been used: 'COVID-19' OR 'SARS-CoV-2' OR 'TDO' OR 'IDO1' OR 'IDO2' OR 'pathway' OR 'Central Nervous System' OR 'metabolism' OR 'immunity' OR 'inflammation' OR 'kynurenine pathway' OR 'ACE-2'. The sixty articles, published independently of the year of publication, in English, with full-text available and relating to, were evaluated for relevance by the author's subjective view. The same was performed with "Phenylalanine" OR 'COVID-19' OR 'tyrosine" OR 'ACE-2', OR 'vitamin B' OR 'gut absorption' OR 'inflammation'. The 'Google Scholar' web search engine was used, with similar search terms, to complete the literature search.
There was a scarcity of articles about amino acids and COVID-19 or neurological symptoms, and it was necessary to search for ACE-2 articles and Try and Phe absorption before the COVID-19 period, which supported the hypothesis by showing that some metabolic pathways exist and that they are likely to be interfered by SARS-CoV-2 infection. Sixty articles were screened (maximum number stipulated by the author) after evaluating the articles based on the author's subjectivity but guided by good practices in science (Appendix), 45 articles composed this study.
Tryptophan is an amino acid whose metabolism can be modified by the enzyme IDO1, expressed by macrophages, monocytes and dendritic cells in processes inflammatory and infectious. The up-regulation of this enzyme happens by stimulating prostaglandins, LPS (lipopolysaccharides) and gamma interferon (IFN)-γ expressed and secreted during the period of inflammation/infection. It turns out that IDO1 modifies the homeostatic pathway of tryptophan catabolism. In homeostasis, Tryptophan is metabolised by the enzyme tryptophan 2,3-dioxygenase (TDO), mainly liver-specific TDO using O2 as co-substrate and heme as co-factor. The extrahepatic IDO is a haemoprotein that also uses O2 as a co-substrate [16-21].
IDO1 has been related to critical pathophysiological changes in the body. When in homeostasis, the role of indoles stimulates an anorexigenic state by stimulating incretin. However, incretin promotes changes in the intestinal epithelium at an inflammatory status, facilitates bacterial translocations, and exerts a critical role in aneurysm development through vascular favouring smooth muscle cell apoptosis and the increase of metalloproteinase expression. At the same time, Kynurenine metabolites stimulate adhesins, chemotactic proteins for inflammatory sites and trigger intestinal diseases and neurological symptoms [22,23].
The metabolism of Tryptophan on the influence of the intestinal microbiota and its products, often derived from Tryptophan, builds the crosstalk between the intestinal nervous system and the brain, with many substances acting on the enterochromaffin cells that are responsible for 5-HT production. IDO1 is also recognised for shifting the immune system towards Treg-mediated tolerance at the expense of the Th17-mediated inflammatory pathway. Kynurenine by-products use vitamin B as a co-factor increasing the demand of this substance to the body [24-27].
COVID-19 has been related to several changes in the central nervous system, including psychiatric and neurological changes such as mood changes, generalised anxiety, insomnia, the opening of acute schizophrenia, difficulty in sedation, intense neuromuscular pain, parkinsonian tremors, delay in returning to a state of wakefulness after a long period of orotracheal intubation. These changes seem to be linked to tryptophan metabolism since sudden depletion of 5-HT can cause acute disorders while maintenance of the hypoxemic state can cause and maintain mental and neurological disorders, as they allow the accumulation of toxic Kynurenine metabolites both in the peripheral nervous and other tissues such as the central nervous system (CNS).
5-HT is formed from Tryptophan in the CNS, pineal gland and intestine. In the pineal gland, 5-HT originates melatonin. This process is initiated by the action of tryptophan hydroxylase (TPH) that exists in 2 isoforms: TPH1 in the periphery and TPH2 in the central nervous system (CNS). Figure 1 summarises the idea contained in this article and the hypothesis that many neuropsychiatric signs and symptoms may originate in the pathways presented, with the main focus being the depletion of Tryptophan due to low intestinal absorption, internalisation of ACE-2 and hypoxemia and inflammation that direct the metabolism of Tryptophan to the formation of toxic by-products to the central nervous system, causing psychiatric and neurocognitive manifestations. It is essential to realise that these manifestations are reversible; when acting on inflammation and replacing substrate to form neurotransmitters, the patient improves the clinical picture, returning to its baseline status (Figure 2).
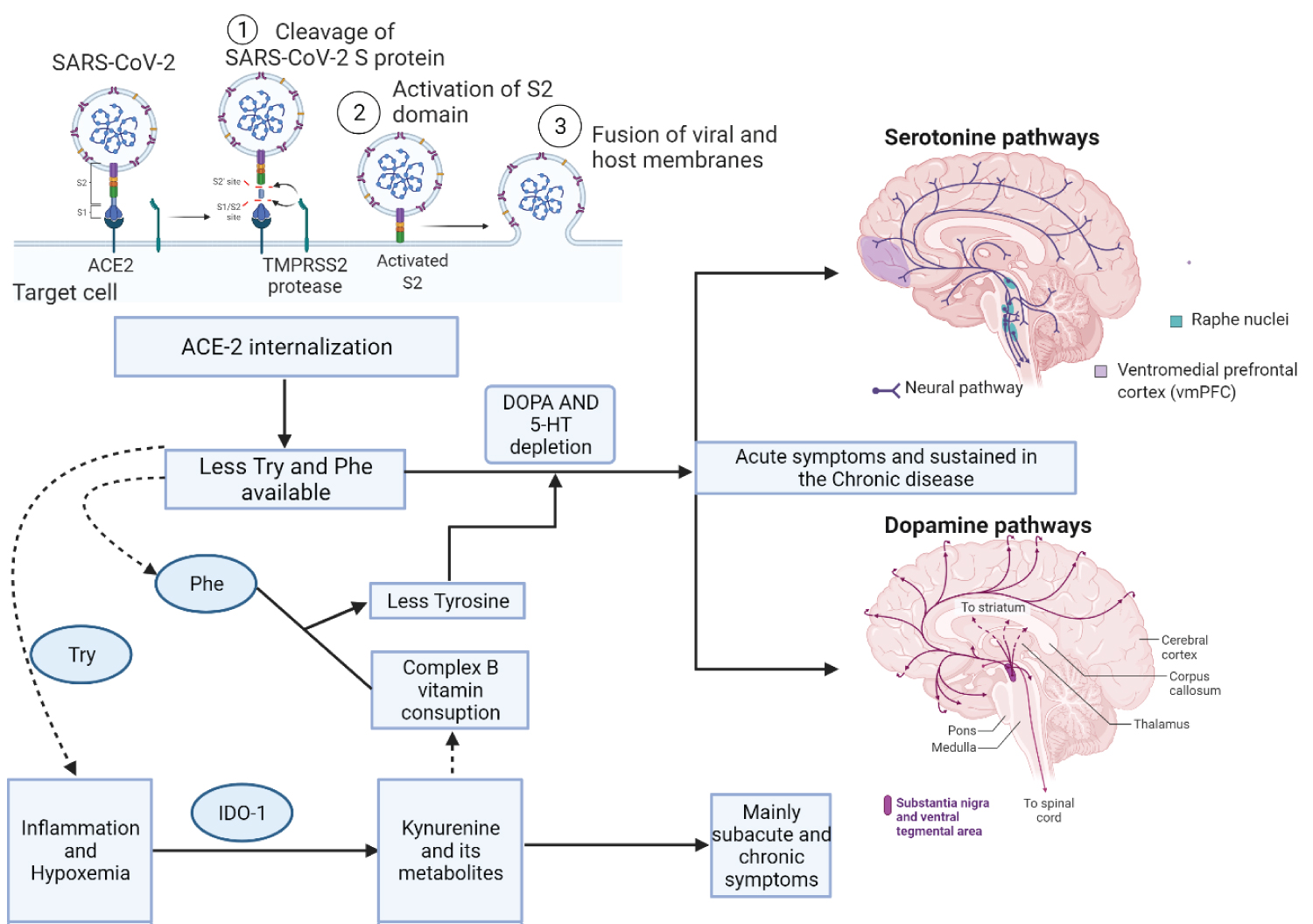 Figure 1: Changes in tryptophan and phenylalanine metabolism due to ACE-2 protein internalization in enterocytes: SARS-CoV-2 infection is systemic, causing many changes in the human body. One of these changes is the decreased absorption of some ACE-2 dependent amino acids. The acute depletion of tryptophan and phenylalanine causes acute psychiatric symptoms frequently observed in patients: mood disorders, generalized anxiety, insomnia. These symptoms are due to the sudden decrease in 5-HT and Dopamine in the central nervous system and the peripheral intestinal serotonin produced by the enterochromaffin cells, causing many metabolic and systemic changes. Lasting hypoxemia blocks many metabolic pathways, for example, the conversion of Try to Niacin (Vitamin B3 = NAD/NADH+). With the metabolism of Try performed by IDO1, there will be more formation of Kynurenine and its metabolites due to the inflammatory status. In the Central Nervous System, these metabolites can simulate rapidly progressive dementia, Alzheimer and Parkinson like symptoms, schizophrenia, somnolence and difficulty in sedation due to these metabolites, GABA and Glutamine receptors are saturated. These metabolites usually use B-complex vitamins as cofactors, promoting depletion of these substances, interfering in the conversion of Phe to tyrosine.
Figure 1: Changes in tryptophan and phenylalanine metabolism due to ACE-2 protein internalization in enterocytes: SARS-CoV-2 infection is systemic, causing many changes in the human body. One of these changes is the decreased absorption of some ACE-2 dependent amino acids. The acute depletion of tryptophan and phenylalanine causes acute psychiatric symptoms frequently observed in patients: mood disorders, generalized anxiety, insomnia. These symptoms are due to the sudden decrease in 5-HT and Dopamine in the central nervous system and the peripheral intestinal serotonin produced by the enterochromaffin cells, causing many metabolic and systemic changes. Lasting hypoxemia blocks many metabolic pathways, for example, the conversion of Try to Niacin (Vitamin B3 = NAD/NADH+). With the metabolism of Try performed by IDO1, there will be more formation of Kynurenine and its metabolites due to the inflammatory status. In the Central Nervous System, these metabolites can simulate rapidly progressive dementia, Alzheimer and Parkinson like symptoms, schizophrenia, somnolence and difficulty in sedation due to these metabolites, GABA and Glutamine receptors are saturated. These metabolites usually use B-complex vitamins as cofactors, promoting depletion of these substances, interfering in the conversion of Phe to tyrosine.
Phe: Phenylalanine; Try: Tryptophan; 5-HT Serotonin; IDO1 indoleamine 2,3-dioxygenase 1
View Figure 1
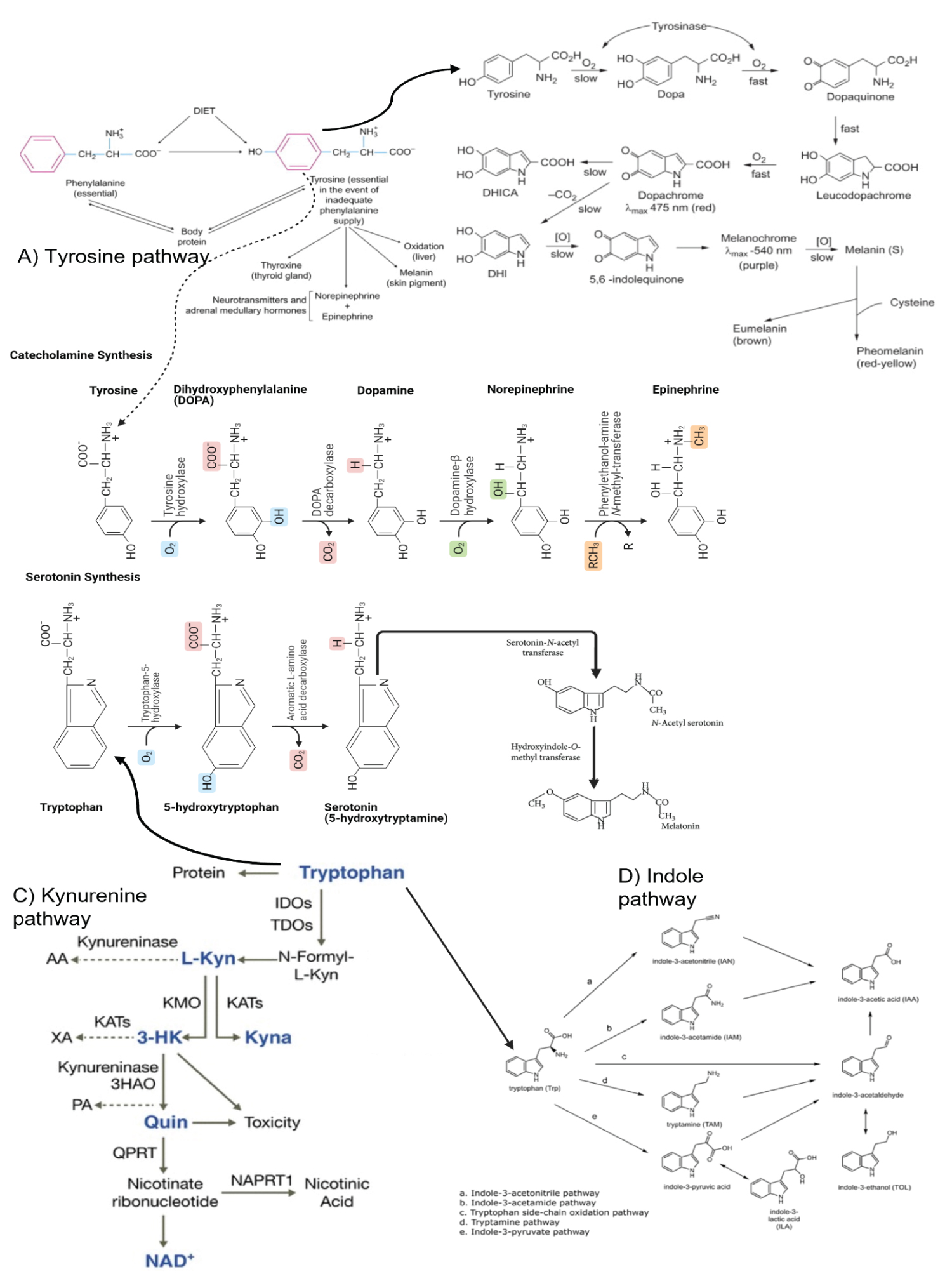 Figure 2: Serotonin and Dopamine Metabolism. In the context of SARS-CoV-2 infection, the internalization of ACE-2 will cause Try and Phe deficiency. Epithelial cells, macrophages, and dendritic cells (DCs) primarily secrete interferon-gamma and IDO1. IDO1 displaces the Try metabolic chain to produce kynurenine and its toxic metabolites. The interaction between the B7 complex on dendritic cells (DCs) with Cytotoxic T-lymphocyte Antigen-4 (CTLA-4) expressed in the membranes of regulatory T cells. This link induces and activates IDO1 in the DCs and is maintained by Transforming Growth Factor-β (TGF-β) and inflammatory mediators via non-canonical actions of Nuclear Factor-κB (NFκB). Hypoxemia further favours this process, as it blocks oxygen-dependent steps, such as for Niacin to be formed. Some toxic products are known to be found in diseases such as Alzheimer's and Parkinson's. QUIN leads acutely to human neuronal death and chronically to dysfunction by at least five mechanisms. Activation of the NMDA receptor; QUIN increases glutamate release by neurons and inhibits glutamate uptake by astrocytes leading to excessive microenvironment glutamate concentrations and neurotoxicity; 3-Hydroxykynurenine can produce neuronal damage by the induction of oxidative stress rather than action on glutamate receptors, also acts synergistically with QUIN and potentiates its toxicity. Kynurenic acid antagonizes all ionotropic glutamate receptors, including N-methyl-D-aspartate (NMDAR), kainic acid and α-amino-3-hydroxy-5-methyl-4-isoxazole (AMPAR) receptors; kynurenic acid non-competitively inhibits α 7-nicotinic acetylcholine presynaptic receptors (nAChRs). All pathways are lagged by the Try or Phe substrate deficit.
Figure 2: Serotonin and Dopamine Metabolism. In the context of SARS-CoV-2 infection, the internalization of ACE-2 will cause Try and Phe deficiency. Epithelial cells, macrophages, and dendritic cells (DCs) primarily secrete interferon-gamma and IDO1. IDO1 displaces the Try metabolic chain to produce kynurenine and its toxic metabolites. The interaction between the B7 complex on dendritic cells (DCs) with Cytotoxic T-lymphocyte Antigen-4 (CTLA-4) expressed in the membranes of regulatory T cells. This link induces and activates IDO1 in the DCs and is maintained by Transforming Growth Factor-β (TGF-β) and inflammatory mediators via non-canonical actions of Nuclear Factor-κB (NFκB). Hypoxemia further favours this process, as it blocks oxygen-dependent steps, such as for Niacin to be formed. Some toxic products are known to be found in diseases such as Alzheimer's and Parkinson's. QUIN leads acutely to human neuronal death and chronically to dysfunction by at least five mechanisms. Activation of the NMDA receptor; QUIN increases glutamate release by neurons and inhibits glutamate uptake by astrocytes leading to excessive microenvironment glutamate concentrations and neurotoxicity; 3-Hydroxykynurenine can produce neuronal damage by the induction of oxidative stress rather than action on glutamate receptors, also acts synergistically with QUIN and potentiates its toxicity. Kynurenic acid antagonizes all ionotropic glutamate receptors, including N-methyl-D-aspartate (NMDAR), kainic acid and α-amino-3-hydroxy-5-methyl-4-isoxazole (AMPAR) receptors; kynurenic acid non-competitively inhibits α 7-nicotinic acetylcholine presynaptic receptors (nAChRs). All pathways are lagged by the Try or Phe substrate deficit.
Tph tryptophan hydroxylase, IDO indoleamine 2,3-dioxygenase, TDO tryptophan 2,3-dioxygenase, L-Kyn L-kynurenine, AA Anthranilic acid, KMO Kynurenine 3-Monooxygenase, KATs Kynurenine aminotransferases, 3HAO 3-Hydroxyanthranilate -Dioxygenase, PA Picolinic acid, Quinolinic acid, QPRT Quinolinate phosphoribosyl transferase, NAD + Nicotinamide adenine dinucleotide, NAPRT1 Nicotinate phosphoribosyl transferase.
View Figure 2
Parkinson's symptoms result from Dopamine depletion by the internalisation of ACE-2, which, intensified by vitamin B6 depletion, causes low Dopamine titters (Figure 1). Dopamine anabolism requires Tyrosine as a substrate, and Tyrosine is formed from Phenylalanine (Phe). When Phe intake is limited, phenylalanine availability may be limited to produce Tyrosine, and Tyrosine can become a conditionally essential amino acid. Phe is used as an energy fuel; its complete oxidation requires biopterin, ascorbate, thiamine, riboflavin, niacin, vitamin B6, pantothenate, lipoate, ubiquinone, magnesium, and iron [28-31].
For these reasons, it is important do not use drugs that inhibit the serotoninergic and dopaminergic pathways. Quetiapine has been widely used in weaning from sedation so that the patient does not have delirium. Nevertheless, would not they present calm to be a reinforcement in the inhibition of already inhibited pathways, triggering a delay in returning to the state and wakefulness? Based on the theory proposed in this article, the NEROCOV/PSYCCOV (PSICCOV) protocol was created, whose purpose is to compare COVID-19 patients, both critical in ICU stay and hospitalized in the wards, taking the usual treatment - using Haloperidol and Quetiapine versus what is proposed in the protocol - using Citalopram, L-Dopa, vitamin B complex replacement and, if necessary, Corticosteroids. (NEUROCOV/PSYCCOV proposed protocol):
The corticosteroid must be given at a high dose, blocking the underlying inflammation and, after returning to baseline status [32-34].
1. Start replacing Tryptophan.
2. Other medications that can be administered are serotonin reuptake inhibitors, justified by the Fluvoxamine trial [35]. Vitamin B complex replacement is necessary since IDO1 and Kynurenine metabolites use vitamin B complex as a co-factor and, for this reason, B complex Niacin (B3) replacement which is NAD/NADH+ which, if depleted, interrupts the intracellular aerobic pathway of respiration, shifting ATP production to beta-oxidation of fatty acids (explanation in another submitted article). Vitamin B6 is also necessary for tyrosine metabolism.
3. L-DOPA replacement.
4. Avoid prolonged hypoxemia and perform orotracheal intubation "in time" as soon as the patient meets the criteria for intubation.
5. Prefer to use Ketamine.
The overlapping of social behaviour also interferes with the biochemical manifestations caused by SARS-CoV-2. It has been recurrent for young people, mainly among men who have sex with other men (MSM) who use methamphetamines in sexual practices -"chemsex"- to present acute psychiatric conditions. The drug itself can cause such symptoms, but methamphetamines can deplete already depleted serotoninergic and dopaminergic pathways. Methamphetamine is a selective agonist of trace amine-associated receptor 1 (TAAR1), whose activation increases cyclic adenosine monophosphate production and inhibits dopamine, norepinephrine, and serotonin reuptake reversing the direction of their transporters. The euphoria triggered by these drugs is well known, and their use in already depleted neurons can cause neuronal damage, apoptosis in addition to acute psychiatric manifestations. For these reasons, it is always important to ask, during the anamnesis, about the use of drugs and when they are part of the patient's behavioural practice [36,37].
COVID-19 still brings many mysteries related to its pathophysiology, but something concrete already exists: COVID-19 is a systemic disease with significant endocrine-metabolic effects. The opening of psychiatric and neurological conditions is constant in our medical practice when dealing with the patient COVID-19. The difficulty in sedating patients shows that the Kynurenine metabolites may be coating the receptors where the central sedatives and inducers act to perform orotracheal intubation. Kynurenic acid (KYNA), quinolinic acid (QUIN) and 3-hydroxykynurenine (3-HK) are toxic metabolites derived from Kynurenine, whose effects on the CNS are known, mimicking symptoms of schizophrenia, Parkinson's disease, and Alzheimer's disease [38,39]. An increase in the levels of the NMDA receptor agonist quinolinic acid and a simultaneous decrease in neuroprotective metabolites have been observed in suicidal patients; for this reason, I consider it essential not only to justify psychiatric symptoms as being caused by issues of social isolation but also by biochemical changes where we can and must act. Furthermore, we have to consider autoimmune meningoencephalitis due to SARS-CoV-2 infection and autoantibodies produced against NMDA receptors [40].
Based on clinical observation and the literature review, this theory (Kynurenine metabolites) is plausible and may warrant what we have observed daily dealing with COVID-19 patients. Deficient absorption of Try happens in Hartnup disease, an autosomal recessive disorder caused by impaired neutral amino acid transport in the apical brush border membrane of the small intestine and the proximal tubule of the kidney. In Hartnup disease, many skin lesions can occur: vesiculobullous eruption with exudation, photosensitivity, dry, scaly, and well-defined eruptions, sometimes resembling chronic eczema [41]. Lesions on the face like lupus erythematosus. COVID-19 patients have Pellagra disease.
The use of corticosteroids, personally preferably Methylprednisolone, acts on inflammation through multiple pathways. Because it is a nonspecific drug, it brings this positive feature when we do not yet know all the pathways that activate or perpetuate inflammation in the "Long COVID-19". Blocking inflammation in the patient and replacing substrates, such as Tryptophan, vitamin B complex and using medications such as Selective serotonin reuptake inhibitors (SSRIs)and L-DOPA, have also shown satisfactory results in an observational way in hospitalised patients and those undergoing outpatient follow-up.
I have no idea when patients will restore their homeostasis after affected by COVID-19. However, from the reviewed literature, it is possible to infer that by inhibiting NF-κB, the body might express ACE-2 again. For this reason, I reaffirm the importance of using corticosteroids or the association of anti-inflammatory drugs to use corticosteroids in smaller amounts and for less time.
These drugs need multicentric trials, in addition to my subjective experience in these nearly two years of COVID-19. I reinforce that we must recognise our limitations in the face of the new, as this is how we will need to study to understand what challenges us. If we assume that we know everything, we are doomed to failure.
Finally, it is essential to consider the general depletion of catecholamines can intensify and aggravate the vasoplegic and inflammatory shock in COVID-19, as the natural supply to maintain vasoconstriction is exhausted. In addition, the lack of Tryptophan can reduce the production of protective indoles by bacteria in the intestinal microbiota, favouring, for example, the growth of Candida sp. There are many theories and hypotheses, but the exciting thing is that all this is plausible, and we must think about these hypotheses and discuss them [42,43].
This article reinforces the need to open our eyes to a new disease that can mimic symptoms of already known diseases, and if we use the usual treatments, we can cause damage instead of promoting the process of resolution and healing. We must not be iatrogenic, and for this not to happen, it remains for us to study and recognise that our knowledge is limited.
To all COVID-19 patients who have died from COVID-19 and those living with, as yet ununderstood, sequelae caused by SARS-CoV-2.
To the HSPE (Hospital do Servidor), who offered me the treasure of observing patients with COVID-19 and allowing me to act on the disease.
I do not have any conflict of interest to disclosure. I am a public servant of the State of São Paulo, Brazil and my interest is focused on working to improve the public health by researching and making the care of the patients sustained by good medical practices.
This manuscript did not receive any funding to be made.
This manuscript has a unique author, Luiz Zanella, and I am responsible for all the content written in this manuscript; also, I built the figures on the BioRender.com platform.
1. Covid-19 patients, regardless of disease severity, have presented neuropsychiatric manifestations.
2. There are reports of mood disorders, depression, acute schizophrenia, generalized anxiety, long-lasting neuropathy in critically ill patients after an ICU stay.
3. In COVID-19, neuropsychiatric symptoms are much more intense and lasting than they were usually seen, and difficulty in sedating the patient COVID-19, which is not seen recurrently, except in rare exceptions, such as in addicted patients to illicit drugs.
4. The ACE-2 protein is responsible for absorbing Tryptophan and Phenylalanine in the intestine.
5. The ACE-2 internalization occurs when SARS-CoV-2 infects the cell, generating a deficit of these two cited amino acids in the body.
6. Tryptophan originates serotonin, melatonin, vitamin B3, Kynurenine, whose metabolites are toxic to the Central Nervous System. Phenylalanine is the precursor of Tyrosine, the basis for forming catecholamines such as Dopamine, Norepinephrine and Epinephrine.
7. Some of the Kynurenine's toxic metabolites are founded in diseases such as Parkinson's and Alzheimer's.
8. SARS-CoV-2 also allows an intense formation of immune complexes and autoantibodies, which can induce specific antibodies for receptors in the Central Nervous System, causing meningoencephalitis.
9. Tryptophan is a substrate to form Niacin (Vitamin B3). Niacin is NAD/NADH+, whose production is reduced by the lack of Try and hypoxemia that inactivates enzymatic processes necessary for its formation.
10. Hypoxemia and inflammation shift the Tryptophan metabolism towards the formation of Kynurenine and its metabolites, resulting in less availability of the already deficient Try to the Central Nervous System and the Niacin pathway. Thus, ACE-2 interferes in the absorption of Try and Phe, resulting in NPSs in COVID-19.
11. Avoid prescribing medicines that inhibit 5-HT or DOPA pathways or both ones. This review shows the theoretical necessity of prescribing Vitamin B complex, Selective Serotonin Reuptake Inhibitors (SSRIs), L-DOPA, Tryptophan, and performs Clinical Trials to prove these efficacy drugs versus the current use of Quetiapine.