Receptor for advanced glycation end products (RAGE) activation is known to play an important role in the development of diabetes complications by amplifying the inflammatory process. Herein, we examined the effect of polyclonal antibodies against human RAGE (Ab anti-hRAGE) on "primed" peripheral blood mononuclear cells (PBMNC by hyperglycemia in type 2 diabetes mellitus (T2DM) patients in comparison to healthy control. ROS generation and IL-1β, IL-6 and TNF-α secretion were studied.
PBMNCs were purified utilizing Ficoll-hypaque gradient. ROS was quantified by luminol-dependent chemiluminescence. Antibodies anti- human RAGE (AGE-like) was purched from Sigma Co. The cytokines were from PBMNC culture in presence or in the absence of anti-hRAGE. IL-1β, IL-6 and TNF-α were quantified in the supernatant of Ab anti-hRAGE-stimulated PBMNCs trough ELISA.
Ab anti-hRAGE (AGE-like) significantly inhibited ROS production in unstimulated or stimulated PBMNCs from T2DM and healthy controls in a similar way. The percentage of inhibition was greater in primed PBMNC by hyperglycemia in diabetes (T2DM patients). In contrast, anti-hRAGE increased secretion of IL-1β, IL-6 and TNF-α (p < 0.05) in the supernatant of Ab anti-hRAGE-stimulated PBMNCs from T2DM patients compared with healthy controls (p < 0.05).
The effect of antibody anti-hRAGE (AGE-like) in primed cells by hyperglycemia in diabetes may activates different signaling network when interact with RAGE on cells surface. Dual results induced by anti-hRAGE (AGE-like) associated with oxidizing response and pro-inflammatory cytokine secretion suggest activation of several signaling network in AGE-RAGE interaction. It may have consequences on innate immunity.
Diabetes, RAGE, Antibody anti-human RAGE, ROS, Cytokines, Inflammation
Diabetes mellitus is a immunological disease characterized by metabolic deregulation and inflammation [1]. Hyperglycemia of diabetes is associated with increase in the generation of reactive oxygen species (ROS) and the formation of advanced glycation end products (AGEs) [2-6]. AGEs interact with their respective receptors (RAGEs), a multiligand member of the immunoglobulin superfamily of cell surface molecules, inducing activation of cascade of several signaling pathways associated with diabetes vascular complications [7-10]. In addition to AGEs, RAGE is a receptor for amyloid fibrils, S100/calgranulins and high mobility group box 1 (HMGB-1) protein [11-13]. It is expressed in many types of cells at low levels, such as endothelium, monocytes, macrophages, T-lymphocytes, neuronal cells, and glomerular epithelial cells [14-16]. But, in pathological conditions, RAGE is upregulated after interaction with respective ligands [17]. Activation of RAGE transduces activating signals for: i) NADPH oxidase system; ii) reactive oxygen species (ROS) production; iii) p38 MAPK; iv) extracellular-signal-regulated kinase 1/2; v) Rac; vi) protein kinase C; vii) phosphatidylinositol-3 kinase (PI3K); viii) intracellular calcium (Ca2+) mobilization; and ix) IκB kinase-β (IKKβ) in vascular endotheliums, smooth muscle cells and monocytes. These signaling pathways iniciate and sustain the activation NFkappaB target genes leading to the production and secretion of proinflammatory cytokines, chemokines and adhesion molecules, such as IL-1β, IL-6, TNF-α, E-selectin, vascular endothelial growth factor (VEGF), vascular cell adhesion molecule (VCAM)-1 and intercellular adhesion molecule (ICAM) [18-28]. There are few reports with cells directly associated with innate immunity and it is possible to suggest consequences on signaling of AGE-RAGE interaction induced by hyperglycemia in diabetes. RAGE activation is suggested to be associated with development of diabetes complications by amplifying the inflammatory process [10]. Soluble RAGE (sRAGE) and monoclonal antibodies anti RAGE have used as therapeutic resource for neutralize AGE and to block the interaction AGE-RAGE on cell surface. It has been suggested that hyperglycemia in diabetes may prime cells metabolically inducing altered cellular reactivity [29]. Herein, we examined peripheral blood mononuclear cells (PBMNC) primed by hyperglycemia from T2DM patients in comparison to PBMNC from healthy non-diabetic control. We use polyclonal antibodies against human RAGE (Ab anti-hRAGE) as AGE-like. ROS generation and IL-1β, IL-6 and TNF-α secretion in hyperglycemia "primed" PBMNCs from Type 2 diabetes mellitus (T2DM) patients and healthy controls were studied.
This study was approved through the Ethical Committee of Santa Casa Hospital (Belo Horizonte - MG, Brazil), and written informed consent was obtained from all participants prior to the commencement of the study.
T2DM patients (n = 20), diagnosed according to the criteria of the American Diabetes Association [30], and healthy controls (n = 20), ranging 47-70 years of age, were recruited from the Endocrinology Department of Santa Casa Hospital. Type 2 DM patients were treated with statins and beta-blockers in addition to hypoglycemic drugs. Prior to the study, all volunteers received complete physical examinations, and detailed evaluations of medical histories and laboratory analyses were performed (Table 1). Pregnant women, individuals suffering from alcoholism, infection, inflammation, dementia or malignant diseases and smoking addictions were excluded from this study.
Table 1: Clinical and biochemical characteristics of the studied population. View Table 1
The following reagents were purchased from Sigma-Aldrich (St. Louis, Mo, USA): Rabbit antibody anti-human RAGE (Ab anti-hRAGE, cat. #SAB1401326, Lot. 09112), NADPH oxidase inhibitor [diphenyliodonium chloride (DPI); cat. # 43088] and Phorbol 12,13-dibutyrate (PDB, cat. # P1269).
PBMNCs were purified from 10.0 mL of heparinized venous blood, using a Ficoll-Hypaque gradient as previously described [31], with slight modifications. The trypan blue exclusion test showed that the cell viability in all samples was of > 95%.
Modulations in the generation of ROS were estimated using the luminol quantitative chemiluminescence assay in a Magic Lite luminometer, (Ciba Corning Co., Medfield, MA, USA). The PBMNCs sample was washed in phosphate buffered saline (PBS) and a suspension containing 1 × 106 PBMNCs/mL PBS was transferred to an unsealed luminescence tube. Luminol (200 μL) dissolved in 0.4 M dimethyl sulphoxide was added to the sample, the final volume of the mixture was adjusted to 500 μL with PBS. The chemiluminescence [expressed in relative ligh units (RLU)/min] of each assay mixture was measured for 15 min (basal ROS production), following which addition of the Ab anti-hRAGE (100 ng/100 μL) to the reaction mixture and chemiluminescence measured for 40 min. Finally, NADPH oxidase inhibitor (DPI) (10 μM; 100 μL) was added to the reaction mixture and chemiluminescence measured for additional 15 min. The chemiluminescence was also made in the presence of PDB. In this case, basal ROS production was measured for 15 min, following which 20 μL of 10-4M PDB was added and ascendant ROS production was determined for 25 min; finally Ab anti-hRAGE (100 ng/100 μL) was added and chemiluminescence recorded for a further 20 min.
Aliquots (100 μl) of PBMNCs suspension (1 × 106/ mL) from T2DM and healthy controls in Dulbecco's modified Eagle's medium (DMEM), supplemented with 10% fetal bovine serum (FBS), were incubated in the absence or presence of Ab anti-hRAGE (100 ng/100 μL) for 72 hours at 37°C under 5% CO2. The final volume was adjusted to 300 μL in DMEM supplemented with 10% FBS. After incubation, the cells were centrifuged and the supernatant collected. The interleukin (IL) 1-β (IL-1β human EIA Kit - Enzo Life Sciences, Inc., New York, USA ), IL-6 (IL-6 human EIA Kit - Enzo Life Sciences, Inc., New York, USA) and tumour necrosis factor-alpha (TNF-α human EIA Kit - Enzo Life Sciences, Inc., New York, USA) concentrations were determined through enzyme-linked immunosorbent assay (ELISA).
The values are presented as the means ± standard deviation (SD). The nonparametric Kolmogorov-Smirnov test was used to assess the normal distribution of the continuous variables. Comparisons between groups were performed using unpaired Student's t-tests. In some experiments, we also used the chi-square test. All analyses were considered significant at p-values < 0.05 using Origin 6.0 (Microcal Software Inc., Northampton, MA, USA).
ROS, expressed as RLU/min, produced by unstimulated PBMNCs from T2DM patients produced higher levels of ROS (186.6 ± 39.0) compared to healthy controls (96.6 ± 11.0) (p < 0.05). The use of polyclonal antibodies against human RAGE suppressed ROS generation 29% and 19% in cells from T2DM and healthy controls, respectively (p > 0.05 by chi-square test). Similar suppression of ROS production was observed in PBMNCs assayed in the presence NADPH oxidase inhibitor (DPI). The percentages of inhibition were 48.0% for T2DM and 31.0% for healthy control (p > 0.05) (Table 2 and Figure 1). PBMNC from patient were more sensitive to inhibition than that from healthy control.
 Figure 1: Effects of Rabbit antibody anti human RAGE (Ab anti-hRAGE) and NADPH oxidase inhibitor [diphenyliodonium chloride (DPI)] on ROS production by human peripheral blood mononuclear cells (PBMNCs) from Type 2 diabetes patients (T2DM) and healthy controls. Each point represents the average of 10 experiments ± SD. RLU = Relative Light Units.
View Figure 1
Figure 1: Effects of Rabbit antibody anti human RAGE (Ab anti-hRAGE) and NADPH oxidase inhibitor [diphenyliodonium chloride (DPI)] on ROS production by human peripheral blood mononuclear cells (PBMNCs) from Type 2 diabetes patients (T2DM) and healthy controls. Each point represents the average of 10 experiments ± SD. RLU = Relative Light Units.
View Figure 1
Table 2: Effect of human RAGE antibody and NADPH oxidase inhibitor diphenyliodonium chloride on reactive oxygen species (ROS) production in human peripheral blood mononuclear cells from T2DM and healthy controls. View Table 2
The role of Ab anti-hRAGE on ROS production by PBMNCs PDB-stimulated is shown in table 2 and figure 2. The phorbol ester activated PBMNCs-ROS derived from T2DM and healthy contols, the results, expressed as percentage of activation, were 166% and 67% respectively (p < 0.05). The PBMNCs-ROS derived PDB-stimulated was inhibited by Ab anti-hRAGE. The results expressed as percentage of inhibition were 55% and 53% for T2DM and healthy controls, respectively. However, the kinetics studies (Figure 1 and Figure 2) demonstrate that the effect of anti-RAGE was more effective in PBMNC from patients, suggesting possible effect of hyperglycemia.
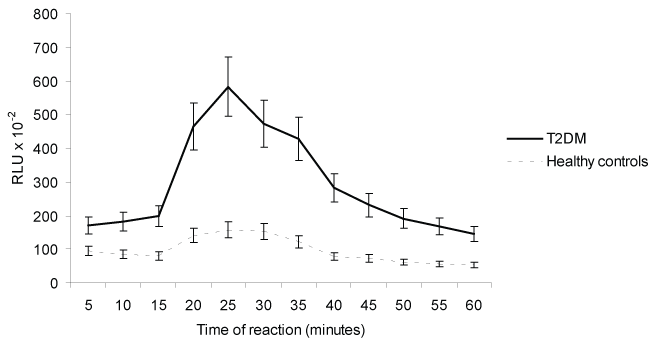 Figure 2: Effects of Rabbit antibody anti-human RAGE (Ab anti-hRAGE) on ROS production in PDB-stimulated human peripheral blood mononuclear cells from T2DM and healthy controls. Each point represents the average of 3 experiments ± SD. RLU = Relative Light Units, PDB = phorbol 12,13-dibutyrate ester.
View Figure 2
Figure 2: Effects of Rabbit antibody anti-human RAGE (Ab anti-hRAGE) on ROS production in PDB-stimulated human peripheral blood mononuclear cells from T2DM and healthy controls. Each point represents the average of 3 experiments ± SD. RLU = Relative Light Units, PDB = phorbol 12,13-dibutyrate ester.
View Figure 2
As depicted in figure 3, hRAGEab activated the secretion of IL-1beta and IL-6 in PBMNCs from T2DM, and stimulated the secretion of TNF-alpha in cells from both studied groups (p < 0.05). The results of the induced effect of hRAGEab in PBMNCs, expressed as mean ± SD, were: IL-1beta: 61.1 ± 13.0 and 22.8 ± 12.4; IL-6: 446.1 ± 71.0 and 159.0 ± 59.9; TNF-alpha: 171.7 ± 42.0 and 108.7 ± 35.7, for T2DM and healthy controls, respectively. Figure 3 also demonstrated that PBMNCs from T2DM produced significantly (p < 0.05) higher amount of IL-1beta (33.9 ± 12.4), IL-6 (312.3 ± 81.2) and TNF-alpha (105.6 ± 13.1) as compared with cells from healthy control (IL-1beta: 17.2 ± 5.0, IL-6: 128.1 ± 37.2, TNF-alpha: 68.9 ± 10.0).
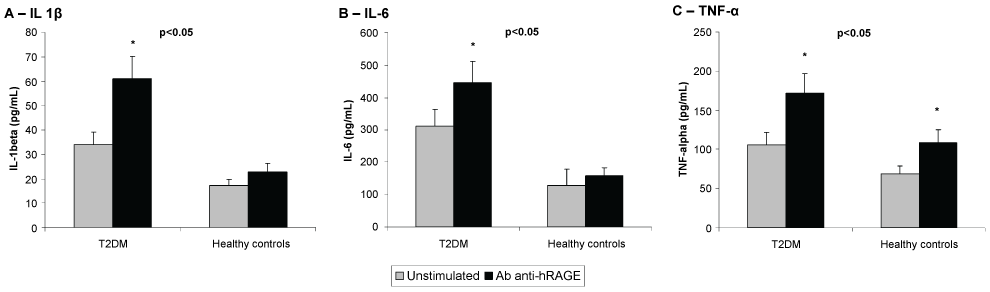 Figure 3: Effects of Rabbit antibody anti human RAGE (Ab anti-hRAGE) on the secretion of IL-1β (A), IL-6 (B) and TNF-α (C) by human peripheral blood mononuclear cells (PBMNCs) from Type 2 diabetes patients (T2DM) and healthy controls. Data as means (pg/mL) ± SD. p < 0.05 T2DM vs. healthy controls; *p < 0.05 vs. unstimulated (Student's t-test). n = 10 for each group.
View Figure 3
Figure 3: Effects of Rabbit antibody anti human RAGE (Ab anti-hRAGE) on the secretion of IL-1β (A), IL-6 (B) and TNF-α (C) by human peripheral blood mononuclear cells (PBMNCs) from Type 2 diabetes patients (T2DM) and healthy controls. Data as means (pg/mL) ± SD. p < 0.05 T2DM vs. healthy controls; *p < 0.05 vs. unstimulated (Student's t-test). n = 10 for each group.
View Figure 3
The present study demonstrated that polyclonal antibodies against human RAGE (AGE-like) enhanced cytokines secretion and inhibited ROS generation in PBMNC from type 2 diabetic patients. It has been reported that the pathological consequences of interaction between RAGE with respective ligand leads to the generation of oxidative stress and upregulation of inflammatory pathways [9,32-34]. The blockade of RAGE with ligands has been suggested as a possible therapeutic target for controlling inflammatory process. In diabetic animal models, the blockade of RAGE, using soluble RAGE (sRAGE), demonstrated reduction in vascular inflammation and atherosclerotic lesion area and suppression in periodontitis-associated alveolar bone loss [10,35-37]. RAGE deficient animal models showed improvement of nephropathy, suppression of kidney injury and diabetes-accelerated atherosclerosis [38-40]. Moreover, anti-RAGE antibodies increased survival in experimental models of severe sepsis, protected against AGE-mediated podocyte dysfunction and suppressed pro-inflammatory activities of human umbilical venular endothelial cells (HUVECs) induced by HMGB1 [41-44]. Thus, theoretically, the use of antibodies anti-RAGE could down-regulate inflammatory signaling pathways.
ROS overproduction hyperglycemia-induced result in oxidative stress and it plays a central role in the pathogenesis of diabetes complications [5,6]. Activation of RAGE results in the generation of ROS in dependence of NADPH oxidase [45]. Our results (Table 2 and Table 3) are not in agreements with other authors [18,27,45]. Antibodies anti-hRAGE down-regulated ROS production in PBMNC either from T2DM or from healthy controls (Table 2, Table 3, Figure 1 and Figure2). It can be due to experimental design we have used. Polyclonal antibodies anti-hRAGE could interact simultaneously with several epitopes on RAGE molecule while monoclonal antibodies or natural AGE interacts with a specific region or epitope. Several metabolic signaling simultaneously activated resulted in different metabolic response. In order to confirm our present results, we performed experiments using an NADPH inhibitor (DPI). The addition of NADPH oxidase inhibitor to PBMNC challenged with antibodies anti-hRAGE showed an addictive effect in reducing ROS production in PBMNC (Table 2 and Figure1). Both PBMNC from T2DM patients and from healthy controls showed similar and comparable metabolic response in the presence of Ab anti-hRAGE. However the inhibition in hyperglycemia primed PBMN showed greater when compared to healthy control (p < 0.05) It suggests that the effect of Ab anti-hRAGE on RAGE can affected by hyperglycemia in diabetes. We studied down regulation of ROS production mediated by anti-hRAGE on PBMNC previously stimulated with a protein Kinase C (PKC) activator. Our results demonstrated that the activation of ROS production induced by PDB ( phorbol ester) was greater in PBMNMC from patients and was fully reversed by Ab anti-hRAGE in cells from T2DM and healthy controls in similar percentage of inhibition (55 and 53%, respectively) (Table 3 and Figure 2). It may suggests that the effect of antibodies anti-hRAGE (AGE-like) depends on PKC and /or NADPH-oxidase signaling pathways. Our present data demonstrated that polyclonal antibody anti-hRAGE induced ROS (oxidizing response) inhibition and increased cytokine secretion by PBMNC from T2DM. It suggests a dual effect for antibodies anti-hRAGE (AGE-like). Inflammatory changes observed in the presence of hyperglycemia have been associated with NF-kappaB activation [46,47]. It is also described that RAGE activation leads to sustained and chronic activation of NF-kappaB [32-34]. Surprisingly, our results showed that antibodies anti-hRAGE (AGE-like) increased secretion of pro-inflammatory cytokines (IL-1β, IL-6 and TNF-α) in cultured PBMNCs from T2DM, but inhibited ROS generation. Secretion of TNF-alpha was stimulated in cells from both studied groups (Figure 3). The increase of IL-1β may suggest the involvement of inflammasome in diabetic complications. It has been reported that increased levels of TNF, IL-1β and expression of NRLP3-inflammasome are associated with endothelial dysfunction and progression of atherosclerosis [48-50]. Ruscitti et al. [50] demonstrated that patients with diabetes and Rheumatoid Arthritis (T2D/RA) increase IL-β when compared with other groups. Antibodies anti-RAGE has no effect on periodontitis, but down-modulated renal complication in diabetic rats and improves neovascularization in the ischemic leg treatment in diabetic mice [50-52]. The use of antibodies anti-RAGE leads to a controversial results and needs further studies.
Table 3: Effect of human RAGE antibody on reactive oxygen species (ROS) production in PDB-stimulated human peripheral blood mononuclear cells from T2DM and healthy controls. View Table 3
Our results suggests that antibodies anti-hRAGE act as AGE-like ligand potentiating the inflammatory response activating different signaling pathways associated to cytokine secretion in cells from patients in comparison to that from healthy control.
It suggests a very complicated phenomenon with mechanism still not fully known. Thus, the use of experimental and therapeutic antibodies, such as, mono or polyclonal antibodies anti-RAGE, need to be used carefully and reinforce the suggestion that the role of AGE-RAGE in pathogenesis of diabetic complications is more complex than it seems.
In conclusion, the effect of antibody anti-hRAGE (AGE-like) in primed cells by hyperglycemia in diabetes may activates different signaling network when interact with RAGE on cells surface leading to a dual results associated with oxidizing response and pro-inflammatory cytokine secretion. It may have consequences on innate immunity.
The authors confirm that there are no conflicts of interest.
The authors would like to thank FAPEMIG, CNPq, CAPES and Rede Mineira de Toxina Terapêutica 26/12.