Trypanosomosis is a vector-borne, tropical disease that causes mortality and morbidity in livestock and humans. In this study we investigated the risk factors for trypanosome infection in cattle in the Maasai Steppe of northern Tanzania. We assessed the influence of age, sex, herd size and history of treatment against trypanosomosis as risk factors of trypanosome infection. Cattle blood samples were collected from 150 cattle in three villages in the vicinity of Tarangire National Park, which acts as a reservoir of tsetse flies, the trypanosome vector. Parasite species were identified using a nested Polymerase Chain Reaction (n-PCR). Smaller herd sizes, young age (1-2 years), and male sex significantly increased the risk of trypanosome infections. Efforts to control trypanosome infection should be strategically based on location and season while considering age, treatment and herd size as risk factors.
Risk factors, Trypanosome infection, Northern Tanzania
AAT: Animal African Trypanosomosis; HAT: Human African Trypanosomiasis; DNA: Deoxyribonucleic Acid; QGIS: Quantum Geographical Information System; PCR: Polymerase Chain Reaction; NMAIST: Nelson Mandela African Institution of Science and Technology; SUA: Sokoine University of Agriculture; PEHPL: Program for Enhancing Health and Productivity of Livestock
Northern Tanzania, in particular the Maasai Steppe, is an area where African Animal Trypanosomosis (AAT) is endemic amongst the cattle of pastoralists [1,2] due to the presence of wildlife and high tsetse fly abundance [3-6]. The effects of AAT on cattle are well documented, and can have substantial impacts on pastoral livelihoods. Depending on the relative severity of the disease, the impacts include: reduced herd fertility, increased abortions, low birth weights, slow calf growth, reduced milk yield [7] and beef quality. This results in increased treatment costs and low revenue for the owners, and can substantially impact rural livelihoods [7]. Despite the significant burden of the disease, little is known about the associated risk factors in cattle.
Different Trypanosome species are responsible for infections in cattle, small stock, and also in humans. Trypanosoma congolense, T. brucei and T. vivax cause trypanosomosis in cattle while humans are infected only by two subspecies: T. brucei rhodesiense (common in East Africa) and T. brucei gambiense (common in West and Central Africa). Trypanosome infections in cattle have been reported in different countries inside and outside Africa. For instance, in Kenya it was observed the prevalence of trypanosome infection in cattle goes up to 41% for T. vivax which is infective in cattle [8]. Similar trends have also been reported in Nigeria where T. vivax and T. brucei were observed to infect cattle [9,10]. Like other countries, Tanzania is also at high risk of trypanosome infection in cattle and other animals, including humans. Several studies in Tanzania showed that cattle acquire trypanosome infections, which impairs cattle productivity by causing mortality to cattle [11]. For instance, the finding from Simanjiro district by [12] revealed that, 24244 cattle had trypanosomosis disease while 10.51% died in the year 2011 to 2013.
Previous studies have revealed that trypanosome infections in cattle could have been enhanced by the co-existence of wildlife, humans and livestock in the ecosystem. This is because the wildlife are reservoirs for the parasite, also due to the continued circulation of parasites among the host species (wildlife, livestock and human) [2]. Our study focused on the Maasai Steppe, which is dominated by transhumant Maasai pastoralists, who move with their cattle to search for pasture and water. In many cases, this increases the interaction of livestock with wildlife, known to be reservoirs of trypanosome parasite [1]. The management system of their cattle varies among age groups, sex and herd size. For instance, calves are left at home while older age groups are taken further afield to search for pasture and water. Also, treatment against trypanosomosis by pastoralists is based on animal age, herd size, sex and season, and is often done by the pastoralists themselves. However due to improper administration of trypanocides by pastoralists, there is risk of drug resistance developing in the cattle [13].
Targeted, effective control strategies to reduce trypanosome infections in cattle can be developed if we first know the risk factors that explain variation in infection among cattle. Studies undertaken at other locations have noted several factors associated with increased prevalence of trypanosome infection. For instance, a study done in Ogun state Nigeria showed that prevalence of trypanosome infection varied among breeds of cattle and sex with a significantly higher prevalence in female than male animals. The same study also revealed that animals aged 5 years or older showed higher prevalence than younger age groups, which the authors associated with changes in grazing behavior which exposed the animals to the vectors [14]. In contrast, a study undertaken in Morogoro, Tanzania revealed that sex, breed, grazing system, farm size and acaricide application were not significant risk factors influencing likelihood of trypanosome infection [15]. In Barinas State, Venezuela it was observed that age, dirt floors as socio-economic indicator, distance from houses to palm trees and congenital transmission are among the risk factors for Trypanosoma cruzi [16]. The variation in risk factors revealed in the different studies indicates the need to assess risk factors within locally-relevant contexts. The Maasai Steppe is an important area for livestock production, and is known to have a high burden of AAT [2,3,17] and it is therefore necessary to assess the risk factors that influence infections in cattle, to better combat the negative effects of the disease.
This study focused on assessing the influence of sex, age, treatment history against trypanosomosis, previous infection, and herd size on trypanosome infection rates in cattle in three villages near a protected area in the Maasai Steppe. The findings from this study are essential for evidence-based trypanosomosis control strategies aimed at minimizing the acquisition of new infections in areas where humans, livestock and wildlife interact and share resources. Proper control of trypanosomosis in cattle would improve the productivity of cattle, socio-economic welfare, and food security of pastoralists.
This study was conducted in three villages in the Maasai Steppe of Northern Tanzania: Sukuro, Kimotorok and Oltukai (Figure 1), in the months of July and October of 2017 and January of 2018. The Maasai Steppe ecosystem is located in northeastern Tanzania with wildlife-livestock -human interaction. The area covers approximately a total of 40,000 square kilometers (15,444 square miles) which is slightly larger than the state of Maryland, USA. The villages were chosen for their proximity to Tarangire National Park which is home to wildlife species that act as hosts for the trypanosome parasite [18] and an important food resource for the tsetse vector. The area is occupied mainly by Maasai pastoralists who keep livestock such as cattle, goats, sheep and chickens for subsistence and commercial purposes. Common wildlife species found in Tarangire National Park and other protected areas in the Maasai Steppe (i.e Lake Manyara National Park, Wildlife Management Areas and ranches) include the eastern blue wildebeest/white-bearded wildebeest (Connochaetes taurinus albojubatus), cape buffalo (Syncerus caffer), common eland (Taurotragus oryx), zebra (Equus quagga), impala (Aepyceros melampus), african elephant (Loxodonta africana), lion (Panthera leo), cheetah (Acinonyx jubatus), warthog (Phacochoerus africanus) and Wild Dog (Lycaon pictus) [19]. Wildlife typically use the protected areas seasonally, moving into surrounding pastoral lands during the rainy season, and thereby increasing interactions with humans and livestock, and facilitating the circulation of trypanosome infections in cattle.
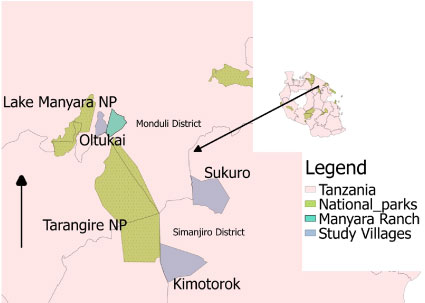 Figure 1: Map showing the three study villages: Sukuro and Kimotorok in Simanjiro District and Oltukai in Monduli District.
View Figure 1
Figure 1: Map showing the three study villages: Sukuro and Kimotorok in Simanjiro District and Oltukai in Monduli District.
View Figure 1
With the help of local government leaders, a list of pastoralists who keep cattle in each village was obtained, and random sampling was used to obtain the five bomas selected for cattle blood sampling. A boma is a traditional compound consisting of a variety of "houses" which look like small huts built with mud and cow dung.
In each boma, 10 cattle were selected randomly at the beginning of the study (July 2017), and then bled, making a total of 150 cattle sampled. Blood samples were collected from the same 10 cattle in each boma in October 2017 and January 2018. However, if any animal sampled at the initial session was no longer in the herd during the second or third sampling sessions, such an animal was excluded from the study. This led to the following sample sizes being included in the analyses after all three sampling periods: 137 blood samples from Kimotorok village, 135 from Sukuro, and 139 from Oltukai. Three milliliters of blood was collected from the jugular vein by venipuncture into an EDTA-containing vacutainer tube. The blood was kept in ice boxes and transported to the laboratory at the Nelson Mandela African Institution of Science and Technology (NMAIST) where it was kept at -20 °C until DNA extraction.
During each sampling session, the cattle were given a prophylactic dose of diminazene aceturate to clear any trypanosome infections. This was done each time after blood sampling in order to clear existing trypanosome infections and monitor newly-acquired infections. Diminazene aceturate is known to be effective for 20-21 days in cattle [20,21], and therefore the cattle could only acquire new infections beginning 21 days after the drug.
DNA was extracted from cattle blood as described in the protocol by Quick-gDNA blood mini prep kit (D3017, Zymo Research, USA) and stored at -20 °C until further analysis. ProFlex™ PCR System Thermocycler (Applied Biosystems®, Life technologies) was used to run the nested ITS-PCR for detection of trypanosome infections. The extracted DNA (2.5 μL) was added to 6.25 μl of PCR master mix (Quick-load® Taq 2 × Master mix, BioLabs, New England, USA) containing the outer ITS primers ITS1 (5' - GAT TAC GTC CCT GCC ATT TG - 3') and ITS2 (5' - TTG TTC GCT ATC GGT CTT CC - 3') each at 0.25 μl and 3.25 μl of nuclease free water. The second round of the PCR contained the inner ITS primers ITS3 (5' - GGA AGC AAA AGT CGT AAC AAG G - 3') and ITS4 (5' - TGT TTT CTT TTC CTC CGC TG - 3') and used 0.5 μl template DNA from the first round. The reaction comprised of an initial heating at 95 °C for 7 minutes followed by 35 cycles of denaturation at 94 °C for 1 min, annealing at 55 °C for 1 min and extension at 72 °C for 2 minutes. Known DNA of T. vivax/T. congolense was used as positive control while water was used as a negative control. The PCR products were separated on 1.5% agarose gels stained in GR Green (Excellgen) and images visualized and documented on a gel imaging system (E-Box C X 5, Vilber Lourmat). Positive PCR products were identified based on DNA fragment size relative to a 100bp molecular weight marker (Quick-Load DNA Ladder, Quick-Load®, New England Biolabs, Inc). Trypanosome infection positivity or negativity was defined as a binary (yes/no) variable based on actual DNA fragment size observed on gels as specified previously [22-24].
Along with sampling of cattle blood, observed and reported risk factors for trypanosome infection were recorded. The risk factors included age, sex, herd size, previous infection and treatment intervention against trypanosomosis (in case where cattle owners treated their animals with anti-trypanocidals in-between sampling sessions). Age groups were classified as proposed by [25] with minor modification (Table 1). The treatment histories of all the sampled cattle were recorded in all the three sampling periods. If the animals were treated within 21 days of blood sampling, they were considered treated. These factors were classified as shown in Table 2.
Table 1: Number of infected cattle in each boma for the three study villages. View Table 1
Table 2: Classification of risk factors for trypanosome infections in cattle. View Table 2
Data were analyzed at both the herd and individual levels. Non-parametric statistical tests such as Mann-Whitney U test (U) and Kruskal-Wallis (H) were used at individual and herd level to compare the variation in trypanosome infections among variables such as age, group and treatment.
Generalized linear mixed models (GLMM) with package 'lme4' version 1.1-17 in R software (R Development Core Team 2011) were used to determine the association existing between risk factors and trypanosome infections, using maximum likelihood with binomial as family at individual level. The Odds Ratio (OR) values were calculated from the coefficient values from the model to predict the direction of effect if it was negative or positive. In this case, if OR > 1 the impact was considered positive while OR < 1 showed negative effect of the risk factors. Trypanosome infection rate was calculated at two time intervals, t1 and t2. The two time intervals were July 2017 to October 2017 (t1) and November 2017 to January 2018 (t2). The infection rate was calculated using the following formula, IR = 1-(Log (N-n) N), where IR = infection rate, N = Original number not infected, n = Number that become infected.
Trypanosome infection rates in cattle were analyzed at the herd level (15 herds) and compared between the two time intervals, ti and t2, and between villages. The largest number of sampled individuals in each herd was 10 cattle while the least was 8 cattle. The herd with highest infection rate had 3 out of 9 cattle while two herds showed no infections for both periods t1 and t2 (Table 1). The rate of infection was observed to be higher in the time interval between November and January (t2) than the time interval between July and October (t1) (Figure 2). The Mann-Whitney U test statistic revealed this difference to be statistically significant (U = 7.5, P < 0.0001, df = 1).
 Figure 2: Trypanosome infection rate in cattle herds during the two time intervals t1 (July 2017 to October 2017) and t2 (November 2017 to January 2018).
View Figure 2
Figure 2: Trypanosome infection rate in cattle herds during the two time intervals t1 (July 2017 to October 2017) and t2 (November 2017 to January 2018).
View Figure 2
Comparison of infection rates between the three study villages indicated that Kimotorok village had highest infection rate during both time intervals, ti and t2. The lowest infection rate during ti (July 2017 to October 2017) was observed in Sukuro village, while Oltukai village had the lowest infection rate during t2 (November 2017 to January 2018) (Figure 3 and Figure 4). However, the variation in infection rate among villages was not statistically significant in neither of the two time intervals; July to October 2017 (Kruskal-Wallis statistical test (H) = 3.70, P = 0.157, df = 2) and November 2017 to January 2018 (Kruskal-Wallis statistical test (H) = 4.90, P = 0.086, df = 2).
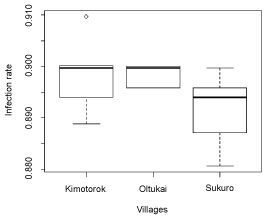 Figure 3: Trypanosome infection rates in cattle in the three study villages for the time interval of July 2017-October 2017.
View Figure 3
Figure 3: Trypanosome infection rates in cattle in the three study villages for the time interval of July 2017-October 2017.
View Figure 3
 Figure 4: Trypanosome infection rates in cattle in the three study villages for the time interval of November 2017 to January 2018.
View Figure 4
Figure 4: Trypanosome infection rates in cattle in the three study villages for the time interval of November 2017 to January 2018.
View Figure 4
A total of 141 cattle in 15 herds which generated a sum of a total of 423 for all the sampling periods were included in the final analyses (after removal of individuals not present at all sampling periods), of which 24 (17%) were observed to be infected with trypanosomes across all 3 time periods. The number of individuals infected by trypanosome parasites varied in each category. For instance, the adult cattle age group had the highest number of infected individuals compared to the calves age group (Table 3).
Table 3: Number of individuals infected with trypanosome species in each category. View Table 3
Cattle that had previously been infected, younger individuals, males, and cattle from medium and small herds were all more likely to have trypanosome infections (Table 4), though these effects varied among the study villages (Table 5).
Table 4: The overall influence of risk factors on trypanosome infections in cattle*. View Table 4
Table 5: Linear mixed effect models among risk factors which showed significant effects in the study villages (Kimotorok and Oltukai, none reveled significant effects in Sukuro)*. View Table 5
Small herd size and male sex in Oltukai showed significantly greater likelihood of being infected with trypanosome parasites than medium herd size and females. In Kimotorok, male sex, medium herd size and previous infection positively influenced the likelihood of being infected with trypanosomes (Table 4).
In this study we present results showing the influence of risk factors for trypanosome infections in cattle in the Maasai Steppe. In this study, we found that cattle aged less than one year were more likely to have trypanosome infections compared to older cattle. This is in agreement with a study done in western Kenya which found that, trypanosome infection is more prominent in young age [26]. This may be explained by different management practices observed during data collection. Young calves were normally grazed near the homesteads, and received infrequent treatments unlike adult cattle. For instance, out of all animals that were treated by the herders in-between sampling periods, only 10% and 12.5% were from the calf and young age groups, respectively, while 86.5% were adult cattle. The more infections detected in calves and younger age groups could also be related to development of the immune system at this age, as well as lesser exposure to of these age groups to frequent treatment, as practiced by the Maasai herders. Previous studies on bovine babesiosis showed that calves have less immunity compared to adult cattle, thus making them more susceptible to new infection [27]. In contrast, a study by [28] in Tororo District, south-eastern Uganda showed that cattle above 3 years were more likely to be infected, contrary to our findings. The discrepancies between our findings and the other studies can be explained either by cattle breed differences or by the varying management practices in the Maasai steppe compared to the study sites in Uganda. For instance the breeds of cattle involved in Uganda reported by [28] were Boran × short horn Zebu cross, Boran × Holstein Friesian cross and African short horn Zebu (Nkedi) under either zero grazing or semi-zero grazing while in our study only Zebu (Bos indicus) cattle were sampled. Furthermore, in the Maasai steppe, older cattle are trekked long distances for pasture and water, what exposes them to tsetse-infested areas, close to wildlife like the Tarangire national park, hence increasing chances of more trypanosome infections in older cattle.
Herd size was also observed to have a significant effect impact on trypanosome infections in cattle. Medium size (51-100 cows) and small size herds (≤ 50 cows) were observed to positively influence the rate of infection with trypanosomes in cattle. This finding is supported by a study done by [29] in cattle to determine the influence of herd size on the prevalence of trypanosomosis, which found that the highest infection rates were reported in smaller and medium size herds than larger size herds. For instance, in our study it was observed that, out of the 22 cattle treated with anti-trypanocidals, 2.08% and 8.33% were from small size herds and medium size herds, respectively, while the rest were from large size herds. Cattle owners in Kimotorok village kept larger herds (> 100 cows) and also adopted frequent treatment regimes against trypanosome infections. The majority of farmers with medium (51-100) and small herds (≤ 50 cows) were from Sukuro and Oltukai villages, where we observed less frequent treatments against trypanosomosis. As the number of cattle is strongly related to wealth in Maasai communities, the higher prevalence of infections in smaller herd sizes could be explained by lack of funds to purchase drugs for treatment [30]. Other studies have also shown similar trends, with highest prevalence of infection reported in medium size herds.
Trypanosome infection rates varied significantly between the two sampling periods, ti (July-October 2017) and t2 (November 2017-January 2018). Higher infection rate was observed during the t2 period. The variation in rate of infection between the two periods might be explained by change of vegetation cover associated with seasonality. It is expected that during the dry season lack of rainfall and higher temperature favor loss of leaves on woody plants which provide shelter for tsetse flies, hence affecting vector dynamics. In addition, the interaction between cattle, wildlife reservoirs and tsetse flies changes from low in July-October 2017 to high interaction in November 2017- January 2018 and this can positively explain the seasonality of infection rates between the two periods. Our findings are in agreement with a study done in the same area [31], which showed that tsetse fly abundance peaks in December, corresponding to our t2 time interval. Also during this time interval (t2), wildlife including reservoirs for trypanosomes were observed to come out from the Tarangire National park thus increasing the cattle-wildlife interaction. Proximity of our study villages to the protected areas enhanced the interaction of cattle with wildlife as was often observed between November 2017 and January 2018.
Unlike the studies done by [29] and [25] which showed that female cattle had significantly more infections than males due to stress of lactation and successive pregnancies, in our study there was a significant influence of male sex on the rate of trypanosome infections in cattle. This agrees with a study done by [32] who found high cases of trypanosome infection in male humans in the Democratic Republic of the Congo and [33] who found positive influence of male sex on trypanosome infection rate in wildlife reservoirs in Zambia. We ascribe our observations to behavior of Maasai pastoralists to value and give more care to cows than bulls when it comes to animal health care, such as treatment or vaccination. In our study, the male cattle which were found to have trypanosome infection were also shown to have received less treatment attention compared to female cattle. The pastoralists give higher value to cows as capital required for herd perpetuation.
Understanding the risk factors for trypanosomosis is essential for developing proper control strategies of the infection. In our study we focused on the influence of age, sex, treatment intervention, and herd size as potential risk factors. We also examined the influence of season and land cover in grazing areas. However, further studies need to be done to address other factors for the prevalence of trypanosome infection in the Maasai Steppe. For instance, other factors reported by researchers to influence the prevalence of trypanosome infection in cattle were body condition score of cattle, breed of cattle [1], location, tsetse flies abundance [25], immunity and drug resistance [34], use of water point (source) particularly during dry season [32], range of time between last spray and the next, distance moved by animal from settlement [35].
Trypanosomosis in cattle will continue to be a challenge among the pastoralist communities in Tanzania due to ecological and other risk factors. For instance, in this study risk factors such as herd size of 51-100 cattle was observed to positively correlate with prevalence in Maasai Steppe which could be due to low drug purchasing power to handle all diseased animals, resulting in no or improper treatment for affected animals. However, when different villages were considered, age of 0-1 years in Oltukai village and age between 1-2 years in Kimotorok village were positively correlated with prevalence of trypanosome infection probably due to low immunity and less treatment priority in this age.
We are thankful to the Bill & Melinda Gates Foundation for funding this study under the Program for Enhancing Health and Productivity of Livestock (PEHPL, ID: OPP1083453). We appreciated the Government authority through their Districts veterinary personnel's from Simanjiro and Monduli Districts for granting us opportunity to carry out the study in their villages, particularly Oleshongoni Boniphace and Edward Kumbi. We would like to thank the Village local government leaders for the support and cooperation which was provided during our study. We would like to thank the laboratory technicians from Nelson Mandela African Institution of Science and technology and those from Genome Science center at Sokoine University of Agriculture, Asteria Butuyuyu and Fatuma Kindoro, for their advice and assistance during our laboratory work. We thank the field assistants, Mokia Mirimbi and Ukweli Kidonya, who were supportive during our field work.