Sarcoidosis is a chronic inflammatory disease involving multiple organ systems for which the inciting stimulus is unknown. It is characterized by elevated markers of inflammation and disseminated granuloma in the affected organs. Factor V Leiden deficiency has been long associated with an elevated risk of thrombosis, however, it has been observed in the patients having sarcoidosis, a secondary inciting stimulus is required to trigger the thromboembolic phenomenon, as to date only a few cases of Primary thrombosis have been reported in Factor V Leiden deficient patients culminating in an increased incidence of thromboembolic disease. We are presenting a case of a patient presenting with cerebrovascular events and pulmonary embolism, who was diagnosed later diagnosed to have sarcoidosis with an associated Protein S deficiency and factor V Leiden mutation making her predisposed to thromboembolic disease. It has been speculated that steroids as a treatment elevate the risk of thrombosis in such patients instead of the inflammatory process per se. however; this patient experienced a thrombotic episode before the start of medications. Additionally, an anatomical relationship has been reported in the literature between the site of sarcoidosis and the development of thrombosis, but the patient had repeated episodes of cerebrovascular events, despite no evidence of involvement of cranial vasculature or any heart defect, that could cause paradoxical embolization. This case report can help to elicit the relation of sarcoidosis with the increased tendency towards thromboembolic disease.
Intubation, Endotracheal tube, Ultrasound, Doppler, Cadaver
Sarcoidosis is a chronic inflammatory condition characterized by multiple organ involvement. It was first described by Professor Caeser Peter Moller Boeck, who initially identified the disease based on the skin lesion and later on confirmed the multiorgan involvement. To date, no specific inciting agent has been identified that causes Sarcoidosis. Sarcoidosis is characterized as a chronic granulomatous disease in the organ involved with macrophages and monocytes being the hallmark of the disease. Active disease simultaneously remits over years; however active disease has been associated with increased incidence of loco-regional venous thromboembolism and vascular stenosis.
We present a case of a 41-year-old Female who presented to pulmonology clinic for management of pulmonary embolism. Ten days before the presentation, she took a flight from New York City to Miami for an abdominal liposuction procedure. During the preparation for surgery, the patient experienced sudden onset of fever, cough, and shortness of breath. Examination revealed bilateral crepitation with bilateral absent breath sounds at bases. Chest X-ray showed pleural effusion, so with suspicion of pneumonia with parapneumonic effusion, she was started on co-amoxiclav and symptomatic care, and surgery was deferred. The following day, the patient's SOB, and chest pain worsened, and she started to experience weakness in her right arm, for which in the emergency department, a CT chest was performed. CT scan showed right-sided pneumonia with a single filling defect in the right pulmonary artery. CT angiogram of head and neck showed patent anterior and posterior circulation. The patient was treated initially with Lovenox and symptomatic care which stabilized her condition, and later she was switched to Eliquis. A week later, during her return flight to New York City, she started to experience headache, slurring of speech, along with right arm weakness, that resolved in an hour. In ED, no source of thrombosis could be found, lower extremity Venous Doppler could not elicit any evidence of DVT. She admitted to having shortness of breath with exertion, associated with dull chest pain, but denied any fever, hemoptysis, leg swelling, and palpitation. The patient's history was significant for a single episode of deviation of angle of the mouth, headache, and slurring of speech, a year before the recent presentation, for which she received Alteplase at that incidence, which alleviated her symptoms. Despite extensive workup, no source of thromboembolic phenomenon could be traced. Slurring of speech improved over time, but she had continued to experience waxing and waning fatigue, shortness of breath, and headaches. A repeat CT chest showed multiple nodules in the right lung parenchyma, along with a filling defect in the right pulmonary artery, Left lung was sparsely involved except for old basilar atelectasis or scarring. The patient had no history of smoking or a family history of thrombotic capacity. Examination revealed bilateral normal pulmonary auscultation with equal and comparable air entry. Neurological examination was nonfocal and muscle power was equal in all four limbs (Table 1).
Table 1: Pulmonary function tests. View Table 1
The patient's exercise capacity was within normal limits. Basic metabolic profile, EKG, and LFTs were within normal limits (Table 2).
Table 2: Lab results for procoagulant status. View Table 2
CT chest, abdomen, and pelvis showed multifocal nodular lung infiltrates, right upper lobe predominant with peripheral confluence/consolidation in conjunction with multi-station lymphadenopathy, worse in the mediastinum/hila raising the strong possibility of sarcoidosis, for which tissue sampling was advised. Borderline cardiomegaly with patent central, main, and segmental pulmonary artery lacking any evidence of acute or chronic pulmonary embolism. The spleen was normal in size with several indeterminate lesions raising the possibility of sarcoidosis. Several indeterminate hepatic lesions were found which could be hemangiomata, for which contrast-enhanced MRI was suggested. It was suggested that alternatively, these findings could be due to lymphoma or metastatic disease of indeterminate primary source. But no evidence of lymphoma or primary tumor could be found on CBC, mammogram, abdominal or pelvic imaging. MRI confirmed hepatic hemangiomata, splenic lesions. Bronchoscopic biopsy of the lesion from the right lung showed findings consistent with sarcoidosis. Based upon the radiological evidence and Siltzbach classification system, the patient was having stage II disease and was started on albuterol for symptomatic SOB, and steroid for sarcoidosis control. Eliquis was continued with all the precautions for falls. Referral to hematologist was provided for evaluation and assessment of low Protein S activity. Further plan for a follow-up assessment after 6 months to document disease status was planned (Figure 1, Figure 2, Figure 3 and Figure 4).
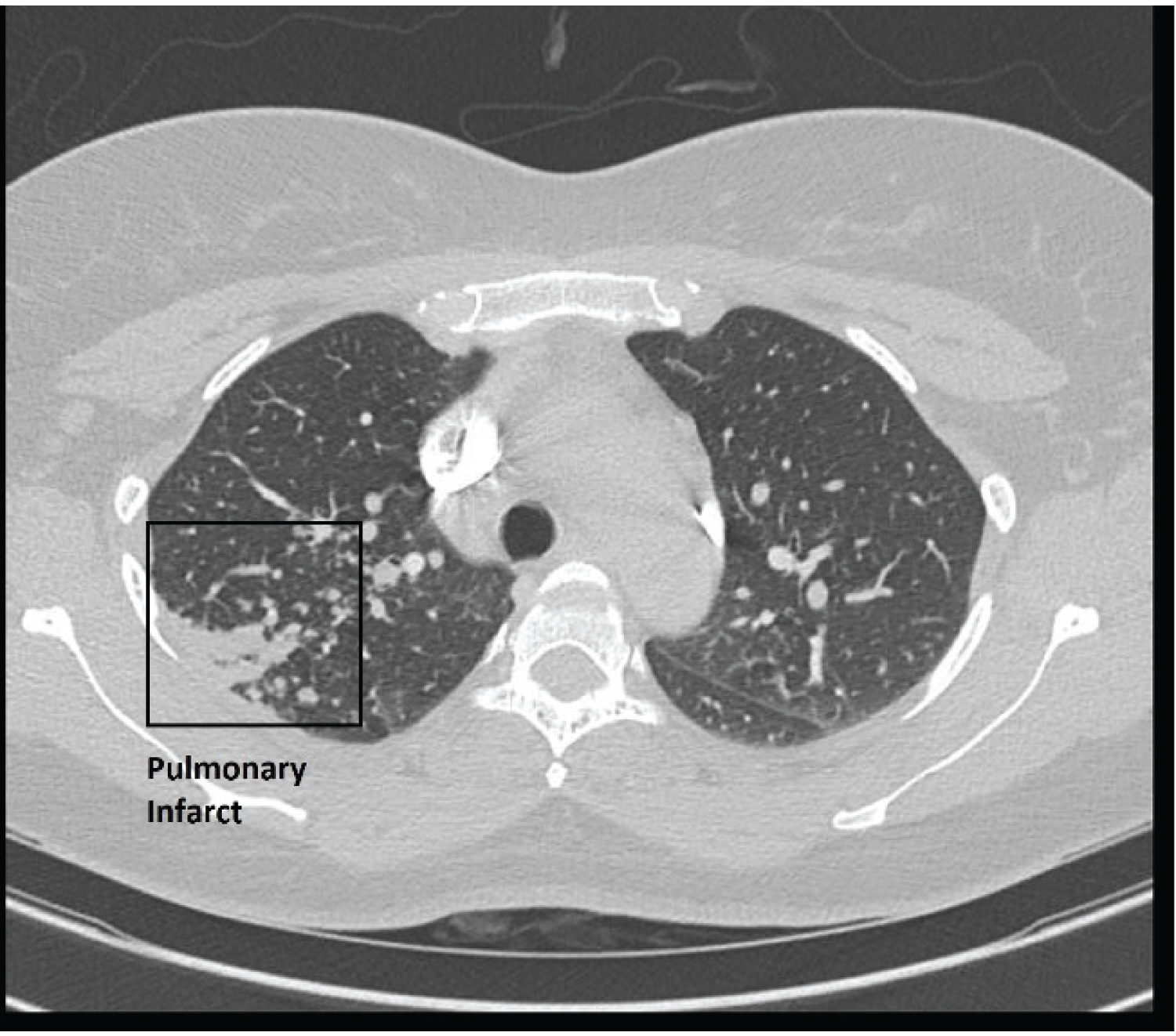 Figure 1: CT chest showing pulmonary infarct. View Figure 1
Figure 1: CT chest showing pulmonary infarct. View Figure 1
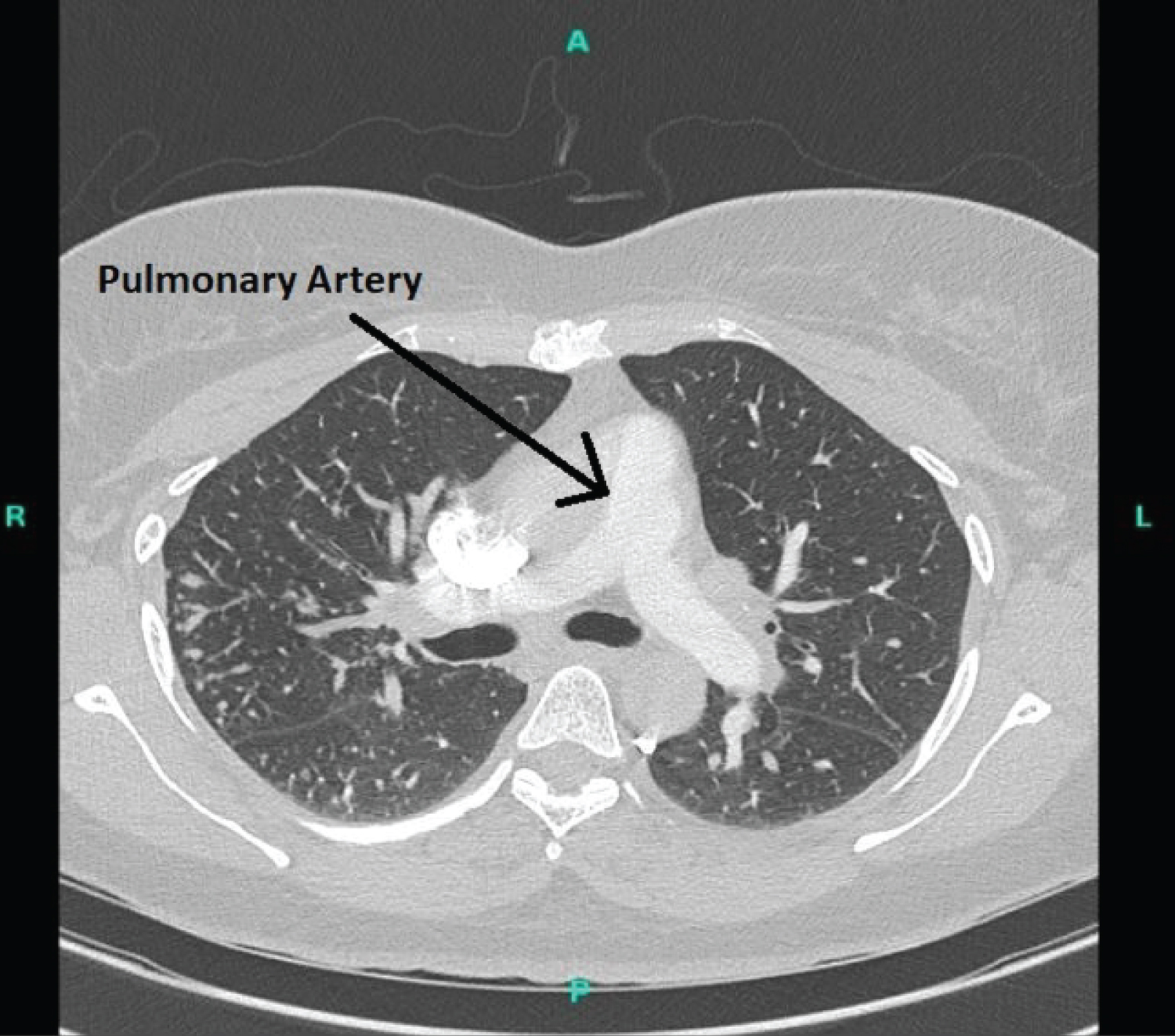 Figure 2: CT chest showing pulmonary artery. View Figure 2
Figure 2: CT chest showing pulmonary artery. View Figure 2
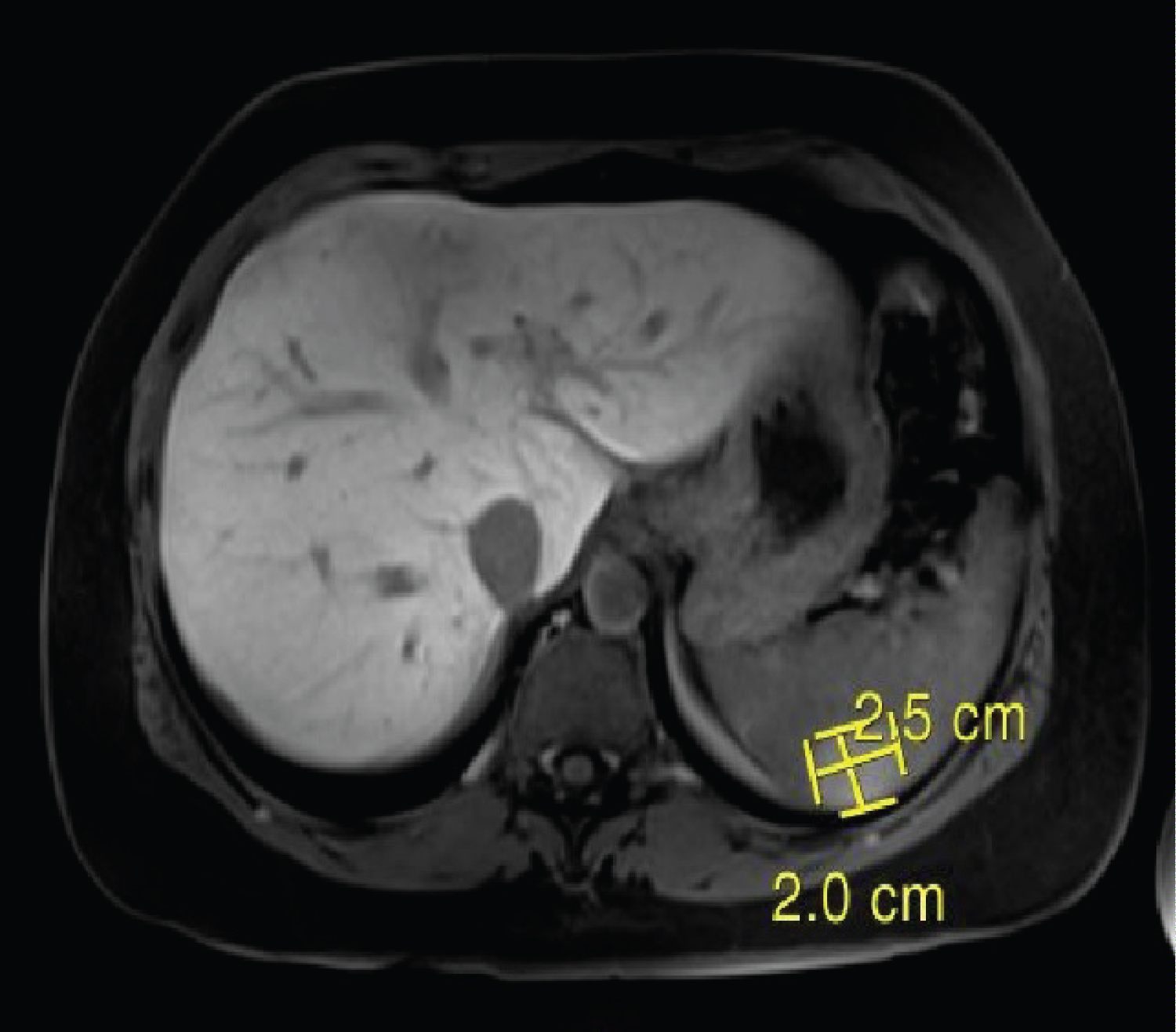 Figure 3: MRI abdomen showing splenic granuloma (Axial section). View Figure 3
Figure 3: MRI abdomen showing splenic granuloma (Axial section). View Figure 3
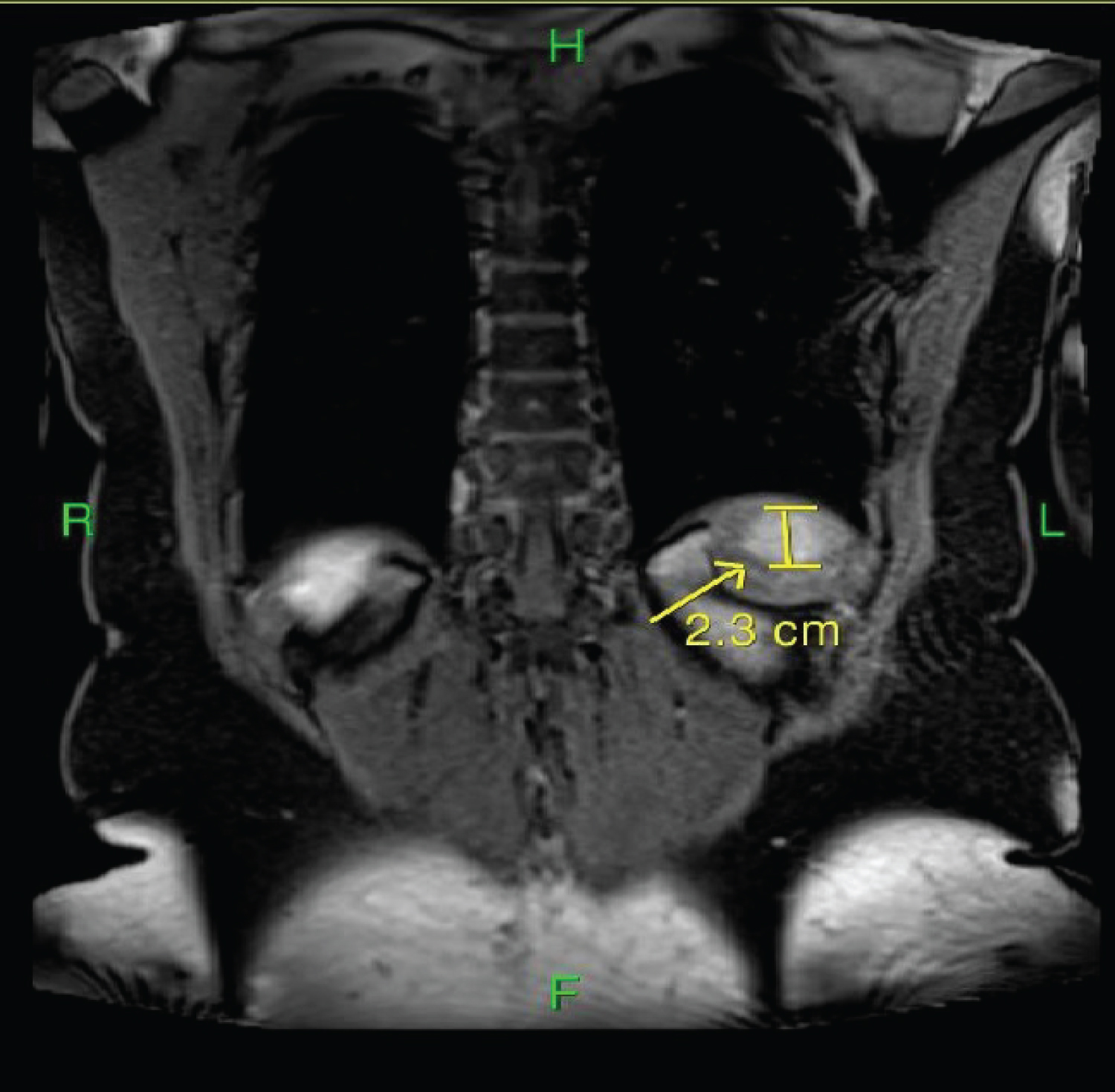 Figure 4: MRI abdomen showing splenic granuloma (Coronal section).
View Figure 4
Figure 4: MRI abdomen showing splenic granuloma (Coronal section).
View Figure 4
Incidence of sarcoidosis among the US population varies between 5-40 cases per 100,000, being more prevalent in Patients from African American ethnicity having 34 cases per 100,000 population [1]. Crawshaw AP, et al. elicited that patients with sarcoidosis had an increased risk of Pulmonary Embolism (Rate Ratio 2.0, 95% CI: 1.1 to 3.4, under age of 65 years) [2]. A similar risk for PTE was found by Swigris and colleagues [3]. Nevertheless, it has not been established whether sarcoidosis is a risk factor for thromboembolic disease. In literature, an anatomical relationship between the incidence of venous thromboembolic disease and the site of active sarcoidosis has been described, exemplified by mural thrombosis with myocardial sarcoidosis [4], cerebral vein thrombosis with neurosarcoidosis [5,6], thoracic vein thrombosis with mediastinal sarcoidosis [7], and Portal vein thrombosis in Hepatic Sarcoidosis [8]. Factor V Leiden mutation has been long affiliated with increased risk of thrombophilia, however, studies have shown that these individuals, even though tend to thrombophilia, need a second acquired stimulus to develop a clinically significant thrombotic event [9]. Smoking has also failed to show a significant risk of association with pulmonary thromboembolism in patients having sarcoidosis [10]. Kolluri, et al. in a retrospective study found that with multivariate regression analysis, the steroid therapy for the treatment of sarcoidosis, rather than the inflammatory disease process, were the most significant risk factor for pulmonary thromboembolism (HR = 3.12, p = 0.007) [11]. There have been various other studies where anti-inflammatory therapy was found to be the actual risk for thromboembolic disease rather than the inflammatory process itself. Tandon, et al. reported a case series of seven cases, where sarcoidosis presented with pulmonary hypertension due to chronic thromboembolic phenomenon, however, no evidence of pulmonary hypertension could be found in this patient [12].
The patient had experienced repeated episodes of thrombosis involving cranial vasculature, but her investigations showed only weak evidence of related deficiencies. Even though the patient was heterozygous for Factor V Leiden, but had normal Protein C activity. Prolonged air travel could be a possible inciting factor for the thrombotic event, but there was no evidence of deep vein thrombosis on imaging. The patient's presentation of waxing and waning headaches and right arm weakness can be explained by transient Ischemic attacks. Sarcoidosis was the only clinical risk factor to explain the patient's propensity to have thrombotic events. The patient had thrombotic episodes before the start of the treatment which contradicts the finding of steroids being the risk factor for thrombosis instead of the inflammatory process.