PPE: Personal Protective Equipment; SARS-CoV-2: Severe Acute Respiratory Syndrome Coronavirus 2; COVID-19: Corona Virus Disease 2019
During the evolving SARS-CoV-2 pandemic, occupational exposure and infection for first responders and health care workers remains a major concern across the globe [1]. Regional shortages in personal protective equipment (PPE), particularly high-level particulate filter masks such as N95, has led to creative interventions to reduce airborne viral exposure while caring for infected patients. Procedures that result in patient-generated aerosols such as nebulizer medication treatments, intubation, bronchoscopy and other manipulations of the upper airway and glottis are among the highest risk encounters. In addition, there is increasing evidence supporting a high community incidence of asymptomatic and pre-symptomatic subjects who actively shed virus, making procedures in patients not suspected of having COVID-19 also risky. Pre-procedural rapid testing would address many of these concerns, but such assays are in limited supply making routine testing all but impossible for high volume medical centers, particularly for non-routine cases (hemoptysis, symptomatic lung masses, etc.). Furthermore, serial testing of ill patients demonstrates an apparent clearing of detectable virus in upper airway specimens with persistent viral nucleic acid detectable in lower airway secretions for up to many weeks [2]. How long such patients actually shed infectious virus is unknown, but this creates a drive to not deem a patient who still needs inpatient level care as "COVID-19 free" until lower respiratory tract specimens have been tested as negative for viral RNA. Many patients in the recovery phase are unable to produce sputum leading to requests for nasotracheal suctioning or bronchoscopy to obtain lower respiratory tract samples. This creates a need to facilitate access to the lower airway at the lowest achievable risk.
To protect proceduralists and assistants during tracheal intubation, a containment "box" has been proposed with anterior access holes for the operator to introduce the tube and laryngoscope [3]. This seems well designed to protect from expelled droplets generated during cough, but may be less suited for avoidance of true airborne particles. Furthermore, it is large, expensive and applying negative pressure has proven challenging as the system is inherently leaky; there is no top access for bronchoscopy.
We have assembled, from materials on hand, a containment "tent" that may offer an improved, low cost approach to protecting a bronchoscopist and assistants from true aerosolized infectious material (Figure 1A). We defined desirable features as being lightweight, portable enough to take to negative pressure patient care rooms, able to contain droplet emissions, and allow for improved aerosol containment. Yet, we still wanted good patient visibility and communication, access for oral suctioning and supplemental oxygen, and rapid disengagement in event of clinical deterioration. Our preliminary design is comprised of a neonatal oxygen tent house (http://novahealthsystems.com/tenthouse.html). This provides access ports above for scope insertion that can be further sealed using a standard ventilator elbow attachment with grommet. There are snap flaps on the sides for operator arm access to the scope and a patient chest "sleeve" that provides a relative seal against bulk particle movement while still allowing for oxygen tubing and oral suction access. The flexible sleeve around the patient's shoulders may be lifted and the mouth accessed for topical anesthesia by gargle and spit, bulb spray, and/or posterior pharyngeal pledgets. During simulated aerosol generation using a standard nebulizer with backlighting to highlight particles, we were unable to detect any droplet escape. In an abundance of caution, we added a filtered suction system in case positive pressure during coughing might drive aerosol escape. This could either be fed through a port at the head of the tent or laid alongside patient head via sleeve. Total cost for this setup is under $20 and weighs less than five pounds. It is fully disposable.
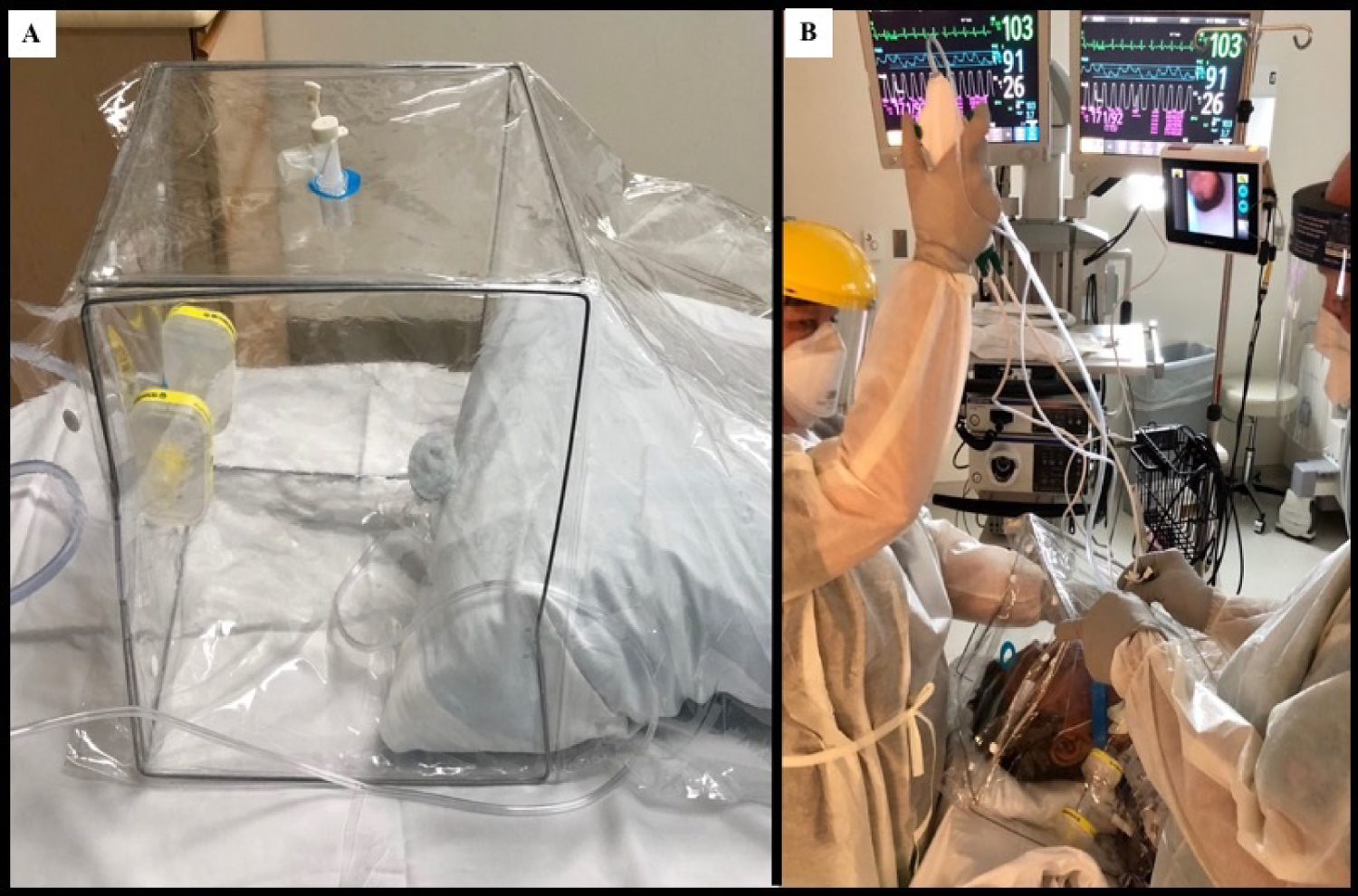 Figure 1: (A) Basic components of the protective bronchoscopy tent include a neonatal oxygen tent "house" with upper port for scope access and port at head for application suction line for negative pressure application; (B) Shows tent in use in suspected COVID-19 patient. A ventilator access elbow used in upper port to help with scope seal and stabilization. The hood at the front creates a relative seal against bulk air flow, yet provides ready access to patient's face for suction, anesthesia and supplemental oxygen. For patient comfort, in this case we fed filtered suction line in from sleeve and sealed off front port.
View Figure 1
Figure 1: (A) Basic components of the protective bronchoscopy tent include a neonatal oxygen tent "house" with upper port for scope access and port at head for application suction line for negative pressure application; (B) Shows tent in use in suspected COVID-19 patient. A ventilator access elbow used in upper port to help with scope seal and stabilization. The hood at the front creates a relative seal against bulk air flow, yet provides ready access to patient's face for suction, anesthesia and supplemental oxygen. For patient comfort, in this case we fed filtered suction line in from sleeve and sealed off front port.
View Figure 1
As you can see in the attached image, there is ready access to the mouth or nose for deployment of the bronchoscope (Figure 1B). In this case, the filtered suction was introduced through the shoulder sleeve for patient comfort. This does not provide ideal conditions for procedures needing delicate tool manipulation or access to very distal sites in the airway. It is also not ideal for cases with a high likelihood of deterioration needing bagging or intubation.
Ideally, all aerosol generating procedures during a pandemic would be performed after upper airway SARS-CoV-2 screening (which are in short supply and may miss those that have cleared the nasopharynx) and with full PPE including N95 equivalent masks for the operator and assistants in the room as detailed by Feroli, et al. [1]. However, inventories at many centers are running critically low and the supply chain is lagging for anticipated needs. Continuous evaluation for de-escalation of isolation status, particularly conservation of N95 masks and CAPR shields, is a critical part of filling this gap in PPE availability [4]. In recovering COVID-19 patients who require ongoing inpatient care, or in suspicious cases presenting late in the clinical course where upper tract specimens may be negative, this set up may offer a safer opportunity for a simple airway washing or BAL to establish the persistence or absence of SARS-CoV-2 RNA in the lower respiratory tract. We acknowledge and concur with societal guidelines detailing that the use of bronchoscopy to diagnose COVID-19 should be discouraged and rare [4-6]. Atypical times sometimes call for creative temporary solutions. We hope the need for such modified techniques will prove to be short lived.
There are no funding disclosures for this work.
Both Mr. Recker and Dr. Gross contributed equally to the design and implementation of this technique at our institution.