A 53-year-old Caucasian nurse with a history of emphysema presented with 6 months of recurrent fever, generalized fatigue, exertional dyspnea, and non-productive cough. She reported a 15 pack-year smoking history but quit 3 months prior to evaluation. She previously worked with patients with chronic tracheostomies in a long-term acute care facility. She denied hemoptysis, sputum production, chest pain, pleurisy, abdominal pain, or unintentional weight loss. She reported no recent travel, inhalational exposures, or history of incarceration. She received several courses of oral antibiotic therapy from local urgent care facilities for presumed acute exacerbations of chronic obstructive pulmonary disease during this period with initial mild improvement but symptomatic recurrence soon after treatment cessation.
At her initial outpatient visit, vital signs were temperature 98.0 °C, blood pressure 157/89, heart rate 93, respiratory rate 18, oxygen saturation 96% on room air. Exam was notable for body mass index 17.4, clear lung fields bilaterally, and no cardiac murmurs, peripheral edema, or digital clubbing. Pulmonary function testing demonstrated obstructive physiology with ratio of Forced expiratory volume in 1 second (FEV1) to Forced vital capacity (FVC) of 0.40 and FEV1 of 0.93 liters (31% predicted). Computed tomography (CT) of the chest demonstrated a complex-appearing right upper lobe airspace opacity (Figure 1). She was started on a prolonged course of amoxicillin-clavulanate for management of presumed lung abscess. However, the patient's symptoms progressed despite this therapy and she was admitted approximately three weeks later for further diagnostic work up and treatment.
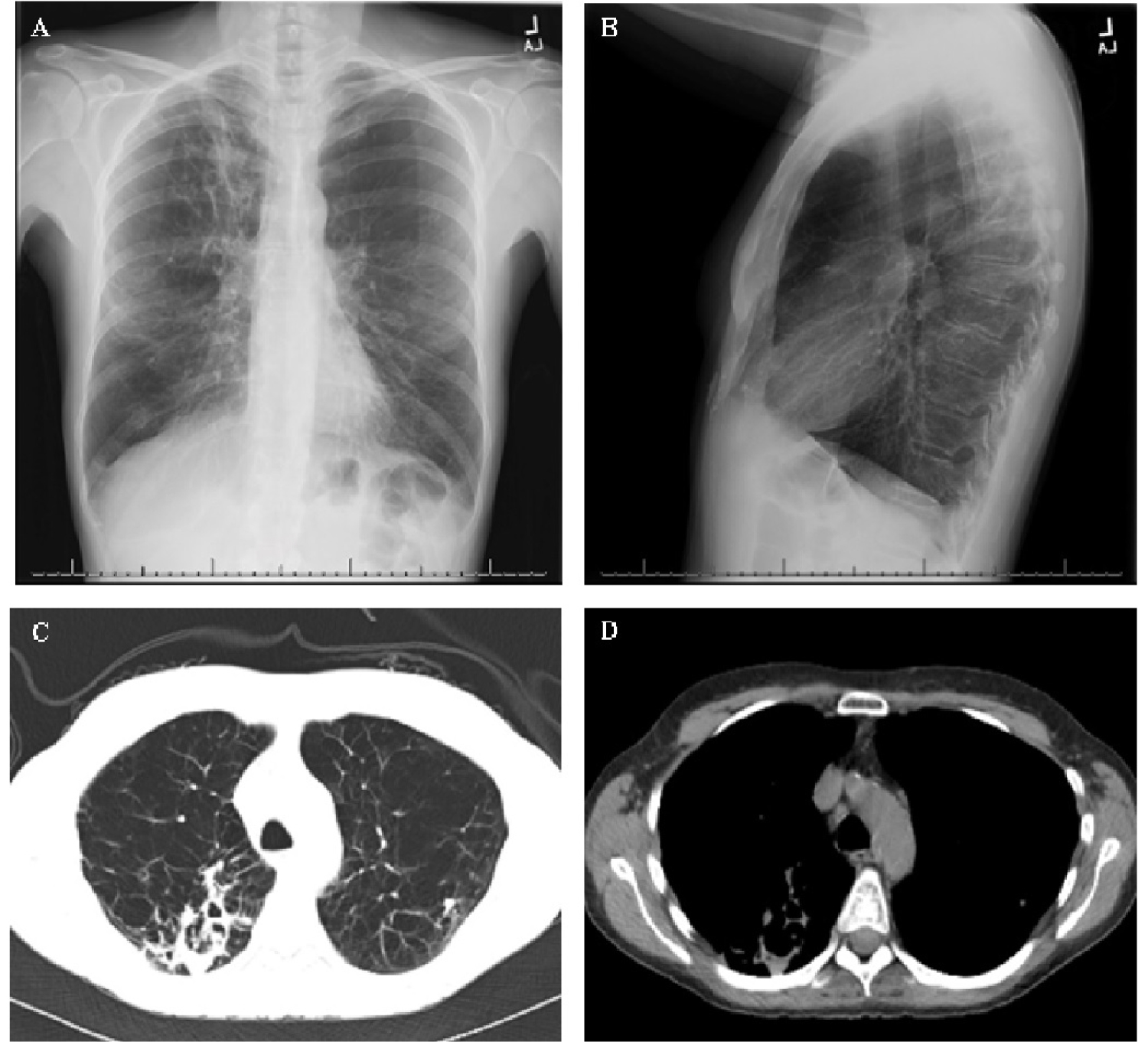 Figure 1: Radiology obtained at time of initial outpatient visit. (A) Posteroanterior chest radiograph; (B) Lateral chest radiograph; (C) Computed tomography axial cut (lung window); (D) Computed tomography axial cut (mediastinal window).
View Figure 1
Figure 1: Radiology obtained at time of initial outpatient visit. (A) Posteroanterior chest radiograph; (B) Lateral chest radiograph; (C) Computed tomography axial cut (lung window); (D) Computed tomography axial cut (mediastinal window).
View Figure 1
Admission laboratory data was notable for white blood cell count 8.0 × 103/μL (72% neutrophils, 18% lymphocytes, 1% eosinophils), hemoglobin 12.7 g/dL (mean corpuscular volume 83.6 fL), C-reactive protein 4.7 mg/d, and erythrocyte sedimentation rate 56 mm/hr. Additional testing was negative for human immunodeficiency virus, interferon-gamma release assay, urine Legionella antigen, and serum Cryptococcal antigen. Alpha-1 antitrypsin level returned within normal range. Repeat CT chest with contrast revealed interval rapid progression of right upper lobe airspace disease and multiple new subcentimeter pulmonary nodules on background severe upper lobe predominant emphysematous changes (Figure 2).
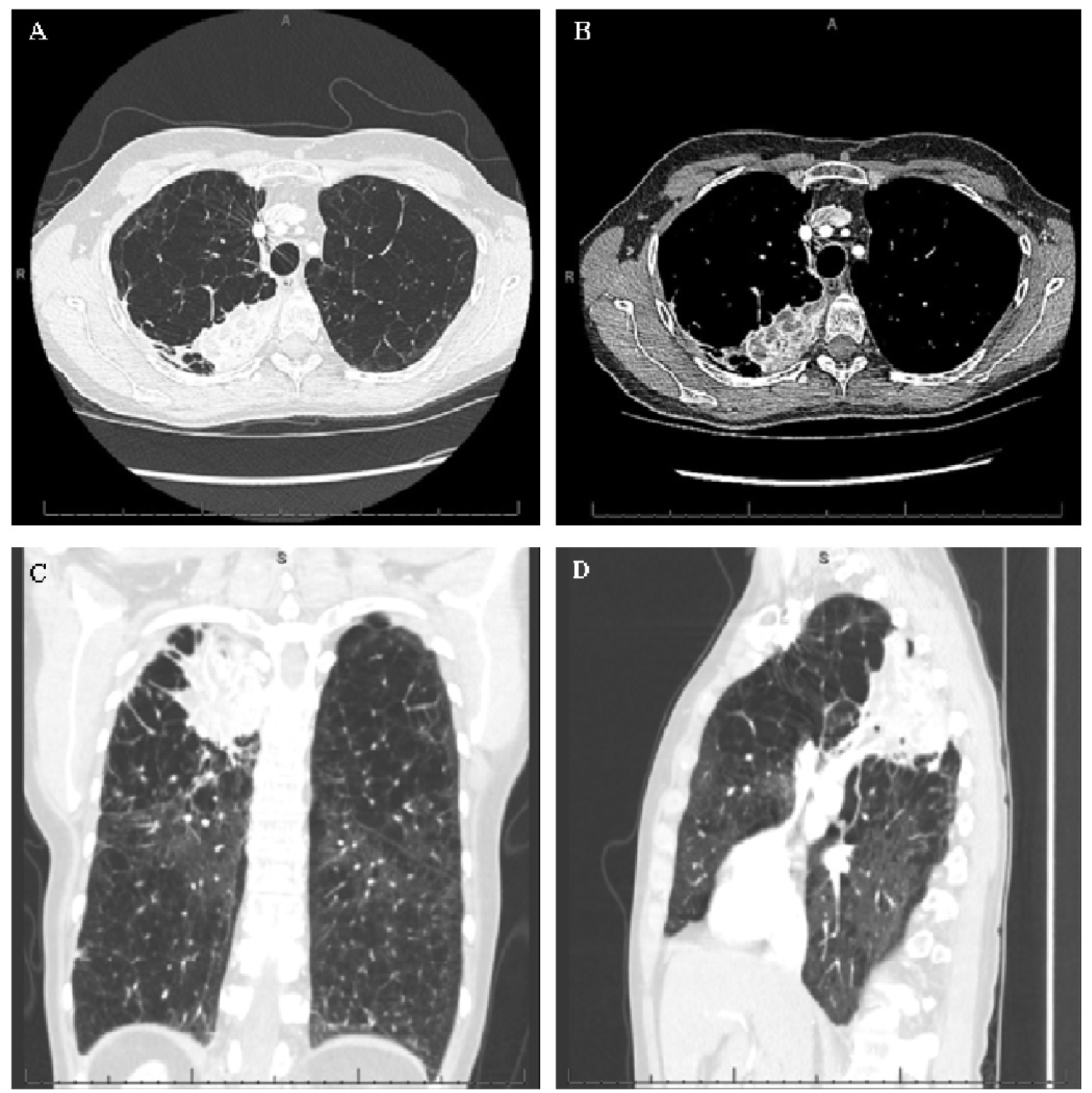 Figure 2: Radiology obtained approximately 4 weeks after initial outpatient visit. (A) Computed tomography axial cut (lung window); (B) Computed tomography axial cut (mediastinal window); (C) Computed tomography coronal cut (lung window); (D) Computed tomography sagittal cut (lung window).
View Figure 2
Figure 2: Radiology obtained approximately 4 weeks after initial outpatient visit. (A) Computed tomography axial cut (lung window); (B) Computed tomography axial cut (mediastinal window); (C) Computed tomography coronal cut (lung window); (D) Computed tomography sagittal cut (lung window).
View Figure 2
Based upon the clinical and radiographic presentation, the most likely diagnosis is:
a) Multifocal bacterial pneumonia
b) Non-small cell carcinoma of the lung
c) Mycobacterium avium complex
d) Pulmonary aspergillosis
e) Mycobacterium tuberculosis
Patient's sputum was sent for acid-fast bacilli stain and mycobacterial culture. The rapidity of symptomatic and radiographic progression, in addition to minimally productive cough, did not allow for a diagnosis to be made by non-invasive measures. Ultimately, the patient underwent fiberoptic bronchoscopy (performed 1 month after the initial outpatient visit and 7 months after the initial chest radiograph) that revealed patent segmental and subsegmental airways without endobronchial lesions or purulence. Bronchoalveolar lavage was remarkable for 20 white blood cells per mm3 (87% neutrophils, 10% lymphocytes, 1% eosinophils) and 8,900 red blood cells per mm3. Cytology brushing demonstrated numerous bronchial epithelial cells without evidence of malignancy. Due to continued symptoms despite intravenous vancomycin and piperacillin-tazobactam, the patient underwent transbronchial biopsy under endobronchial ultrasound guidance for definitive tissue diagnosis 3 days later. Surgical pathology returned with fibrinopurulent exudate, abscess formation, active granulation tissue, and granulomatous inflammation Acid-fast bacilli stain was positive for rare acid-fast organisms and Fite stain was positive for rare non-branching rods (Figure 3).
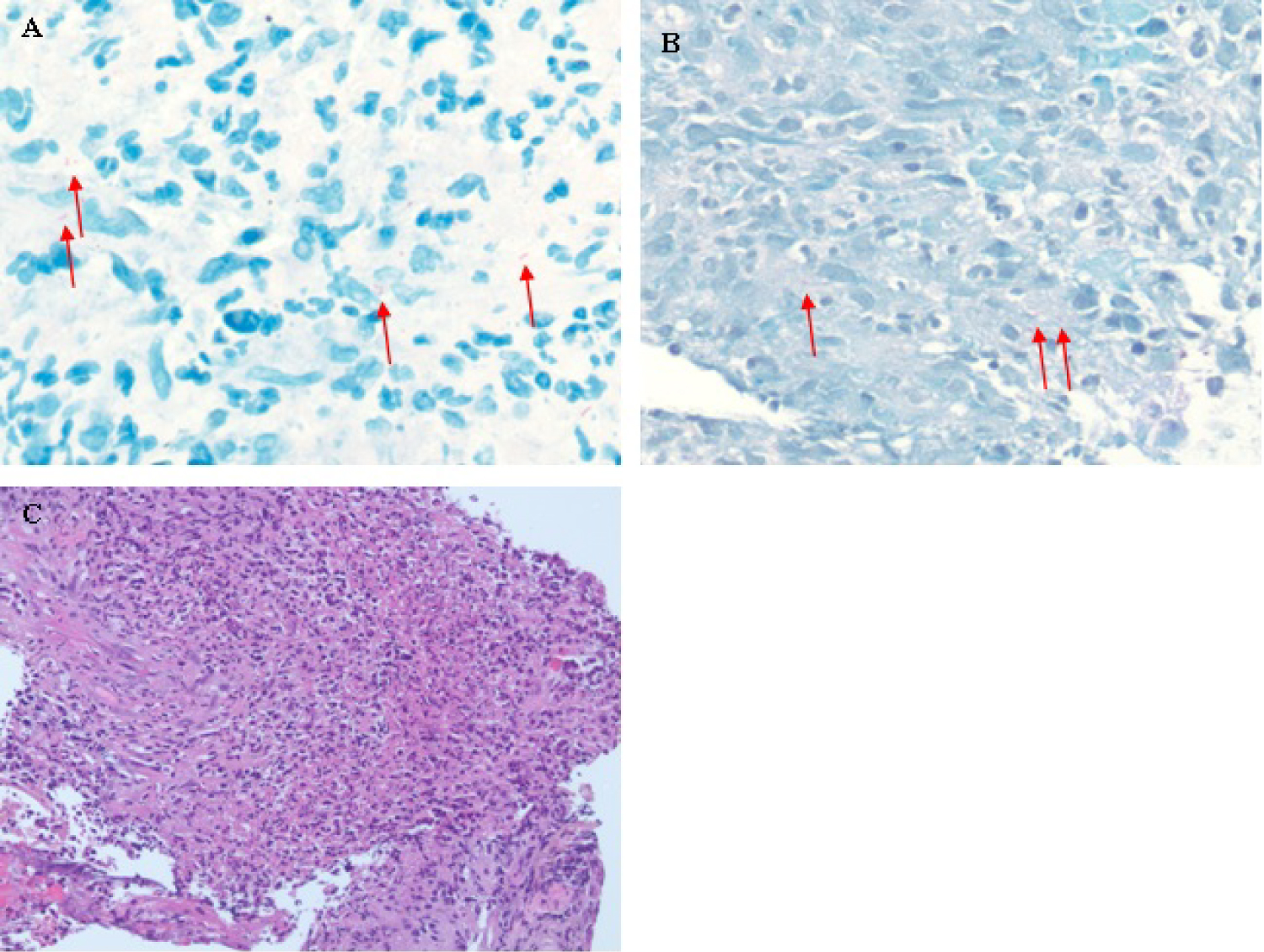 Figure 3: Pathology of right upper lobe mass. (A) Acid-fast bacilli stain. Red arrows indicate rare organisms; (B) Wade-Fite stain. Red arrows indicate rare non-branching rods; (C) Hematoxylin and eosin stain.
View Figure 3
Figure 3: Pathology of right upper lobe mass. (A) Acid-fast bacilli stain. Red arrows indicate rare organisms; (B) Wade-Fite stain. Red arrows indicate rare non-branching rods; (C) Hematoxylin and eosin stain.
View Figure 3
Diagnosis: Mycobacterium avium complex.
Clinical Discussion: The acid-fast bacilli smear from the bronchoalveolar lavage returned positive for Mycobacterium avium complex (MAC) by deoxyribonucleic acid probe. Broad-spectrum intravenous antimicrobial therapy was discontinued, and she was started on combination therapy with azithromycin, rifampin, and ethambutol while awaiting antimicrobial susceptibilities. She endorsed rapid improvement in clinical symptomatology within 1 to 2 weeks. Follow-up CT chest without contrast 3 months into medical therapy revealed interval improvement of right upper lobe consolidation (Figure 4).
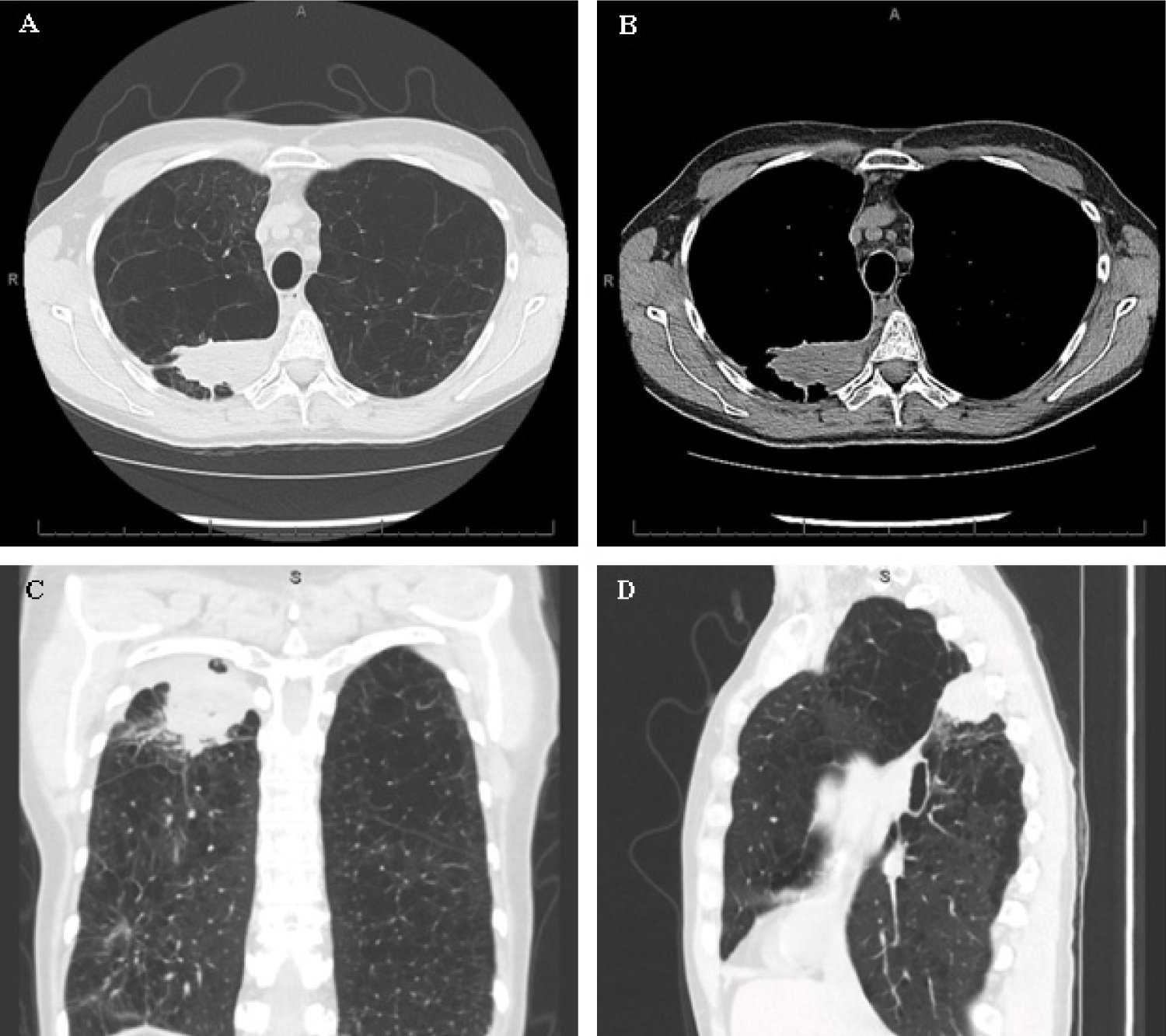 Figure 4: Radiology obtained approximately 3 months after initiation of antimicrobial therapy. (A) Computed tomography axial cut (lung window); (B) Computed tomography axial cut (mediastinal window). (C) Computed tomography coronal cut (lung window); (D) Computed tomography sagittal cut (lung window).
View Figure 4
Figure 4: Radiology obtained approximately 3 months after initiation of antimicrobial therapy. (A) Computed tomography axial cut (lung window); (B) Computed tomography axial cut (mediastinal window). (C) Computed tomography coronal cut (lung window); (D) Computed tomography sagittal cut (lung window).
View Figure 4
MAC is the most common cause of Non-tuberculous mycobacterium (NTM) in the non-HIV population. In the immunocompetent population, it is often seen in elderly Caucasian men with histories of tobacco use and underlying lung diseases, such as chronic obstructive pulmonary disease. Patients often present with non-specific clinical symptoms such as fever, generalized fatigue, cough (which may be productive or non-productive), dyspnea, and unintentional weight loss. Due to the typically indolent nature of this disease, those with chronic underlying lung diseases may already have extensive lung parenchymal destruction by the time of initial diagnosis.
The unique aspect of this patient's case was the extremely rapid (< 1 month) radiographic progression of disease associated with symptomatic deterioration in an otherwise immunocompetent host. Current literature suggests risk factors associated with disease progression in the non-HIV population include fibrocavitary disease, low body mass index, and systemic symptoms on presentation [1,2]. However, disease progression is typically more indolent. The rapidity of clinical and radiographic progression precluded outpatient evaluation.
The American Thoracic Society/Infectious Diseases Society of America (ATS/IDSA) diagnostic criteria for MAC include both clinical and microbiologic requirements [3]. Clinically, there must be pulmonary symptoms with typical imaging findings (described below) in addition to appropriate exclusion of other diagnoses. Microbiologically, there must be positive culture data from ≥ 2 expectorated sputum samples, positive culture data from bronchoalveolar lavage or washing, or biopsy with typical Mycobacterium histopathologic features (described below) with concomitant positive culture data.
First-line treatment for MAC consists of triple therapy with a macrolide (typically azithromycin), a rifamycin (typically rifampin), and ethambutol. Treatment duration typically ranges between 15 to 18 months, as sputum conversion requires 3 to 6 months and antimicrobial therapy is continued until negative sputum culture for ≥ 12 months. Pre-treatment susceptibility testing is imperative due to variable sensitivities to several first- and second-line antimicrobial therapies. In cases with macrolide resistance, quadruple therapy with rifampin, ethambutol, clofazimine, and an aminoglycoside (typically amikacin or streptomycin) is preferred [4]. Indications for surgical management include medication intolerance or resistance, failure to convert sputum after ≥ 6 months of continuous therapy, and localized upper lobe cavitary disease.
Radiographic discussion: Chest imaging may be highly variable in immunocompetent patients. Previously, Levin and colleagues described two distinct radiographic presentations of pulmonary MAC: Classic or atypical [5]. The classic appearance, most commonly in elderly Caucasian males with underlying chronic pulmonary diseases, may be indistinguishable from pulmonary tuberculosis [6]. This form of disease is often described on CT as fibrocavitary with upper lobe predominant nodules, often with contiguous pleural involvement. The atypical appearance, more common in middle-aged women and classically described as Lady Windermere syndrome, presents with centriacinar nodules and bronchiectasis affecting the right middle lobe or lingula [7]. A third form of pulmonary MAC combines features from both classic and atypical forms.
Pathologic discussion: Fluid or tissue from suspected sites of involvement should be collected for culture and histopathology. The presence of fibropurulent exudates and non-caseating granulomas on pathology are suggestive of active inflammation and supportive of, but not specific for, MAC [8]. Ziehl-Neelsen staining, more commonly known as acid-fast staining, differentiates acid-fast bacteria and their resistance to decolorization by certain acids. Mycobacterium would have characteristic bright red or purple, straight or slightly curved rods. Wade-Fite stain is a modified technique of the standard Ziehl-Neelsen stain that is specifically used to detect Mycobacterium leprae. All specimens should be sent to microbiology specifically for acid-fast bacilli culture and smear. If positive, deoxyribonucleic acid probes are reflexively performed for identification of Mycobacterium species.
1. Mycobacterium avium complex (MAC) can cause a rapidly progressive disease in immunocompetent patients.
2. MAC should be included in the differential diagnosis of unresolving and/or progressive airspace disease.
3. Fibrocavitary disease in patients with MAC is associated with a worse prognosis. This radiographic finding should elicit a prompt diagnostic evaluation for MAC and, if found, prompt institution of therapy.