Acoustic neuromas, also known as vestibular schwannomas are benign and slow-growing tumors arising from neural crest-derived Schwann cells. Treatment of acoustic neuromas targets to achieve local control while preserving hearing without comprimising cranial nerve functionality. In this context, radiosurgery in the form of Stereotactic Radiosurgery (SRS) or fractionated stereotactic radiotherapy (FSRT) offer viable therapeutic options for effective management. Multimodality imaging has gained utmost priority for improved target defnition for radiosurgery. In this study, we assessed the utility of Magnetic Resonance Imaging (MRI) for target volume definition for acoustic neuroma radiosurgery.
Twenty patients treated with radiosurgery for acoustic neuroma at our institution were included. Radiosurgery target definition was performed by using CT simulation images only or by using fused T1 gadolinium-enhanced MR images acquired within 1 week before treatment day. A comparative evaluation was made including volumetric analysis of target volumes.
Target volume definition based on CT-only imaging and CT-MR fusion based imaging were comparatively evaluated for 20 patients receiving SRS for acoustic neuroma at our institution. Mean target volumes were 5.7 cc (range: 2.1-13.9 cc) and 6.2 cc (range: 2.3-14.1 cc) with CT-only imaging and CT-MR fusion based imaging, respectively.
MRI may be used as a viable imaging modality for acoustic neuromas and may improve target definition for radiosurgery despite the need for further supporting evidence.
Stereotactic radiosurgery (SRS), Fractionated stereotactic radiotherapy (FSRT), Acoustic neuroma, Vestibular schwannoma, Target volume definition
Acoustic neuromas are benign tumors which are also named "vestibular schwannomas". Acoustic neuromas commonly originate from the transition zone between central oligodendroglial cells and peripheral schwann cells within the vestibular part of cranial nerve VIII. Acoustic neuromas account for the overwhelming majority of tumors occurring in the cerebellopontine angle in the adult population [1-3]. Acoustic neuromas may be associated with neurofibromatosis type II in some patients who present with bilateral lesions and have a typically worse outcome [4,5].
Asymptomatic acoustic neuroma lesions are being increasingly detected by use of contemporary imaging techniques, increased adoption of magnetic resonance imaging (MRI) and high resolution CT [6,7]. Involvement of the cochlear nerve may result in hearing loss and tinnitus as common syptoms of acoustic neuroma.
Management of acoustic neuromas aims at maintaining cranial nerve functions and hearing preservation while achieving improved local tumor control. Observation with serial imaging as a conservative management option has been suggested due to slow growth pattern of smaller acoustic neuromas [8-11]. Nevertheless, a considerable proportion of observed patients may require intervention during the course of their disease. Therapeutic modalities for management of acoustic neuromas primarily include radiosurgery and surgical resection. In the scarcity of highest level of evidence to dictate therapeutic decisions, deciding on optimal management is typically based on individualized assessment taking into account several factors including lesion size and growth pattern, presentation symptoms, age, patient preferences and availability of treatments. Although surgery remains to be a traditional therapeutic option, complications may be substantial in some patients [12,13].
Stereotactic Radiosurgery (SRS) has been judiciously used for management of a variety of indications with considerable success [14-32]. Radiosurgery offers a viable treatment modality for both primary or complementary management of acoustic neuromas [33-35]. Accuracy of radiosurgery for acoustic neuromas may be improved with incorporation of MRI [36,37]. In this study, we assessed the utility of Magnetic Resonance Imaging (MRI) for target volume definition for acoustic neuroma radiosurgery.
Twenty patients treated with radiosurgery for acoustic neuroma at our institution were included in the study. All patients gave written informed consent for radiosurgical treatment, and decision for radiosurgery was taken after thorough evaluation of patients by a multidisciplinary team of experts on neuroradiology, radiation oncology and neurosurgery.
After fixing of the stereotactic head frame to the patients' skull with 4 pins under local anesthesia, all patients underwent Computed Tomography (CT) simulation at CT simulator (GE Lightspeed RT, GE Healthcare, Chalfont St. Giles, UK) available in our institution. A slice stickness of 1.25 mm was used for acquisition of contrast-enhanced planning CT images, and acquired images were sent to the delineation workstation (SimMD, GE, UK) for contouring of the acoustic neuroma lesion along with critical structures in close vicinity of the target. To make a comparison of target definition with CT only and CT-MR fusion, target volumes were determined by using CT simulation images only or by fusion of T1 gadolinium-enhanced MRI acquired within 1 week before radiosurgical treatment. Target volumes generated by using CT-only imaging and CT-MR fusion imaging were comparatively evaluated. Definition of ground truth target volume was decided after colleague peer review and consensus of treating radiation oncologists for every patient. Radiosurgery planning system was ERGO ++ (CMS, Elekta, UK) and treatments were delivered with Synergy (Elekta, UK) Linear Accelerator (LINAC) using 3 mm thickness head-on micro multileaf collimator (micro-MLC) available at our institution.
A median dose of 12 Gy (range: 10-13 Gy) was prescribed to the 85%-95% isodose line to encompass the target volume for radiosurgery. Image Guided Radiation Therapy (IGRT) techniques including kV-CBCT (kilovoltage Cone Beam CT) and XVI (X-ray Volumetric Imaging, Elekta, UK) was used for treatment verification. All patients were delivered dexamethasone with H2-antihistamines after completion of radiosurgical treatment.
Twenty patients treated using SRS at our institution for acoustic neuroma were evaluated for definition of target volume using CT-only imaging and CT-MR fusion based imaging. Mean target volumes were 5.7 cc (range: 2.1-13.9 cc) and 6.2 cc (range: 2.3-14.1 cc) with CT-only imaging and CT-MR fusion based imaging, respectively. Definition of ground truth target volume decided after colleague peer review and consensus of treating radiation oncologists was identical to target determination based on CT-MR fusion based imaging in the overwhelming majority of the patients. Optimal windows and levels were selected to improve target contouring on planning CT and MR images for radiosurgery treatment planning. Axial, coronal and sagittal images were used for achieving improved precision in delineation. Arc Modulation Optimization Algorithm (AMOA) was utilized to achieve improved coverage of the target volume without comprimising normal tissue sparing. Singe session SRS was delivered using the LINAC with 6-MV photons. Planning CT and MR images of a patient are shown in Figure 1.
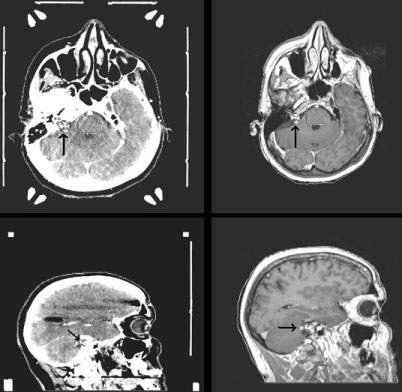 Figure 1: Planning CT and MR images of a patient with acoustic neuroma showing the lesion (black arrow). View Figure 1
Figure 1: Planning CT and MR images of a patient with acoustic neuroma showing the lesion (black arrow). View Figure 1
Target volume definition is a critical part of radiosurgery planning. Due to step dose gradients, part of the target lesion may be left outside of the target in case of incomplete target determination. While CT may typically be successful in evaluation of the bony anatomy and detecting larger acoustic neuroma lesions, some intracalicular lesions with smaller volumes may go unnoticed on CT. Improved contrast resolution of MRI renders it a viable imaging modality for definition of acoustic neuroma lesions. Contrast enhancement on T1 weighted MRI is typical for acoustic neuromas (Figure 1), however, inhomogeneous enhancement may be observed in the setting of larger acoustic neuroma lesions. Also, T2 weighted MR images may be used for improved definition of the optic apparatus and cochlea.
There is extensive research on improving the toxicity profile of radiation delivery in the modern era. Contemporary imaging techniques improve delineation accuracy for a variety of tumor sites. In the context of acoustic neuroma radiosurgery, our study adds to the growing body of evidence supporting the use of MRI for target definition. In our study, definition of ground truth target volume decided after colleague peer review and consensus of treating radiation oncologists was identical to target determination based on CT-MR fusion based imaging in the overwhelming majority of the patients.
In conclusion, MRI may be used as a viable imaging modality for acoustic neuromas and may improve target definition for radiosurgery despite the need for further supporting evidence.
There are no conflicts of interest and no acknowledgements.