Rheumatic diseases can be a paraneoplastic syndrome for an occult neoplasia. Some syndromes are more characteristic than others. In this case report, we present a patient with musculoskeletal symptoms suggestive of both carcinomatous arthritis and polymyalgia rheumatica that was shown to have adenocarcinoma of the lung. The musculoskeletal symptoms accompanied the course of the disease, disappearing with the treatment and re-occurring when it relapsed.
A variety of rheumatic syndrome are associated with neoplasia [1]. They can result from bone and joint invasion by the tumor (metastasis to the musculoskeletal system) [2-4], synovial reaction to neoplastic cells nearby [3,4], hemorrhage into the joint [4], secondary gout [3,4], complications from cancer treatments [2], neoplasia secondary to rheumatic diseases [2], malignancies secondary to immunosuppressive [2], and finally paraneoplastic syndromes [2-4].
This paper will focus on the last one: rheumatic diseases as paraneoplastic syndrome, which consist on symptoms caused by the malignancy, not directly related to the tumor mass or the metastasis [2]. They occur in 10% at the time of diagnosis, but almost 50% on the course of the neoplasia [2,3]. Most paraneoplastic syndromes are endocrine, but rheumatic, as well as others, are also observed [5].
The underlying mechanism of paraneoplastic rheumatic syndromes is unknown but has been extensively hypothesized: 1) Both neoplasia and rheumatic disease have the same trigger [2], 2) Toxins produced by the tumor trigger inflammation and therefore the rheumatic disease [2], 3) Paraneoplastic rheumatic syndromes consist on hypersensitivity reactions, against the intracellular antigens eliminated by apoptotic cancer cells [6], 4) Rheumatic paraneoplastic diseases represent a successful tumor immunosurveillance [7].
Some features of the rheumatic disease should raise suspicion of paraneoplastic syndromes and start a prompt study [3]. Those are: personal or familiar cancer history, exposition to carcinogens, or immunosuppressive therapy, concomitant constitutional symptoms (e.g. Weight loss, loss of appetite, fever, asthenia), atypical presentation, specific manifestations that epidemiologic studies have shown association to neoplasia, age of diagnosis over 50-year-old, acute onset, refractoriness to standard therapy, concomitant paraneoplastic syndromes, anemia, monoclonal band on protein electrophoresis and tumor biomarkers positivity [4,8].
Clinically, the musculoskeletal symptoms follow the course of the malignancy. So, treatment of the neoplasia results in regression of the symptoms, and their recurrence coincides with tumor relapse [4].
On this case report, we will present an example of rheumatic disease as a paraneoplastic syndrome.
We present a 63-year-old male, native from Ukraine but living in Portugal, who works in constructions and smoker (15 pack-years). He presents on the emergency department with a 3 months course of polyarthralgia on small and medium joints. He also complained of reduction on proximal strength on scapular belt and significant weight loss > 15 kgs.
On the clinical examination, he presented with arthritis of the small and medium joints of hands and wrist (red, hot, swollen joints with palpable synovitis), and inability to elevate upper members. He had no palpable lymph nodes or other significant change on the clinical observation.
Laboratory revealed slight anemia (hemoglobin 11.3 g/L, hematocrit 34.4%, normocytic normochromic), with elevation of inflammatory parameters such as leukocytosis of 14.900/L (neutrophils 10.650/L, eosinophils 0.99/L, basophils 0.14/L, and monocytes 1.24/L), C-reactive protein (CRP) 59 mg/L and erythrocyte sedimentation rate (ESR) of 62 mm3. Iron kinetic showed free iron 17 ug/dL (reference values 65-175 ug/dL), transferrin 1.61 g/L (reference values 1.63-3.44 g/L), transferrin saturation 8.8% (reference values 20-40%), and ferritin 259.3 ng/mL (reference values 21.81-274.66 ng/mL). B12 vitamin was below 125 pg/mL (reference values 187-883 pg/mL), with normal level of folic acid 4.3 ng/mL (reference values 3.1-20 ng/mL). Muscle enzymes were below normal with creatinine kinase 10 U/L (reference values 30-200 U/L) and myoglobin of 16.3 ng/mL (reference values below 154.9 ng/mL). Kidney and liver function enzymes were between normal levels. Thyroid function normal with thyroid-stimulation hormone (TSH) 1.92 uUI/mL (reference values 0.35-4.94 uUI/mL) and free thyroxine (FT4) 1.06 ng/dL (reference values 0.7-1.48 ng/dL). Total prostate-specific antigen (PSA) 1.76 ng/mL (reference values below 4 ng/mL). Protein electrophoresis revealed an alpha 1 peak suggestive of inflammation, of 17.7% (reference values 7.1-11.8%). Immunology showed a positive anti-nuclear antibody of 1/160 titer, speckled fine granular pattern. Rheumatoid Factor, anti-citrullinated protein antibodies were negative, as well as anti-ds DNA antibody and extractable nuclear antigen antibodies. Infectious serologies were also negative for hepatitis B and C, human immunodeficiency virus (HIV), and cytomegalovirus (CMV) (IgG positive revealing previous contact, but IgM negative).
Chest radiography had an opacity on right hemithorax, on the lung base (Figure 1). The patient also performed a hands radiography that revealed no erosions (Figure 2). A full body computerized axial tomography (CT) scan was done to exclude neoplasia revealing "hypercaptant lymph node on supra-clavicular fossa 18 × 14 mm. Alveolar condensation without the bronchogram sign, rounded with hipocaptant regions suggestive of necrosis with pleural contact on right basal pyramid. Bronchi caliber reduction. Lymph node conglomerates and hipocaptant lymph nodes pre-vascular and para-tracheal on the right. These aspects are suggestive of primary lung lesion (Figure 3).
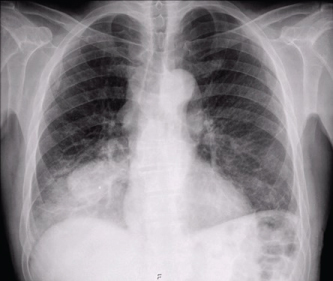 Figure 1: Chest radiography revealing an opacity on right hemithorax lung base. View Figure 1
Figure 1: Chest radiography revealing an opacity on right hemithorax lung base. View Figure 1
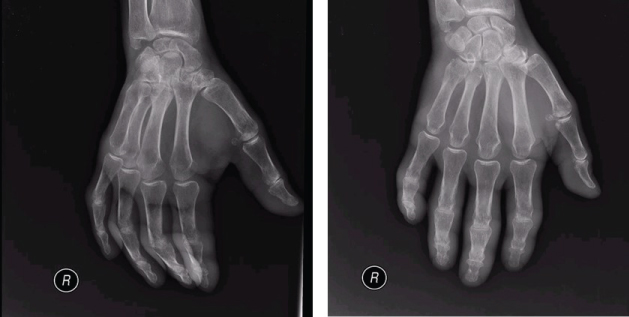 Figure 2: Hands and wrists radiography (right hand) with no erosions. View Figure 2
Figure 2: Hands and wrists radiography (right hand) with no erosions. View Figure 2
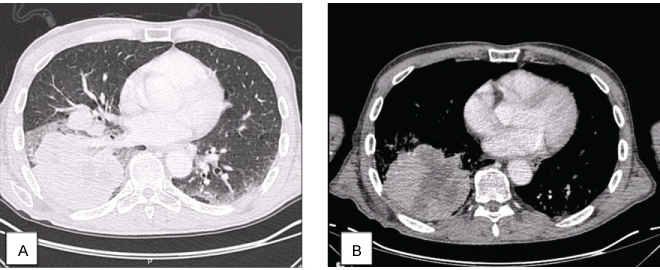 Figure 3: Chest CT: Alveolar condensation without the bronchogram sign, rounded with hipocaptant regions suggestive of necrosis, with pleural contact on right basal pyramid, lung window (A), and liver window (B). View Figure 3
Figure 3: Chest CT: Alveolar condensation without the bronchogram sign, rounded with hipocaptant regions suggestive of necrosis, with pleural contact on right basal pyramid, lung window (A), and liver window (B). View Figure 3
A bronchofibroscopy was performed revealing "Reddish and irregular mucosa. Occlusion of inferior right and medium bronchi. Abundant secretions which were aspirated. Indirect signs of neoplasia".
Bronchi cytology was positive for neoplastic cells characterized as non-small cell carcinoma. A biopsy was performed on the right inferior lobe mass which was positive for thyroid transcription factor 1 (TTF-1) and negative for cytokeratin 5/6 favoring adenocarcinoma of the lung as final diagnosis.
A Positron emission tomography/Computer tomography (PET-CT) staged the malignancy as T3/4N3M0 - IIIB, with an Eastern Cooperative Oncology Group (ECOG) performance status of 2 due to the musculoskeletal symptoms. He was started on chemotherapy with cisplatin and vinorelbin. His musculoskeletal symptoms regressed completely with cancer directed therapy.
Five months after the diagnosis, he was rushed to emergency hospital with tonic-clonic seizures and a head CT revealed a "right subcortical temporal hyperdense expansive lesion, with central necrosis and peri lesional edema" (Figure 4). This was further confirmed by magnetic resonance (MRI), which also excluded secondary lesions (Figure 5).
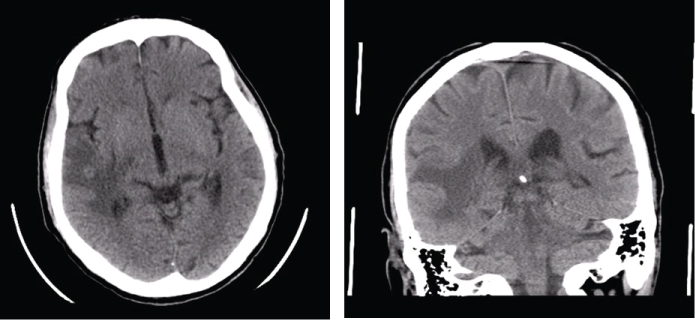 Figure 4: Head CT: right subcortical temporal hyperdense expansive lesion, with central necrosis and peri lesional edema. View Figure 4
Figure 4: Head CT: right subcortical temporal hyperdense expansive lesion, with central necrosis and peri lesional edema. View Figure 4
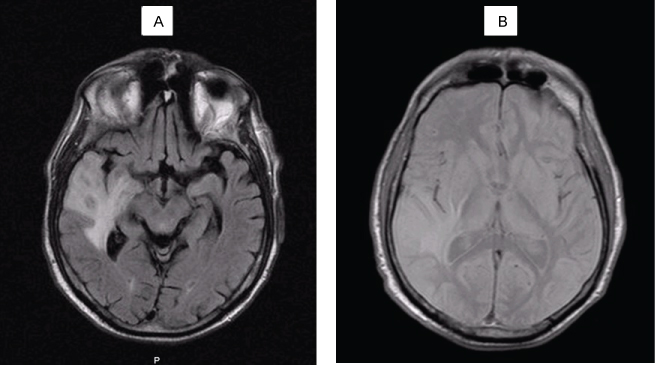 Figure 5: Head MRI: intra-axial lesion, round, 13 mm diameter, cortical-subcortical transition of anterior region of the right superior temporal circumvallation. The aspect is solid, hipointense on T1 (A) and isointense with grey substance on T2 (B), with moderate reinforce with gadolinium injection. Extensive perilesional vasogenic edema on the right temporal lobe. View Figure 5
Figure 5: Head MRI: intra-axial lesion, round, 13 mm diameter, cortical-subcortical transition of anterior region of the right superior temporal circumvallation. The aspect is solid, hipointense on T1 (A) and isointense with grey substance on T2 (B), with moderate reinforce with gadolinium injection. Extensive perilesional vasogenic edema on the right temporal lobe. View Figure 5
The tumor was restaged as stage IV. He was submitted to metastasis ablation with gamma knife radiosurgery, however there was progression of the lesion on subsequent head CT’s. Once again, he presented with polyarthritis of hands and wrists and proximal muscle weakness of the scapular belt, which maintained.
He had several episodes of pneumonia with admission to the hospital. The patient had increasing functional limitation and was admitted to a continued care unit where eventually died.
This patient presented with a rheumatologic paraneoplastic syndrome with musculoskeletal symptoms, which prompt etiologic study heralded the discovery of an asymptomatic adenocarcinoma of the lung. Despite the rapid diagnosis and treatment, the patient succumbed to the malignancy.
The musculoskeletal symptoms were suggestive of both carcinomatous polyarthritis and polymyalgia rheumatica.
Carcinomatous polyarthritis (or paraneoplastic polyarthritis) is a seronegative form of inflammatory arthritis [2,4,9]. The association between the arthritis and cancer is well known [9], but a temporal relationship is necessary for a paraneoplastic syndrome to be assumed [3,4,10]. Usually the symptoms appear 8-12 months before the malignancy development [3,11]. It has an average age of onset of 54, 2 years with a male to female ratio of 1.7:1 [12]. The most frequent malignancies are hematologic (about 1/3), but lung (in men) [3,4,9], breast (in female) [3,4,9], colon, ovarian and gastric have also been described [2,12]. The pathogenesis seems to be related to immune mechanisms such as cytokines and cytotoxic lymphocytes [13] and circulating immune complexes [3,4,14].
With a variable clinical presentation, it often resembles to rheumatoid arthritis (RA) [2-4], particularly late-onset RA [15], however some features set them apart (Table 1). It usually has an acute, rapid onset, with asymmetrical lower extremity involvement, with no nodules or deformities [2-4]. It is accompanied by elevation of inflammatory markers (CRP and ESR), and usually seronegative for anti-citrullinated protein antibodies (ACPAs) but may be positive for rheumatoid factor and antinuclear antibodies [2,3,9]. Synovial fluid has nonspecific inflammatory changes and histology shows and nonspecific synovitis [3,4]. Radiographically, no erosions are visible [2,4]. Constitutional symptoms of weight loss, fever, enlarged lymph nodes and hepatosplenomegaly may be present [2,9]. The arthritis will most often be refractory to non-steroid anti-inflammatory drugs, corticoid therapy and disease modifying drugs [2,16], but responsive to the tumor treatment [3,6,17].
Table 1: Laboratory results. View Table 1
Polymyalgia rheumatica (PMR) can be confused with the previous [18]. It occurs in older people and can have arthralgia of the scapular and pelvic region [18] but also hands and wrist with swelling [19], associated with elevated inflammatory parameters [18]. It can also present with headache, in which case temporal arteritis, a type of granulomatous medium vessel vasculitis, should be suspected [20] (Table 2).
Table 2: Comparison between RA and carcinomatous polyarthritis features [2-4,9]. View Table 2
PMR is often associated to malignancy, but its relationship is questionable, as some symptoms of occult neoplasia can mimic PMR [4,21], such as older age, diffuse myalgia, weight loss, and ESR elevation [4]. Atypical forms are more likely related to malignancy: age < 50 years, asymmetrical involvement, associated arthralgia, ESR < 40 or > 100, and partial response to corticosteroids [4,8]. PMR is associated to hematologic and solid neoplasia [4,21], usually preceding it 1-13 months [8].