Hydroxychloroquine, originally used as an antimalarial agent, is now commonly prescribed for the treatment of rheumatic diseases such as Rheumatoid Arthritis (RA) and Systemic Lupus Erythematosus (SLE). Hydroxychloroquine associated hypoglycaemia is a rare but documented adverse effect, usually noted within the first few months of starting the medication [1]. Here we describe the case of a patient with Rheumatoid Arthritis who developed hypoglycaemia after 7 years of treatment with a stable dose of Hydroxychloroquine.
In 2009 a 70-year-old man was diagnosed with seropositive Rheumatoid Arthritis which mainly manifested as hand deformities. He had been solely taking methotrexate, 15 mg weekly, and hydroxychloroquine, 200 mg twice daily, for 7 years. He had no other relevant past medical history, was an ex-smoker and consumed approximately 4 units of alcohol per week.
In December 2017, he complained of feeling intermittently weak and light headed. The patient who has a daughter with Type 1 diabetes mellitus, likened his symptoms to his daughter's hypoglycaemic events, and used his daughter's blood monitor to record his own blood sugars during these symptomatic episodes. He did this over a twenty-four-day period (Figure 1 and Figure 2). During two of these episodes, his blood glucose was recorded as very low (1.9 mmol/l).
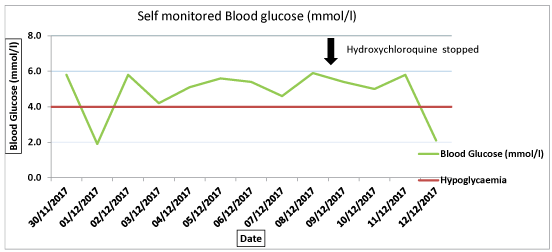 Figure 1: Patient's self-reported blood glucose monitored between 30/11/2017 - 12/12/2017.
View Figure 1
Figure 1: Patient's self-reported blood glucose monitored between 30/11/2017 - 12/12/2017.
View Figure 1
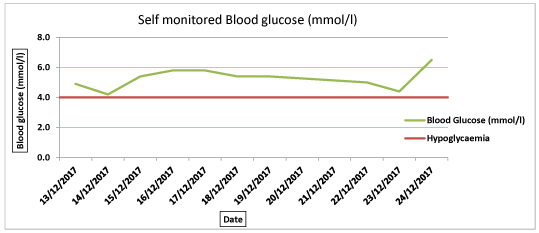 Figure 2: Patient's self-reported blood glucose monitored between 13/12/2017 - 24/12/2017.
View Figure 2
Figure 2: Patient's self-reported blood glucose monitored between 13/12/2017 - 24/12/2017.
View Figure 2
After reviewing the hydroxychloroquine consumer information [2], which mentions hypoglycaemia as a possible adverse reaction, the patient decided to stop this medication. The discontinuation of hydroxychloroquine was nine days after he had commenced glucose self-monitoring, and eight days after recording low blood glucose. He continued to record for a further 15 days and had another symptomatic episode of hypoglycaemia 72 hours after stopping hydroxychloroquine (Figure 1 and Figure 2). For a further 12 days he monitored his blood sugars and felt well and had normal blood sugars (Figure 2).
Formal serological investigations, which included an extended (15 hour) fasting blood glucose (5.0 mmol/L; normal range 3.5-8.0 mmol/L), a serum insulin level (7.6 miu/L; range 3-15 miu/L), C-peptide (709 pmol/L; normal range 330-1400 pmol/L), a morning cortisol level (435 nmol/L; normal range 171-618 nmol/L), HbA1C (36 mmol/L) and short-synacthen test, were all normal. These tests were done six weeks after hydroxychloroquine was discontinued. A fasting blood glucose, done within 72 hours of discontinuation, was normal (5.5 mmol/L). An electrocardiogram did not detect any abnormalities and in particular prolongation of the QT interval was not seen. Magnetic resonance brain imaging was negative for a structural pathology.
Hydroxychloroquine belongs to the aminoquinoline class of medications. There are several theories as to its mechanism of action in treating autoimmune inflammatory conditions. One is that it increases the pH within the intracellular cytoplasmic compartments which is thought to interfere with the antigen presenting function of B cells and macrophages [3]. Failure to acidify the endosome also blocks activation of Toll Like Receptors (TLRs), important to autoimmune processes [4]. Another proposed mechanism of action relates to the ability of aminoquinolines to increase apoptosis in specific cell types which is thought to be its mode of action in SLE. It is suggested that beta-cell activating factor (BAFF), a serum factor which plays a significant role in autoantibody production in SLE, is reduced [3].
Commonly recognized side effects associated with Hydroxychloroquine include nausea, abdominal discomfort, headaches and skin rashes [2]. It is also essential to inform patients of the rare potential development of retinal toxicity [2].
An ophthalmology screen for hydroxychloroquine related complications is advised within the first year of in line with the American Academy of Ophthalmology [5]. This includes a fundal examination and if maculopathy is present visual field and spectral-domain optical coherence tomography (SD-OCT) are recommended [5].
There have been at least three previous case reports of hydroxychloroquine induced hypoglycaemia in non-diabetic patients with RA [1,6,7]. The first case of hydroxychloroquine induced hypoglycaemia in a non-diabetic patient with RA was recognized in 2007 [1] and the first case in a diabetic patient was reported in 1999 [8].
In this case, appropriate investigations were undertaken and failed to find any alternative explanation for the patient's hypoglycaemic episodes. The main difference between our case and previous case reports is the 7-year delay in onset in this side effect compared to the earlier cases.
Although this is an unusual finding in our case, the Hydroxychloroquine packaging insert provides a warning that hypoglycaemia could occur [9]. There has been a recent placebo-controlled study involving pre-diabetic patients randomised to receive hydroxychloroquine or placebo. This found a reduction in blood glucose levels in the hydroxychloroquine group after 12 weeks [10]. From the patient's record of blood glucose levels there was a recorded hypoglycaemic episode recorded three days after treatment cessation. This can be explained by the prolonged terminal half-life of hydroxychloroquine which can be between 40 and 50 days [11]. The patient recorded chart shows there were no further hypoglycaemic episodes in the 12 days after this and he remained well.
The proposed mechanisms of hypoglycaemia related to hydroxychloroquine have been extrapolated from studies on chloroquine, a structurally similar compound. In vitro, chloroquine reduces intracellular insulin degradation, increases intracellular insulin accumulation, delays receptor recycling and activates insulin-mediated glucose transport [12]. A reduction in insulin resistance has also been observed [12,13].
Quattro (1990) and Petri (1996) recognised the anti-hyperglycaemic properties of hydroxychloroquine in patients with type II diabetes mellitus and the benefits relating to glucose metabolism and insulin sensitivity in patients with SLE [14,15]. In diabetic patients, hydroxychloroquine use can result in a reduction in insulin requirements via these mechanisms [3]. This has been also observed in a recent study of patients with Rheumatoid arthritis and SLE and observed in a case report from 2009 [16,17].
In a large prospective study published in 2007, of 4905 adults with rheumatoid arthritis, there were statistically significant lower incidence rates of diabetes in patients taking hydroxychloroquine compared to those who were not [16]. Regression analysis showed that the risk of developing diabetes was significantly lower when taken for more than 4 years [18].
As the number of cases being reported demonstrating an association with hydroxychloroquine induced hypoglycaemia in non-diabetic patients with rheumatoid arthritis is increasing further research into this is necessary.
Recommendations:
• In symptomatic patients taking hydroxychloroquine random glucose testing may be helpful. This should be considered after the first few months if patients describe symptoms of hypoglycaemia, where no other cause has been identified.
• Recorded low blood glucose levels should be appropriately investigated with formal, laboratory tests.
• Patients should be informed about this potential side effect prior to initiating treatment and be aware that if they take blood glucose lowering medication that hydroxychloroquine may potentiate these effects. This may be helpful in lowering insulin requirements.
• Where hydroxychloroquine is suspected as a cause for symptoms, resolution of symptoms after discontinuation of this medication, with or without laboratory correlates, is suggestive of an association.
The frequency of monitoring required remains unclear and further research to answer this is warranted. We also would suggest that more research into the understanding of the mechanism by which Hydroxychloroquine results in hypoglycaemia is needed as in this case hypoglycaemic episodes developed after a long period of stability on this medication.