There is growing interest in the health effects of dietary polyphenols on cardiovascular risk factors. In this context, the effect of hydroxytyrosol on lipid status was investigated in healthy subjects. Hydroxytyrosol, a polyphenol especially known from olive oil, is available on the market from different sources. Absorption from those sources is prerequisite for its effects.
Here we report a study on the absorption and the effects of a pure, synthetic form of hydroxytyrosol compared to an olive extract and a placebo. The phenols hydroxytyrosol and homovanillic acid were chosen as markers in urine. The study was conducted as a mono-center, randomized, double-blind, placebo-controlled, three-way cross-over design with 30 healthy subjects. The four-week oral supplementation phases were separated by two-week wash-out phases.
Neither total cholesterol levels nor HDL (high-density lipoprotein)-cholesterol levels were significantly affected by the study preparations. However, a significant LDL (low-density lipoprotein)-cholesterol reduction was seen after intervention with the pure, synthetic hydroxytyrosol in comparison to placebo (p = 0.0003). LDL cholesterol levels were significantly reduced between the beginning and end of intervention with the pure hydroxytyrosol (delta LDL cholesterol: -9.4 mg/dL ± 10.9 mg/dL, p = 0.0004), whereas no LDL-lowering effects were seen after the intervention with the olive extract or placebo (olive extract: delta LDL cholesterol: +2.1 mg/dL ± 15.2 mg/dL; placebo: delta LDL cholesterol: +4.1 mg/dL ± 15.4 mg/dL). The excreted amounts of hydroxytyrosol and homovanillic acid confirm the uptake of the study products in comparison to placebo (p < 0.0001). Overall the tolerability of the study products was very good.
Our study supports the beneficial effects of pure hydroxytyrosol from a synthetic source on LDL cholesterol.
Hydroxytyrosol, Olive extract, Olive oil, LDL cholesterol, HDL cholesterol, Lipid status, Homovanillic acid
ACC: Acetyl-CoA Carboxylase; AE: Adverse Event; BMI: Body Mass Index; DGAT: Diacylglycerol Acyltransferase; FAS: Full Analysis Set; GCP: Good Clinical Practice; HDL: High Density Lipoprotein; HMG CR: 3-hydroxy-3-methylglutaryl-CoA Reductase; ICH: International Conference on Harmonisation; IRB: Institutional Review Board; LC-MS/MS: Liquid Chromatography Tandem Mass Spectrometry; LDL: Low Density Lipoprotein; oxLDL: Oxidized Low Density Lipoprotein; SAE: Serious Adverse Event
Cholesterol is an important lipid for the body, both as a constituent of cell membranes and as a precursor of several hormones, bile acids and vitamin D [1]. Serum cholesterol, especially elevated serum LDL cholesterol, is a known risk factor for diseases caused by atherosclerosis (e.g., coronary heart disease or cerebrovascular disease) [2]. Lipid-lowering therapies are prescribed to reduce this risk factor [2]. At present, application of statins is the primary and secondary prevention for treating cardiovascular diseases [3]. This therapy, however, is limited by poor tolerability in some cases and by a number of side effects, with the result that one-third of statin users discontinue the therapy within one year [4]. Another potential approach to decreasing the risk of cardiovascular disease is the Mediterranean diet [5,6]. The literature has shown a lower incidence of cardiovascular diseases in individuals consuming a Mediterranean diet [7,8]. It has been suggested that these effects are partly attributable to the olive oil component of the diet [9]. Furthermore, the leaves of the olive plant contain phenolic compounds at a much higher concentration than the olive fruit and oil [10-12]. Olive leaf extract could influence cardiovascular risk via its potential to induce anti-atherosclerotic, hypotensive, antioxidant, anti-inflammatory and hypocholesterolemic effects [13]. A recent study in humans has shown that a phenolic-rich olive leaf extract reduced a number of cardiovascular risk factors, such as plasma total cholesterol, LDL cholesterol and triglycerides. Systolic and diastolic blood pressure, also a cardiovascular risk factor, could be reduced through the intake of the phenolic-rich olive leaf extract [14]. Furthermore, the biological activity of the (poly)phenol components contained within the water-soluble fraction of extra virgin olive oil is more pronounced than in refined olive oil [14,15]. Hydroxytyrosol is one of the main phenolic components of olive oil and is present in the fruits and leaves of the olive tree (Olea europaea L.). Furthermore, hydroxytyrosol is also a known component of red wine. In olive oil, hydroxytyrosol is found both in its free form and as part of more complex compounds such as oleuropein. Oleuropein is responsible for the bitter taste of olives and decreases as the fruit ripens. It is converted into its unglycosylated form (oleuropein aglycone) by enzymatic hydrolysis and finally into hydroxytyrosol, which indicates maturation of the olives [16]. Once absorbed, the phenols undergo first-pass intestinal/hepatic metabolism in the human body. This leads to the formation of sulfate and glucuronide conjugates, as well as metabolites such as homovanillic acid [17-20]. Homovanillic acid and hydroxytyrosol can both be determined in urine and allow conclusions to be drawn about the bioavailability of hydroxytyrosol [21].
The health benefits of the phenolic compounds in olive oil and olives have been well documented in recent decades [22]. Investigations of different olive preparations with different formulations and phenolic content can be found in the literature [13,15,23-29]. Whether these health benefits are also exerted by individual phenolic compounds is a subject of debate. Product specific investigations are needed because of different composition and formulations which might impact the health benefit. This study investigated the uptake and the lipid-lowering effects of hydroxytyrosol from different sources (synthetic and natural extract).
The study was a mono-center, randomized, double-blind, placebo-controlled, three-way cross-over design with 30 healthy subjects. Intervention periods of four weeks were separated by wash-out phases of two weeks. The study was conducted in keeping with ICH-GCP (International Conference on Harmonisation-Good Clinical Practice) guidelines and in compliance with the declaration of Helsinki, and was reviewed by the Institutional Review board (IRB) of the Landesärztekammer Baden happen Württemberg without any concerns.
The nutrition study was performed from July 2016 to December 2016 at BioTeSys GmbH, Esslingen, Germany, an independent study site with a focus on nutritional research.
After having given written informed consent and after completion of the screening procedures, subjects were invited for study visits. Subjects were randomly assigned to the intervention phases. For further details, see the study flowchart in (Figure 1).
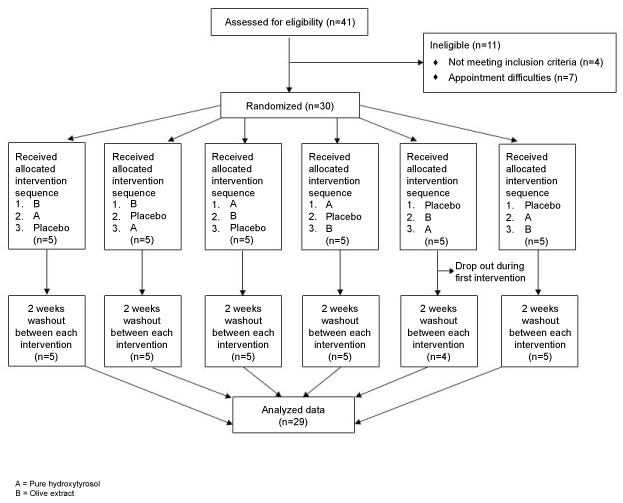 Figure 1: Study flowchart. View Figure 1
Figure 1: Study flowchart. View Figure 1
For standardization, subjects consumed standardized meals (lunch and dinner) on the days prior to the visits and were asked not to consume olives or foods containing olives for the duration of the study. At each visit, lipid status was determined after at least 8 hours of fasting. At the end of the four-week intervention phases, blood samples were collected 8 hours after having ingested the study product. Furthermore, subjects collected their urine in sampling containers 8 hours prior to each visit to the study site in order to analyze hydroxytyrosol and homovanillic acid in their urine. As a safety precaution, each visit also included monitoring for adverse events, concomitant medication and blood pressure measurement.
Thirty (30) subjects were recruited to participate in the study (15 women and 15 men). Twenty-nine (29) subjects finished the study in its entirety. The following inclusion criteria were used: Healthy women and men aged 35 to 65, BMI (body mass index) between 19 and 30 kg/m2, non-smoker, no planned changes in lifestyle regarding activity and nutrition, good physical and mental health, normal clinical findings for hematology and clinical chemistry. The main exclusion criteria were dyslipidemia (defined as cholesterol > 250 mg/dL, LDL cholesterol > 160 mg/dL and triglycerides > 150 mg/dL), history or presence of significant cardiovascular disease or comorbidities, known allergies to the ingredients of the study preparation, the use of drugs or dietary supplements with potentially interfering lipid-modifying, anti-oxidative properties (up to two months prior to the study and throughout its duration), the use of hypertensive drugs, pregnancy or lactation, fat malabsorption, gastrointestinal diseases or conditions, gall bladder resection, chronic intake of anticoagulants, and alcohol or drug abuse.
Hydroxytyrosol from two different sources and a placebo were used as study products during the course of the study. All substances were provided in capsules. A spray-dried powder containing 20% hydroxytyrosol on maltodextrin for use in dietary supplements was used as a source of pure synthethic hydroxytyrosol (HTEssence®, Wacker Chemie AG). The olive extract used was a polyphenol formulation made from olive fruit concentrate spray-dried on maltodextrin with a minimum of 15% hydroxytyrosol content for use in dietary supplements. The natural extract contained hardly any other phenolic compounds like oleuropein or tyrosol. The dosage of the two products used in the study was equivalent to an intake of 30 mg hydroxytyrosol daily. Maltodextrin with no active substances was used as the placebo. Two capsules of the study preparations were ingested twice a day together with a meal (morning and evening).
The current study evaluates the effects of hydroxytyrosol from different sources (a pure, synthetic preparation or an olive extract) on oxLDL (oxidized LDL) (primary endpoint) and on lipid status (total cholesterol, LDL cholesterol, HDL cholesterol and triglycerides) (secondary endpoints) in comparison to placebo. Furthermore, the study also investigates hydroxytyrosol and its metabolites (homovanillic acid) in urine. The current publication focuses on the following secondary endpoints: the effect of hydroxytyrosol on lipid status and hydroxytyrosol and its metabolites in urine. The chosen sample size of 30 subjects evaluated in the cross-over design, which was determined on the basis of oxLDL reduction, was also sufficient for detecting lipid-lowering effects.
Blood was collected in S-Monovette® serum tubes from Sarstedt (7.5 mL Z-Gel for serum), the serum was separated by centrifugation at 3000 × g for 10 min. The lipid status was determined at an accredited routine lab (Synlab laboratories in Leinfelden) using the ADVIA2400 clinical chemistry analysis system from Siemens.
The total volume of excreted urine over approximately 8 hours was determined, and an aliquot of urine stored at -80 ℃ until analysis. For the analysis of hydroxytyrosol and its metabolites, urine samples were treated with glucuronidase prior to extraction. The analysis of hydroxytyrosol and homovanillic acid was performed using LC-MS/MS (liquid chromatography tandem mass spectrometry) technology (WATERS XEVO TQS-micro). In brief, an aliquot of urine was diluted 1:1 with 0.05 M ammonium acetate buffer (pH 5). Glucuronidase treatment was performed for 90 min at 37 ℃. The incubation was stopped by adding acetic acid, cooling down and adding water. Samples were processed by SPE (Oasis HLB 3 cc (60 mg) extraction cartridges) after equilibration with methanol, conditioning with 0.05 M ammonium acetate buffer and elution with ethyl acetate. After drying under nitrogen, samples were reconstituted in 5% methanol and filtered through spin cups prior to LC-MS/MS injection.
A gradient elution using two solvents (10 mM ammonium formate at pH 5 and acetonitrile) was employed for the separation of hydroxytyrosol and homovanillic acid using an Acquity UPLC BEH C18 1.7 µm, 2.1 × 50 mm column. The coefficient of variation was determined to be 13.5% for hydroxytyrosol and 12.6% for homovanillic acid.
To investigate any effects that ingestion of the study preparations may have on lipid status, the parameters of lipid status were evaluated using a linear mixed model that took sequence, period and treatment into account. Due to the secondary nature of the analysis, the difference between the two active treatments and the placebo was assessed by means of pairwise tests performed without adjusting for multiple comparisons. Pre-post differences (compared to baseline) after four weeks of supplementation were used for the analysis. The baseline values were used as covariates. To further evaluate the LDL cholesterol findings, outliers for the delta levels were detected by calculating the interquartile range and applying a factor of 1.5. Because the values of two subjects were outside of this range during the intervention with the pure hydroxytyrosol, the data for LDL cholesterol are depicted additionally without these two outliers.
A linear mixed model with end-of-intervention values was used for evaluating the uptake of the study preparations by comparing the excretion of hydroxytyrosol and metabolites from one study product to another. Hydroxytyrosol and homovanillic acid in urine were evaluated after correcting for the volume of urine excreted over the period of 8 h.
Data are presented for the full analysis set (FAS) for the intervention. The analysis was performed with SAS Version 9.3 and Graph Pad Prism Version 5.04.
In total, 41 subjects were screened for eligibility, of which 30 were enrolled in the study. One subject dropped out prior to the end of the first intervention phase due to personal reasons. Baseline characteristics for the 29 subjects finishing the study are depicted in (Table 1).
Table 1: Baseline characteristics of subjects. View Table 1
All subjects showed normal lipid status, with the upper limit for inclusion for total cholesterol defined at 250 mg/dL.
Overall, the compliance with study products was very good, averaging 97.6-98.9% during the intervention periods and not differing between interventions (p = 0.8517, Friedman test). Only one subject showed a compliance rate < 80% in one of the interventions.
An overview of lipid status during the different intervention phases is presented in (Table 2).
Table 2: Lipid status at baseline (pre) and end of intervention (post) for the three intervention periods. View Table 2
Total cholesterol was very stable during the study. In none of the intervention phases was a significant change found between the levels at baseline and end of intervention. The minor changes summarized in (Table 2) can be attributed to normal physiological variability. The delta changes did not show significant product effects (p = 0.5470).
In terms of LDL reduction, the intervention with pure hydroxytyrosol showed a benefit that was significant in comparison to placebo (p = 0.0249), as well as a trend towards the olive extract (p = 0.0556). These effects were strengthened after outliers were excluded (pure, synthetic source vs. placebo: p = 0.0003; pure, synthetic source vs. olive extract: p = 0.0011, see (Figure 2)).
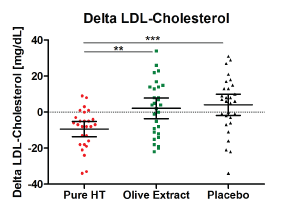 Figure 2: Distribution of delta LDL cholesterol as post-pre difference after intervention with study products (mean ± 95% CI; **p < 0.01; ***p < 0.001); (pure HT (hydroxytyrosol): n = 27, olive extract: n = 29, placebo: n = 29). View Figure 2
Figure 2: Distribution of delta LDL cholesterol as post-pre difference after intervention with study products (mean ± 95% CI; **p < 0.01; ***p < 0.001); (pure HT (hydroxytyrosol): n = 27, olive extract: n = 29, placebo: n = 29). View Figure 2
LDL cholesterol was significantly reduced during the intervention with the pure hydroxytyrosol formulation (by 9.4 mg/dL ± 10.9 mg/dL, p = 0.0004, see (Figure 3)).
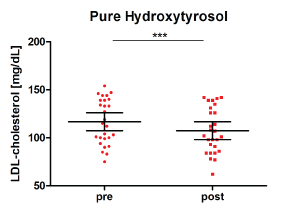 Figure 3: Distribution of LDL cholesterol as pre and post levels after intervention with pure hydroxytyrosol n = 27; (mean ± 95% CI; ***p < 0.001). View Figure 3
Figure 3: Distribution of LDL cholesterol as pre and post levels after intervention with pure hydroxytyrosol n = 27; (mean ± 95% CI; ***p < 0.001). View Figure 3
Intervention with the study preparations showed no impact on HDL cholesterol (p = 0.7073).
The comparison of all three intervention periods showed no significant difference in the delta change for triglycerides (p = 0.1492). No significant change over time could be seen in triglycerides between the beginning and the end of the intervention. Data are summarized in (Table 2).
Hydroxytyrosol was not detectable in samples collected prior to intervention periods. At the end of intervention, urine was sampled after intake of the evening dose of study products containing 15 mg of hydroxytyrosol (for the pure hydroxytyrosol and the olive extract). Unexpectedly, one subject showed detectable amounts of hydroxytyrosol after placebo intervention. This might be explained by the intake of olive-containing products as part of the participant’s normal diet und thus represents a violation of the study restrictions.
After intake of hydroxytyrosol-containing products, hydroxytyrosol was excreted in the urine by a majority of subjects, thus confirming product uptake. Hydroxytyrosol was below the limit of detection only in two subjects after intake of pure hydroxytyrosol and in five subjects after intake of olive extract.
Homovanillic acid is not only a metabolite of hydroxytyrosol but also a major catecholamine metabolite. As such, detectable amounts of homovanillic acid were measured in baseline urine samples. No differences in baseline levels were seen for the three intervention periods. At the end of intervention, the amount of homovanillic acid excreted increased significantly after intake of hydroxytyrosol-containing products. The data presented in (Table 3) indicate that hydroxytyrosol is metabolized quickly and is predominantly excreted as the metabolite homovanillic acid. When corrected for urine volume, the amounts of hydroxytyrosol and homovanillic acid excreted confirm the significant uptake of hydroxytyrosol from study products in comparison to placebo (p < 0.0001), but show no difference between hydroxytyrosol-containing products (excreted hydroxytyrosol: p = 0.6487; homovanillic acid: p = 0.6419).
Table 3: Excreted amount of hydroxytyrosol and homovanillic acid at baseline (pre) and end of intervention (post) for the three intervention periods. View Table 3
The tolerability of the study preparations was overall very good.
Of the subjects undergoing intervention with the olive extract, 13 reported 20 AEs (adverse events). Ten subjects undergoing intervention with pure hydroxytyrosol reported 13 AEs, and 15 subjects undergoing intervention with the placebo reported 28 AEs. One of the AEs was judged to be an SAE (serious adverse event) due to hospitalization. None of these AEs were related to the study products.
Overall, headache and common cold followed by musculoskeletal problems such as back pain were the most commonly reported AEs.
No changes in blood pressure were observed during intervention periods (data not shown).
The health benefits of the phenolic compounds in olive oil/olives have been well documented in recent decades. However, studies have mainly been performed with virgin olive oils naturally rich in phenols or with olive oils enriched with phenolic extracts [23], whereby the resulting lack of consistency is possibly due to variability in the doses, formulations and study designs. The aim of this project was to evaluate the effects on lipid status of four-week supplementation with two different hydroxytyrosol preparations in comparison to placebo. Different products of different origin, composition and formulation are on the market which limits comparability. Therefore, product specific data are necessary. Focus of this study was the evaluation of a synthetic hydroxytyrosol formulation in comparison to placebo. Additionally, a natural hydroxytyrosol extract was integrated in the study design. The study indicated a reduction in LDL cholesterol for the synthetic extract in comparison to placebo. No effects on total cholesterol, triglycerides or HDL cholesterol were seen. The extent of LDL lowering was remarkable, averaging 8% even in the normolipidemic population. The primary target of statins is a 15% LDL reduction [30] in patients with increased LDL cholesterol levels. Triglycerides are more prone to high variability, which was also observed in the investigated study population, despite standardizing external factors to a considerable degree, including standardized meals prior to blood sampling. Nevertheless, some subjects were not compliant with these standardization efforts. This may have limited the ability to detect possible effects on triglycerides the normotriglyceridemic population. No significant impact on lipid status was seen during the intervention with the olive extract.
The lipid-modifying effects of hydroxytyrosol have also been reported in the literature. Pure hydroxytyrosol has been shown to reduce blood triglycerides, and total and LDL cholesterol, and to increase HDL cholesterol in a diet-induced model of atherosclerosis performed in hypercholesterolemic rabbits [29]. Other studies using animal models have also shown reductions in total and LDL cholesterol with olive oil polyphenol extracts or purified hydroxytyrosol [27,31]. LDL cholesterol-lowering effects have also been confirmed in human studies [32], both with extra virgin olive oil consumption and with olive leaf extract [33,34]. By contrast, the in vivo study published by Lopez-Huertas, et al. [35] using 45 mg of purified hydroxytyrosol (99.5%) showed no effect on blood lipids after 8 weeks of supplementation. The sample size of 14 subjects was very small and the study did not include a placebo group. Furthermore, stability problems affecting the study preparations could have been a factor as well, as the product was transported daily on ice to subjects for administration. Hydroxytyrosol was purified from an olive mill waste water extract using food grade solvents, preparative chromatography and further desiccation resulting in a purity of 99.5%. In a study of Colica, et al. [36] with a comparable study design (cross-over with three-week intervention) investigating 15 mg of a water soluble olive fruit extract also no lipid modulating effects and even a slight but significant increase were identified which nevertheless remained in the normal state in the healthy investigated subjects [36]. Such effects were also seen in the present study for the natural extract and placebo phase.
Overall, current evidence of the lipid-modifying effects of olive polyphenols is somewhat controversial, with some studies demonstrating positive lipid-modifying effects such as an increase in HDL cholesterol [15,25,26] while others have failed to show any effects on lipid status [35,37-39]. All the more, product specific data are necessary as so far influencing factors of lipid-modifying properties are not fully understood due to the complex physiology. Anyhow, Priore, et al. investigated the mechanisms underlying the lipid-modifying effects of hydroxytyrosol. A direct, short-term inhibitory effect of hydroxytyrosol on fatty acid, cholesterol and triglyceride synthesis in the liver was seen in rat-liver cells [40]. As key enzymes, acetyl-CoA carboxylase (ACC), 3-hydroxy-3-methylglutaryl-CoA reductase activity (HMG CR) and diacylglycerol acyltransferase (DGAT) were involved in inhibition. The early and direct down-regulatory effect of hydroxytyrosol on fatty acids and cholesterol synthesis was also confirmed in glioma cells [41].
Physiological variability and external confounding nutritional factors could possibly counteract the results in human studies. This might also be one explanation for slightly differing baseline values and why slightly elevated LDL cholesterol levels were seen even after the placebo intervention. Furthermore, it is assumed that confounding factors might have obscured the possible effects of the olive extract. For each intervention phase, baseline parameters were determined after a two-week wash-out phase. Due to the rapid turnover of hydroxytyrosol, no carry-over effects were expected. A cross-over design controls for inter-individual differences and has also been used in other studies investigating the effects on lipid patterns [42]. A parallel design, however, is used predominantly in studies investigating longer term effects.
One strength, but also possibly one limitation of the study was that it investigated lipid-modifying effects in subjects with normolipidemic status, thus limiting the expected effects. In any case, pure hydroxytyrosol did have a physiologically relevant effect on LDL cholesterol.
Different biotransformation pathways have been described for hydroxytyrosol. In this study and given the LC-MS/MS methodology used, evaluation of the uptake of the study preparation focused on an analysis of homovanillic acid and hydroxytyrosol in 8 h urine samples.
Hydroxytyrosol was not detectable in samples collected prior to intervention periods. By contrast, after intake of hydroxytyrosol-containing preparations, hydroxytyrosol could be detected in most urine samples, and highly significant amounts were excreted for both preparations in comparison to placebo (p < 0.0001). No difference in the amounts of hydroxytyrosol excreted were identified between the hydroxytyrosol products (hydroxytyrosol: p = 0.6487, comparison of pure hydroxytyrosol vs. olive extract). Both formulations were provided in the form of a highly water-soluble powder, where the pure hydroxytyrosol was of synthetic origin and the olive extract was of natural origin. Because homovanillic acid is a major catecholamine metabolite, detectable amounts of homovanillic acid were measured in baseline urine samples and during placebo intervention. No differences in baseline levels were seen for the three intervention periods. After intake of hydroxytyrosol-containing study products, the homovanillic acid concentration increased 16.9-fold for the synthetic extract and 10.7-fold for the olive extract in comparison to baseline measurements. No differences in the amounts of homovanillic acid excreted were detected between the pure hydroxytyrosol and the olive extract (p = 0.6419). Homovanillic acid is also one of the main metabolites of hydroxytyrosol, which explains the significant increase in excreted amounts after intake of hydroxytyrosol. Within the study no concentration time curve in blood of hydroxytyrosol from the different sources and formulations were investigated. The uptake of both hydroxytyrosol containing products was only confirmed indirectly in urine by evaluating cumulative excreted amounts within 8 hours. Nevertheless, the uptake kinetic might differ from the two different products and thus also contribute to differing metabolic effects.
This study investigated the lipid-lowering effect of hydroxytyrosol from different sources (a pure, synthetic product and an olive extract) in comparison to placebo in a healthy normolipidemic population. The dosage used was standardized to 30 mg/day hydroxytyrosol, which was administered over an intervention period of four weeks each, with a two-week wash-out phase in between. The investigation of lipid-modifying properties revealed a significant LDL cholesterol reduction for pure hydroxytyrosol in comparison to placebo but was not confirmed for the olive extract. The uptake of both formulations was confirmed by the excretion of hydroxytyrosol and its major metabolite homovanillic acid in urine. Both the pure hydroxytyrosol and the olive extract were administered in the form of a water-soluble powder, and proved to be safe and well tolerated. Overall, the data support the ability of pure hydroxytyrosol to maintain healthy LDL cholesterol levels.
Wacker Chemie AG funded this study and provided the study products. Wacker Chemie AG had no part in the running or the data analysis of the study. BioTeSys GmbH conducted the study as an independent contract research organization. S. Siefer, R. Wacker and C. Schoen are employees of BioTeSys GmbH. Authors declare no conflict interest.