This overview focuses on how to overcome the treatment challenges posed by COVID-19 infection and reduce mortality with antioxidant therapy. The rationale for this treatment approach is explored by addressing the following objectives:
1. To review the pathophysiology of COVID-19 infection in the respiratory tract.
2. To review how the infection and inflammation caused by COVID-19 results in oxidative stress with progression to multiple organ damage and possible death.
3. To review the body's natural response to oxidative stress.
4. To explain how increasing Glutathione level with L-Glutamine treatment could improve surfactant regeneration and reverse the sequelae of COVID-19 infection.
COVID-19 is the viral pandemic caused by the novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) (WHO). SARS-CoV-2 is an enveloped positive-sense virus in the RNA beta Coronavirus family [1]. Identifying a definitive treatment for COVID-19 infection and complications has been challenging considering the rapid and devastating progression for the 20% of patients who develop severe infection. A lot of ongoing research is focused on producing effective antiviral treatment and vaccine. However, it appears that enhancing alveolar fluid clearance and reducing lung inflammation are key to recovery in moderate to severe COVID-19 infection. Based on my previous research on oxidative stress in trauma and surgical patients, I propose that managing the inflammatory process in COVID-19 infection with L-Glutamine could reduce morbidity and mortality thus improving outcome.
There are 2 types of alveolar cells including Alveolar epithelial type 1 cells (AT I) and Alveolar epithelial type 2 cells (AT II) [2,3]. Alveolar epithelial type 1 cells (AT I) are responsible for gas exchange. Alveolar epithelial type 2 cells (AT II) are progenitor cells for type 1 pneumocytes [4,5]. Alveolar type 2 epithelial cells contribute to lung epithelial repair. They also synthesize and secrete all components of the pulmonary surfactant that regulate surface tension in the lungs [6].
Pulmonary surfactant is a membrane-based system, a complex of lipids and proteins (90% lipid and 10% proteins) assembled and secreted by AT II into the thin layer of fluid coating the respiratory surface. Pulmonary surfactant fulfills two simultaneous functions: defensive role as the first barrier against the entry of pathogens; and a biophysical role of stabilizing the air-exposed interface to prevent alveolar collapse. The absence or deficiency of surfactant produces severe lung pathologies [7,8].
The literature is evolving on the pathophysiology of this novel infection [9] with presumed similarity to the SARS-COV, though COVID-19 appears to be more aggressive. Robert J Mason [2] has described three stages of COVID-19 infection based on SARS-COV model summarized below in words and Figure 1.
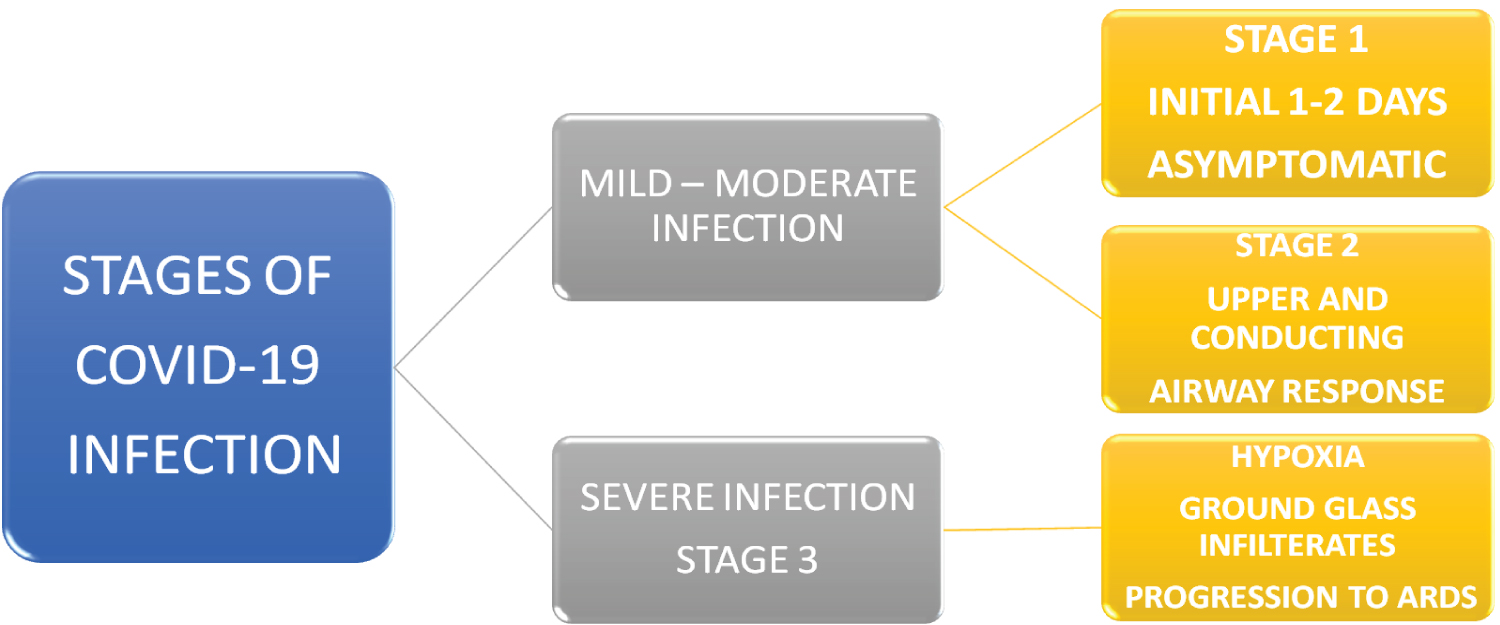 Figure 1: Stages of COVID-19 Infection.
View Figure 1
Figure 1: Stages of COVID-19 Infection.
View Figure 1
1. Stage One: The asymptomatic stage in the initial 1-2 days when COVID-19 virus likely binds to nasal epithelial cells by attaching to cell surface receptor angiotensin-converting enzyme 2 (ACE2) followed by viral replication. This process involves attachment, penetration, biosynthesis, maturation, and viral release [10-12]. ACE2 expression is high in lungs, heart, ileum, kidneys, and bladder. There is limited innate immune response, but infected individuals are infectious. Nasal swabs might be more sensitive than throat swabs in identifying individuals [13-15].
2. Stage Two: This occurs in the next few days with upper and conducting airway involvement. COVID-19 virus propagates and migrates down the lower respiratory tract triggering innate immune response. C-X-C motif chemokine 10 (CXCL10), an interferon receptive gene is elevated, and it is a useful disease marker for COVID-19 infection [9,16,17]. It also predicts the duration of mechanical ventilation [16]. Clinical features are evident with mild to moderate symptoms in 80% of affected individuals, and nasal swabs are usually positive.
3. Stage Three: This involves the lower respiratory tract, 20% of infected patients progress to this stage with about 2% fatality [18]. COVID-19 virus preferentially infects type II alveolar cells compared to type I alveolar cells [19,20]. The virus propagates in type 2 cells and large number of viral particles/toxins are released. The alveolar cells undergo apoptosis and death. The effect of the invasion and unrestrained inflammation [13] is the likely loss of most type II alveolar cells [2,17,21]. This inflammatory response results in elevated CRP [22], depletion of alveolar glutathione, pulmonary oedema, pneumonia, ARDS. Depletion of alveolar glutathione leads to Systemic Inflammatory Response Syndrome (SIRS) and oxidative stress [23]. The chest X-ray and CT scan reveal bilateral pneumonia with ground glass infiltrates.
All viral infections have been associated with the release of reactive oxygen species (ROS), accumulation of lipid peroxidation products and oxidized glutathione (GSH) [17]. The resultant effect on the body is severe oxidative stress with massive release of free radicals and significant depletion of antioxidants [15]. The lung damage in severe COVID-19 infection occurs through inflammatory mediated excessive secretion of proteases and reactive oxygen species (oxidative stress), as well as direct damage from viral replication [24,25] (Figure 2).
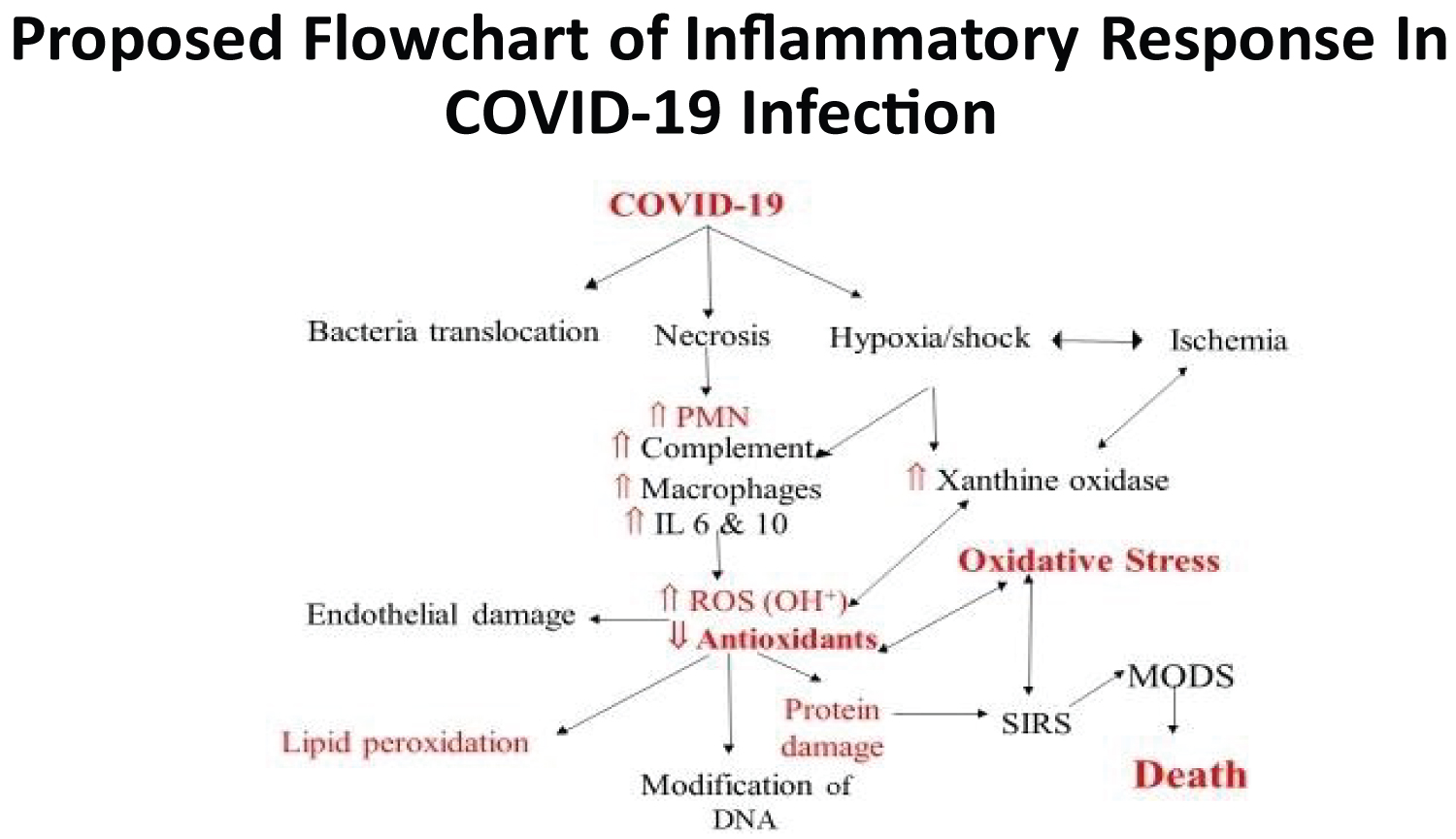 Figure 2: Mechanism of Oxidative Stress in Trauma- Adapted from Obayan, 2004.
View Figure 2
Figure 2: Mechanism of Oxidative Stress in Trauma- Adapted from Obayan, 2004.
View Figure 2
The body maintains oxidative homeostasis by using the antioxidant system to neutralize released free radicals (Figure 3 and Figure 4). The most important antioxidant in the body is glutathione (GSH), others include vitamin E, vitamin C and quercetin. The pathway for the body's response to free radical production is illustrated in the flow chart (Figure 5). Oxidative stress is an imbalance between free radicals in the body and the antioxidant defense system (Figure 6). Oxidative stress can be measured the bedside with Urine Carbonyl Test (UCT) and the oxistress assay developed by Obayan, et al. [23] or other available tests.
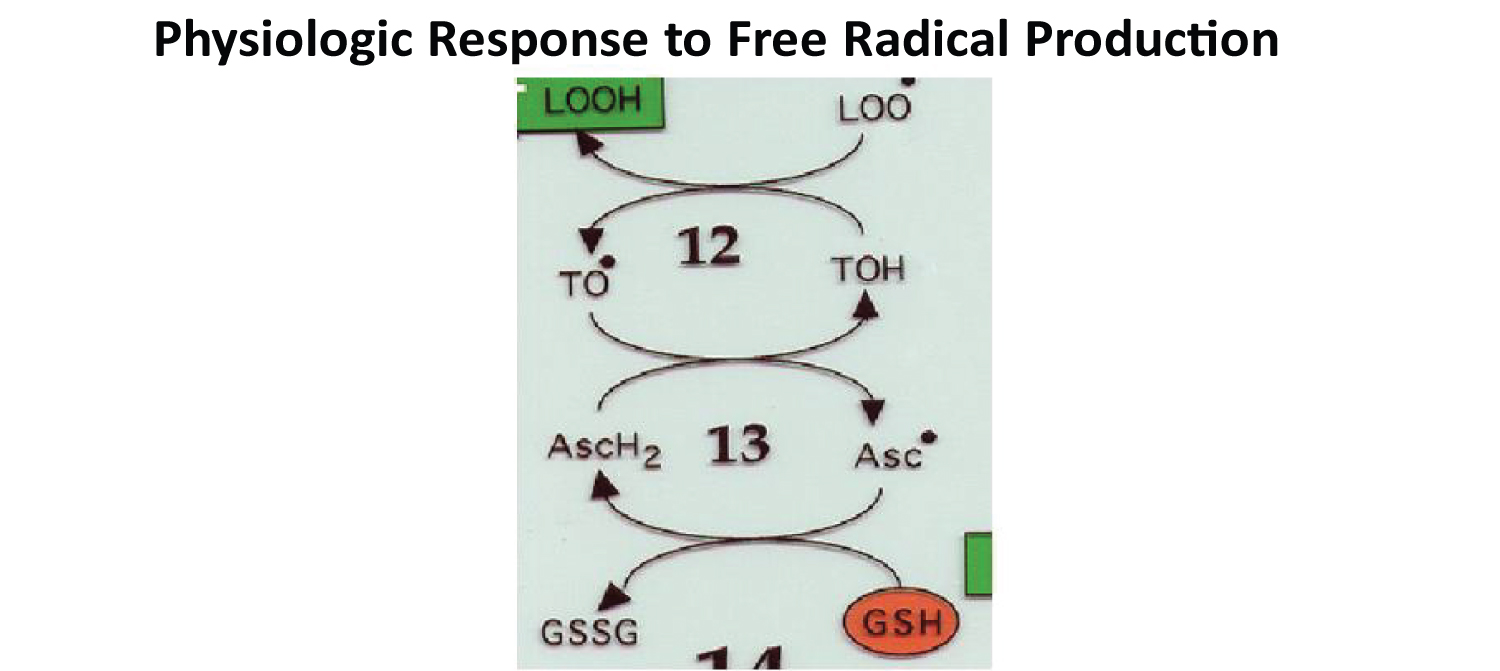 Figure 3: LOO (free radical), TOH (vitamin E), AscH2 (vitamin C) and GSH (Glutathione) - (Obayan, 2004).
View Figure 3
Figure 3: LOO (free radical), TOH (vitamin E), AscH2 (vitamin C) and GSH (Glutathione) - (Obayan, 2004).
View Figure 3
 Figure 4: Oxidative Stress (Obayan, 2020).
View Figure 4
Figure 4: Oxidative Stress (Obayan, 2020).
View Figure 4
 Figure 5: Oxidative Homeostasis (Obayan, 2020).
View Figure 5
Figure 5: Oxidative Homeostasis (Obayan, 2020).
View Figure 5
 Figure 6: Glutathione Cycle (Obayan 2004).
View Figure 6
Figure 6: Glutathione Cycle (Obayan 2004).
View Figure 6
Glutathione is a tripeptide thiol present in all animal cells and made from three amino acids namely, glutamine, cysteine, and glycine (Figure 7). Glutathione is the most important cellular antioxidant in the body. It reverses the effect of free radicals directly and indirectly by regenerating vitamins C and E. High doses of vitamin C have reduced mortality and morbidity in COVID-19 patients [20]. Glutathione has been shown to be effective in reversing the dyspnea and further relieving respiratory symptoms in severe COVID 19 [26,27]. Glutathione plays a role in the recirculation of zinc in the body and acts as an intracellular transporter for zinc into the cell. Zinc has been shown to diminish the intracellular replication of viruses including COVID -19 [28]. The in-transportation of zinc into the mitochondria is significantly improved by cysteine which is a component of glutathione. The participation of zinc in antioxidant protection, redox sensing, and redox regulated signaling is done in collaboration with glutathione [10,29]. Glutathione regenerates surfactant producing alveolar cells [30]. The primary role of free radicals is to attack the virus. However massive free radical release in the absence of neutralizing antioxidants causes significant endothelial damage, lipid peroxidation, protein damage and DNA modification resulting in systemic inflammatory response (SIRS). The body's antioxidant system neutralizes regularly released free radicals that make up 5% of every breath. The initial antioxidant response to excessive free radical release due to inflammation and infection is not different from baseline. The antioxidant system subsequently recruits more potent antioxidants including glutathione to neutralize excessive free radicals [23]. The recruitment process takes about 5-7 days [15], which coincides with the clinical manifestations of COVID-19 infection. Individuals with premorbid medical conditions have a diminished antioxidant reserve and are more at risk of oxidative stress [15,31] (Figure 7). The management of severe COVID-19 infection will require regeneration of alveolar cells and reversal of the inflammatory response. A good antioxidant reserve will likely prevent SIRS and death [32]. However, the antioxidant reserve is likely depleted in most patients with severe COVID-19 infection, therefore exogenous antioxidant therapy is indicated for improved outcome [33].
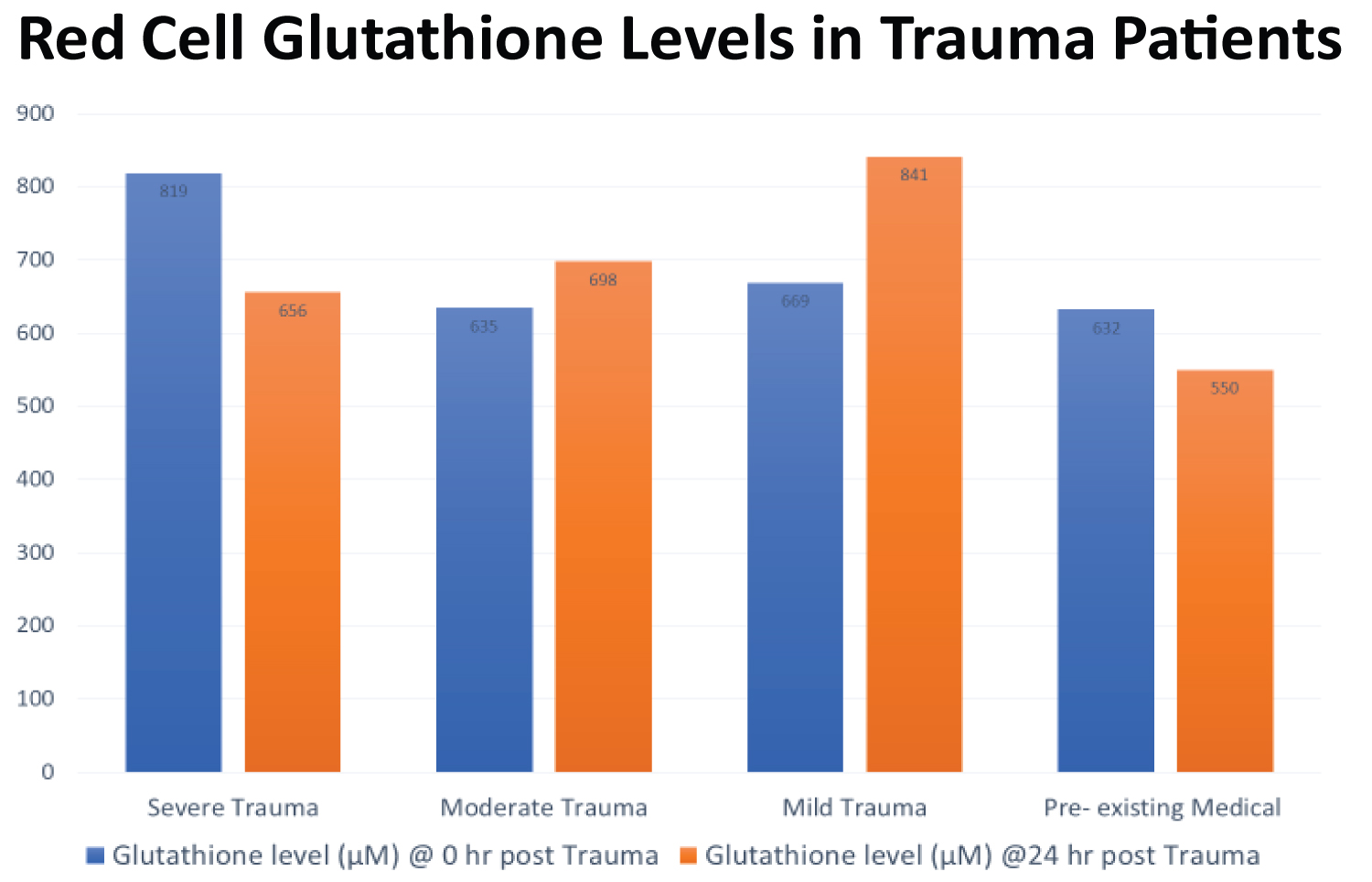 Figure 7: Red Cell Glutathione Levels in Trauma Patients (Obayan, 2004).
View Figure 7
Figure 7: Red Cell Glutathione Levels in Trauma Patients (Obayan, 2004).
View Figure 7
The main source of glutathione is glutamine, a conditional essential amino acid. It is also the most accessible exogenous precursor of glutathione that has been used in critical care, sepsis, trauma, burns and other conditions with oxidative stress [21], known to suppress viral infection such as Herpes simplex virus reactivation in addition to glutamines benefit in colorectal cancer and radiation enteritis [34-36]. Glutamine also plays a vital role in cell proliferation, tissue repair and acts as an energy substrate [21,22]. There is increased production and release of glutamine in response to severe metabolic stress from any cause. Glutamine provides the substrate for rapidly dividing cells of the immune system and gastrointestinal tract [15]. Endogenous glutamine production mostly from skeletal muscles is inadequate in severe metabolic stress [15]. Exogenous glutamine treatment of oxidative stress in major surgery patients produced the following observations: increased plasma glutamine level; increased total plasma antioxidant level (p = 0.05); increased red cell glutathione level; decreased free radical level in the treatment versus non-treatment group (p= 0.036 @ 24 hr) [23].
The benefit of exogenous glutamine administration for oxidative stress in surgery and critical care patients is well documented in the literature. Exogenous glutamine treatment will likely increase antioxidant reserve and modulate the excessive inflammatory process reported in moderate to severe COVID-19 infection [37]. Therapeutic doses of oral or parenteral glutamine between 0.3-0.75 g/kg body weight improved clinical outcome of surgical and intensive care patients [21,23,38]. These doses can be adopted for COVID-19 patients. Prophylactic glutamine treatment has also been employed in different conditions and may also be beneficial in some COVID-19 patients [6,34,37,39].
Glutamine treatment should be considered in the management of moderate to severe COVID-19 infection based on the benefits observed in previous studies with similar pathophysiology.