Health care workers (HCWs) have been disproportionately impacted by the COVID-19 pandemic, but this specific population's infection risk remains unknown. The purpose of this study was to determine the incidence of COVID-19 infection and associated predictors among select hospital staff in the Bronx, New York City.
919 SARS-CoV2 RT-PCR results were retrospectively reviewed from 890 HCWs seeking testing between March 18th and April 24th, 2020. Demographic data, job title, clinical history, PCR results, and morbidity were analyzed.
Of all patients tested, 26.7% (237) tested positive for SARS-CoV2. 17.7% of positive patients demonstrated a temperature of at least 37.5 °C, compared to 6.0% of negative patients (p < 0.00001). 12.2% (29) of patients were clinically asymptomatic. Cough, malaise, fatigue, headache and anosmia were significantly more prevalent in the SARS-CoV2 positive group. Only one patient required intubation, and there were no fatalities recorded. No significant difference was seen in positive rates between clinical, conditionally clinical, and non-clinical HCWs.
We identified an infection rate among HCWs during the height of the COVID-19 consistent with local epidemiological data from the surrounding region. Overall, HCWs who tested positive displayed vital signs congruent with viral illness. The data also suggest that all HCWs, including those with non-clinical roles, had an equivalent risk of COVID-19 infection during the height of the pandemic. As the surge has now affected other cities, this awareness may help shape future policies for protection of exposed health care personnel.
Health care workers (HCWs) have been disproportionately impacted by the COVID-19 pandemic. As of April 9, 2020, of the 49,370 COVID-19 cases reported to the Centers for Disease Control and Prevention (CDC), 9,282 (19%) were among HCWs. Moreover, within a cohort of 1,423 HCWs who tested positive for infection, 55% endorsed likely contact with an infected individual or patient at the workplace [1]. In the era of COVID-19, HCWs must acknowledge the potential risk of personal harm to both themselves and their families while at work.
HCWs continuously expose themselves to potentially infectious bodily fluids, surfaces, and aerosolized secretions while in the hospital setting. As of early March, the National Health Commission of China indicated that > 3,300 HCWs had been infected (of whom 22 died) [2]. Daily occupational exposure, particularly among registered nurses, physicians, patient care assistants, and other employees with continuous access to patients has inevitably increased as communities have become overwhelmed with widespread infection.
HCWs have also been exposed to rising psychosocial and physical demands during this crisis. A recent Korean study noted that during the early days of the epidemic, some personnel did not leave the hospital in order to have time to rest or due to fear of potentially transmitting the infection to their family members if they became infected [3]. In another recent multinational study on stress among 906 HCWs due to the current global crisis, 48 (5.3%) workers screened positive for moderate to very-severe depression, 79 (8.7%) for moderate to extremely-severe anxiety, 20 (2.2%) for moderate to extremely-severe stress, and 34 (3.8%) for moderate to severe levels of psychological distress [4].
Understanding the transmission risk of COVID-19 among various hospital employees and HCWs is particularly important for guiding evidence-based protective measures within hospitals as the US economy recovers from the pandemic. It is also vital to define the incidence of COVID-19 infection among HCWs in New York City as it can possibly be used for anticipatory guidance for regions that are currently experiencing a surge in cases. The purpose of this study was to determine the incidence of COVID-19 and associated predictors of infection among select hospital staff at Lincoln Hospital, New York City.
Lincoln Medical and Mental Health Center (New York City Health and Hospital Corporation) is an acute care hospital providing primary, secondary, and tertiary care services to the South Bronx community. In the fiscal year 2019, Lincoln had 21,269 inpatient discharges, 356,921 outpatient visits, 124,194 emergency room (ER) "treat and release" visits, and 1,782 deliveries [5]. Lincoln is located in the High Bridge-Morrisania section of the Bronx. The population served by Lincoln Hospital suffers from higher rates of economic stress than the rest of the City, with approximately 32% of residents experiencing poverty and 14% unemployment. As of April 4th, 2020, a total of 2,171 individuals treated at Lincoln were confirmed positive for COVID-19, and a total of 1,105 hospital staff were involved in the care of at least one COVID-19 patient [6]. We created a comprehensive clinical database to examine risk factors for acute illness, including demographic factors, prior medical history, and occupational exposure over a 6-week period of employee RT-PCR testing. Given current concerns for a continued wave of infection throughout the country, an understanding of infection rates among HCWs may also be crucial in planning for future public health responses in high incidence regions.
A total of 919 nasopharyngeal swabs were retrospectively examined from employees seeking SARS-CoV2 reverse-transcriptase polymerase chain reaction (RT-PCR) testing between March 18th and April 24th, 2020. After specimen collection, nasopharyngeal swabs were placed in 3 mL viral transport media and processed by BioReference Laboratories (Opko Health Inc., New York). Inclusion criteria included staff of the Hospital (both Lincoln Hospital employees and Lincoln Hospital employees - i.e. temporary staff) tested onsite by Lincoln Hospital's primary care service. Employees presented to the clinic who desired testing due to either close contact with an individual with confirmed infection, or due to clinical symptoms consistent with viral infection. However, testing was not mandatory for all employees and was not consistent during the outbreak. Rather, testing was initially available based on symptoms or high exposure, then on April 1, 2020 was expanded to cover all Lincoln staff including those who were asymptomatic. For many hospital departments, repeat testing was desired from employees who had tested positive previously and had been placed under self-quarantine before returning to work. There were no restrictions as to the number of tests per individual, nor were there required intervals between tests. Demographic data, employee job title, clinical history (including relevant comorbidities), quality and duration of symptoms, positive SARS-CoV2 RT-PCR, results of repeat testing, and morbidity/mortality were analyzed. HCWs tested were stratified into the following categories at the authors' discretion in order to determine the risk profile of each group based on clinical exposure: clinical (medical doctors, registered nurses, physician assistants and patient care associates), conditionally clinical (those working on patient units without direct contact, i.e. social workers, custodial staff, and unit clerks), and non-clinical (i.e. administrators). Human Resource (HR) data from Lincoln Hospital were obtained to confirm mortality and additional demographics not available in our chart review. All of the nasopharyngeal swabs were performed by two volunteer faculties and one resident physician within the department of Otolaryngology/Head and Neck Surgery. Statistical analysis was conducted using SPSS statistical analysis software. Comparison of proportions was employed using Chi-squared and 2-tailed student t-test for statistical analysis with an alpha level of 0.05. All elements of this study were approved by the NYC Health and Hospitals Corporation and Lincoln Hospital Institutional Review Board.
A total of 890 HCWs charts were queried for a total of 919 nasopharyngeal swabs. The information was cross-referenced to the HR data as stated in the Methods. 17 swab specimens were cancelled due to inconclusive results. Among all HCWs tested, 26.7% (235) were positive for SARS-Co-V2. Within the study cohort, 30.4% were male and 69.6% were female. The mean age was 46.6 ± 12.7 years. 32.2% of males tested positive, while only 24.2% of females tested positive (p = 0.01). While the greatest number of patients resided in the Bronx compared to any other NYC borough (441/890 or 49.6%, Figure 1), the greatest proportion of positive patients lived in Manhattan (43/130 or 33%).
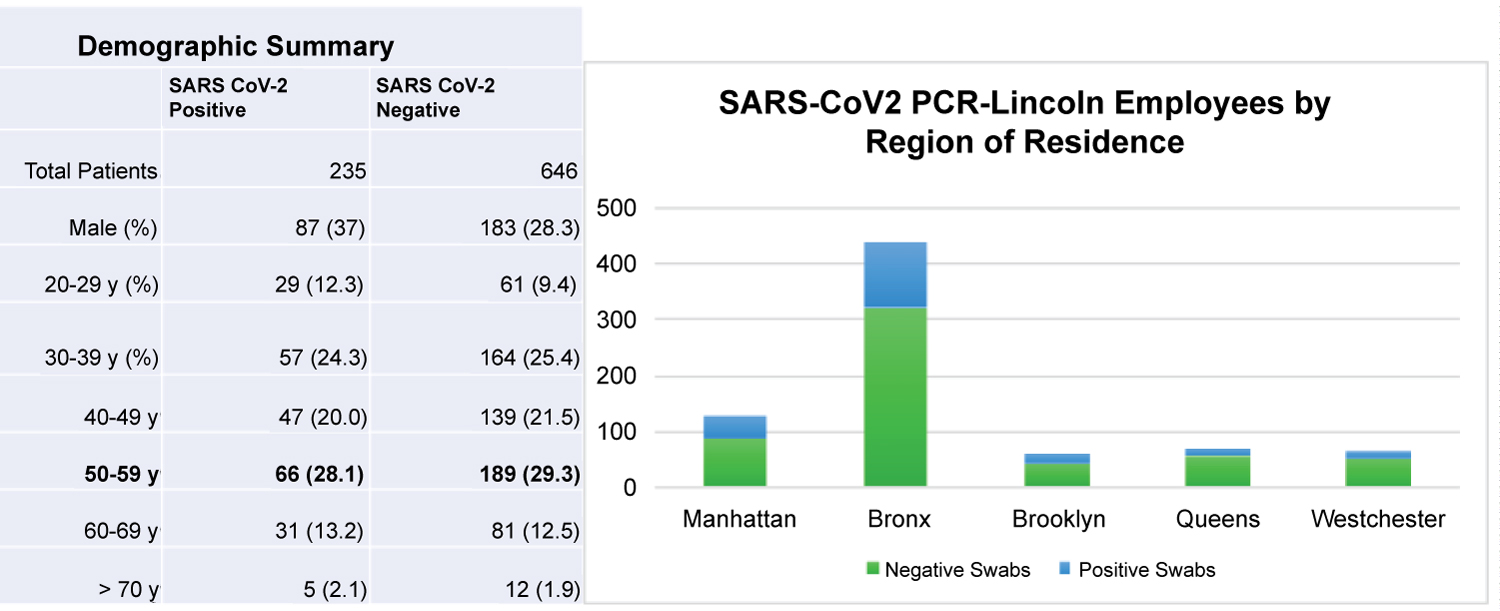 Figure 1: Demographic Summary and PCR Testing by Region of Residence.
Figure 1: Demographic Summary and PCR Testing by Region of Residence.
The most prevalent age demographic presenting for testing both SARS-CoV2 positive and negative patients was 50-59 years. HCWs who resided in the Bronx presented the most to be tested.
View Figure 1
Vital signs significantly differed between tests with a positive vs. a negative nasopharyngeal swab result. 17.7% of positive patients demonstrated a temperature greater than or equal to 37.5 °C, compared to 6.0% of negative patients (p < 0.00001). The mean SpO2 was 97.9% in the positive group compared to 98.3% in the negative group (p = 0.04). The average heart rate on presentation was 86.8 beats per minute (bpm) in the positive group, 4.8 bpm higher than the negative group. The most prevalent demographic by age in either group was the cohort between 50 and 59 years of age (Figure 1).
Of all 237 patients testing positive for SARS-CoV2, 12.2% (29) were asymptomatic (Figure 2). The most common symptoms among positive patients were cough (49.8%, n = 118), myalgia (38%, n = 90), and fatigue (17.3%, n = 41). These symptoms were all significantly more prevalent compared to the SARS-CoV2 negative group (p < 0.001). Headache and anosmia/dysgeusia were also significantly more prevalent among positive patients (Figure 2b). Of note 83% (20/24) of those who endorsed anosmia or dysgeusia tested positive for SARS-CoV2 infection. There were a total of 32 visits to the emergency room among patients tested, and 53.1% (17/32) patients were positive. One patient in the SARS-CoV2 positive group was intubated after admission and later discharged. There were no additional hospital admissions or fatalities in the study cohort.
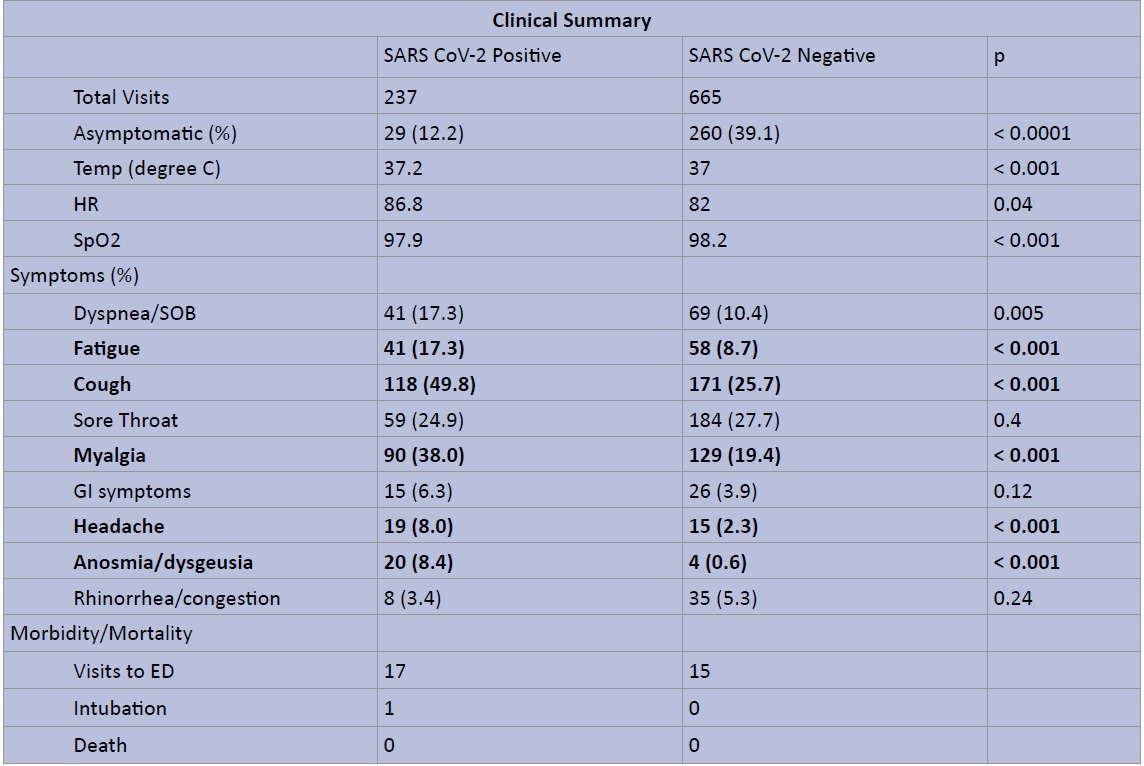 Figure 2a: Clinical Summary of SARS CoV2 RT-PCR Testing - Symptoms, Vital Signs, and Morbidity/Mortality.
Figure 2a: Clinical Summary of SARS CoV2 RT-PCR Testing - Symptoms, Vital Signs, and Morbidity/Mortality.
The data table illustrates total number of clinic visits and average vital signs for SARS-CoV2 positive vs. negative patients, as well as prevalence of symptoms at presentation and noted morbidity or morality.
View Figure 2a
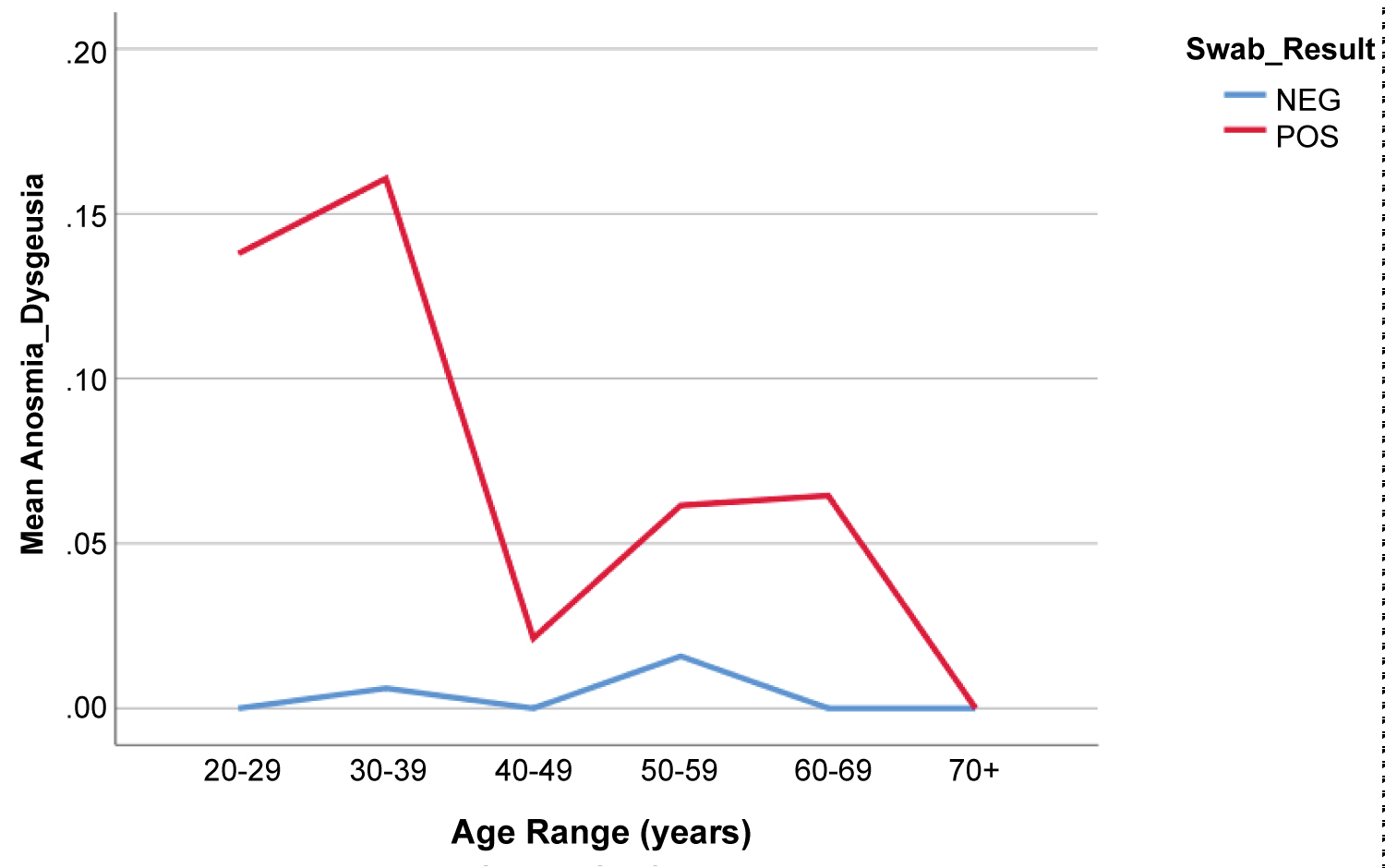 Figure 2b: Percentage of Patients Endorsing Anosmia by Age and SARS-CoV2 RT-PCR.
Figure 2b: Percentage of Patients Endorsing Anosmia by Age and SARS-CoV2 RT-PCR.
Percentage of patients reporting anosmia on presentation as a function of age and SARS-CoV2 PCR result.
View Figure 2b
The most common comorbidity among both positive and negative SARS-CoV2 groups was hypertension (18.1% in positive group vs. 15.9% in negative group), followed by asthma (13.9% vs. 11.4%). There was no significant difference in prevalence of chronic comorbidities between genders or across age groups. However, the total number of chronic diseases was significantly higher across all age groups for those patients who presented to the ED who tested SARS-CoV2 positive compared to those who tested negative (Figure 3).
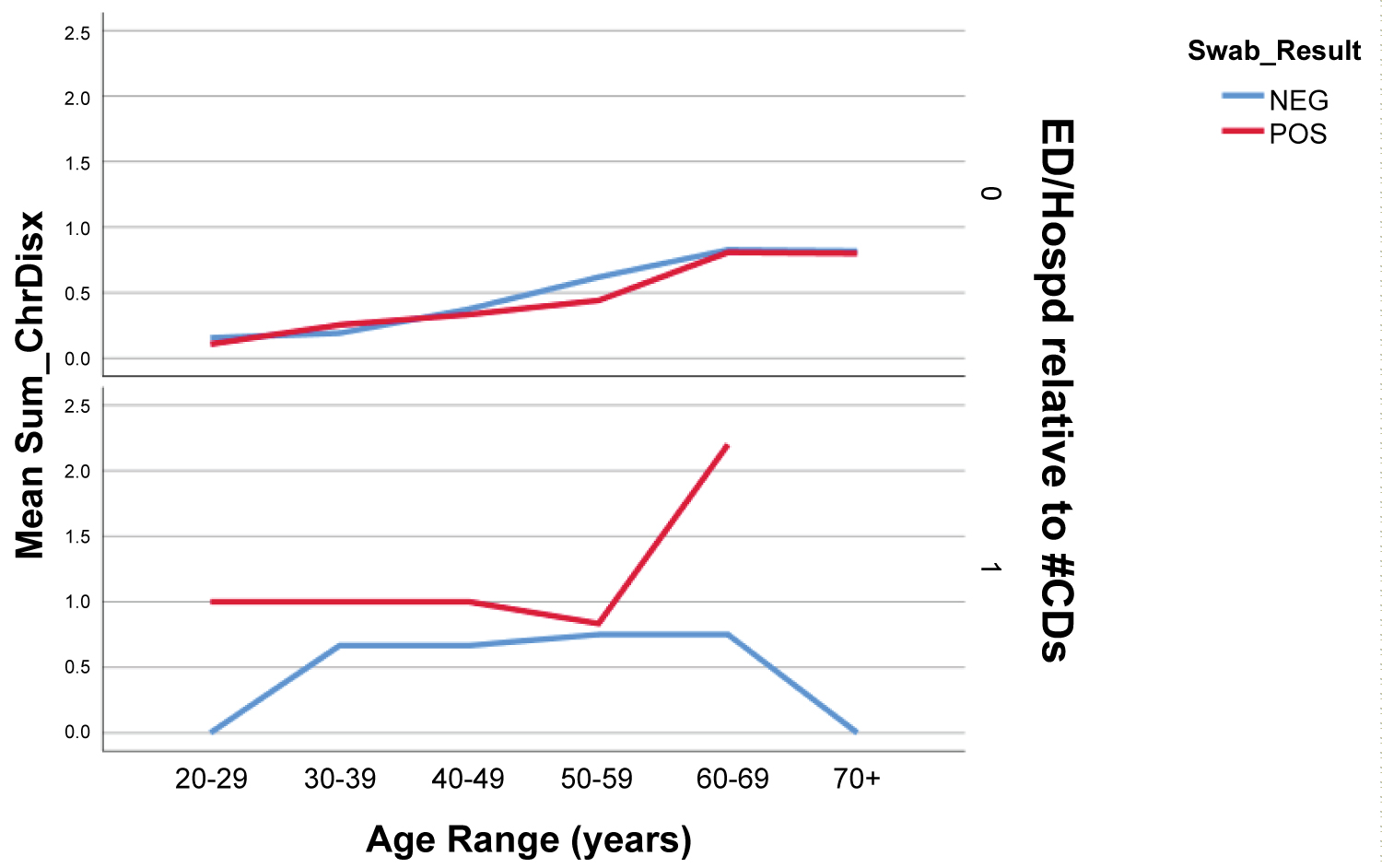 Figure 3: Total # of Chronic Diseases for ED Presentation among SARS-CoV2 Patients.
Figure 3: Total # of Chronic Diseases for ED Presentation among SARS-CoV2 Patients.
Top row - Patients who did not present to emergency room. Bottom Row - patients who presented to emergency room for any complaint after testing. There is a sharp rise in mean sum of chronic disease among patients with positive swab result who presented to ED > 50 y of age.
View Figure 3
When stratified into occupational groups, those groups with the highest proportion of patients testing positive included employees working in nutrition (66%; 4/6 swabs), surgical supply (50% 2/4 swabs), pharmacy (46%, 6/13 swabs), facilities management (44%, 4/9 swabs), and housekeeping (35%, 11/31 swabs) (Figure 4a and Figure 4b). Of note, patient care associates (241), nursing staff (197) and resident physicians (116) were the most highly tested employees during the study period. Employee titles were stratified into clinical (MD, RN, PA, PCA), conditionally clinical (those working on patient units without direct contact, i.e. social workers, custodial staff, and unit clerks), and non-clinical (i.e. administrators). No significant difference was seen in SARS-CoV2 positive test rates between clinical, conditionally clinical, and non-clinical employees.
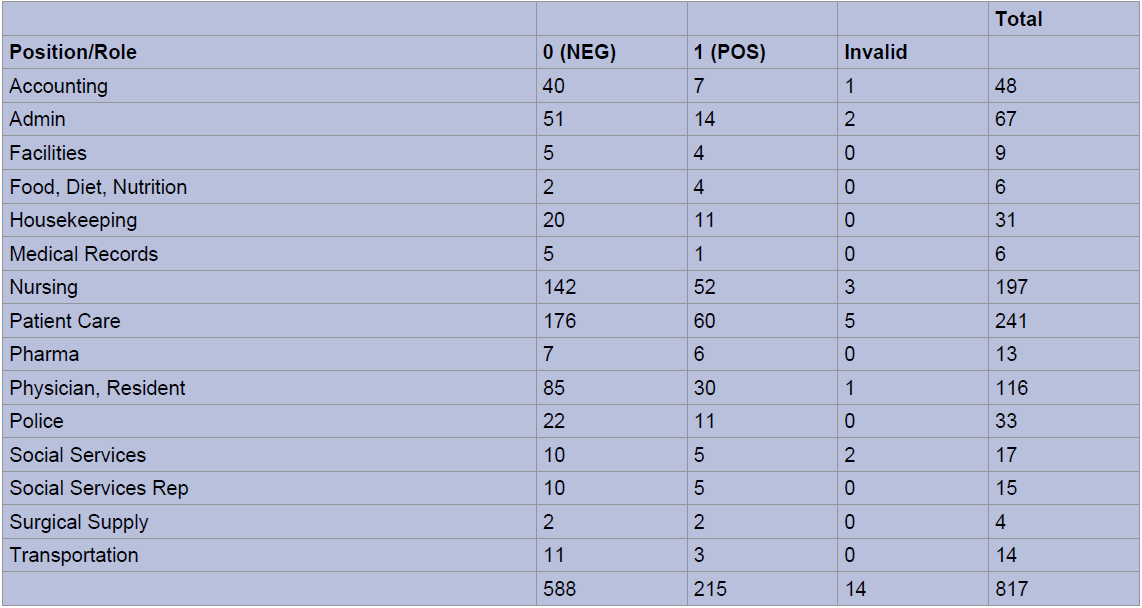 Figure 4a: Nasopharyngeal RT-PCR Testing by Employee Position/Role.
View Figure 4a
Figure 4a: Nasopharyngeal RT-PCR Testing by Employee Position/Role.
View Figure 4a
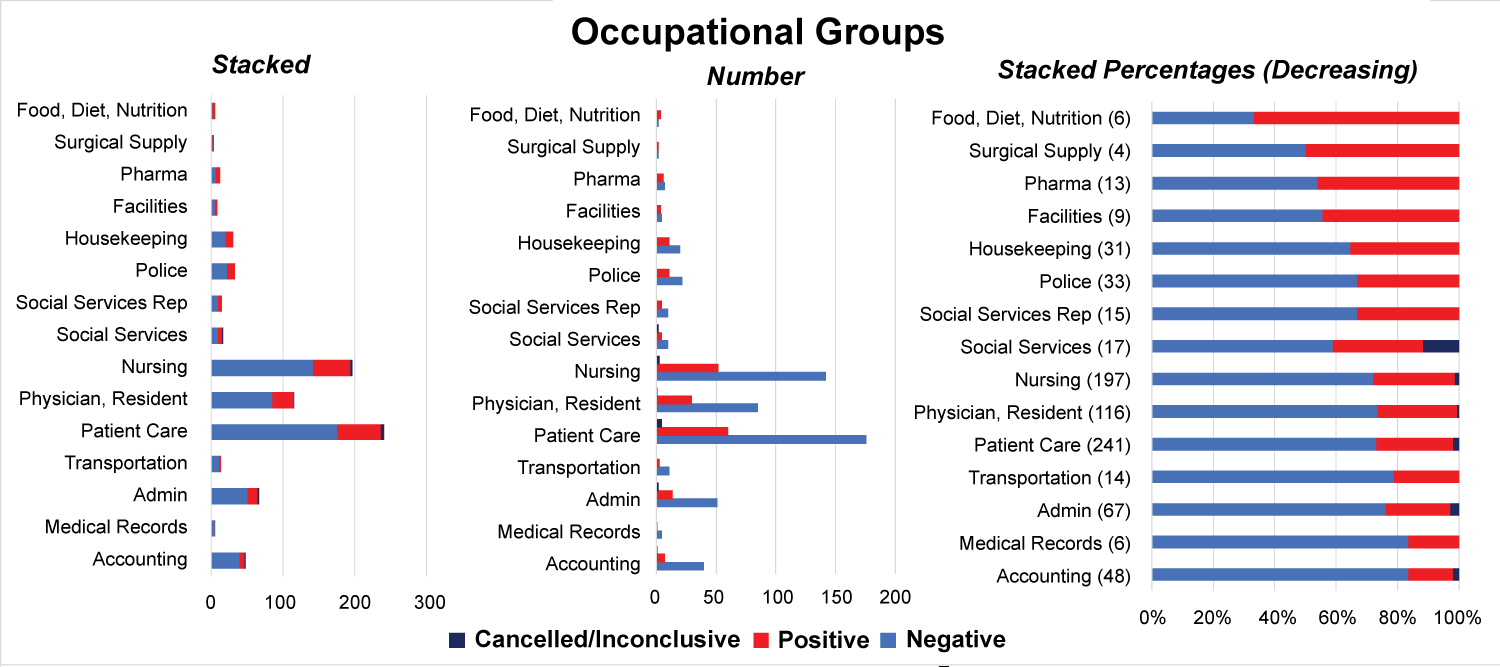 Figure 4b: Percent Positive SARS-CoV2 RT-PCR by Employee Position, Descending Order.
View Figure 4b
Figure 4b: Percent Positive SARS-CoV2 RT-PCR by Employee Position, Descending Order.
View Figure 4b
As cases of COVID-19 continue to rise across the United States, HCWs have been recognized as a particularly high-risk group. In a case series of 138 patients treated in a Wuhan hospital, 40 patients (29% of cases) were HCWs. Among the affected HCWs, 31 (77.5%) worked on general wards, 7 (17.5%) in the emergency department, and 2 (5%) in the intensive care unit (ICU) [7]. Occupational exposure to co-employees, infected patients, and surfaces where viral particles persist collectively present a transmission risk in the healthcare setting. This study is one of few investigations to exclusively define the rate of SARS-CoV2 infection rate among healthcare workers in a high incidence region.
We found a 26.7% positive test rate among all employees who presented for testing in a highly affected region of the Bronx NY, with a higher prevalence among males. Zip code specific data for the Bronx cite a rate of 29.1% infection (with 1,698 deaths as of May 18, the highest in NYC) which is generally consistent to the data seen in this study [8]. While the CDC has reported that nearly 20% of infected Americans work in healthcare, it is unclear if the rate of transmission is significantly higher in the workplace for these individuals. In a recent Chinese study examining viral shedding patterns in COVID-19 patients with acute respiratory distress syndrome, over 60% of samples from the toilet, sink and door handles of an individual with SARS-CoV2 were positive for viral RNA [9]. This may have suggested an increased risk of acquiring viral particles among hospital employees. However, the data from our study show an infection rate that closely approximates that of the general population in the region.
The utility of N95 masks as well as other personal protective equipment (PPE) has been shown to significantly decrease risk of COVID-19 among HCWs in many case reports, including in the context of aerosolizing procedures [10]. Interim guidance for the usage of PPE in the Interim Report from WHO emphasized the importance of simple preventative measures such as hand hygiene, N95 or appropriate facial mask, and social distancing whenever feasible [11]. These practices were enforced throughout the hospital during the time period for this study and likely attenuated transmission rates despite increased exposure among employees tested.
From our cohort, 12.2% of patients who tested positive were asymptomatic. While the CDC has cited a nearly 40% asymptomatic carrier rate [12], this study cohort was a self-selected group of HCWs who chose to undergo testing. This is likely to have increased the overall proportion of symptomatic HCWs tested, which would have the effect of lowering the incidence of asymptomatic carriers in this cohort.
Vital sign differences between positive and negative SARS-CoV2 groups are consistent with the known systemic inflammatory response associated with COVID-19 infection, including tachycardia, fever, and hypoxia. Of note, differences in vital sign values were not large, but statistically significant given the number of patients in the study. Of importance was the statistical difference between patients who demonstrated a temperature greater than 37.5 degrees Celsius between positive and negative groups. Although absolute differences in vital signs were small between positive and negative HCWs, many institutions have utilized temperature as a means to screen hospital staff for COVID-19 on a daily basis.
Medical comorbidities were seen in both groups, and the rates of hospitalization/ED presentation were higher among COVID-19 positive patients with history of medical comorbidities. Hypertension was seen most often in both positive and negative groups followed by asthma, but with no significant difference in prevalence. According to data from the COVID-19 Associated Hospital Surveillance network, older patients with obesity, hypertension, diabetes, cardiopulmonary disease are at a high risk of hospitalization [13]. There were no mortalities during the study and this was confirmed with the Department of Human Resources of the hospital four weeks after the study period.
Patients who tested positive were more likely to complain of cough, headache, fatigue, myalgia, and anosmia/dysgeusia. Of note anosmia was a very specific finding, with 83% of patients endorsing anosmia having a positive test result. Mao, et al. analyzed the frequency of neurological manifestations in 214 patients with coronavirus disease 2019 (COVID-19), identifying anosmia in 11 (5.1%) patients and dysgeusia in 12 (5.6%) patients [14]. It is unclear how long these symptoms persist among COVID-19 patients. Of note, rhinorrhea and congestion were not correlated with positivity, and in fact were more prevalent in patients who did not test positive (Figure 5). These findings correlate with a recent review on COVID-19 by Ear, Nose and Throat (ENT) specialists in March 2020, which emphasized that ENT symptoms were uncommon with COVID-19 as nasal congestion and rhinorrhea were observed in less than 5% of cases [15].
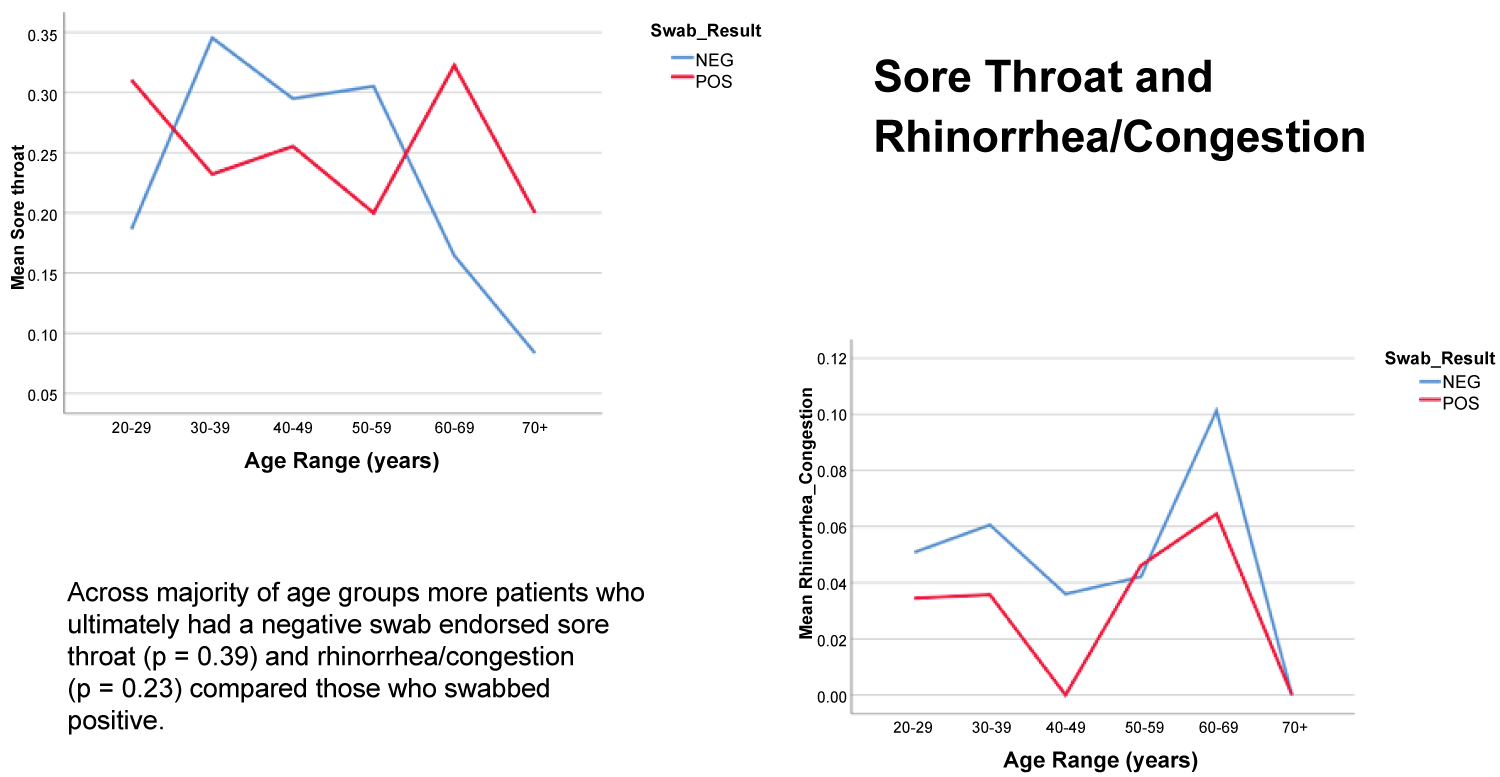 Figure 5: Percentage of Patients Endorsing Sore Throat and Rhinorrhea/Congestion by Age and SARS-CoV2 RT-PCR.
View Figure 5
Figure 5: Percentage of Patients Endorsing Sore Throat and Rhinorrhea/Congestion by Age and SARS-CoV2 RT-PCR.
View Figure 5
While employees directly involved in patient care (clinical HCWs) were the most tested in the study, the highest proportion of positive employees were those who worked in facilities, food preparation, pharmacy, and surgical supplies (Figure 4). The significance of this difference is difficult to establish due to overall low numbers of non-clinical employees tested. However, this may suggest an equal if not elevated risk of infection among non-clinical workers, many of whom are equally exposed to potential infection but who may not be prioritized to receive PPE. There have been no large-scale studies examining the cumulative risk of non-clinical healthcare workers during COVID-19, and so further investigation is warranted.
Our study has the limitations of a large-scale retrospective review. Although a limited number of individuals performed testing, variations in method of nasopharyngeal swabbing may have increased false negative rates. However, the false negative rate was likely low given that all of the physicians performing tests were adequately trained faculty within the Otolaryngology department. The study is also subject to the inherent acquisition bias of database review which may have altered results. Furthermore, it is difficult to ascertain the source of infection for most, if not all, of positive cases as many employees may not have had adequate resources to socially distance outside of the hospital, increasing the likelihood of disease transmission outside of the workplace. The equivalent infection rate between the hospital setting and surrounding Bronx community suggests that perhaps HCWs are not at an increased risk of infection at work, but determining the relative environmental risk of acquiring infection at home vs. work for HCWs is beyond the scope of this study.
In this study we identified an infection rate of 26.7% among HCWs during the height of the COVID-19 pandemic in a highly affected region of NYC, which is generally consistent with local epidemiological data from the surrounding region. Overall, HCWs who tested positive displayed vital signs congruent with viral illness, and HCWs presenting with anosmia were much more likely to test positive. Males were more likely to test positive than females. HCWs with medical comorbidities that tested positive for COVID-19 were more likely to present to the emergency room. There was no significant difference between positive test rates between clinical and non-clinical HCWs. However, those in traditionally non-clinical roles demonstrated a higher proportion of HCWs testing positive. Future studies are necessary to elucidate the timeline of seroconversion among PCR positive patients in regards to serum antibodies, the effect of socioeconomic status, education, and race on testing results, as well as the comparative rates of repeat infection among clinical vs. non-clinical health care workers.
No external funding resources were utilized in this study.