Coronavirus disease 19 (COVID-19), originated at Wuhan city of China in early December 2019 has rapidly widespread with confirmed cases in almost every country across the world and has become a new global public health crisis. The etiological agent was designated as Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The virus was originated in bats and human transmission primarily occurs through direct, indirect, or close contact with infected people through infected secretions such as respiratory secretions, saliva or through respiratory droplets that are expelled when an infected person coughs, sneezes, or speaks. The World Health Organization coined the term COVID-19 and declared this novel coronavirus disease as a pandemic on March 11, 2020. The virus is highly contagious and the incubation period ranges between 2-14 days. The virus infects the human respiratory epithelial cells by binding through Angiotensin-Converting Enzyme 2 (ACE2) receptors. Many infected people are either asymptomatic or develop a mild respiratory illness. The major clinical symptoms of the disease are fever, non-productive cough, fatigue, malaise, and breathlessness. Severe illness such as pneumonia, acute respiratory distress syndrome (ARDS), and death occurs in the elderly and patients with comorbid conditions. The case fatality rate is estimated to be 2-3%. The rapid surge was observed in new cases and COVID-19 related deaths outside of China since the beginning of March-2020. As of June 8, 2020 more than 7 million confirmed cases and > 400 thousand deaths were reported from 213 countries and territories. The disease is mainly diagnosed by the detection of viral RNA in nasopharyngeal swab or Broncho-alveolar lavage (BAL) by polymerase chain reaction reverse transcriptase-polymerase chain reaction (RT-PCR). Treatment is basically symptomatic and supportive. Several vaccines are still under various stages of clinical trials. Remdesivir was the first antiviral drug approved for treatment but its efficacy is yet to be determined. At present preventive measures such as contact, droplet, and airborne precautions are the main strategy to control the spread of the disease.
Coronaviruses, COVID-19, Pandemics, Pneumonia, Polymerase chain reaction
The coronaviruses of zoonotic origin have resulted in several outbreaks of severe respiratory infections in the past twenty years and were notified as major pathogens of public health importance [1-3]. The zoonotic coronaviruses outbreaks that have occurred in 21st century namely SARS (Severe Acute Respiratory Syndrome) in 2002 and MERS (Middle East Respiratory Syndrome) in 2012 have resulted in several cases of pneumonia, Acute Respiratory Distress Syndrome (ARDS), multiorgan dysfunction, and approximately 800 deaths in each [4-7]. The detailed investigations during the outbreaks of SARS and MERS have confirmed that civet cats and dromedary camels are the sources of infection to human respectively [6,7]. Added to these is the recent outbreak of novel coronavirus infection that originated in early December-2019 in Wuhan city of China. It has spread rapidly across the globe and has become the major pandemic disease of the 21st century. This article focus on the review of the current COVID-19 outbreak emphasizing etiology, epidemiology, clinical presentation, and diagnosis, treatment, and prevention strategies. The knowledge concerning this virus is evolving rapidly and hence the readers are encouraged to update themselves regularly.
Coronaviruses belong to a large family of enveloped RNA viruses that possess crown-like surface glycoprotein projections (hence the name Coronaviruses). They have a non-segmented single-stranded positive-sense RNA genome [8-10].There are four main sub-groups of coronaviruses, namely alpha, beta, gamma, and delta. Genomic studies have shown that animals (humans, bats, camels, rodents, etc.) are the gene sources of alpha and beta coronaviruses, while birds seem to be the sources of gamma and delta coronaviruses [8-10]. Common human coronaviruses (Figure 1) namely 229E, NL63, OC43, and HKU2 which have been in circulation are generally known to cause mild self-limiting upper respiratory tract infection. However, the recently identified strains of animal origin such as SARS-CoV-2, MERS-CoV, and the present SARS-CoV-2 which belong to beta-coronaviruses have resulted in large outbreaks with varying clinical severity ranging from mild self-limiting to life-threatening illness [8-10].
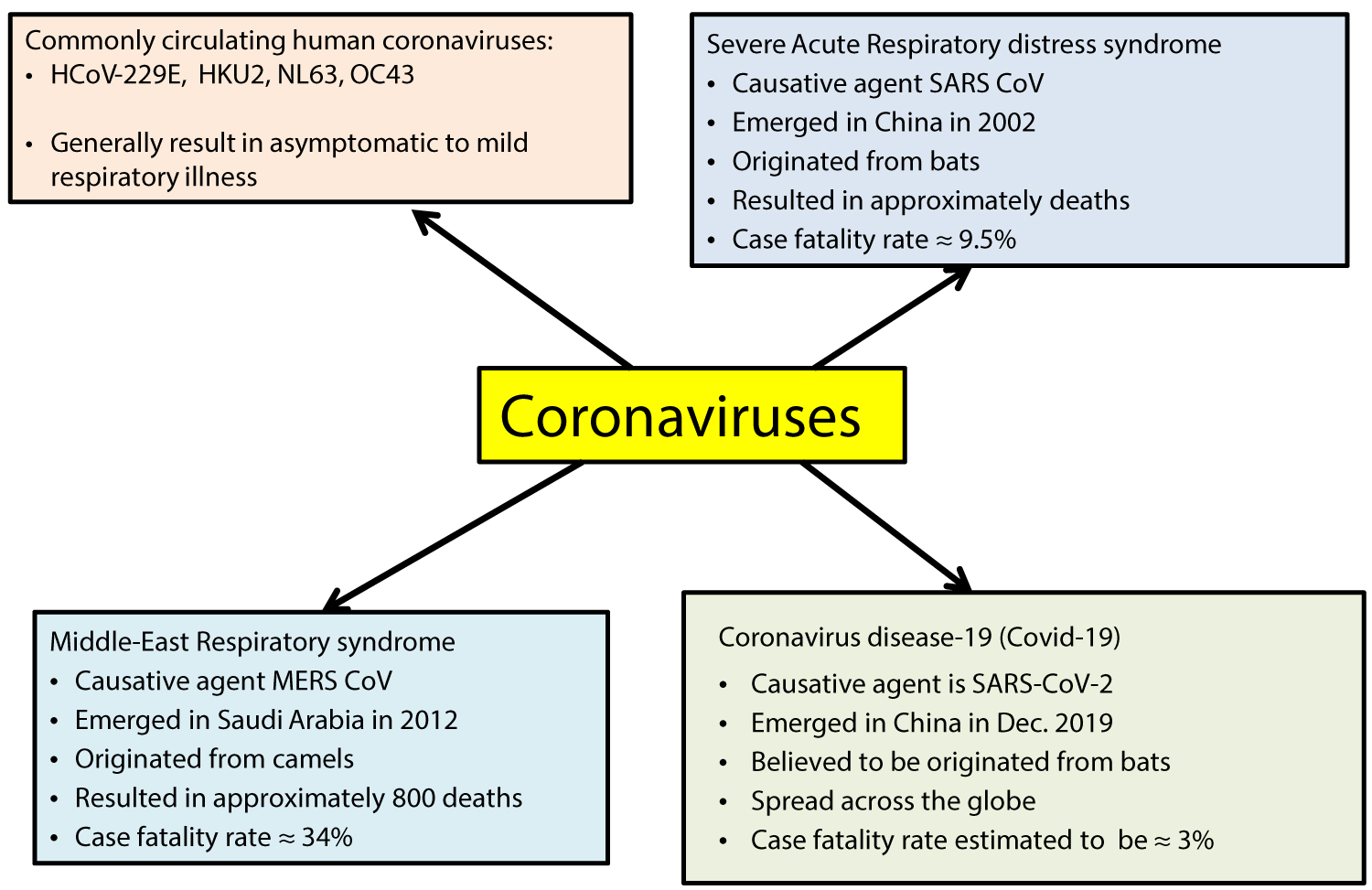 Figure 1: Summary of Coronaviruses.
View Figure 1
Figure 1: Summary of Coronaviruses.
View Figure 1
The expansion of genetic diversity among coronaviruses is primarily attributable to its wide animal host range and ability to undergo a high rate of genetic recombination and mutation. The difference in cell tropism, host range restriction, and pathogenicity among coronaviruses are primarily due to the specificity of viral surface attachment glycoprotein (S glycoprotein) [7]. The complete genome sequencing studies on RNA extracted from nasopharyngeal and sputum samples of COVID-19 affected patients have demonstrated phylogenetic similarities with the SARS-CoV-2 [9,10]. Therefore, 2019-nCoV was recently renamed as SARS-CoV-2 by the International Committee on Taxonomy of Viruses (ICTV). Though the primary source for SARS-CoV-2 is not very clear, it is believed that bats seem to be the source, and infection might have transmitted to humans from the infected bats [9,10].
A cluster of patients presented with pneumonia of unknown etiology in Wuhan city of China was reported to the Chinese Center for Disease Control (CDC) on 31, December 2019. Initial investigations identified a novel coronavirus designated as SARS-CoV-2 (previously known as 2019-nCoV) as the etiological agent [9]. The infection was originated in Huanan seafood and animal market in Wuhan city of Hubei province and it provided some link to an animal to human transmission through the sale of seafood and live animals [6,7]. Subsequently, the infection was noticed in increasing number of patients inside as well as outside the Wuhan city, who did not have exposure to animal markets and this suggested person to person transmission [6,7]. The novel coronavirus was found to be highly contagious and has rapidly spread across the world within a span of 2-3 months. The number of COVID-19 related cases and deaths increased exponentially outside of China since the beginning of March 2020 [11,12]. So far around 213 countries and territories have reported novel coronavirus cases [12]. On January 30, 2020, the International Health Regulations Emergency Committee of the World Health Organization declared the outbreak as a public health emergency of international concern (PHEIC) [13]. Further, on February 19, 2020, WHO coined the term COVID-19 and declared this novel coronavirus disease as a pandemic on March 11, 2020 (Figure 2: Timeline in the key events of SARS-CoV-2) [1,10]. As of June 8, 2020, more than 7 million cases and 400 thousand deaths were reported globally. The COVID-19 has resulted in unprecedented human and health crisis and the measures undertaken to contain the frightening pandemic has resulted in the global financial crisis.
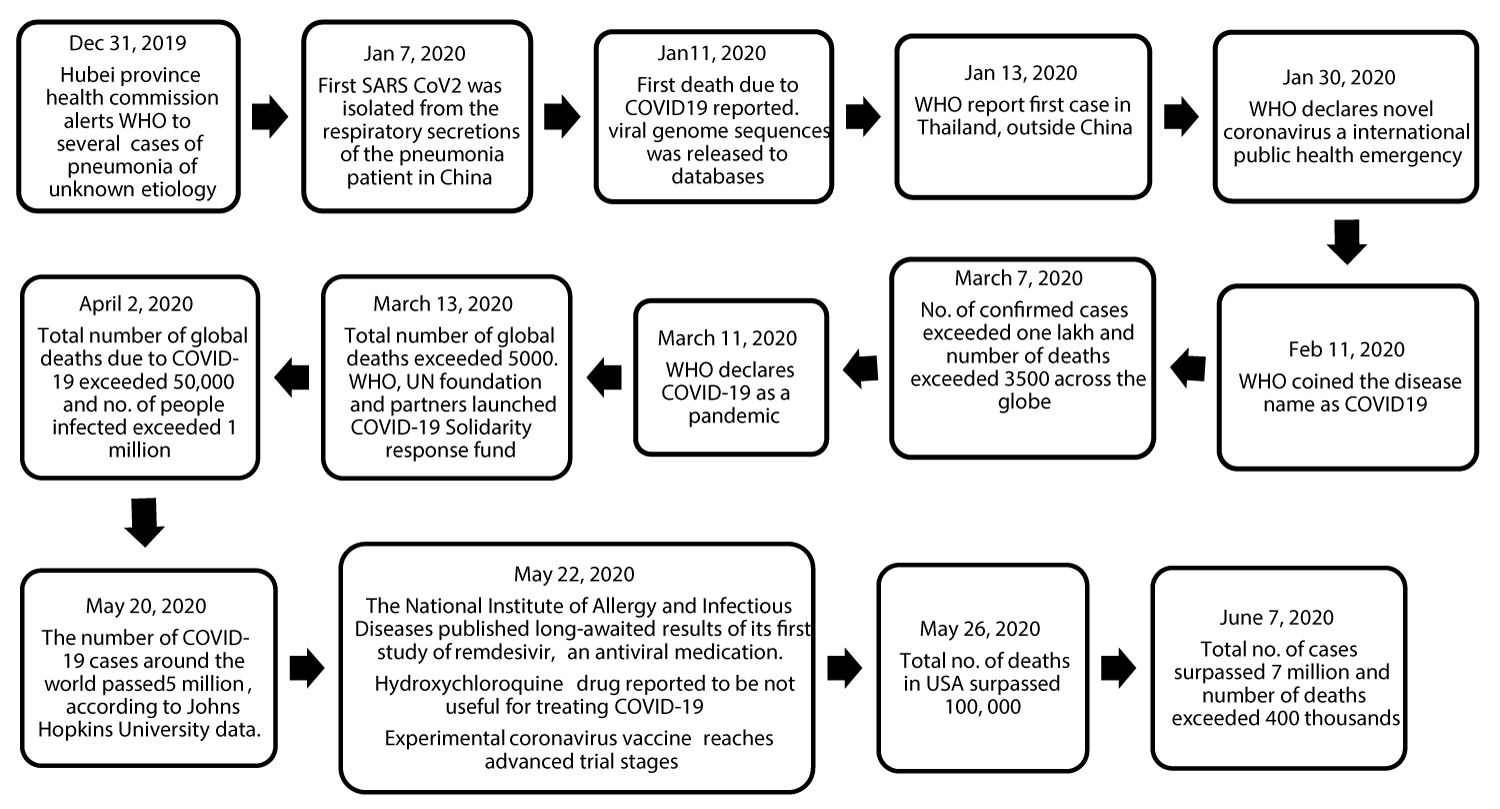 Figure 2: Timeline in the key events of SARS-CoV-2 outbreak.
View Figure 2
Figure 2: Timeline in the key events of SARS-CoV-2 outbreak.
View Figure 2
China had observed the maximum number of cases and COVID-19 related deaths in January and February-2020 but the rapid declining trend was noticed from the beginning of March-2020 [12]. Owing to their immediate and substantial countermeasures the outbreak control was achieved quickly [12]. However, the number of cases and COVID-19 related deaths outside of China (across the globe) rose exponentially from the beginning of March 2020 (Figure 3). Currently, the number of cases and deaths due to COVID-19 across the globe has exceeded that of China considerably [12]. The worst affected countries were the United States of America, Brazil, European countries, India, Russia, Iran and others [12]. As of June 8, 2020, the USA had experienced highest number of deaths followed by the United Kingdom, Brazil, Italy, Spain, France, and others (Figure 4) [12]. The case-fatality rate of the ongoing COVID-19 pandemic has increased more than what was expected at the beginning of the spread with the predominance in European countries and the USA (Figure 5) [14]. The exact reasons for high mortality in these countries yet to be identified. However, several hypotheses were put forwarded to explain this high case-fatality rate. Demographics, cultural behavior especially in Italy and Spain, and delay in response and implementing nationwide lockdown procedures following the detection of COVID-19 cases early in January 2020 is believed to be the major factors that would have contributed for rapid dissemination of infection and COVID-19 related deaths [15]. A recent study after analyzing the death cases from 66 administrative regions of Italy, France, Spain, and Germany reported the high case-fatality rate in the industrial regions of these countries that are heavily polluted with nitrogen dioxide. The long term exposure to nitrogen dioxide has been associated with the development of comorbidities such as hypertension and cardiovascular diseases. This may be an important contributing factor for COVID-19 related fatality in these regions [16]. Additionally, the highest concentration of old age population (23% of residents 65 or older with a median age 47.3) in Italy next only to Japan might have resulted in a large number of deaths [17]. Lack of availability of adequate medical facilities to manage the sudden overwhelming increase in the number of severe or critically ill patients due to the COVID-19 outbreak might have compounded the problem [17]. Interestingly, the case fatality rate in some countries such as New Zealand, South-Korea, Germany, Hong Kong, and others were low compared to other European countries and the United States. This relatively low COVID-19 fatality rate was believed to be due to the nation's early and high level of testing among a wide sample of the population and swift preventive measures taken by these countries to limit the spread of the infection [12,18]. Another study reported the significance of BCG vaccination in reducing the COVID-19 related mortality [19]. In most of the countries with high COVID-19 case fatality rate (European countries, the USA, and Iran), Universal BCG vaccination was either never implemented or abandoned long before [20]. In contrast heavily populated countries like India, Pakistan, Russia, and others, the case fatality rate is relatively low though the number of confirmed cases are more [12]. BCG vaccination is known to significantly induce production of pro-inflammatory cytokines, particularly Interleukin-beta 1(IL-B1) which plays a vital role in anti-viral immunity [21]. However, there is no conclusive evidence with respect to BCG induced immune defenses which needs to be further investigated [21]. Surprisingly, Japan, though has the highest old age population, hit early by COVID-19 and not having adopted more restrictive measures found to have very low COVID-19 related mortality in contrast to Italy and other European countries. This might be due to their extensive universal BCG vaccination and their cultural behavior [20].
 Figure 3: Daily Global deaths since March 1, 2020.
View Figure 3
Figure 3: Daily Global deaths since March 1, 2020.
View Figure 3
 Figure 4: Daily vs. total number of death due COVID-19 in countries with > 100 thousand confirmed cases and China as of June 13, 2020.
View Figure 4
Figure 4: Daily vs. total number of death due COVID-19 in countries with > 100 thousand confirmed cases and China as of June 13, 2020.
View Figure 4
 Figure 5: COVID-19 - Case fatality rate of the ongoing COVID-19 pandemic in countries with > 100 thousand confirmed cases and China as of June 13, 2020.
View Figure 5
Figure 5: COVID-19 - Case fatality rate of the ongoing COVID-19 pandemic in countries with > 100 thousand confirmed cases and China as of June 13, 2020.
View Figure 5
COVID-19 is the major pandemic that the world has witnessed in the 21st century. In view of its rapid pandemic spread, it is fortunate that the disease has been asymptomatic to mild in the majority (> 80%) of the patients [6,22,23]. Moreover, the majority of the affected individuals recover completely. However, SARS-CoV-2 appears to be highly contagious and has rapidly spread worldwide within a span of 3-4 months with bewilderingly varying impact on different countries [6]. The infection spreads through the respiratory droplets generated through coughing or sneezing by symptomatic or asymptomatic patients [10,24]. The infectious droplets can spread to a distance of 1-2 meters and may remain viable on surfaces for days in a favorable environment [10,24]. However, they are readily destroyed by chemical disinfectants such as sodium hypochlorite, ethyl alcohol, hydrogen peroxide, and others [25]. Patients can be infectious during the incubation period, as long as clinical symptoms persist, and even after clinical recovery. The incubation period varies between 2-14 days (median 5 days) and the infection is transmitted mainly by inhalation of droplets or touching contaminated surfaces and then touching mouth, nose, and eyes [10]. It has been also hypothesized that the virus transmission can also occur by fecal-oral route since it has been identified in a stool sample and contaminated water supplies [26]. Congenital transmission is not yet known but the occurrence of neonatal infection through the post-natal transmission has been described [27]. It has been identified that SARS-CoV-2 acts through ACE 2 receptor alike SARS-CoV-2 . Additionally, respiratory epithelial cells have shown to provide better growth conditions for SARS-CoV-2 compared to SARS-CoV-2 and MERS-CoV [7,10,28]. In various modeling studies it is estimated that basic case reproduction (BCR) rate to be ranging from 2 to 6.7 in SARS-CoV-2 compared to 2 in SARS-CoV-2 [12]. A recent study found that children have less angiotensin-converting enzyme 2 (ACE2) in nasal epithelium than older populations, which might confer some protection against SARS-CoV-2 and this explains the low frequency of infection and case fatality among the children [29].
The incubation period ranges between 2 to 14 days. The median incubation period in a study conducted on 1099 confirmed cases of COVID-19 was 4 days [30]. Another study reported a median incubation period as 5.1 days [31]. The clinical features varied from asymptomatic to mild symptomatic illness to life threatening pneumonia and multiorgan dysfunction. The common clinical features are fever, cough, malaise, fatigue, headache, and breathlessness. Hence they are indistinguishable from other common respiratory viral infections [10]. Additionally, gastrointestinal symptoms such as diarrhea appear to be less frequent in SARS-CoV-2 infected patients as opposed to SARS-CoV-2 [22]. Wang, et al. in their study on 138 hospitalized patients in China reported fever (98.6%), fatigue (69.6%), and dry cough (59.4%) as the most common clinical features at the onset of illness [30]. Similar ranges of clinical manifestations were reported by many cohort studies conducted on COVID-19 confirmed patients in Wuhan [10,32]. Severe disease and adverse outcome such as pneumonia, acute respiratory distress syndrome, acute kidney injury, and death are seen in elderly and individuals with comorbid conditions such as hypertension, respiratory and cardiac diseases, diabetes, cancer, etc [10,30,31]. The severe manifestations are due to a cytokine storm resulting from extreme rise in inflammatory cytokines such as interleukin-1 and TNF-alpha [23]. The median time from the onset of clinical symptoms to breathlessness was 5 days, hospitalization was 7 days, and development of ARDS was 8 days [10]. About 25-30% of the patients may need intensive care admission and the median duration of hospital stay in those who recovered was 10 days [10]. A recently published report by the Chinese CDC revealed that majority had a mild illness (81%), while only 5% developed critical illness with overall mortality rate of 2.3% while no deaths occurred among non-critical patients [33]. It also revealed that higher case-fatality among old age people; > 80 years (14.8%), 70-79 years (8%), 60-69 (3.6%), 50-59 (1.3%) and < 0.4% in people below 50 years of age [33]. A similar study from Italy reported highest number of deaths among elderly people of age 81-90 years (42.7%) and 71-80 years (35.6%) [15]. Furthermore, the case-fatality rate was much higher (6-10.5%) among patients with underlying comorbidity compared to patients (0.9%) without comorbid conditions [30]. The case fatality rate among hospitalized patients ranged between 4-11% [10,12].
As per the guidelines of WHO and CDC, the possibility of COVID-19 should be suspected in all patients presented with fever and/or features of lower respiratory tract infection who reside in or have recently (within prior 14 days) traveled to geographical affected areas or who have had close contact with a confirmed or suspected case of COVID-19 [34,35]. All individuals who meet the criteria for suspected cases should undergo testing for SARS-CoV-2, in addition to other respiratory pathogens [34,35].
With respect to the specimen for investigation, CDC advocates collecting samples from the upper respiratory tract (nasopharyngeal or oropharyngeal samples), and if possible from the lower respiratory tract. The nasopharyngeal swabs are advocated to be collected by using synthetic fiber swabs with plastic shafts [34,35]. The use of calcium alginate swabs is not advisable as it is inhibitory to viruses and interferes with PCR testing [35]. The collection of a sputum sample from patients with cough and the Broncho-alveolar lavage (BAL)/endotracheal tube aspirates in mechanically ventilated patients is recommended [35]. The samples are needed to be stored at 2-4 °C. The detection of SARS-CoV-2 viral nucleic acid in the clinical samples by reverse transcriptase-PCR test (RT-PCR) is highly sensitive and specific [10]. However, there were reports of negative RT-PCR results on oropharyngeal swabs of some patients who showed CT findings suggestive of pneumonia in the early part of the infection and subsequently tested positive by RT-PCR [36]. Rapid antigen test and antibody tests are also available. However, there are higher chances of missing an active infection with the antigen test. Antibody tests only detect antibodies the immune system develops in response to the virus. It generally takes one to two weeks to develop enough antibodies to be detected by a test and hence it should not be used to diagnose an active infection [14].
Other laboratory investigations are generally nonspecific. Chest computed tomography (CT) may aid in making the diagnosis. The presence of multiple areas of consolidation or ground-glass appearance bilaterally is the typical findings observed in multiple studies [10,30]. Variable leucocyte count (leukocytosis/leucopenia/lymphopenia), elevated liver enzymes and C-reactive protein have also been reported [10,30]. In many patients with pneumonia serum prolactin level found to be normal; however, they are found to be elevated in critically ill patients [30]. One study revealed the strong association between mortality and high D-dimer levels and severe lymphopenia [10].
Currently, there is neither specific anti-viral therapy nor vaccine is available. Therefore treatment is entirely symptomatic and supportive therapy including advanced life support if necessary [10]. However, several antiviral drugs and vaccines are under stages of clinical trials. Recently clinical trials of SARS-CoV-2 messenger RNA vaccine mRNA-1273 have shown promising signs [37]. In all trial participants, the vaccine candidate led to seroconversion with binding antibody levels either at or above levels seen in convalescent sera [37]. Remdesivir was the first antiviral drug that was approved for human use recently. A study has shown faster recovery of hospital admitted COVID-19 patients treated with remdesivir compared with placebo treatment [38]. Other antiviral drugs such as favipiravir, and nitazoxanide, nafamostat, and interferon beta-1a have shown a promising effect on SARS-CoV-2 in-vitro studies [39]. Hydroxychloroquine, an antimalarial drug is reported to be effective in treating COVID-19 associated respiratory complications. However, several scientists expressed ambiguity about benefits in the use of hydroxychloroquine in treating COVID-19 and it requires further large scale studies before it is encouraged for use [40]. Some pilot studies have shown the benefits of using corticosteroids, tocilizumab (an anti-IL-6 receptor antibody) and etoposide in selected COVID-19 patients with cytokine storm [41].
At this time prevention is crucial since there is no approved highly effective treatment for COVID-19. The current strategy to limit the spread of the disease is by implementing effective control measures. It focuses on patient isolation and adhering to strict infection control practices during the diagnosis and patient care. The prime strategy is a droplet, contact, and airborne precautions. Certain chemicals such as 70% ethanol and 0.1% sodium hypochlorite solutions were found to reduce viral infectivity significantly by 1-minute exposure [10]. Hence, they are helpful in early containment and further spreading of the virus. WHO and national health regulatory bodies strongly recommend all individuals to frequently wash their hands with soap and water and use of portable hand sanitizer. Maintenance of social distancing of more than 2 meters, avoiding contact with confirmed cases, avoiding needless travel, covering mouth with an elbow while coughing and sneezing and use of face mask as per the local guidelines are the other recommended measures for reducing transmission and flattening the curve. Patients with acute respiratory infection should wear the mask and keep a distance from the contacts, cover coughs or sneeze with disposable tissues or clothes, and wash their hands. Elderly and people with underlying serious comorbidities should avoid public gatherings. Healthcare workers caring for infected people need to adhere strictly to contact and airborne precautions including the use of personal protective equipment (PPE) such as N95 mask, gowns, gloves, goggles, and others [42].
SARS-CoV-2 that showed similarity to SARS-CoV, probably originated from bats has rapidly spread across 213 countries and territories resulting in a major and devastating pandemic. Due to the severity of infection, the World Health Organization declared it as a public health emergency of international concern. Such life-threatening viral outbreaks have become more frequent in the last two decades and that emphasizes on implementing effective public health strategies to negate the never-ending threats inflicted by emerging pathogens.
Authors declare that there are no conflicts of interest.