Hepatitis B infection causes significant morbidity and mortality worldwide. Chronic hepatitis B infection has been on the rise since 1990 with the highest prevalence reported in sub-Saharan Africa. Health care workers, intravenous drug users, commercial sex workers and men who have sex with men (MSM) are high risk groups for Hepatitis B virus (HBV) infection. Due to similar routes of transmission, Human Immunodeficiency Virus (HIV) infected individuals are also at high risk for Hepatitis B infection. The aim of this study was to define the prevalence and describe risk factors of Hepatitis B among high-risk groups.
This cross-sectional study was carried out in 2014 to 2016 among high-risk groups within western Kenya. A total of 860 participants were tested for Hepatitis B surface antigen (HBsAg). Populations studied included were People living with HIV/AIDS (PLWHA), substance users, MSM, female sex workers and patients presenting with signs of chronic liver disease. Data analysis was carried out using Stata version 15.
The overall prevalence of Hepatitis B across all risk groups from this study was 10.7% (95% CI 8.6 to 12.8%) out of 860 persons screened. The MSM population had the highest HBV prevalence of 19/90, 17.4% (95% CI 10.2 to 24.7%). Hepatitis B and HIV coinfection prevalence was 53 /802 (8.8%). Reported contact with jaundiced persons showed independent association with Hepatitis B infection after adjusting for other factors OR 1.87 (95% CI 1.18 to 2.96).
The high prevalence of HBV infection shows the need for ongoing screening of high-risk populations to inform planning for vaccination and preventive measures. HIV coinfection was also found to be high among those who were screened which indicates need to test for both viruses.
Hepatitis B, HBV, Western Kenya, High risk groups
AMPATH: Academic Model Providing Access to Healthcare; DNA: Deoxyribonucleic Acid; HBeAg: Hepatitis B Early Antigen; HBsAg: Hepatitis B Surface Antigen; HBV: Hepatitis B Virus; HIV: Human Immunodeficiency Virus; IDU: Intravenous Drug Users; IREC: Institutional Research and Ethics Committee; MSM: Men Who Have Sex With Men; MTRH: Moi Teaching and Referral Hospital; SSA: Sub Saharan Africa; WHO: World Health Organization; FCSW: Female Commercial Sex Workers; PCR: Polymerase Chain Reaction; UOR: Unadjusted Odds Ratio; AOR: Adjusted Odds Ratio
Hepatitis B infection, which is caused by Hepatitis B Virus (HBV), is a major public health problem with 2 billion people infected worldwide and more than 400 million chronic carriers worldwide. Globally it causes 1.2 million deaths per year and various complications including chronic hepatitis, cirrhosis and liver cancer [1-4]. At present, viral hepatitis kills more people than HIV, tuberculosis or malaria with mortality reported to have increased by 63% since 1990 [5]. HBV is a highly infectious virus transmitted mainly via blood, body-fluid contact, and vertical transmission [6]. The HBsAg in serum is the first serology marker to indicate active HBV infection, either acute or chronic [7]. A hepatitis B vaccine, available since 1982, has a high efficacy in the prevention of HBV transmission and has brought about remarkable changes in the global epidemiology of HBV infection [6]. The Kenya expanded program for immunization included Hepatitis B vaccination for children in 2013. The immunization program however only supports free vaccination for children under the age of 5 but not for the other categories [8]. Monovalent Hepatitis B vaccine is also recommended for health workers and other risk groups at 0, 1 and 6 months. Untreated chronic hepatitis B infection can result in liver cirrhosis and hepatocellular carcinoma [9], both of which have high morbidity and mortality. The World Health Organization (WHO) reports the burden of HBV in Africa is difficult to assess due to underreporting and inaccurate records [10].
Reviews of HBV epidemiological studies conducted in sub-Saharan countries in the last three decades show that HBV infection is highly endemic in sub-Saharan Africa (SSA) [11-14]. Due to its largely asymptomatic nature, chronic, viral hepatitis is a silent epidemic; most people are unaware of their infection [15]. Health care workers, intravenous drug users, commercial sex workers, HIV infected persons, and Men who have sex with men (MSM) are high-risk groups for Hepatitis B infection [16]. The prevalence of HBV infection varies widely by region and risk group. The prevalence of chronic HBV infection in Kenya has been reported to be 6-12% in the general population [17,18]. Biko, et al. found a prevalence of 9.7% among 247 HIV infected persons in care in western Kenya [19] . A study among individuals presenting with chronic liver disease described 44% prevalence of HBV infection. A survey of the regional blood transfusion data found a low prevalence 1.6% of HBV infection in Western Kenya. Blood donors are a highly selected population with low risk of Hepatitis B and therefore do not represent general population prevalence. The blood donor study also showed no demonstrable change in the prevalence of HBV infection a three-year period [20,21].
Knowledge of the burden of Hepatitis B infection is important for planning vaccination programs at a national level. Risk factors for HBV infection also need to be described for planning prevention strategies to prevent further spread of Hepatitis B. We set out to determine the prevalence and to describe the risk factors of hepatitis B infection among high-risk groups in the geographic area surrounding Eldoret.
This was a cross-sectional study of data from a Hepatitis clinical care screening project.
The hepatitis care project was conducted within Academic Model Providing Access to Healthcare (AMPATH) program. AMPATH is a partnership between the Moi University, Moi Teaching and Referral Hospital, the Kenyan government and North American universities led by Indiana University. AMPATH provides clinical care to over 85,000 PLWH [8].
The Hepatitis care project was carried out between 2014 and 2016. Screening for Hepatitis started in 2015 January and ended in 2016, January. This funded project was carried out to screen high risk participants for Hepatitis infection and to provide clinical support to those found to be infected, which included testing for Hepatitis B viral load levels, other viral markers and liver function tests. The lab tests were run at the AMPATH reference lab, results communicated to the participants; those found positive were then linked to the MTRH liver clinic for further care. High risk persons were tested HBsAg negative were referred to the MTRH vaccination clinic for vaccination.
The high-risk groups included in this project were known HIV positive persons in care - people living with HIV/AIDS (PLWHA), substance users, MSM, female commercial sex workers (FCSW) and chronic liver disease patients. PLWHA were already attending AMPATH HIV care clinics. These participants were known beforehand to be HIV infected and were already enrolled into care. The diagnosis of HIV was confirmed from the AMPATH database after the participants consented.
Substance users were defined as those who reported use of any type of substance use within the categories - Intravenously injected drugs, inhaled substances and glue sniffing. These participants had a pathological set of behavior associated with the substance use and were recruited either from the MTRH inpatient drug and alcohol rehabilitation unit or the street. A subset of street connected children and youth who formed part of the substance users were screened at the adolescent clinic in AMPATH and at venues set up in the community.
MSM and FCSW were recruited through meetings and primary health care facilities where they receive primary health care. Liver disease patients were inpatients identified to have chronic liver disease as determined by the primary clinician and documented in the patient chart.
All patients who did not know their HIV status were counseled and tested for HIV if they consented. Those who were found to be positive were referred for further care at the AMPATH clinics or to the primary health care facility from where they were recruited.
All persons enrolled in the Hepatitis care project filled out a risk assessment profile questionnaire adapted from CDC [22]. Those who were unable to read or write were assisted by care coordinators. Blood was drawn for HBV screening by a care coordinator. Those who did not know their HIV status were counseled and consented for a rapid HIV test. Blood samples collected were subjected to Hepatitis B surface antigen test (HBsAg) and HBV panel testing (HBV DNA (viral load) and Hepatitis B early Antigen (HBeAg) for those who were HBsAg positive. Laboratory methods used for screening of HBsAg and HBeAg were ELISA while DNA polymerase chain reaction (PCR) was used for viral load testing. For purposes of analysis and clinical referral, any person who tested positive for HBsAg was considered HBV infected.
The study was carried out in Eldoret, a town in western Kenya and its environs. Eldoret town is the largest growing cosmopolitan town situated on the highway to Uganda and Rwanda with an estimated population of 289,380 as per 2009 National Census.
Ethical approval was obtained from Institutional Research and Ethical Committee (IREC) of Moi University/Moi Teaching and Referral Hospital (MTRH) to analyze the data from the clinical care project. The privacy of persons was protected through de-identification of data.
All persons who were screened in the Hepatitis care project were included in the study. These included known HIV positive persons in care - people living with HIV/AIDS (PLWHA), substance users, MSM, female commercial sex workers (FCSW) and chronic liver disease patients.
Data was collected into a Redcap database. All variables were inspected for outliers, inconsistencies, missing data and distribution. Continuous variables were summarized while categorical variables were tabulated to identify inconsistencies and outliers. Visual inspection using scatter plots and histogram was also used to identify outliers and distribution of the data. No inconsistencies or outliers were identified in the data.
The primary exposure variable in this study was the high-risk group (MSM, PLWH FCSW, Liver disease and substance users) while the outcome variable was HBV infection. Other risk factors for HBV infection were also explored and have been described in the results section below. These risk factors were extracted from CDC hepatitis screening tool and are a priori known to increase risk of hepatitis B infection.
Data was analyzed using Stata version 13 for windows [23]. Missing data (2.3%), was excluded in multivariable analysis but this was not expected to introduce any bias to the analysis because it was a small percentage. Univariate analysis was based on all available data while missing data were excluded in multivariable analysis. Descriptive data were described using means and medians for continuous variables and frequencies for categorical variables. Further analysis was carried out using Chi square and student t-test to assess the association between the variables. The 95% confidence intervals were calculated using Clopper-Pearson method binomial confidence intervals [24].
Logistic regression was used for univariate analysis and to model the relationship between HBV infection and risk group, adjusting for confounders. Univariate logistic regression was performed to assess the likelihood of HBV infection compared to participant characteristics. The characteristics which had a p value of less than 0.20 in the univariate analyses were further explored in a multivariate logistic regression model. These factors were; age, gender, marital status and previous history of contact with a jaundiced person.
A total of 860 persons were screened for Hepatitis B; 402 were known HIV-infected, 109 were MSM, 21 were liver disease patients, 249 substance users and 79 FCSW. The overall mean age was 35.2 (SD 11.6) years, 471 (55%) were female. About 167/402 (42%) of PLWHA, 141/249 (57%) of substance abusers, 29/109 (27%) of MSM, 26/79 (33%) of FCSW and 8/21 (38%) of liver disease patients were married (Table 1).
Table 1: Participant sociodemographic characteristics and risk factors for HBV infection presented by risk group. View Table 1
Risk factors for HBV infection were sexual activity (> 90%), tattoos and traditional marks (> 30%) and traditional circumcision (> 40%) reported among all the groups. Less reported risk factors were intravenous injection drug use (< 12%) and history of liver disease (< 6%) in all risk groups (Table 1).
The overall HBV infection (as defined by detectable HBsAg) prevalence was 10.7% (95% CI 8.6% to 12.8%) across all the risk populations. MSM had the highest prevalence of HBV infection (17.4%) compared to PLWHA (10.2%), with a significantly higher Odds of HBV infection of 1.86 times compared to PLWHA group (Table 2). The prevalence of HBV in Liver disease, substance users and FCSW groups was 9.5%, 9.2%, 8.9% for respectively. None of these groups had significantly higher or lower odds of HBV infection compared to the PLWHA group.
Table 2: Prevalence of HBV infection by risk group and unadjusted odds ratios. View Table 2
HBV DNA Viral load and HBeAg was performed for 50% (46) of the participants who were HBsAg positive due to lab challenges. The median (IQR) HBV DNA viral load was 3,138.5 (353 - 351,990) cp/ml. HBeAg was reactive in 39 (45.9%) who were tested.
Proportion of persons co-infected with HIV who were diagnosed during screening for HBV was 62/402 (57%) MSM, 9/21 (43%) Liver disease, 93/249 (37%) substance users and 33/79 (42%) FCSW (Figure 1). Overall, 53/802 (8.8%) participants were HIV/HBV coinfected inclusive of those who were PLWHA. Out of the 92 participants who were HBV positive, 53 (57.6%) were HIV co-infected.
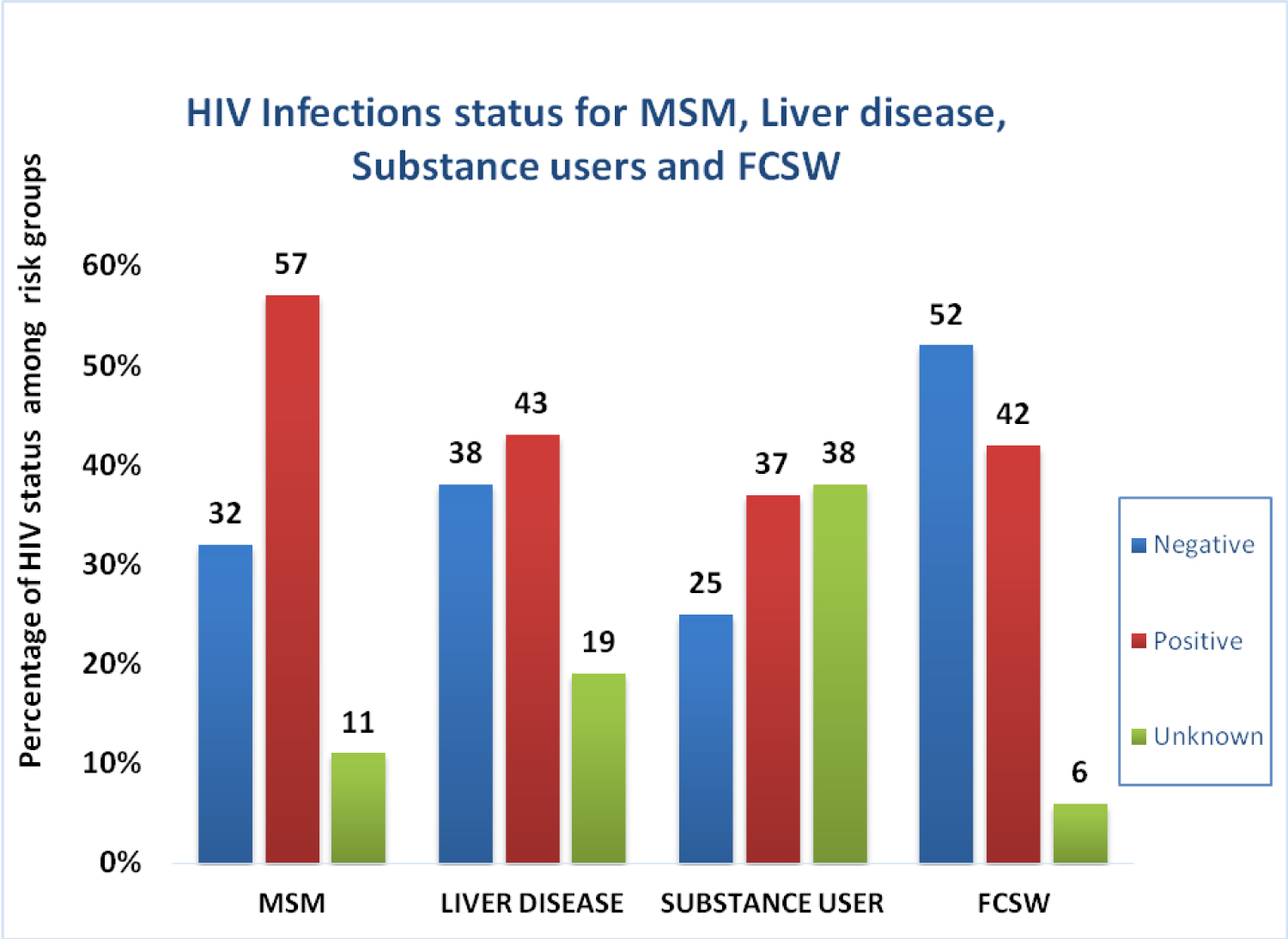 Figure 1: HIV infection status for MSM, Liver disease, substance users and Female commercial sex workers.
View Figure 1
Figure 1: HIV infection status for MSM, Liver disease, substance users and Female commercial sex workers.
View Figure 1
Further analysis for the association between HBV infection and participant characteristics such as age, sex, marital status and previous contact with jaundiced persons were carried out (Table 3). Younger persons (UOR 0.97, 95% CI 0.95 to 0.99), married persons (UOR 0.60, 95% CI 0.38 to 0.95) and females (UOR 0.63, 95% CI 0.41 to 0.97) were less likely to HBV infected. Persons who reported a history of contact with jaundiced persons had a 93% (UOR 1.93, 95% CI 1.24 to 2.99) higher likelihood of HBV infection. There was no significant association between the other risk factors and HBV infection.
Table 3: Comparison of participant characteristic by HBV infection. View Table 3
The results of the multivariate model described in Table 4 show that contact with jaundiced persons remained significantly associated with HBV infection, AOR 1.80 (95% CI 1.15 to 2.81).
Table 4: Adjusted OR for the relationship between HBV infection and participant characteristic after adjusting for other factors. View Table 4
This study found a high prevalence of HBV infection among high risk populations within western Kenya. MSM group had a higher odds of HBV infection compared to PLWHA. The Center for Disease Control and prevention reports that among adults 20% of new Hepatitis B cases occur in MSM [25]. HIV infection was also high among these risk groups probably because HIV and Hepatitis B have similar routes for transmission. Few HIV-infected persons are able to clear HBV infection [26].
HIV-HBV coinfection has been found in similar studies to this one. Muriuki, et al. in 2008 and Biko, et al. in 2011 in reported a HBV prevalence of 10% in Nairobi and 9% in western Kenya respectively [27,28].
This study found 17.4%, 9.2% and 8.9% prevalence among MSM and FCSW groups respectively. This is comparable to other studies; Van Houdt, et al. in 2012 reported 23% prevalence of HBV among MSM and 28% in drug users while Lopamudra reported 9.7% prevalence among injection drug users in India [29,30]. HBV prevalence of 32.5% was reported in female sex workers in Turkey in 2009 by Pinarbasi, et al. [31].
Some of the differences in the prevalence reported might be due to mixing of the injection and inhalation drug use among the substance users' group. Other variations may have resulted in lower prevalence in the present study arose from the fact that the persons screened in this study were recruited through their primary care facility. This could have resulted in the screening of persons who had a tendency for health seeking behavior.
The present study found that a history of contact with jaundiced persons was significantly associated with HBV infection after adjusting for other factors. A study in Saudi Arabia found contact with HBV infected persons had the highest association with HBV infection OR 2.5 (95% CI 1.6 to 3.8). Other important predictors for HBV infection described in the Saudi Arabian study were dental procedures (OR 2.0) and blood transfusion (OR 1.7) [32]. This points to the highly infectious nature of Hepatitis B virus where some of the transmission could be explained by close nonsexual contact with HBV infected persons.
De Almeida Pereira, et al. in 2006, found significant association between male gender (OR 1.6), older age (OR 3.9), tattooing (OR 1.6) and multiple sexual partners (1.4) and HBV infection after adjustment for confounding. Our study did not find these factors to be associated with HBV infection. Part of the reasons for the lack of association could be social desirability bias in reporting risk behaviors.
Further variances in prevalence between other studies and this study may be difficult to explain the studies may not be exactly comparable to this one.
This study was the first of its kind to be done within western Kenya covering a range of high-risk populations. Female sex workers, MSM and substance users have not been previously screened for HBV infection in Kenya. Overall, many persons were screened for Hepatitis B although some groups were smaller than others depending on the available numbers from these groups. The persons selected into the risk group were all screened, this represented 100% of all persons presenting to primary care or otherwise. This project was important in showing feasibility of screening.
The small number of liver disease patients limited the robustness of analysis within this group. There was a high rate of HIV coinfection which made the multivariate analysis difficult because these groups were initially selected based on their known risk. Persons may have failed to report all their risk factors due to social desirability bias. This may have led to underestimation of the effect when determining associations. We were not able to carry out a viral load or HBeAg test on every infected person due to limitations of the lab in our setting.
There is high prevalence of HBV infection amongst high-risk population in western Kenya. There is need for education and vaccinations of the negative high-risk individuals to prevent spread to the general population. Contacts of these groups should be screened and if negative advised on vaccinations. Those infected should be followed up and put on treatment as per national and international guidelines. Primary prevention strategies on safe sex practices which include minimizing the sex partners and use of condoms can reduce the rate of HBV transmission.
Ethical approval for the study was obtained from Institutional Research and Ethical Committee (IREC) of Moi University/Moi Teaching and Referral Hospital. The ethical committee waived the need for written consent since this analysis was done retrospectively. All the clinical data has been de-identified as per the ethical board requirement.
Not applicable.
Data is available on request from the author.
The authors declare that they have no competing interests.
The Hepatitis Care project which generated data for this study was funded by the AbbVie Foundation grant number IU KC IP ID # 00362241.
MK and AG participated in the conception of the study and carried out the data collection and analysis together with FFS, HIL, EW and LKM. All authors participated in interpretation of data, drafting and approval of the final manuscript. All authors read and approved the final manuscript.
We acknowledge the AMPATH reference lab, clinicians at the AMPATH clinics, the hospital administration and all persons of the care screening project.