Thanks to the widespread use of antiretroviral therapy, the number of older patients living with HIV, usually defined as individuals over 50 years of age, has markedly increased everywhere in the world.
The aim of this review is to describe the epidemiology and special needs of older persons with HIV in Canada now and in the near future.
It is estimated that, by 2025 in Canada, one in six of all HIV-infected adults will be over 50 years of age. Most will reside in Ontario, Quebec, and British Columbia and will require a number of specialized health services.
These three provinces need to train extra multidisciplinary manpower and establish appropriate public health policies in order to adequately prepare for this new health care challenge.
HIV, Aging, Health care needs, Canada
The human immunodeficiency virus (HIV) epidemic can be divided into three eras: 1) 1981-1995, the era from emergence of the virus to the advent of antiretroviral therapy (ART) [1]; 2) 1995-2010, the era of ART introduction and expanding use [2], and 3) 2010-present, the post ART era [3]. Prior to the introduction of ART, infection with HIV inevitably resulted in the development of the acquired immune deficiency syndrome (AIDS), which was uniformly fatal. Antiviral therapy prevents HIV infection from progressing to full-blown AIDS and, thus, extends life. This fact has ushered in the post ART era, with large numbers of people now living and aging with HIV. Once such individuals reach the age of 50, they are referred to as HIV50+.
Across the globe, the numbers of HIV50+ individuals are growing exponentially [4-7]. They live with a chronic illness because, despite its many benefits, ART does not fully restore health and well-being. Treated HIV infection is characterized by persistent low-grade inflammation, residual immunodeficiency, and a variety of ART-induced toxicities [8,9]. The aging process itself plus lifestyle behaviors such as smoking and alcohol or drug use [10] result in the growing frailty of this population. Frailty refers to an impaired ability to fight off health challenges and to recover from bouts of illness. It is associated with a gradual decline in everyday function. While ART can completely suppress viral load, current data show that the life expectancy of persons with HIV is still 13 years shorter than that of HIV-negative age peers [11]; it is, however, improving [12].
Persons with HIV today face two major challenges. The first challenge is to suppress the original virus and fight off all opportunistic infections. The introduction of ART reduced the mortality rate from 28.4 per 100 person-years in 1995 to 8.8 per 100 person-years in 1997 [2]. The incidence of opportunistic infections such as pneumocystis jirovecii pneumonia, mycobacterium avium complex disease, and cytomegalovirus infection declined from 21.9 per 100 person-years in 1994 to 3.7 per 100 person-years [2,13-15].
The second challenge is more difficult to meet. It is to slow the pace of the aging process, which continues to be exceptionally rapid in person with HIV [16,17]. HIV50+ individuals experience prematurely high rates of cardiovascular disease, cancer and cognitive decline. It remains unclear whether HIV itself, its treatment and associated side effects, or other risk factors are responsible, but the natural aging process appears to be accelerated [18,19]. Premature age-related co-morbidities [16] and the increased risk for adverse outcome of illness, or frailty [20] and event mortality [21] remain characteristic of this population. One study found that co-morbidities were the most prevalent in individuals aged 60 years or older [22]. Geriatric syndromes including frailty are also seen at higher rates among individual HIV50+ [23].
The pathophysiology of inflammation resulting from HIV infection is impossible to distinguish from the inflammation associated with natural aging [9]. Both consist of cell damage, then tissue damage, followed by organ damage and eventual organ failure. Initially, damage can be reversed, but once organs begin to degenerate, the progress to death appears inevitable. The pathways in chronic HIV and aging are identical; the combination adds speed to the process. Firstly, the diagnosis of HIV tends to be delayed in older patients [24]. Additionally, HIV50+ may be extra vulnerable to the toxic effects of treatments and the pharmacological interactions of combinations of medications [25].
Figure 1 depicts the natural history of normal human aging versus the natural history of aging with HIV. As persons with HIV age, they battle HIV-associated immunosenescence, which is characterized by low-level chronic inflammation and impairment in the ability to mount adequate immune responses to infection, which accelerates the natural aging process. HIV50+ also need to deal with ART toxicities and ART resistance, as well as any residual harms from the initial HIV infection [8,9]. As a result of these battles, they become frail at a relatively young age [26]. They live with chronic disability and increasing numbers of co-morbidities [16,27] and eventually they die at relatively young ages [11,28].
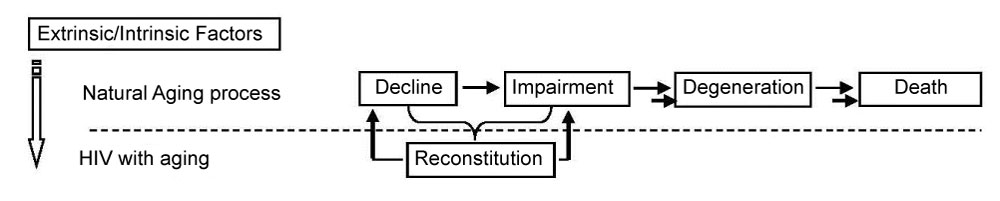 Figure 1: Aging process and diseases (e.g., HIV). View Figure 1
Figure 1: Aging process and diseases (e.g., HIV). View Figure 1
Data from the Joint United Nations Programme on HIV/AIDS (UNAIDS) shows an increase in the over 50 HIV population from 2.8 million in 2006 [29] to 4.2 million in 2014 [30]. In sub-Saharan Africa alone, the estimate in 2014 was that more than 2 million individuals over 50 were living with HIV [30]. A recent study reports that, globally, there were 5.7 million (4.7 million - 6.6 million) HIV individuals aged 50 or older in 2016 and this number was expected to increase to an estimated 7.5 million (6.3 million - 8.8 million) by 2020 if the UNAIDS treatment target of 81% ART coverage were met Figure 2 [4]. These figures may well be an underestimate [31].
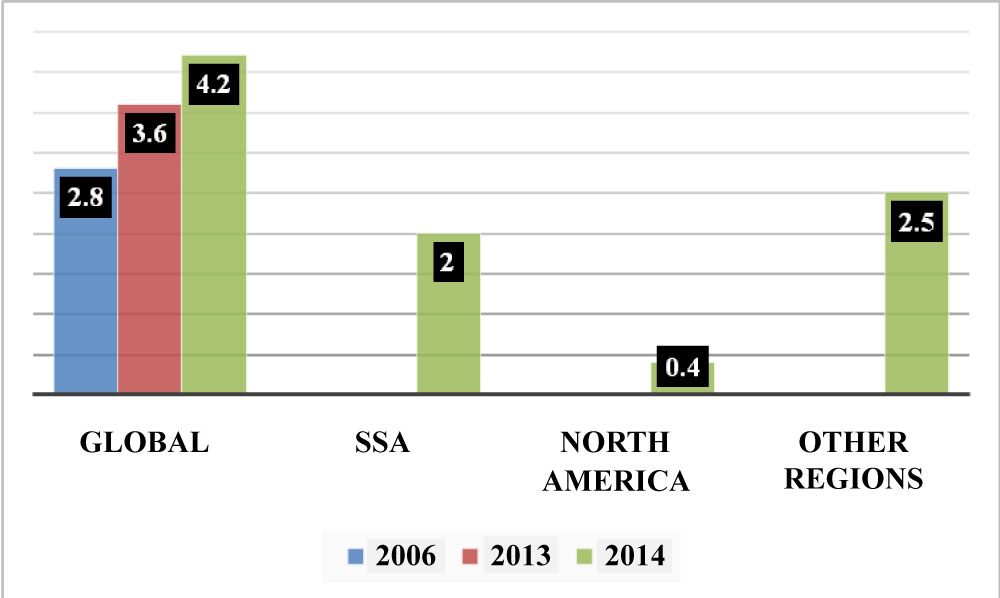 Figure 2: Numbers of HIV50+ worldwide (Trends in million).
Figure 2: Numbers of HIV50+ worldwide (Trends in million).
Graph complied from numbers found in UNAIDS 2014. SSA = Sub-Sahara Africa. View Figure 2
An earlier survey estimated that 3 million HIV50+ (14% of the total 21 million HIV-infected individuals aged 15 years or older) could be found in sub-Saharan Africa [7]. The prevalence of HIV in those aged 15-49 years in this region was 5% in 2010; the prevalence among those over age 50 almost reached 4%.
Five countries in sub-Saharan Africa had the highest number of older adults with HIV: Mozambique, Nigeria, South Africa, Zambia and Zimbabwe. These five countries account for 54% of the total number of HIV50+ [7].
There are few data on the demographics of HIV50+ in Europe. In France, in 2003, an estimated 19% of patients who were enrolled in the French Hospital Database on HIV (a nation wide hospital-based cohort of > 100,000 HIV-infected patients) were over age 50 (21% of men and 13% of women fit into this category) [32]. In Italy, of the total number of HIV-infected adult cases, 9% were over 50-years-old. This proportion increased over time, from 5% in 1982-1990 to 16% in 2000-2005 [33]. The United Kingdom (UK) underwent a relatively small, concentrated HIV epidemic, with an estimated 101,600 people living with HIV in 2017 [34]. HIV prevalence was higher among men, approximately 3.0 per 1,000, compared with women, approximately 1.3 per 1,000. In 2012, it is estimated that 24,510 people living with HIV in the UK were aged 50 or over [35]. At the end of 2017, it is estimated that 36,278 (85.07% males; 14.93% females) were HIV50+ [36] 98.70% of whom received HIV care. One study reported that over 10% of the whole sample (184/1,687, 10.9%) were aged 50 years or above [37]. Gay men made up 13.1% of the total, Anglo-African heterosexual men 8.5% and Anglo-African heterosexual women 6.9%. One third of the HIV-positive gay men over 50 years were first diagnosed with HIV in their 50s or 60s (33.3%, 32/96).
In Australia, at the end of 2017, 27,545 people were estimated to be living with HIV. Of people living with diagnosed HIV in 1986, 5% were aged over 50 years. In 2017, those over 50 constitute 46% of the HIV population [38].
In 2008, HIV50+ in the United States accounted for 24% of HIV infections, an increase from 17% in 2001. It was estimated that by 2025 more than half of all persons with HIV would be older adults [5,39]. But, by 2016, nearly half the people living with HIV in the U.S. were already aged 50 or older. For instance, in New York City, Xia, et al. [40] report that more than 50% of HIV+ individuals are currently over 50-years-old.
The increasing proportion of older people among the HIV population is due to a number of factors. In regions of the world where better public health measures are in place, because of ART, there are fewer potentially contagious people, all of which results in the total number of HIV cases having significantly dropped. Among the HIV+ population, there is now a growing percentage of older individuals who were infected before the advent of ART and who have survived because of ART. Older adults are rarely prioritized for HIV prevention or testing and are, therefore, at more risk than younger adults for HIV acquisition [41]. Their use of condoms tends to be relatively low; since the advent of ART, they may feel that they do not need to use precautions. There is also evidence that physicians do not regularly discuss and provide advice on sexual issues with patients aged 50 and older [41].
There are limited data on individuals who first become infected after age 50, and their numbers vary from region to region. It is estimated that 100,000 people aged 50 and older acquire new HIV infections every year in low-and middle-income countries [30]. Approximately 74% of this subgroup live in sub-Saharan Africa.
Among the total HIV50+ population, the proportion of persons with new HIV infections approaches 12.9% in Western Europe, 10% in Central Europe, and 4% in Eastern Europe [6]. In the United Kingdom, new diagnoses among older adults more than doubled between 2001 and 2012, rising from 497 in 2002 to 990 in 2012 [42].
The latest reports have found a significant increase in new HIV diagnoses in older adults in 16 countries between 2004 and 2015. This is especially the case in central and eastern (EU/EEA) countries. These new HIV50+ were more likely than not to have been infected via heterosexual contact and to show relatively advanced disease at the time of diagnosis [43,44].
In Australia, new HIV infection in persons 50 years of age or older accounted for 17.23% of all new HIV diagnosis from 2008 to 2017. In 2017, the largest aged group with new HIV infection was 30-39 years, followed by ages 20-29 years, and then by ages over 50 Figure 2 [38].
The incidence of HIV in Canada differs depending on era [45]: 1) Between 1996 and 2000 there was a significant decrement of HIV cases following the introduction of ART, 2) Between 2001 and 2008, the incidence was stable, and 3) From 2009 till the present, there has been a gradual decline in incidence. In 2013, the national HIV diagnosis rate for all ages was 5.9 per 100,000 [45]. The province of Ontario accounted for the highest proportion of reported HIV cases (39.6%, n = 827), followed by Quebec (21.7%, n = 453), and then British Columbia (13.0%, n = 272), Alberta (12.5%, n = 261), and Saskatchewan (6.1%, n = 126).
In 2015, Patterson, et al. [46] conducted a study in Canada of HIV positive individuals aged 18 and above who all initiated antiviral therapy after the first of January, 2000. As predicted, life expectancy for this population improved steadily over time. At the time of analysis, those who were 20 years of age could expect to live another 37.5 more years, 22 fewer years than age peers who were HIV negative.
By the end of 2014, a cumulative total of 80,469 HIV (including AIDS) cases aged over the age of 15 had been reported in Canada. Most were from Ontario, Quebec and British Columbia with 2,044 new cases reported in 2014 [45]. Of this latter group, most were 30-39 years old. Those 40 to 49 were the second largest incident group. Most of the new cases were male; nearly half were men who had sex with other men. Drug users accounted for 13% of new cases.
There are limited data on older persons with HIV. HIV50+ in Canada were first described in 1992 by Ferro & Salit [47]. By 2025 (Figure 3), it is estimated that one in six of all HIV-infected cases (15-years-old or older) will be HIV50+, but this will depend on whether one quarter, one half, or three quarters of 40 - 49-years-old HIV adults will have turned 50. The context is that the percentage of all AIDS seniors (age 65 and older) in the Canadian population is reported to be growing quickly. The proportion was 14.1% in 2010 and is estimated to become 23-25% by 2036 [48].
 Figure 3: Estimated numbers of older adults with HIV in Canada (2014 versus 2025) depending on whether one quarter, one-half, or three quarters of 40 - 49-year-old HIV adults will have turned 50.
Figure 3: Estimated numbers of older adults with HIV in Canada (2014 versus 2025) depending on whether one quarter, one-half, or three quarters of 40 - 49-year-old HIV adults will have turned 50.
Graph complied from numbers found in HIV and AIDS in Canada. Surveillance report, December 2014. View Figure 3
As of December 2008, 2,644 (12.4% of person with AIDS in Canada) were aged 50+ [41]. In 2012, an estimated 7,582 (10.7%) were 50+. The majority (85.4%) were male [49]. The percentage of AIDS reported among HIV50+ increased from 15.9% in 1999 to 21.6% in 2008 [50].
Within risk categories, from 1999 to 2008, the percentage of HIV50+ men who have sex with men (MSM) dropped; heterosexual cases climbed from 25% to 30%. The injection drug risk increased markedly from 5.3% to 26.1%. Most cases were found among heterosexuals, fewer among MSM and fewer still were in the drug injecting group. As to ethnicity/race, the majority of Canadians HIV50+ were Caucasian, followed by First Nations and Black. Among Canadians aged 50 years or older, more tested positive in 2008 than in 1999 (15% vs. 11%).
In the province of Ontario, 32,547 persons were infected with HIV by December 2011 [51]. Of these, 2,882 were HIV50+, (2,421 males and 378 females). It is noteworthy that deaths among those aged 40 and older have increased since 1998, while deaths decreased among those under 40.
In Ontario, the Ontario HIV Treatment Network (OHTN) plays a crucial role with respect to HIV community-based research, institutional collaborations, community-based individual collaborations, and training of young researchers. The OHTN is the HIV national lead of two major observational and longitudinal HIV cohorts, the Positive Spaces, Healthy Places (PSHP) and the OHTN HIV Cohort Study (OCS), both funded by the Canadian Institutes of Health Research (CIHR).
PSHP is a five-year prospective cohort whose goal is secure housing and health for HIV+ persons. The aim of OCS is health and well-being of HIV-infected individuals living in Ontario.
Situated in British Columbia (BC) but serving all of Canada, the Canadian HIV Observational Cohort (CANOC) Collaborative Research Centre has been addressing five main areas: Burden of aging and associated co-morbidities, quality of care, quality of life, cost-effectiveness of interventions for modern HIV management and comparative effectiveness and drug safety of modern antiretroviral treatments [52].
The findings accumulated by these three Canadian research cohorts will lead to many improvements in the care for HIV patients, but none of the three specifically address the needs of older Canadians who are HIV positive.
The numbers of HIV50+ are growing quickly throughout the world. Individuals with HIV are surviving to older ages because of ART, but their health and quality of life suffers because of co-morbidities, inadequate access to health care, inadequate training of health care workers, and inadequate social support.
In Canada, universal health insurance is theoretically able to address all the health care needs of this population, but the aging population with HIV has been insufficiently targeted. Because of the many parallels between natural aging and chronic HIV infection, the specialized skills of gerontologists and geriatric social workers, nurses, physiotherapists, and occupational therapists are needed to better address the health and psychosocial needs of this disadvantaged population.
Not applicable.
Not applicable.
None.
None.
The author wishes to thank Dr. Mary V. Seeman for her English editing and proofreading of the manuscript. I would especially like to thank Dr. Sean B. Rourke for the excellent supervision that he has provided me throughout my Ph.D. program.