The lung is a heterogeneous organ due to the effect of gravity on the arterial supply and lymphatic system, and this leads to a predilection of different pathologies to certain areas of the lung.
The pathophysiological factors that make the upper lobe "vulnerable" to various different pathologies are additive, and not mutually exclusive. They include a tall, thin body habitus with an antero-posteriorly flattened chest as associated with a low BMI, compounded by the poor apical blood supply and concomitant limited lymphatic drainage, possibly accentuated by a physically tall lung and a deep first rib furrow that makes the apex appear prominent.
These factors promote high physical stress with resultant tissue tearing or destruction caused by a negative circumferential stress vector within the visceral pleura of the prominent apex, abetted by large-sized apical alveoli resulting from the distortion of the lung under its own weight.
Adequate repair may be limited by the poor apical blood supply, exacerbated by the presence of congenital apical bullae that are present in approximately 15% of an otherwise healthy population. The elastic fibers within the lung become damaged and replaced by scar tissue, aggravated by exposure to noxious particulates like cigarette smoke that overwhelm the ability of the already meager apical antiprotease defense to prevent elastic fiber damage. Cycles of destruction, repair and healing lead to fibrobullous lesions and cavity formation in the postero-apical area of the lung around the rib furrows; these then turn into a scarred immunological enclave prone to further disease progression.
Pathophysiology, Lung, Apex, Tuberculosis, Biomechanics, Upper lobe-predominant disease
The lung is unlike many other organs in that it is not homogeneous in its structure or function but is heterogeneous with regards to its perfusion, ventilation and perfusion-ventilation ratios [1-4], pleural pressure [5], lymphatic flow [6], mucociliary clearance [7,8] and pleural mechanical stress [9]. Much of the heterogeneity of the lungs is due to the effects of gravity [10], its function as a pressure vessel and of its shape as constrained by the thoracic wall [11,12].
These anatomical and physiological differences affect the lung's responses to various noxious factors that can be extrinsic or intrinsic. These include diverse environmental and congenital factors such as inhalation of injurious gases, particulates and antigens [13-16], infections like tuberculosis (TB) and aspergillosis [17,18], and genetic disorders like mutations in cystic fibrosis transmembrane conductance regulator (CFTR) protein and alpha-1 antitrypsin deficiency [19,20].
The aim of this review is to show how various anatomical and physiological features of the upper lobes and apices of the lungs combine to the make this part of the lung more vulnerable to certain disease etiologies, (Table 1). In so doing, we aim to demonstrate a common pathophysiological pathway for apical and upper lobe lung disease.
Table 1: Classification of upper lobe lung disease by predominant aetiological mechanism. View Table 1
The lungs occupy the pleural cavity. This cavity is not smooth internally but characterized by ridges caused by the ribs. The most prominent rib ridge is that of the first rib. This may be such a deep furrow that a surgeon might feel a sharp edge on intra-operative palpation [21]; (Figure1). Computerized tomography scans in coronal and sagittal views show that the apex of the lung sticks out like a thumb from the rest of the lung; (Figure 2). The result is that the apex geometrically has the shape similar to that of a rugby ball or bullet, that is a prolate or elongated spheroidal shape.
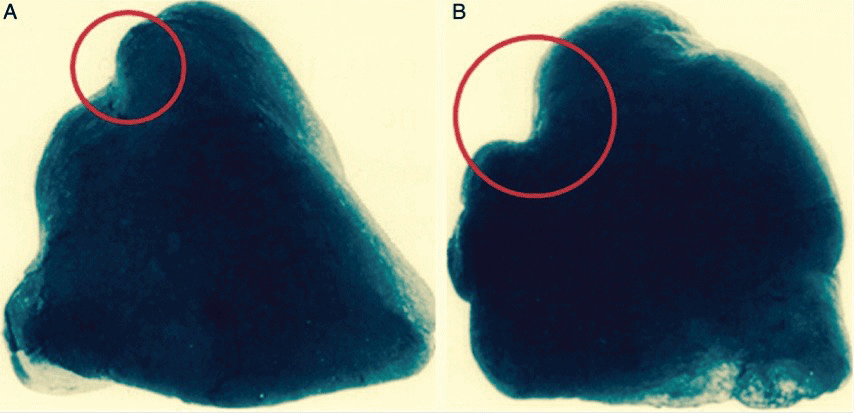 Figure 1: A,B) Anterior and posterior views of moulds of the lung apex showing prominent indentations in the lung parenchyma caused by rib furrows, especially that of the first rib which are deeper posteriorly. Modified after Stephenson [21].
View Figure 1
Figure 1: A,B) Anterior and posterior views of moulds of the lung apex showing prominent indentations in the lung parenchyma caused by rib furrows, especially that of the first rib which are deeper posteriorly. Modified after Stephenson [21].
View Figure 1
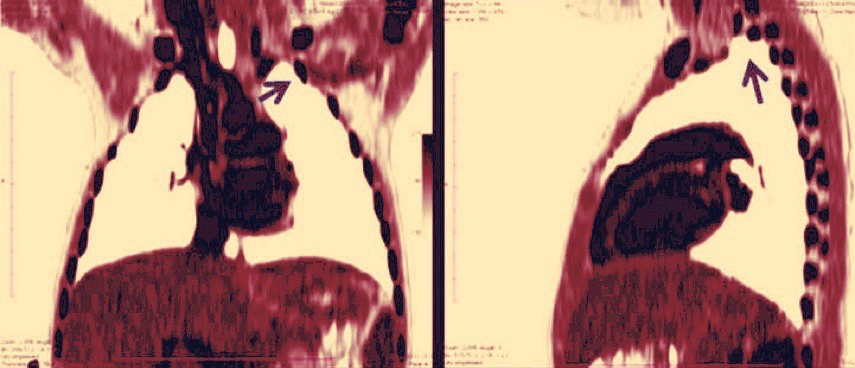 Figure 2: Inverted colorized coronal and sagittal reconstructions of a thoracic CT scan showing how the first rib indentation (arrow) causes the apex of the lung to be very prominent, causing raised levels of pleural stress.
View Figure 2
Figure 2: Inverted colorized coronal and sagittal reconstructions of a thoracic CT scan showing how the first rib indentation (arrow) causes the apex of the lung to be very prominent, causing raised levels of pleural stress.
View Figure 2
Figure 3 shows the ratios of stress in spheres and in ellipsoids with varying ratios of major-to-minor (a/b) axes where S1 is the axial stress and S2 is the circumferential stress. When the ratio of the height to the base of the apex protruding beyond the first rib reaches 1.42, the circumferential stress component becomes zero.
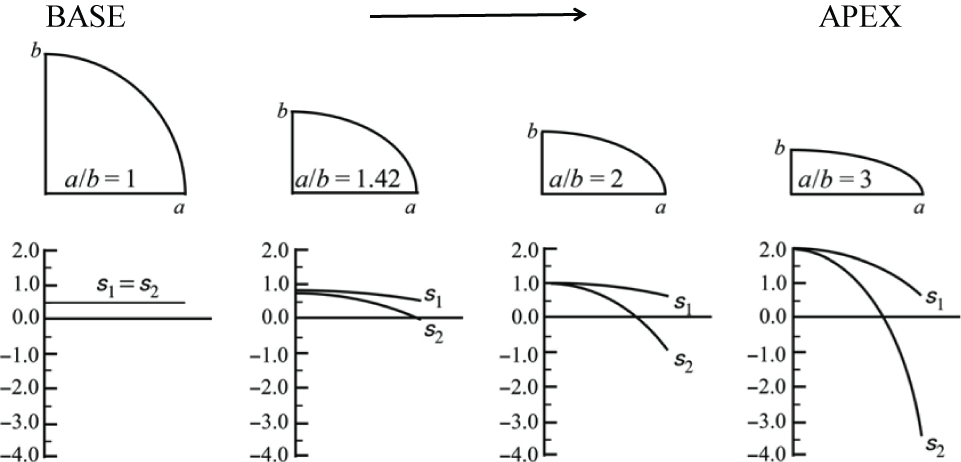 Figure 3: Ratios of stress in spheres and in ellipsoids with varying ratios of major (a) to minor (b) axes showing axial stress S1 and circumferential stress S2. Modified after Fryer [22].
View Figure 3
Figure 3: Ratios of stress in spheres and in ellipsoids with varying ratios of major (a) to minor (b) axes showing axial stress S1 and circumferential stress S2. Modified after Fryer [22].
View Figure 3
When the height to based ratio exceeds 1.42, the circumferential stress becomes negative and instead of containing the lung, it promotes an external traction force, that combined with the high negative pleural pressure present at the apex of between -15 to -20 cm H2O [5], causes an instability at the shoulder area of the prolate spheroid. This may result in tearing as the pressure vessel pulls apart. Thinning of the pleura with porosity may also occur [23]. The tearing or buckling effect results in repair by scarring and fibrosis with repeated cycles of damage and repair that may promote cavity formation [11]. The negative curvature of the rib grooves further aggravates the issue, resulting in the reversal of the normal surface loads (circumferential stresses), leading to tearing in the shoulder areas. The area most at risk is the posterior aspect of the apex where the first rib furrow is most prominent.
Thoracic index is defined as the ratio of anteroposterior and lateral chest diameters. In the fetus, chest cross-section is circular, although fetal thoracic index exceeds unity in early fetal development [24]. As children grow, the ribcage drops with gravity, resulting in a change from the horizontal ribs in the cone-shaped fetal chest with a thoracic index of 0.85 to a flattened rib cage with angulated ribs and a thoracic index of 0.6 by age 3 years.
Gravity acts on ribs to increase their angle of inclination with the horizontal, resulting in drooping of ribs with growth and aging, as the ribs pivot on their costo-vertebral joints. The initial drop in rib angulation in children leads to a drop in thoracic index as children start to walk upright [25]. The chest wall changes with age [26]; in particular, the thoracic index increases as the ribs drop outwards and laterally with age. There is a progressive rounding of the thoracic cage with increasing age-related obesity [27], (Figure 4). Chest anteroposterior diameter increases but the lateral diameter remains static [28]. Females have a smaller chest than males, with smaller anteroposterior and lateral rib cage diameters [28].
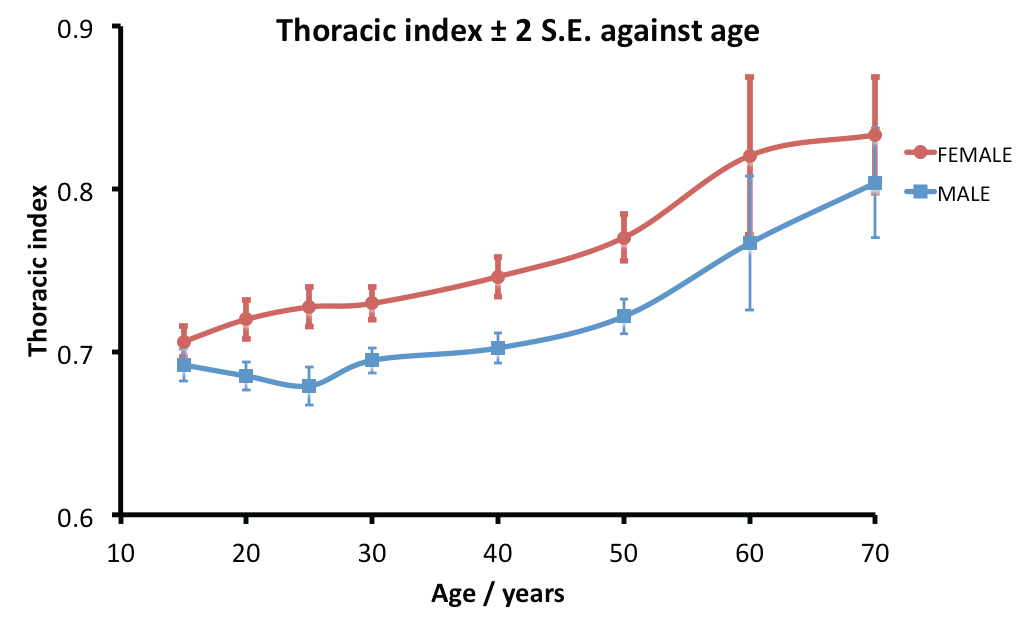 Figure 4: Changes in thoracic index ± 2 SE with age according to gender. Males have a lower thoracic index than females. SE is standard error for the sample mean and 2 SE the upper and lower 95% confidence limits (0.975 quantile of the normal distribution). Redrawn after Takahashi [27].
View Figure 4
Figure 4: Changes in thoracic index ± 2 SE with age according to gender. Males have a lower thoracic index than females. SE is standard error for the sample mean and 2 SE the upper and lower 95% confidence limits (0.975 quantile of the normal distribution). Redrawn after Takahashi [27].
View Figure 4
The predilection for tuberculosis to affect the tall, long-chested individual supported Dock's early work in 1944 on the pathogenesis of TB [13,29]. Similarly, it has long been recognized [30] that typical spontaneous pneumothorax patients are tall, thin males [31,32] with elongated lungs [33], a diminished anteroposterior diameter and a low thoracic index [34].
Low BMI is strongly associated with a low thoracic index (AP flattened chest shape) [28]. Seven cohort studies on tuberculosis demonstrated a clear relationship between BMI and incidence of TB across several countries and different levels of TB burden [35-42]. Though both poor nutrition and low BMI are associated with TB infection, they can be considered as independent factors [43], with low BMI acting as a marker of low AP thoracic diameter.
Low pulmonary pressure in the apex due to gravity has been proposed as a plausible explanation for apical lung disease since 1945 [44]. Although the blood flow in the apices is low, ventilation-perfusion (V/Q) ratios are high, so the partial pressure of oxygen (pO2) in the apex is the highest anywhere in the lung at 132 mmHg which is ideal for the tubercle bacillus that is an obligatory aerobe [45,46]. At the uppermost parts of the lung, the pressure within the vessels may be less than the alveolar pressure, described as West zone 1 [47].
However, evidence now indicates that gravity has a less important role in the pattern of regional distribution of ventilation and blood flow in the lung compared to structural features of the airways and blood vessels [48]. The gravitational model cannot explain lack of postural inequality in the prone position and heterogeneity of ventilation and perfusion distribution at the same vertical level with persistent heterogeneity in the absence of gravity [49]. The non-symmetrical fractal branching of the bronchial and pulmonary vascular anatomy is now considered to be the most important factor causing heterogeneity of perfusion and ventilation in the lungs [50].
Both the gravitational and fractal models result in a relatively over-ventilated apex (V/Q ratio 3:1) and an over-perfused base (V/Q ratio 0.6:1) [1]. The balance between repair, protection by antiproteases and destruction by exposure to cigarette smoke may be altered by the relative hypocapnic alkalosis in the apex. In a rabbit lung model, hypocapnic alkalosis was directly injurious to the lung and potentiated ischemia-reperfusion-induced acute lung injury [51]. The delivery of deleterious gases is also higher in the apex, potentiating the problem. This pathophysiological mechanism is also relevant to diseases like smoking, centrilobular emphysema and cadmium exposure, where the elastic fibers are weakened by antiproteases [15,52,53]. Furthermore, local apical respiratory alkalosis may explain the upper lobe location of metastatic pulmonary calcification secondary to renal failure, secondary hyperparathyroidism, intravenous calcium therapy and osteolysis due to metastases or multiple myeloma due to a decrease in calcium solubility in alkaline tissue [13].
From the low apical pulmonary arterial pressure model follows the greatly diminished regional formation of tissue fluid or lymph. Dock's lymph stasis theory provides an explanation for the apical localization of pulmonary TB [44], chronic pulmonary histoplasmosis and certain inorganic dust diseases [17]. However, the lymph stasis theory fails to explain TB in the apex of the upper lobe [54,55], which the pressure vessel model (see below) can account for [12].
The lymphatic system plays a crucial role in particle clearance. The size of the particle load is important since a large load overwhelms the alveolar macrophage [56-58]. In the literature, particle size ranges are often classified as coarse (> 2.5 microns in diameter), fine (< 2.5 μm) and ultrafine (0.001-0.1 μm) [59]. The increased pulmonary toxicity of ultrafine particles is related not only to the large surface area in relation to their mass but also to their interstitial access as they bypass the mucociliary escalator [60].
Particles greater than 5-10 μm are unlikely to reach distal airways, whereas particles smaller than 0.5 μm move into and out of alveoli by diffusion, often without substantial deposition and injury. Particles between 1-5 μm diameter get lodged at the bifurcation of the distal airways [61,62] near the juxta-alveolar origin of the lymphatic channels, making them more dangerous [63]. At the alveolar duct bifurcations, macrophages accumulate and engulf trapped particulates.
Nanoparticles have a diameter of between 1-100 nanometers (nm) and can translocate easily from lung epithelium to regional lymph nodes, to the blood circulation and then accumulate in various organs of the body. There is yet elementary knowledge about the short- and long- term effects of nanoparticles delivered to the lungs, but some are toxic at the cellular biochemical level [64,65].
Clearance of particles by lymphatics is slow [66] and in the apex is limited by diminished arterial driving pressure and by the decreased apico-posterior chest wall movement, which lessens the effectiveness of the chest wall movement acting as a respiratory pump [67]. Pathophysiologically, decreased lymphatic clearance occurs in chronic granulomatous disease (tuberculosis, sarcoidosis [68], eosinophilic granuloma [69], bronchocentric granulomatosis [70], chronic histoplasmosis [17] and farmer's lung [71]) and pneumoconiosis [13]. Whilst coal dust is relatively inert, silica, asbestos and beryllium are more reactive and cause fibrotic reactions at lower concentrations [72]. Reactive particles trigger the macrophages to initiate an inflammatory response and promote fibroblast proliferation and collagen deposition, leading to predominantly upper lobe fibrosis especially posteriorly.
Modelling the thorax as a pressure vessel, apical pleural stress is ten-fold that of the lung base. In an antero-posteriorly flattened chest there is a further fou-fold increase; when combined this results in a forty-fold increase in pleural stress [10,12,73,74]. A tall, unsupported apex is therefore prone to increased stress leading to apical TB and spontaneous pneumothorax; this geometrical shape may also explain the occurrence of both TB and pneumothorax in the apex of the lower lobe where ventilation-perfusion ratios and blood flow are optimal, lymphatic clearance is normal and ventilation is average [54,55]. It also explains the pathophysiology of post lobectomy air leak, where air leak is more in the upper lobes and also on the right compared to the left due to the loss of chest wall conformational support leading to high pleural stress [75].
The pressure vessel model above differs from that described by West which considered gravity distorting the lung under its own weight, leading to a higher strain in the apices and a fourfold increase in apical alveolar size [76]. With a fixed rib cage such as in ankylosing spondylitis, apical stress was higher [77] and lymphatic clearance diminished due to decreased pulmonary pump action [67], leading to fibrosis and cavitation in the upper lobe [78-80].
The lung is elastic and its elastic recoil is balanced by the tendency of the chest wall to spring outwards. The elastic fibers are weakened by exposure to cigarette smoke, which is a significant source of cadmium exposure [81].
Marfan's syndrome is a genetic disorder affecting fibrillin, a glycoprotein essential for formation of elastic fibers in connective tissue [82]. This can commonly result in pneumothorax [83,84] and rarely cystic changes, emphysema, pneumonia, bronchiectasis, bullae and apical fibrosis [85]. The mechanism is due to repeated cycles of healing of disrupted, highly stressed apical interstitial tissue.
Centrilobular emphysema, the commonest type of pulmonary emphysema, is a routinely found in asymptomatic elderly patients. It is predominantly located in a patchy distribution in the upper zones of each lobe, that is the apical and posterior segments of the upper lobe and the superior segment of the lower lobe [86] and appears as emphysematous spaces of up to 1 cm diameter, located centrally within the secondary pulmonary lobule, replacing the central bronchovascular bundle [86,87]. It is strongly associated with smoking in a dose-dependent manner [88].
Normal alpha-1 antitrypsin is an antiprotease that protects against neutrophil elastase that would otherwise damage elastic fibers. Patients with alpha-1 antitrypsin deficiency develop upper lobe emphysema exacerbated by exposure to cigarette smoke and environmental dust; however, emphysema may develop at an early age even without significant smoking [20].
In contrast, in cystic fibrosis the cystic fibrosis transmembrane conductance regulator (CFTR) protein responsible for efflux of chloride and inhibition of sodium channel activity is non-functional, resulting in normally thin mucus secretions becoming thick and inspissated, thus preventing adequate particle clearance and leading to bronchiectasis of the upper lobes and apical segment of lower lobes [89].
Patients with pleuropulmonary fibroelastosis (PPFE) show clinically progressive refractory upper lobe fibrosis, with clinical data suggesting a link to recurrent pulmonary infection [90]. These patients are slender, with a flat rib cage and narrow anterior-posterior diameter [91].
The lung is a heterogeneous organ with respect to V/Q ratio, lymphatic flow and mechanical factors; these are summarized in Figure 5. Although some factors are related to gravity others are consequent on the chest wall and pleural cavity shape [12]. These differences dictate how the lung responds to extrinsic inhaled stimuli and intrinsic mechanical characteristics. In the normal upright lung, most blood flow and ventilation occurs in the lower lobes [47], yet despite an expectation that most inhaled pathogens or noxious gases would therefore be delivered to the dependent portion of the lung, the opposite often happens, with the upper one third of the lung comprising the upper lobe and upper segment of the lower lobes often involved in pathology [13]. In contrast, where the injurious factor is delivered solely through via the bloodstream, such as with drugs (amiodarone, nitrofurantoin, methotrexate and illicit drugs), the disease predominantly affects the basal part of the lung.
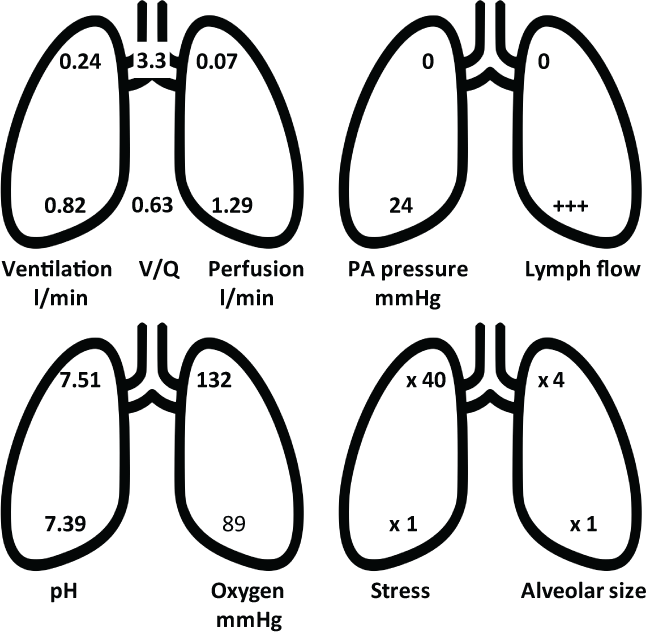 Figure 5: Regional differences in the normal lung, showing apical and basal values for ventilation, ventilation-perfusion ratios (V/Q), perfusion, pulmonary artery (PA) pressure, lymph flow, hydrogen concentration as pH, partial pressure of oxygen, pleural stress and alveolar size. Based on Casha [9], West [47] and Gurney [13].
View Figure 5
Figure 5: Regional differences in the normal lung, showing apical and basal values for ventilation, ventilation-perfusion ratios (V/Q), perfusion, pulmonary artery (PA) pressure, lymph flow, hydrogen concentration as pH, partial pressure of oxygen, pleural stress and alveolar size. Based on Casha [9], West [47] and Gurney [13].
View Figure 5
The upper lung suffers a greater concentration of particulates and pathogens that can overwhelm its meager antiprotease defenses [92] limited by poor blood flow, lymphatic stasis that fail to clear the offending pathogen or particulates and increased mechanical stress (pressure vessel model). This may limit healing and promote scarring, subpleural bleb formation and possibly cavitation [9,11,12]. The increased mechanical stress leads to repair by fibrosis and scarring with the formation of apical vascular adhesions due to relative low blood supply in the apex. At increased risk is the 15% of the population who are born with congenital apical blebs who are at high chance of disease progression should their body habitus be tall, thin with an antero-posteriorly flattened chest typical of a low BMI build [93].
The various factors we have described all contribute to a common pathophysiological pathway. Often in upper lobe pulmonary disease, the patients are described as tall and thin with low BMI [13,42,43]. In a comparative study between colliery workers across the UK, there was a statistical difference in BMI between those who suffered from pneumoconiosis (tall, light men) and healthy co-worker (short, heavy men) controls matched for geographical colliery location, pit purity and level of dust exposure [94].
These pathophysiological factors do not exclude disease in the lower lobe. Only a small change in the balance between repair and injury is required to alter the outcome. For example, the radiological pattern of reactivation of TB, which classically occurs in the postero-apical part of the upper lobe, changes to lower lobe consolidation when there is immunosuppression with HIV [95]. In asbestosis, the size and needle like shape of the asbestos fiber (length 100 μm, diameter 30 μm) favors entrapment in the lower lobes [96-98].
The pathophysiological factors that make the upper lobe "vulnerable" to certain pathologies are additive, and not mutually exclusive. They include a tall, thin body habitus with an antero-posteriorly flattened chest, typically associated with a low BMI, and a poor apical blood supply and concomitant poor lymphatic drainage possibly accentuated by a tall lung, with prominent first rib furrow that makes the apex appear prominent. These factors either promote physical stress with its consequent tissue tearing or destruction caused by a negative circumferential stress vector within in the visceral pleura in a prominent apex or prevent adequate healing due to a poor blood supply, abetted perhaps by the presence of congenital apical bullae present in approximately 15% of an otherwise healthy population. The elastic fibers within the lung become damaged and replaced by scar tissue, aggravated by exposure to noxious particulates like cigarette smoke that overwhelm the ability of the already meager antiprotease defenses to prevent damage of the elastic fibers. Cycles of destruction, repair and healing lead to cavity formation in the postero-apical area of the lung around the rib furrows; these then increasingly become a scarred immunological enclave prone to further disease progression.