A hypothesis for the pathogenesis of pulmonary TB is presented based on the biomechanics of pleural stress in the context of the lung as a pressure vessel. This hypothesis modelled pleural stress as a marker of pleural tissue destruction and subsequent repair. It identified five predictors of TB reactivation, namely adolescent and young adult age, male gender, apical location, low antero-posterior chest diameter or low thoracic-index rib-cage shape and the presence of pre-existing apical blebs.
This hypothesis is complete in that it explains the occurrence of apical lower lobe disease, the age, gender and build of those at risk, the incidence of concurrent apical disease and also Esmail's prediction about the size of the population at-risk of TB reactivation.
Once a subject can be identified as having several of the biomechanical predictors of TB reactivation as explained by the pleural-stress pressure vessel hypothesis, initial screening can be performed based on demographics, Body Mass Index (BMI) and thoracic index.
A second round of screening by imaging can confirm the presence of apical blebs or bullae with an expected pick-up rate of approximately 15% of the screened group. It would therefore be possible to immunize, closely observe or treat this high-risk group. Low-cost, portable MRI has been suggested as a TB screening tool available in the near future as it promises to be a near ideal screening tool in that it is safe, can be delivered in a low-cost, mobile and portable manner using new and emergent Magnetic Resonance Imaging (MRI) technologies, with high sensitivity and specificity.
Tuberculosis, Pathophysiology, Predictor, Reactivation, Screening, Biomechanics
Tuberculosis [TB] still remains a major global health threat especially in middle and low-income countries where it is more prevalent [1] and where multi-drug resistant TB is a serious public health issue [2,3].
TB has long been recognized as having a predilection to the apex of mainly the upper lobe but also to that of the lower lobe [4]. In 1944 Rich noted that "there is no more puzzling circumstance in the pathogenesis of pulmonary tuberculosis, nor one that has excited more interest and speculation, than the peculiar fact that in the adult the disease begins almost invariably in the upper portion of the upper lobe" [5]. Based on Rich's own work, this was put down to higher oxygen tensions in the apex of the lung, important as the TB bacillus is an obligate aerobe [6].
The relatively poor apical blood flow can explain this, since gravity directs two-thirds of blood flow to the lower lobe and the apices have a very low or nonexistent blood flow [7] with a high partial pressure of oxygen [pO2] of 132 mmHg [8]. There is a pattern of increasing and then decreasing blood flow along the lung during different gravitational conditions [9] that correlates well with West's Zones 1-4 [10,11]. However, this pattern persists during weightlessness, suggesting that the effect of gravity was at best limited [12,13]. Instead a new fractal structural model has now replaced the earlier gravitational model with regards to ventilation-perfusion matching with results from a common pattern of asymmetric branching of the airways and blood vessels [12,13].
The apex is relatively hyperventilated with well-expanded alveoli resulting from the combination of a high negative pleural pressure in the apex but with limited compliance due to relative upper rib immobility resulting in a local respiratory alkalosis [14]. This local apical respiratory alkalosis may explain the upper lobe location of metastatic pulmonary calcification [14]. Gravity restricts perfusion to the apex. Apical lymphatic clearance restricted by gravity, low perfusion and diminished respiratory motion, with apical cavitation occurring after tuberculous reactivation [15]. The conventional explanations of high apical oxygen levels and gravity accounting for TB's apical localization appear to be invalid for the lower lobe apex [4,16].
TB infection occurs in two stages: An initial primary infection with Mycobacterium TB, the "Ghon focus", and later progression to active secondary disease [17]. Although the first implant of perhaps 1-5 tubercle bacilli [15] can occur anywhere in the lung (see Figure 1A) [8], the cavitary lesion is often located in the apical regions of the upper lobes (see Figure 1B) [19].
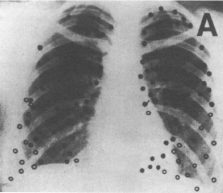 Figure 1A: Site of calcified single primary tuberculous lesions in 105 patients, found throughout lung fields. From Medlar [18].
View Figure 1A
Figure 1A: Site of calcified single primary tuberculous lesions in 105 patients, found throughout lung fields. From Medlar [18].
View Figure 1A
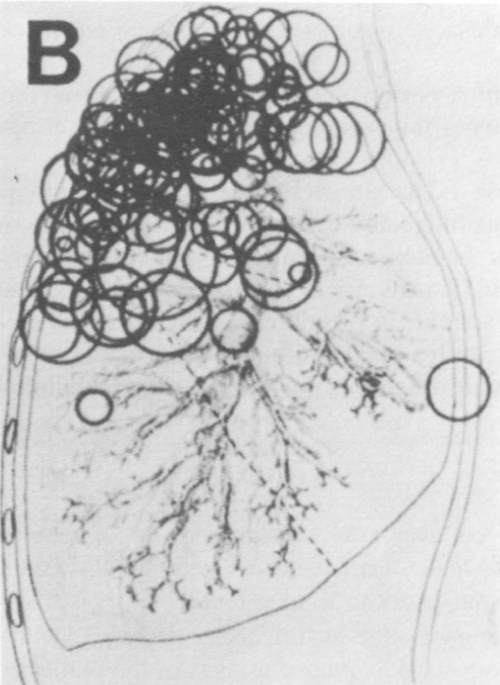 Figure 1B: Site of single cavitary tuberculous lesions in 204 individuals, showing predilection to apico-posterior aspect of upper lobes and apices of the lower lobes. From Sweany [19].
View Figure 1B
Figure 1B: Site of single cavitary tuberculous lesions in 204 individuals, showing predilection to apico-posterior aspect of upper lobes and apices of the lower lobes. From Sweany [19].
View Figure 1B
Reactivation occurs in about 5% of primary infected individuals, resulting in active pulmonary TB developing usually in the apex of the upper lobe, or rarely in the apex of the lower lobe [4]. The reason why TB selects the apex in low-body mass index [BMI] males [20-24] for reactivation remains elusive [23].
As the primary infection occurs anywhere in the lungs, the tubercle bacilli must reach the vulnerable apex of the lungs for infection to progress to secondary active disease. This occurs either by endogenous or exogenous spread; endogenous haematological spread occurs during a bacteremia throughout the first infection whilst endogenous spread occurs via the airway after repeated exposures [15]. Spread to the lung apices leads to cavitation, which is important in that dissemination through these lung cavities, is the major pathway for onward TB transmission [25].
Although both TB and malnutrition are linked with poverty, there is no independent association between primary TB and malnutrition; and the incidence of tuberculin positive tests in the USA, where Bacillus Calmette-Guérin (BCG) immunization is not offered, is not affected by body weight, height or BMI [17,26].
Secondary pulmonary TB is different and unusual in that there is a strong relationship between active TB and BMI occurring in different countries [24] and across various levels of TB burden without an obvious biological mechanism [17]. Cohort studies have shown that active or secondary TB infection is not related to body mass or BMI at the time of the initial primary infection, although high body mass may be protective [22]; suggesting that low-BMI individuals are at higher risk of reactivation and progression to active secondary disease [27].
Whilst high apical oxygen levels together with poor gravitational or fractal apical blood flow and reduced lymphatic clearance give a potential explanation for the apical location of secondary TB in the upper lobe, these conventional explanations for TB's apical localization appear to be invalid for the apex of the lower lobe where blood flow and ventilation-perfusion ratios are optimal, ventilation is average and lymphatic clearance is normal [4,16].
Besides not explaining disease in the apex of the lower lobe, conventional theory does not explain the association with low BMI, or the sudden increase in disease in male adolescents, marking a sudden increase in pulmonary TB, but not in non-pulmonary TB [25]. Furthermore, conventional theory does not explain why tuberculous pneumothorax occurs in apical secondary TB but rarely develops in miliary pulmonary TB [28,29]. The pressure vessel hypothesis expounded here uses pleural stress as a marker of tissue destruction, followed by repair by fibrosis and subsequent repeated cycles of tearing and tissue repair that may lead to cavitation.
Every scientific theory has to be universal in its application, complete, without exceptions, all encompassing [including other pathologies if possible], preferably be logical and simple following Occam's razor, yet allowing for enormous complexity and must be predictive in order to be useful [30]. This paper proposes the biomechanical pleural stress or pressure vessel hypothesis, presenting supportive evidence and predictions, and builds on these to suggest innovative applications to TB screening.
In this hypothesis we consider the lung to be a pressure vessel; this is defined as a vessel that contains gases under pressure and therefore has a transmural pressure gradient. Although Pascal's principle requires that the pressure is identical everywhere within the lung, Laplace's law describes a variation in wall tension or pleural stress (wall tension increases with lower tangential radius). In the lung, a confluent chest wall helps to support the visceral pleural surface. However parasagittal CT scans reveal that the lung does not always reach the chest wall in the rib furrows especially that of the first rib, which is the deepest furrow [31]. Furthermore the apex can be very oblate in shape, which means that it is much higher than the size of its base. In effect when the tangential radius of the lung surface curvature is negative, the longitudinal or axial stresses become negative and therefore unstable and prone to tearing and poor healing, making it "vulnerable" to TB [32].
Finite element analysis (FEA) computer modelling was performed on a lung model based on chest dimensions measured from a small series of 30 randomly chosen anonymised CT scans that had been reported as normal. The model developed was constructed with a groove representing a first rib furrow. FEA is a computer analysis methodology that approximates shapes by simplifying complex geometry by instead substituting a mesh with nodes.
FEA simulations use simplifications of geometry to limit computational time. In order to ensure that the mesh discretization did not influence the solution, a mesh study was performed with three different meshes of increasing refinement to show that the chosen mesh size did not alter the results. The mechanical properties of the material remain a source of error as with all FEA models.
FEA simulation data showed the presence of three areas of high stress activity in the apical region present near the apex and within two grooves on either side of the apex caused by the first rib furrows, with maximal stress fortyfold-higher than basal low stress areas, see Figure 2.
 Figure 2: Finite element analysis of computer simulation of lung model showing that pleural stress is maximal at the apex and first rib furrows. Maximum stress is shown as red, minimum stress as blue.
View Figure 2
Figure 2: Finite element analysis of computer simulation of lung model showing that pleural stress is maximal at the apex and first rib furrows. Maximum stress is shown as red, minimum stress as blue.
View Figure 2
Although this mathematical model cannot be directly validated experimentally since it is not practical to measure pleural stress in-vivo, this model has been used successfully to explain the pathogenesis of primary spontaneous pneumothorax (PSP) and predict those at high risk of developing PSP [31].
In effect the pleural surface gets torn by high-pressure coughing, repair is by fibrosis but the repair is prone to re-tearing. This occurs especially if the rib cage is not confluent, if the apex is oblate as in asthenic individuals or if the pleural surface is thinned by the presence of congenital apical blebs that occur in about 15% of the population [33]. These cycles of repeated tearing and fibrosis may perhaps even lead to development of bullae especially in the context of a high negative apical pleural pressure [34] that may act as a perfect location for the TB bacilli.
Can we predict those individuals that are likely to progress? Finite element computer modelling of a lung model showed that highest pleural stress was found in the apical region (10-fold increase compared to the base) and was related to an antero-posteriorly (AP) flattened chest shape (4-fold increase compared to round chest shape) [35]. This low thoracic index (AP to lateral chest diameter ratio) or low AP diameter acts as a marker for low BMI, since AP diameter is directly related to BMI [31]. Thoracic index was statistically significantly lower in males at all age groups in a study by Takahashi and lowest in young males in their late teens and twenties [35]. Seven cohort studies on TB have linked BMI and incidence of TB in several countries across a spectrum of high, intermediate and low TB incidences [20-23,36-38].
The Pleural Stress model can identify the following five groups as predictors of increased risk of TB reactivation: Apex (high stress and high negative pleural pressure), low thoracic index/low BMI (high stress), young age (marker for low thoracic index), males (marker for low thoracic index) and presence of apical blebs (pleural weakness that can be exploited by high stress to cause cavitation).
For this Pleural Stress hypothesis to be considered valid, the incidence of TB reactivation would be expected to increase in the five aforementioned states where apical pleural mechanical stress is predicted to be high. The epidemiology of TB was investigated through a literature search to determine whether these characteristics were fulfilled.
Reactivation of pulmonary TB occurs in the apico-posterior segments of the upper lobes or in the superior segments of the lower lobes in young adults [4]. In contrast pulmonary TB in the elderly results in lower lobe pneumonia, similar to other bacterial infections [39]. The conventional theory is that these are sites of impaired lymphatic drainage [40] and high pO2 levels [39] however this does not explain the predilection to the apical region of the lower lobe [4], suggesting that oxygen levels and gravity may not be such important factors.
The FEA model showed that the lung apex was an area of high pleural stress levels, ten-fold that of surrounding areas and particularly prone to buckling and tearing of the lung when the apical height to base ratio exceeded 1.42 [41]. The high negative apical intrapleural pressure also aggravates these stresses. The negative curvature of the first ribs furrows results in a further large increase in pleural stress due to a reversal of the normal surface stresses [41].
Lonnroth reviewed seven prospective studies that removed the confounding effect of weight loss caused by TB and showed that the incidence of new pulmonary TB was five times higher in a low BMI group (BMI < 21) as compared to a high BMI group (BMI > 31) [23]. However this marked association between pulmonary TB and BMI was not observed for extra-pulmonary TB [20], suggesting that low BMI predisposes to an increased risk for pulmonary TB only, and not for non-pulmonary TB. Low BMI is strongly associated with a low thoracic index (AP flattened chest shape) [42]. This AP flattened chest shape showed a 4-fold increase compared to round chest shape [35]. In effect this means that the apex of low thoracic index individuals is subject to a fortyfold increase in pleural stress compared to basal areas of the lung.
Reactivation of pulmonary TB increases in adolescence and young adults, males more than females whilst in contrast the reactivation of non-pulmonary TB remains constant [25]. The mechanism is most likely the same as low thoracic index, since the lowest thoracic index occurs after adolescence [43], when the U-shaped relationship between thoracic index and age [44] is at its lowest.
The prevalence of TB is identical and low in both genders until reaching adolescence when it increases in males [45] after puberty when there is a "sudden emergence" of cavitating lung disease [25]. The mechanism is most likely the same as low thoracic index since male chests are AP flatter than those of females at all age groups after adolescence [44].
Primary spontaneous pneumothorax (PSP) is a condition that has apical sub-pleural blebs present in almost all (76-100%) patients [46], which are commonly visible on CT scanning [47]. Such a population would be expected to be at high risk of TB reactivation due to presence of weakened pleura from the sub-pleural blebs and also because PSP patients have a low AP chest diameter [48].
The incidence of TB is about 7:100,000 [49], and that of PSP is 18:100,000 in Swedish males [50]. Concurrent disease can be expected be extremely rare as the multiple of the two incidences. However 5.4% of PSP patients had TB and 2.1% of patients with TB had PSP [51], suggesting that the presence of apical blebs is a very strong prognostic factor towards TB reactivation. Since lung bullae or blebs occur in 15% of the population [33], this suggests that the pool of individuals at high risk of TB progression should be of a similar magnitude. This hypothesized proportion fits in with the prediction by Esmail that the population at-risk of TB reactivation must be significantly much smaller the 2.33 billion who are immune-sensitized to TB [52].
Current diagnostics tests (tuberculin skin test and interferon-gamma release assays) only show infection with TB bacteria but do not indicate latent TB infection or progression to TB disease. Together with chest X-ray and sputum investigation, they are poor predictors of those who will develop active disease. Advanced imaging such as computed tomography (CT) and combined positron emission tomography/computed tomography (PET/CT) have been used to identify minimal disease, and together with invasive sampling now play a diagnostic role in selected cases in specialist centres within high-income countries.
The World Health Organization (WHO) Stop TB partnership set a 2050 elimination target which requires a historically unprecedented 20%/year reduction in global incidence [52,53]. The American Thoracic Society lists two main categories that should be screened for latent TB infection; those at risk of exposure to active TB, and medical conditions or other factors associated with progression from latent TB infection to active disease e.g. immunosuppression, diabetes, chronic renal failure etc [54].
The guiding epidemiological rule of thumb for TB is Styblo's rule that states that "an annual incidence of 50 sputum-smear-positive TB cases in a population of 100,000 generates an annual risk of infection of 1%" (probably an overestimate) [55-57]. Seen in this light, new strategies are required to identify and target the group of those likely to reactivate, not simply the conventional categories. Screening remains the cornerstone strategy in the road to elimination by 2050 [52].
A novel strategy proposed on the basis of the biomechanical pleural stress hypothesis elucidated above is to target using the five predictors of TB reactivation described above.
Screening can be targeted as a first stage to adolescent to young adult, low BMI subjects with a low thoracic index. This can be measured simply by using body calipers especially in thin people. Measurement is more difficult in the obese, but these are protected from TB infection anyhow [22]. The second screening stage would be to identify those with apical blebs using ultrasound, CT or magnetic resonance imaging (MRI).
Apical bullae can be diagnosed by thoracic ultrasound, however the procedure is operator-dependent and may be limited by poor ultrasound windows; so experience is limited with few published reports [58,59]. Low-dose CT has been proposed as a safe and effective yearly screening tool for lung cancer [60] as the 0.3 mSe radiation exposure required is much safer than the conventional dose of 7 mSe for contrast CTs. Use of low-dose CT is important since higher dose CT scans are known to result in an increase in lung cancer [61,62].
MRI has also been evaluated for possible lung cancer screening [63,64] and is equal to CT for investigation of thoracic pathology [65]. MRI is usually thought of as an expensive investigation with high peak electricity consumption and requiring liquid helium for superconductor cooling. However new MRI technologies are emerging that can make MRI a portable screening tool with low-cost and high-performance [66]. These include pre-polarized MRI [67,68], hyperpolarized gas MRI [69], permanent Halbach magnets [70], Transmit Array Spatial Encoding (TRASE) coils [71], Diffusion Weighted Imaging (DWI) [72] 2015 and combined modality methods (e.g. positron emission tomography (PET)-MRI and single photon emission CT (SPECT)-MRI) [73]. A working portable MRI, weighing less than 100 kg and at a cost of $USD 10,000, was constructed in 2014 [74]. A similar MRI machine would revolutionize screening for latent TB using the five predictors of TB reactivation identified above, while still maintaining the requirements of an ideal screening tool of safety, low-cost, portability, low-electricity consumption and high sensitivity and specificity.
A hypothesis for the pathogenesis of TB has been elucidated based on the biomechanics of pleural stress in the context of the lung as a pressure vessel. This hypothesis was complete in that it explained the occurrence of apical lower lobe disease, the age, gender and build of those at risk, explained the incidence of concurrent apical disease and the 15% incidence of congenital apical blebs also explains Esmail's prediction about the size of the population at-risk of TB reactivation. The computer-based pressure-vessel model has provided additional and important information to support the hypothesis for the pathogenesis of tuberculosis.
Once a subject can be identified as having several of the biomechanical predictors of TB reactivation discussed here, using initial screening based on BMI and thoracic index, then confirming the presence of apical blebs or bullae by imaging in an expected approximately 15% of the screening group [33], it would be possible to direct immunization, close observation or treatment of this high-risk group [52]. Low-cost, portable MRI has been suggested as a promising screening tool in the near future and may well be what is required to hasten reaching the 2050 target of global TB eradication.