Tinea capitis is a superficial fungal infection of the scalp and hair caused by dermatophytes such as Trichophyton and Microsporum. Tinea capitis is very rare in adults, and may affect those with immunosuppressive diseases or menopausal elderly women. Clinical manifestations along with trichoscopy and Wood’s light, can help the clinician to determine the correct diagnosis, in order to reduce irreversible sequelae and decrease multiple contagion. KOH direct exam and culture confirm diagnosis and aetiology. We report a 61-year-old female with a 50-year history of tinea capitis. This is an atypical case in a postmenopausal elderly woman who was treated as seborrheic dermatitis.
Tinea capitis, Adult, Trichoscopy, Corkscrew hairs, Trichophyton tonsurans, Terbinafine
Tinea capitis is a superficial fungal infection of the scalp and hair caused by dermatophytes such as Trichophyton and Microsporum. It can be caused by any pathogenic dermatophyte except for Epidermophyton floccosum and Trichophyton concentricum. In developed countries, Trichophyton tonsurans is the most common causative agent. Trichophyton rubrum, the most commonly isolated dermatophyte worldwide, is rarely the causative agent of this infection. Tinea capitis is the most common cutaneous mycosis in children whilst is uncommon in adults. It represents 4 to 10% of dermatophyte infections, and 69 to 90% affects mainly children between 3 and 8 years of age; boys are more affected than girls [1-3].
The non-inflammatory clinical presentation can be found in 90% of affected children. It is more frequent in tropical regions with low socioeconomic conditions and affects almost exclusively children (98%). The inflammatory form is observed only in 10% and most commonly seen in prepubescent children. Tinea capitis is caused by dermatophytes that use keratin as a nutrient source. Scalp erythema, scaling, and crusting are typical signs of this fungal infection [2-5].
Tinea capitis is rare in adults, it may affect those with severe immunosuppression such as leukaemia/lymphoma, systemic lupus erythematosus or diabetes mellitus, or patients taking corticosteroids and other immunosuppressive drugs [2,3]. Postmenopausal and elder women can present clinical manifestations due changes in the pH of the scalp and a decrease in quality and quantity in sebum, especially in medium chain fatty acids, as a result of hormonal imbalances that alter these protective effects of sebum [3,4]. Children present with pruritus, scaling, "black dots", hair loss, and posterior cervical lymphadenopathy, whilst tinea in adults is often subtler and may mimic the appearance of seborrheic dermatitis with mild inflammation and scarce scaling [5,6].
The frequency of each aetiological fungus is highly variable depending on the geographical location. In Mexico, Trichophyton tonsurans is presented in 15-28% of infected adults with tinea capitis. Subtle differences in the clinical appearance of tinea capitis may be noted depending upon the causative organism. Trichophyton tonsurans is usually an asymptomatic indolent disease in adults [7-9].
Clinical manifestations of the fungal infection are determined by the type of hair infestation (ectothrix or endothrix). Endothrix infestation owe to Trichophyton tonsurans produces generalized scaling and localized perifollicular inflammation reminiscent of lichen planopilaris, while ectothrix infestation due to Microsporum sp. produces well demarcated erythematous plaques suggestive of psoriasis. Patients are commonly exposed to Microsporum from dogs, cats (M. canis) or farm animals (M. nanum). M. audouinii is present in soil that is rich in keratinous material. Trichophyton tonsurans is an anthropophilic fungus that establishes a symbiotic relationship with its human host. The quantity of inoculation and the immune status of the host are other factors that will give a specific clinical scenario. The evaluation in the type of infestation can guide the final diagnosis, which is confirmed by identifying the aetiological agent [3-5,7,8].
Other available diagnostic tests are Wood's light, KOH direct exam and cultures on Sabouraud dextrose agar or Mycobiotic agar. Wood's light can be useful to screen adults in an elderly nursing home setting when Microsporum sp. are present as it is negative with T. tonsurans. KOH direct exam should include scale and broken hairs to increase sensitivity. Cultures determine the fungal genus and species but take as long as a month to grow [5].
Trichoscopy is the term used for dermoscopic imaging of the scalp and hair, is a non-invasive technique that can be performed rapidly in office evaluation. Tinea capitis most specific trichoscopic features are comma hairs, which were first reported by Rudnicka, et al. in 2008 [10]. Other features well known for tinea capitis are corkscrew and zigzag hairs, black dots and bar code-like hairs [3-9,11-14].
Tinea capitis must be treated with systemic antifungal agents because topical agents do not penetrate the follicle. Even so, concomitant treatment with 1% to 2.5% selenium sulphide or 2% ketoconazole shampoos are recommended at the first two weeks, in order to reduce transmission. For many years, the first-line treatment for tinea capitis has been griseofulvin because of its safety and effectiveness. However randomized clinical trials have confirmed that newer drugs, such as terbinafine and fluconazole have equal effectiveness and safety and shorter treatment courses [15].
Treatment of Trichophyton tinea capitis should be based on systemic griseofulvin or terbinafine for at least 8 weeks. The use of terbinafine 250 mg per day dose considerably shortens the duration of the treatment and is associated with fewer side effects than griseofulvin (gastrointestinal intolerance and headaches). Terbinafine may be superior to griseofulvin for Trichophyton species, whereas griseofulvin may be superior for the less common adult infections with Microsporum sp.
We present a case of tinea capitis with atypical chronic evolution.
A 61-year-old woman presented with a 50-year history of hair loss and a pruritic dermatosis on parietal and occipital region of the scalp. She had been diagnosed with seborrheic dermatitis and treated irregularly with topical preparations with salicylic acid and coal tar presenting partial resolution of scale and pruritus. She lives in a rural mountain zone in the north-west of the country, near the Sierra Madre Occidental. She has long lasting acral vitiligo, hypertension treated with losartan and recently diagnosed type 2 diabetes mellitus. Physical examination revealed a 10 × 15 cm plaque with diffuse alopecia, erythema and scaly scalp. Trichoscopy showed black dots, corkscrew, zig zag, and bar code-like hairs in a scaly and erythematous base (Figure 1 and Figure 2).
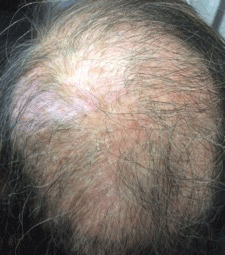 Figure 1: Alopecic plaque in the scalp with erythema and desquamation. View Figure 1
Figure 1: Alopecic plaque in the scalp with erythema and desquamation. View Figure 1
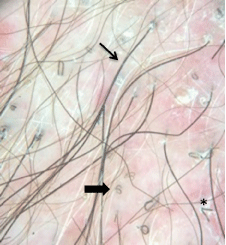 Figure 2: Trichoscopy showed corkscrew hairs (dark arrow), bar code-like hairs (tiny arrow), zig zag hairs (*) and black dots in a scaly and erythematous base. View Figure 2
Figure 2: Trichoscopy showed corkscrew hairs (dark arrow), bar code-like hairs (tiny arrow), zig zag hairs (*) and black dots in a scaly and erythematous base. View Figure 2
KOH examination was positive with endothrix infestation (Figure 3a) and culture was positive for T. tonsurans (Figure 3b). Oral terbinafine 250 mg per day was initiated but the patient was lost during follow up due to geographical distance from our dermatological centre.
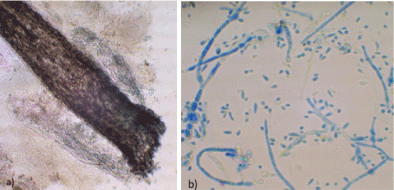 Figure 3: a) KOH direct exam 10x; b) Positive culture on Sabouraud agar. View Figure 3
Figure 3: a) KOH direct exam 10x; b) Positive culture on Sabouraud agar. View Figure 3
Tinea capitis is a frequent scalp infection in children typically caused by Trichophyton and Microsporum species although in adults is not so common, therefore diagnosis can be delayed or erroneous. As our patient, who has had this infection for over 50 years without adequate treatment nor a proper diagnosis. Additional factors such as living in a rural endemic zone and having no access to a dermatologist consultation may have contributed to this atypical chronic clinical presentation. Long lasting cases like this are extremely rare, therefore the importance of informing them.
El-Khalawany, et al. described in 2013 the predisposing factors in adults; contact with animals was a common predisposing factor in rural areas, whereas transmitted infection from other family members was more common among urban patients. They also found a higher incidence of tinea capitis in adults (46.4%) among menopausal women due to the involution of sebaceous glands following decreased blood estrogen levels. Systemic diseases such as diabetes mellitus, use of immunosuppressants drugs and long use of topical or systemic steroids are commonly reported in association with tinea capitis affecting adults [16].
Other source of infection reported in the literature is the use of certain hair care instruments in hair salon services [17].
From 1960 to 1970 tinea capitis was mainly caused by Trichophyton, now a days in Mexico, M. canis is the most frequent aetiological agent causing the 80% of infections and T. tonsurans only the 15% T. tonsurans is found in rural areas but particularly in the northern part of the country. T. tonsurans performs an endothrix type of infestation, which causes hair to break easily [13,14,18].
The epidemiology of tinea capitis varies in different geographical areas. For instance, in the 2014 Cervetti, et al. described the aetiological agents in 13 Italians adults: 7 patients with M. canis, 3 with T. mentagrophytes, 2 patients with T. rubrum, 1 with T. violaceum but no one with T. tonsurans [14].
As Morell, et al. described in 2012, T. tonsurans infection in adults can be manifested atypically, with diffuse desquamation and lesions suggestive of seborrheic dermatitis or another inflammatory dermatosis [13].
Tinea capitis caused by Trichophyton species should be treated with systemic griseofulvin or terbinafine for at least 8 weeks. Terbinafine may be superior to griseofulvin for Trichophyton species, whereas griseofulvin may be superior for the less common adult infections with Microsporum sp.
In conclusion, tinea capitis in elderly women should be considered as a differential diagnosis with other inflammatory scalp diseases as it is usually characterized by atypical alopecia plaques. Medical awareness of this rare entity is mandatory to avoid delay in diagnosis and inappropriate treatment. Mycological samples should be taken in adults with little or no response to common treatments.
Authors have no conflict of interest to disclose.