Fungal pneumonia or pulmonary mycosis is a fungal infection of the lungs [1]. It can be caused by an endemic pathogen in immunocompetent hosts or an opportunistic agent in immunocompromised patients or a combination of both. Among the myriads of pathogens implicated in pulmonary mycosis, Aspergillus spp. Candida spp. Coccidioides spp. and Pneumocystis jiroveci are more prevalent [2]. Pulmonary mycoses present a significant clinical challenge due to the wide-ranging etiology and potential for severe complications.
Pulmonary mycoses, with their elusive nature, can masquerade as primary pulmonary malignancies [3-5]. Diverse clinical and radiological manifestations of fungal pneumonia including low grade fevers, weight loss, pulmonary mass or nodules overlap pulmonary malignancy. This can delay diagnosis and lifesaving treatment in patients afflicted with fungal pneumonia. It is imperative to delineate overlapping features between fungal pneumonia and pulmonary malignancies and develop better and more accurate systemic approach to diagnostic evaluation.
We present a case series of four patients with pulmonary mycoses. These cases include aspergillosis, blastomycosis, coccidioidomycosis, and histoplasmosis highlighting clinical and radiological heterogeneity in patients with pulmonary mycoses. This case series underscores diagnostic pitfalls inherent in differentiating pulmonary mycoses from malignant processes highlighting a clinically unmet need for better diagnostic modalities.
Multiple pulmonary nodules due to aspergillosis in patient with aplastic anemia
A 42-year-old female with history of aplastic anemia on immunosuppression and morbid obesity with gastric sleeve surgery presented with severe fatigue and vaginal bleeding. One month prior, she was diagnosed with aplastic anemia and was started on cyclosporine, eltrombopag and horse anti-thymocyte globulin. On presentation, she was hemodynamically unstable with blood pressure 99/49 mmHg, temperature 101.4 °F and heart rate 128 beats/min. Her laboratory data on presentation is shown in Table 1.
Table 1: Shows laboratory data on presentation. View Table 1
She was admitted to intensive care unit (ICU) with diagnosis of septic shock and diabetic ketoacidosis (DKA). She was treated with broad spectrum antibiotics, vasopressors, fluid resuscitation, multiple blood transfusions, and insulin drip and started to improve clinically. She was subsequently found to have elevated total bilirubin of 4.6 mg/dl (n = 0.2-1.1 mg/dl), which was most likely due to cyclosporine [6]. However, magnetic resonance cholangiopancreatography (MRCP) was done to investigate hyperbilirubinemia which showed hepatic hemosiderosis and left sided pleural effusion concerning for empyema. Pleural effusion (as shown in Figure 1A) was drained along with chest tube placement (Figure 1B) and fluid analysis of pleural effusion confirmed diagnosis of hemothorax, which was most likely to be spontaneous in setting of low platelet count. Patient was requiring almost daily platelets and packed red blood cells (PRBC) transfusions. Computerized tomography (CT) scan of chest was performed after thoracentesis and was suggestive of decreased left sided pleural effusion and four nodular opacities in right upper and lower lobes (as shown in Figure 1C and Figure 1D).
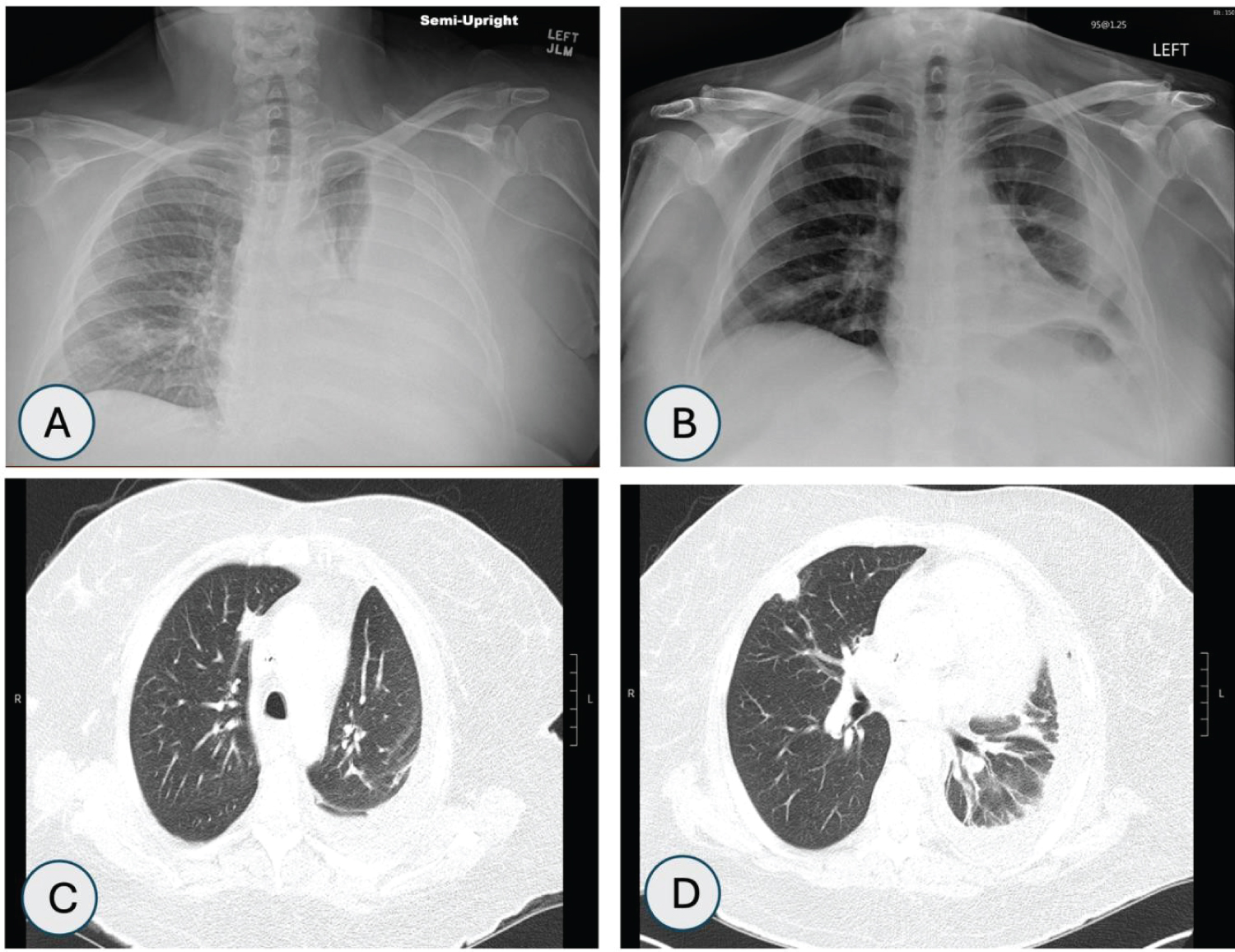 Figure 1: A) Chest X-ray (CXR) shows left sided pleural effusion; B) CXR post left sided chest tube placement; C) Computerized tomography (CT) chest showing right medial upper lobe nodule; D) CT chest showing right subpleural based nodule.
View Figure 1
Figure 1: A) Chest X-ray (CXR) shows left sided pleural effusion; B) CXR post left sided chest tube placement; C) Computerized tomography (CT) chest showing right medial upper lobe nodule; D) CT chest showing right subpleural based nodule.
View Figure 1
Transthoracic echocardiography (TTE) was done to rule out infective endocarditis which was unremarkable for any valvular pathology. Blood cultures did not grow any pathogenic organisms however beta glucan was elevated to 280 pg/ml (n < 80 pg/ml). Results for fungal testing are shown in Table 2.
Table 2: Results for fungal testing for patient with aspergillosis. View Table 2
Confirmatory tissue diagnosis was required to differentiate between benign versus malignant cause of pulmonary nodules which was challenging due to persistently low platelet count around 10,000/uL and patient requiring transfusions daily. Patient was given multiple platelet transfusions to increase platelet count to greater than 50,000/uL and CT guided core biopsies of one of pulmonary nodules was performed. Pathologic examination of tissue specimen established diagnosis of aspergillosis (Figure 2A, Figure 2B, Figure 2C and Figure 2D). Patient was initially treated with amphotericin B which de-escalated to voriconazole after confirmatory tissue diagnosis. It was later on switched to isavuconazole due to blurry vision. Timeline of our patient is shown in Figure 3.
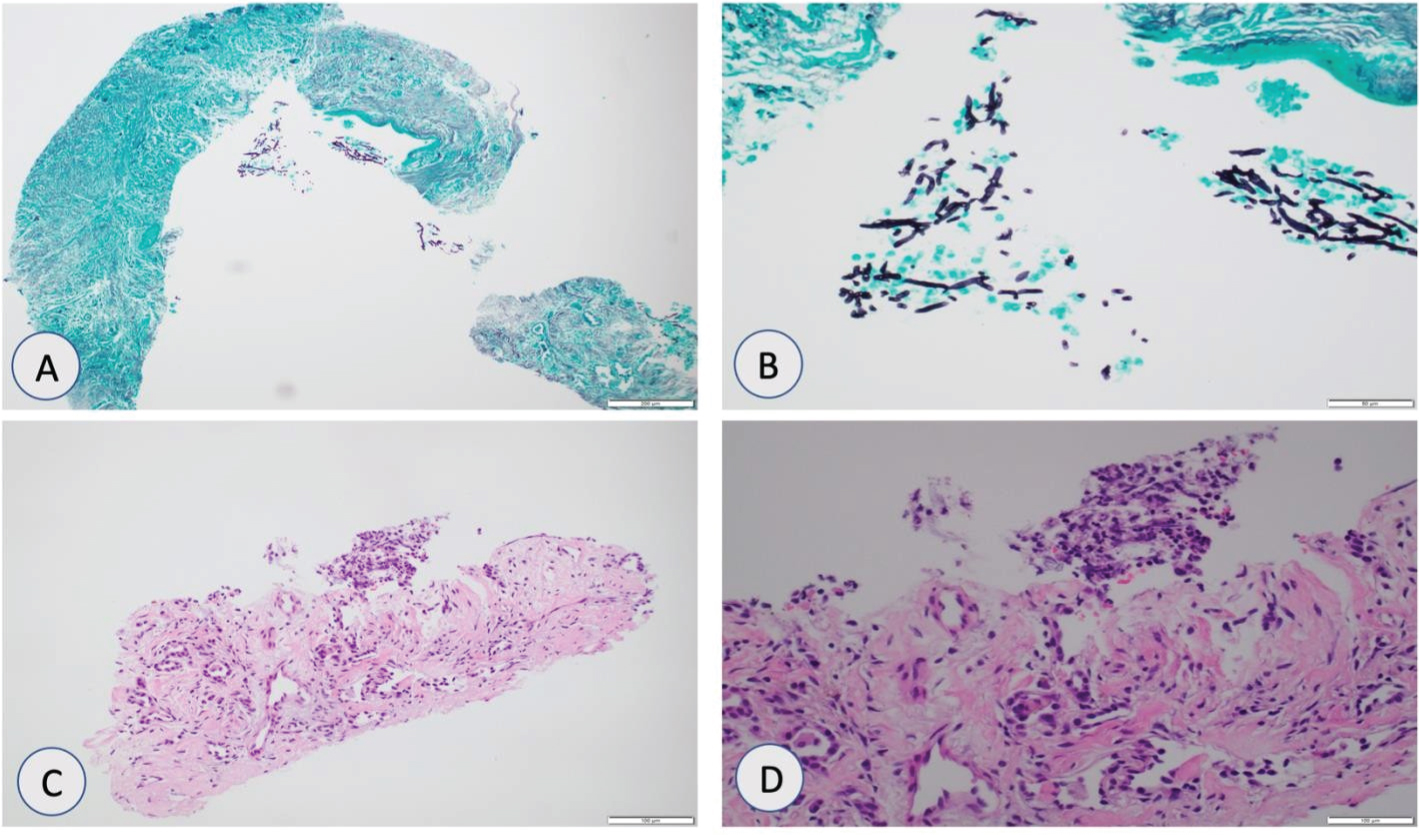 Figure 2: (A & B) Grocott methenamine silver (GMS) stain showing fungal organisms with morphology similar to Aspergillus species at 20x and 40x magnification; (C & D) Hematoxylin and eosin (H&E) stain showing fibrotic airways with mixed inflammation and necrotic debris at 20x and 40x magnification.
View Figure 2
Figure 2: (A & B) Grocott methenamine silver (GMS) stain showing fungal organisms with morphology similar to Aspergillus species at 20x and 40x magnification; (C & D) Hematoxylin and eosin (H&E) stain showing fibrotic airways with mixed inflammation and necrotic debris at 20x and 40x magnification.
View Figure 2
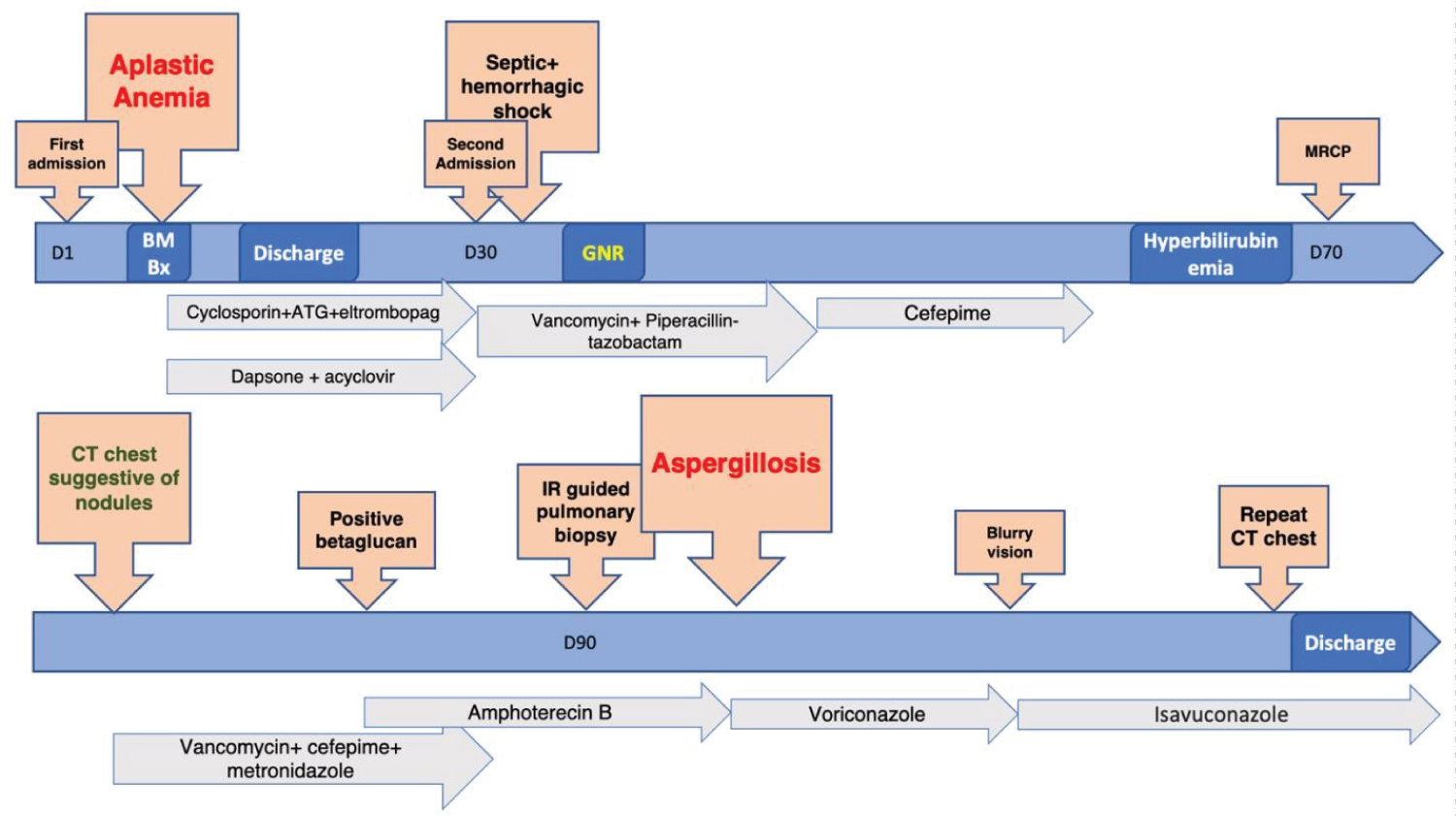 Figure 3: Timeline of our patient.
View Figure 3
Figure 3: Timeline of our patient.
View Figure 3
Multisystem involvement of blastomycosis with rare central nervous system (CNS) involvement in an immunocompetent patient.
A 19-year-old male presented to emergency room (ER) with multiple complaints including left sided neck swelling, draining ulcer on left breast, right sided knee pain, and multiple skin nodules for 2 weeks. On presentation, he was hemodynamically stable. Laboratory data on presentation is shown in Table 3.
Table 3: Shows laboratory data on presentation. View Table 3
CT scan of chest showed right retrohilar mass measuring 2.3 × 3.6 × 7.7 cm obstructing right lower lobe bronchus and osteomyelitis of left clavicular head along with rim enhancing fluid collection as shown in Figure 4A, Figure 4B, Figure 4C and Figure 4D.
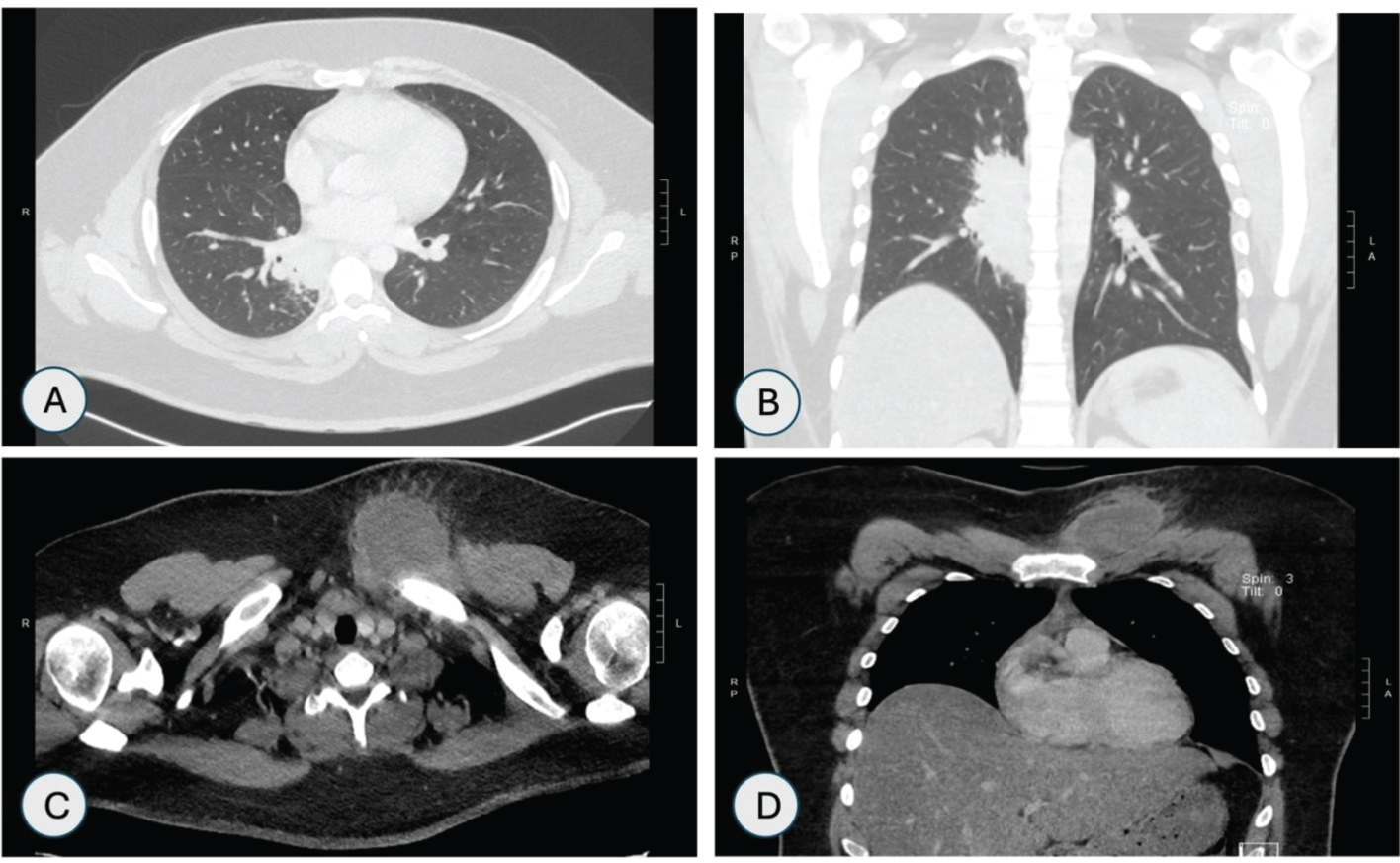 Figure 4: CT Chest-Lung window (A & B) Axial and coronal views showing right retrohilar mass measuring 2.3 × 3.6 × 7.7 cm focally obstructing the bronchus serving the basal medial right lower lobe. CT chest-bone window; (C & D) Axial and coronal views showing lucencies in head of the left clavicle with anterior subcutaneous fluid collection with rim enhancement.
View Figure 4
Figure 4: CT Chest-Lung window (A & B) Axial and coronal views showing right retrohilar mass measuring 2.3 × 3.6 × 7.7 cm focally obstructing the bronchus serving the basal medial right lower lobe. CT chest-bone window; (C & D) Axial and coronal views showing lucencies in head of the left clavicle with anterior subcutaneous fluid collection with rim enhancement.
View Figure 4
CT head was suggestive of small enhancing lesions in the right frontal cerebral lobe and left cerebellar hemisphere (Figure 5A). Magnetic resonance imaging (MRI) of head showed multiple small enhancing foci in bilateral cerebral and cerebellar hemispheres as shown in Figure 5B. MRI of left knee showed enhancement in the medial tibial epiphysis and metaphysis with adjacent soft tissue edema and large suprapatellar effusion (Figure 5C).
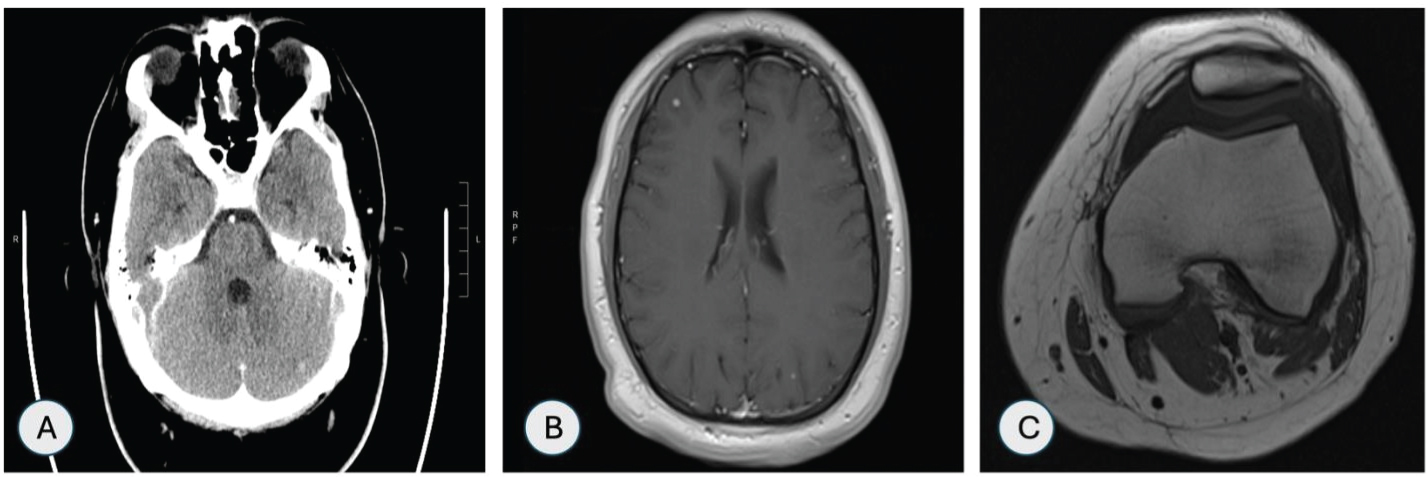 Figure 5: (A) CT head showing 5 mm enhancing lesion in left cerebeller hemisphere; (B) MRI head showing multiple enhancing foci in bilateral cerebral hemispheres; (C) MRI left knee showing large suprapateller effusion.
View Figure 5
Figure 5: (A) CT head showing 5 mm enhancing lesion in left cerebeller hemisphere; (B) MRI head showing multiple enhancing foci in bilateral cerebral hemispheres; (C) MRI left knee showing large suprapateller effusion.
View Figure 5
Patient was admitted with suspicion of widespread malignancy and underwent ultrasound guided biopsy of left breast ulcer (Figure 6A), right wrist subcutaneous nodule (Figure 6B), lumbar puncture and arthrocentesis of left knee.
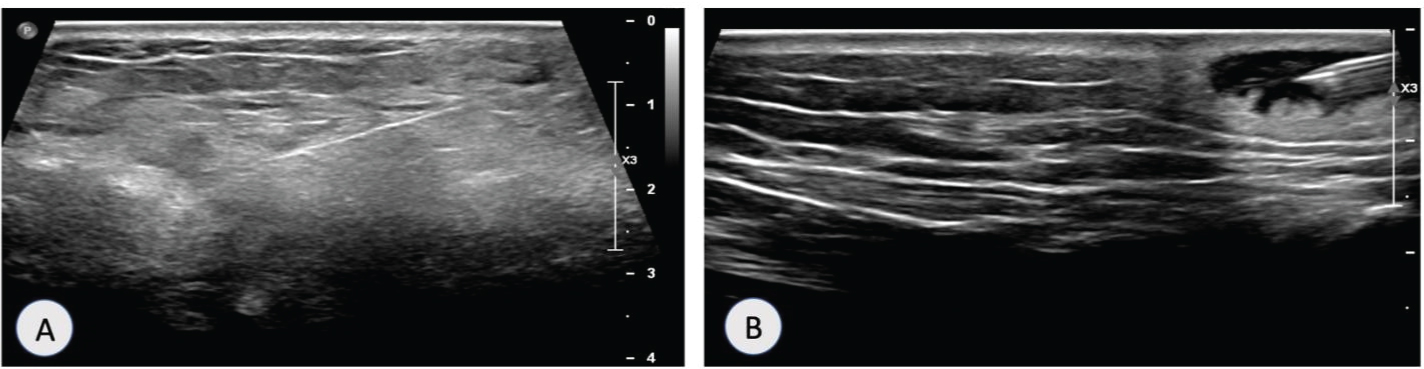 Figure 6: (A) Ultrasound guided biopsy of left breast lesion; (B) Ultrasound guided biopsy of right wrist lesion.
View Figure 6
Figure 6: (A) Ultrasound guided biopsy of left breast lesion; (B) Ultrasound guided biopsy of right wrist lesion.
View Figure 6
QuantiFERON gold, beta glucan, aspergillus galactomannan and fungal isolator blood cultures were unremarkable for any pathogenic organism. Antibody testing was positive for Blastomyces dermatitidis (7.9 (n = < = 0.9 IV)) as shown in Table 4.
Table 4: Shows laboratory data of patient with blastomycosis. View Table 4
Left breast and right wrist biopsy showed broad based budding yeast as shown in pathology slides (Figure 7A and Figure 7 B). Patient had a pet snake and mice (used as food for snake) kept in potting soil in his room which was a risk factor for blastomycosis. Patient was treated with amphotericin B due to rare central nervous system involvement for four weeks and was switched to voriconazole with improvement.
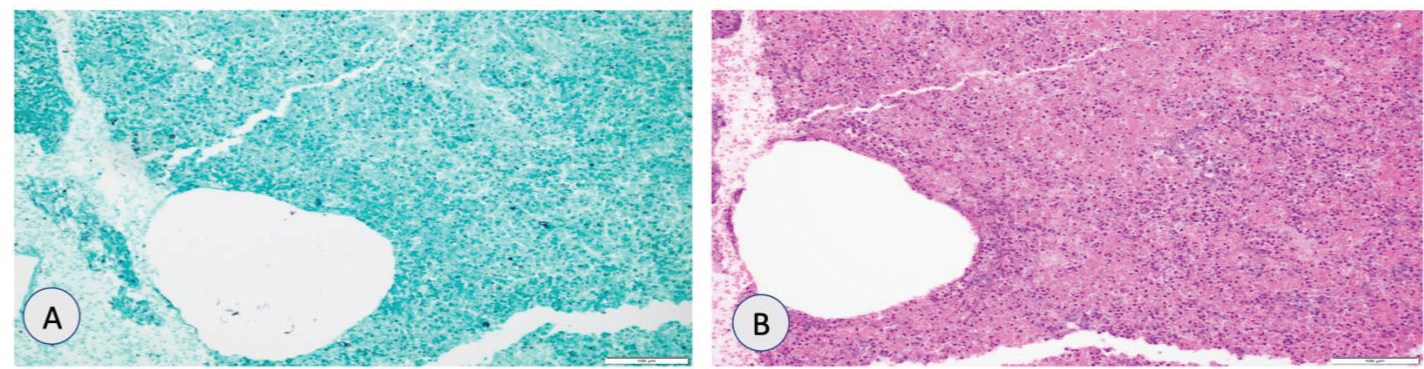 Figure 7: (A) Grocott methenamine silver (GMS) showing rounded yeast forms with broad-based budding suggestive of blastomycoses; (B) Hematoxylin and eosing (H&E) stain showing extensive necroinflammatory debris.
View Figure 7
Figure 7: (A) Grocott methenamine silver (GMS) showing rounded yeast forms with broad-based budding suggestive of blastomycoses; (B) Hematoxylin and eosing (H&E) stain showing extensive necroinflammatory debris.
View Figure 7
Immunocompetent patient with coccidioidomycosis as tracheal mass
A 53-year-old male presented to ER with complaints of having fever, productive cough, and shortness of breath for past few weeks. Patient was recently treated with azithromycin and oral steroids in outpatient settings.
During this admission, CXR was suggestive of consolidation in the right middle lobe as shown in Figure 8A.
Patient was started on ceftriaxone and azithromycin but he continued to spike high-grade fevers despite being on antibiotics. Non-contrast CT scan of chest was done which was suggestive of consolidation in right middle lobe with central areas of necrosis along with mediastinal lymphadenopathy (Figure 8B and Figure 8C).
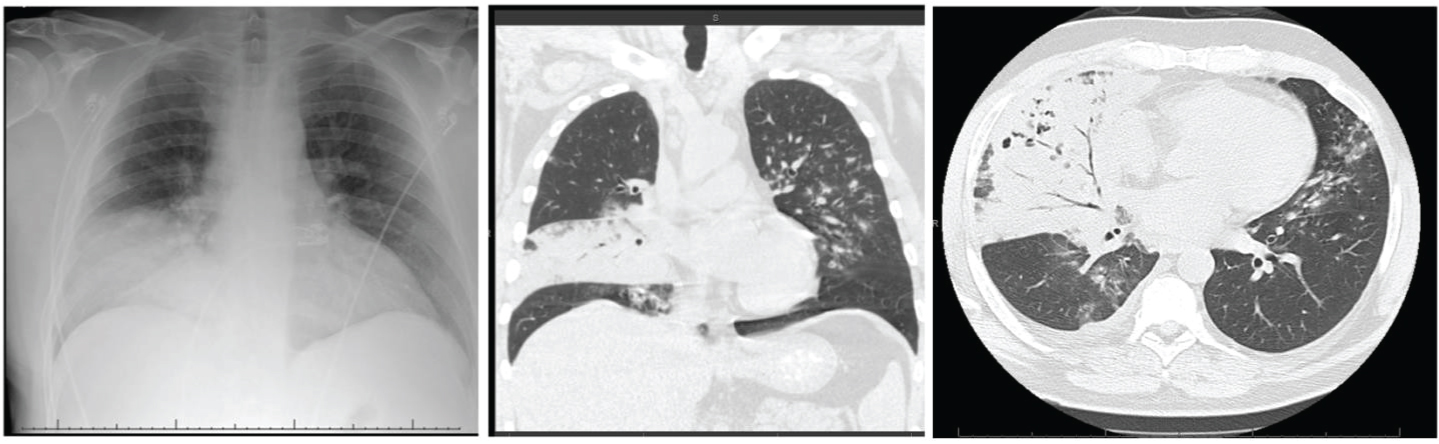 Figure 8: (A) CXR showing right middle lobe consolidation; (B & C) CT chest showing right middle lobe consolidation along with mediastinal lymphadenopathy.
View Figure 8
Figure 8: (A) CXR showing right middle lobe consolidation; (B & C) CT chest showing right middle lobe consolidation along with mediastinal lymphadenopathy.
View Figure 8
Antibiotics were escalated to vancomycin, cefepime, and metronidazole. Blood culture, urinary legionella, and streptococcus antigen were all unremarkable for pathogenic organisms. Beta glucan level was elevated to 73 pg/ml and coccidioides antibody and galactomannan antigen were negative. Sputum culture was remarkable for Coccidioides immitis . Upon a detailed review of history, patient recently went to vacation in California and was golfing there outdoors quite often. Since then, he had been having fevers with night sweats and cough.
Patient was diagnosed with coccidioidomycosis and was discharged on fluconazole. Patient continued to have a persistent cough with hoarseness and low-grade fevers after discharge. Bronchoscopy was suggestive of tracheal mass above carina (as shown in Figure 9) in a concentric fashion which was concerning for malignancy, however biopsy was suggestive of fungal elements confirming previously diagnosed coccidiomycosis.
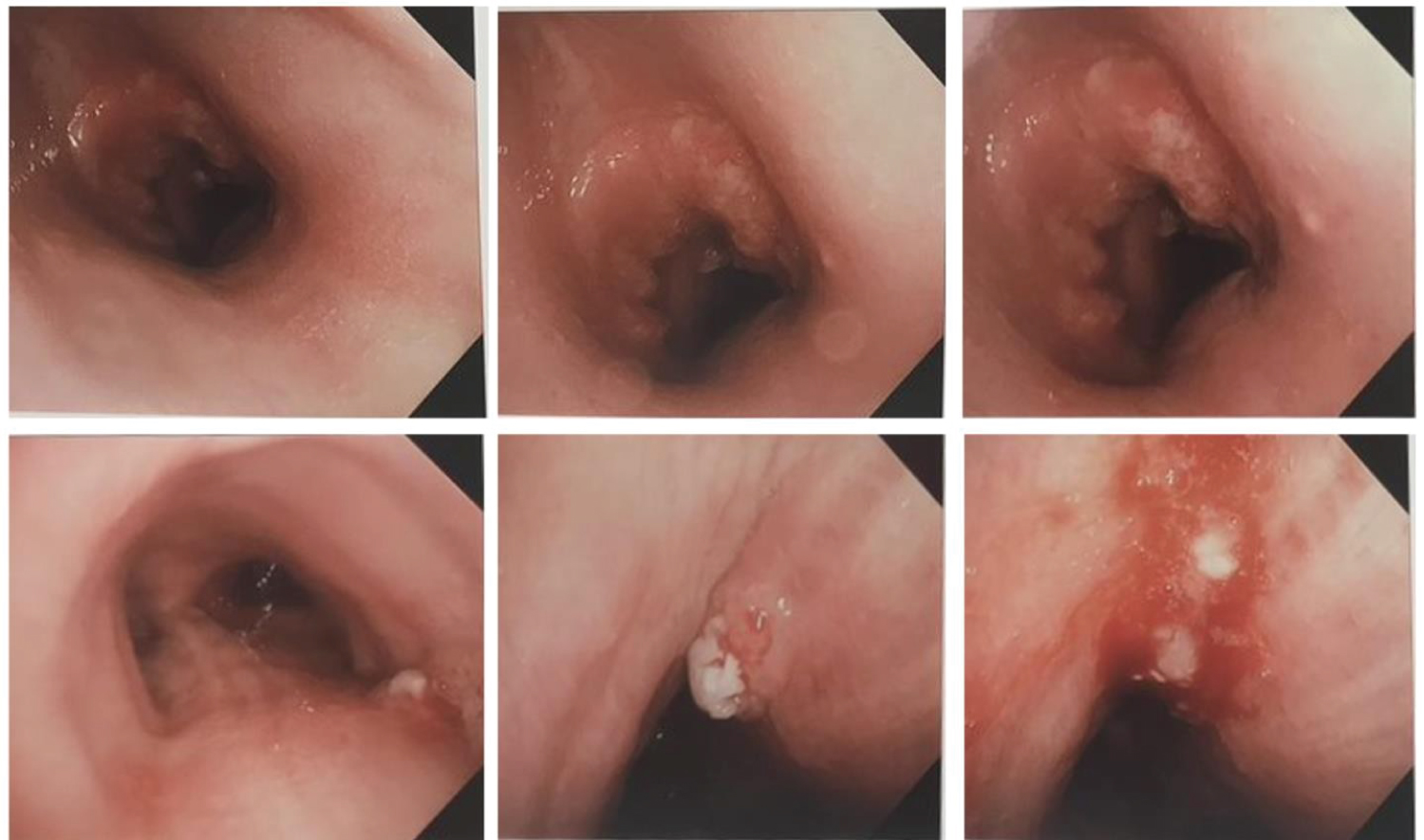 Figure 9: Bronchoscopic images showing tracheal mass.
View Figure 9
Figure 9: Bronchoscopic images showing tracheal mass.
View Figure 9
Considering disseminated coccidioidomycosis and fluconazole induced transaminitis, patient was started on amphotericin B with improvement in symptoms.
Pulmonary masses in an immunocompetent patient due to histoplasmosis
A 64-year-old male with an extensive smoking history (69 pack-year), chronic obstructive pulmonary disease (COPD), nonischemic cardiomyopathy with implantable cardioverter-defibrillator (ICD), and type 2 diabetes presented with complaints of having nonproductive cough, night chills and unintentional weight loss for the past few weeks. Chest X-ray was suggestive of 12 mm smooth density along lateral aspect of right upper lobe as shown in Figure 10A. CT chest showed right upper lobe parenchymal masses with bulky mediastinal and right hilar lymphadenopathy as shown in Figure 10B, Figure 10C and Figure 10D.
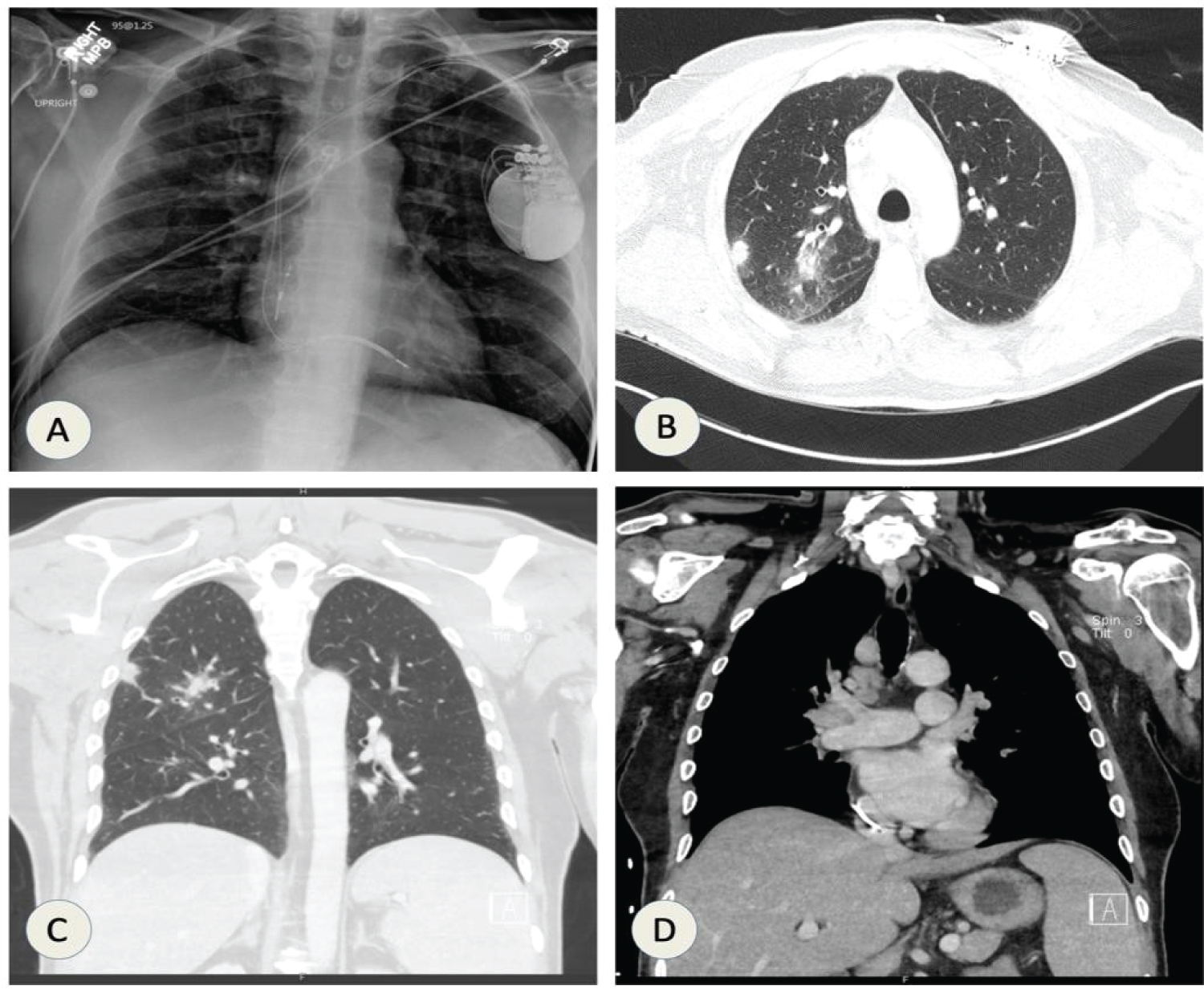 Figure 10: (A) CXR shows 12 mm small rounded density seen along the lateral aspect of the right upper lobe inferior to the posterior aspect of the right fourth rib; CT chest (B & C) showing right peripheral upper lobe mass; (D) Mediastinal and right hilar lymphadenopathy.
View Figure 10
Figure 10: (A) CXR shows 12 mm small rounded density seen along the lateral aspect of the right upper lobe inferior to the posterior aspect of the right fourth rib; CT chest (B & C) showing right peripheral upper lobe mass; (D) Mediastinal and right hilar lymphadenopathy.
View Figure 10
Considering extensive history of smoking and suspicious radiological findings, there was a suspicion of pulmonary malignancy. However, due to history of night chills, fungal serologies were obtained as shown in Table 5. Bronchoscopy with bronchoalveolar lavage (BAL) was unremarkable for malignant cells. Endobronchial ultrasound (EBUS) guided transbronchial sampling was suggestive of budding yeast as shown in Figure 11A, Figure 11B, Figure 11C and Figure 11D. Patient was treated with isavuconazole as he was on multiple antiarrhythmics with resolution of symptoms.
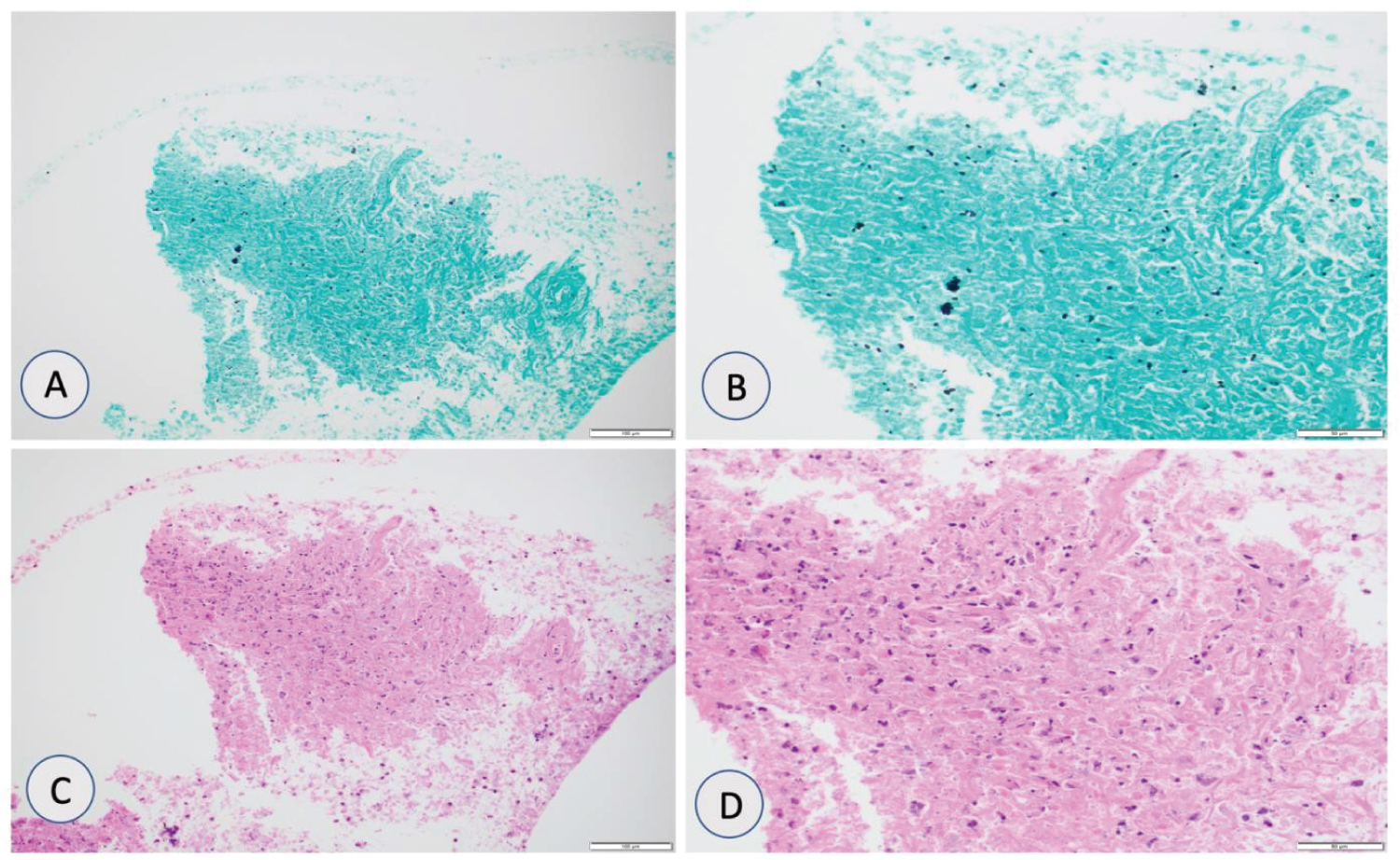 Figure 11: (A & B) Grocott methenamine silver (GMS) stain showing budding yeast suggestive of histoplasmosis species at 20x and 40x magnification; (C & D) Hematoxylin and eosin (H&E) stain showing necroinflammatory debris at 20x and 40x magnification.
View Figure 11
Figure 11: (A & B) Grocott methenamine silver (GMS) stain showing budding yeast suggestive of histoplasmosis species at 20x and 40x magnification; (C & D) Hematoxylin and eosin (H&E) stain showing necroinflammatory debris at 20x and 40x magnification.
View Figure 11
Table 5: Shows laboratory values of patient. View Table 5
Invasive and disseminated fungal infections are associated with high mortality especially in immunocompromised patients [7]. Current estimates of fungal disease incidence and mortality are imprecise for United States (US) [7] as fungal pneumonia is not mandatory reportable for all states. As a result, data is mostly reliable from those states where fungal infections are reportable. As per center of disease control (CDC), coccidioidomycosis causes an estimated 15% to nearly 30% of community-acquired pneumonias in highly endemic areas. There were approximately 200 coccidioidomycosis-associated deaths each year (deaths in which coccidioidomycosis was listed as a primary or contributing cause on a death certificate) in the United States during 1999-2019 [8]. For histoplasmosis, state-specific annual incidence rates ranged from 0 to 4.3 cases per 100,000 population during 2011-2014 [9]. Crude mortality rate was approximately 8% for adults who were hospitalized for histoplasmosis [9]. Similarly, two cohort analyses of blastomycosis cases in US found that between 8-9% of people with blastomycosis died [10]. Total mycologic burden of US includes more than 75,000 hospitalizations and nearly 9 million outpatient visits occur every year and total costs are conservatively estimated at $11.5 billion and could be as high as $48 billion [11]. Thus, it is imperative that fungal pneumonia is diagnosed and treated promptly especially in immunocompromised patients.
Fungal pneumonia or pulmonary mycoses mimic pulmonary malignancy clinically and radiologically. It can be associated with fever, chills, night sweats, and malaise, which can be non-specific and overlap with the symptoms of lung cancer [12]. Radiological features include pulmonary nodules, parenchymal mass showing spiculated margins, thick-walled cavity, cavity showing nodular margins and chest wall invasion [13]. Case 1 with aspergillosis had multiple pulmonary nodules with history of aplastic anemia. There was a concern for leukemic progression of aplastic anemia presenting as pulmonary nodules radiologically. On CT scan, there appeared to be feeding vessels supplying these nodules and radiologist raised suspicion of septic pulmonary emboli however, TTE was not suggestive of infective endocarditis. Patient 2 with blastomycosis had right retrohilar mass with radiological features highly suggestive of malignancy. There were multiple brain lesions which were overlapping with lung cancer as CNS involvement is rare in blastomycosis. Moreover, initial non- culture based testing including galactomannan and beta glucan were unremarkable which was confusing. Patient 3 had tracheal involvement of coccidiomycosis which is rare and there was suspicion of tracheal cancer initially until diagnosis of disseminated coccidioidomycosis was confirmed by tissue biopsy. Patient 4 with histoplasmosis had extensive tobacco use history along with clinical and radiological features suggestive of malignancy until histoplasma urinary antigen was found to be positive. This led to tissue biopsy confirming diagnosis of histoplasmosis.
These cases highlight that distinguishing infectious fungal pulmonary lesions from lung cancer is challenging for both clinicians and radiologists. Detection of fungal antigens in body fluids, including galactomannan and beta glucan can aid in diagnosis. However, these non-culture based serum diagnostics have variable sensitivity and specificity, and limitations such as false positives and decreased sensitivity in the setting of mold-active antifungal prophylaxis in immunocompromised patients [14]. Gold standard for the diagnosis of fungal pneumonia remains tissue histopathology confirmation of the invasion of hyphae in lung tissue [15]. This has significant limitations in itself including inability to collect specimens, risk associated with procedures especially in immunosuppressed patients with thrombocytopenia (as seen in patient 1), delayed diagnosis associated with waiting for culture results and probability of inconclusive results [16].
The need of hour is to develop more efficient testing technologies and sampling techniques which can diagnose fungal infections with rapid turnover, have better sensitivity and specificity, and avoid invasive sampling interventions. Multiple molecular diagnostic methods to diagnose pulmonary mycoses are being developed currently. Clinical validation of these tests is challenging as accuracy is difficult to determine when gold standard tests are far from perfect [17]. These diagnostic methods include testing technologies based on polymerase chain reaction (PCR), DNA sequencing, whole genome sequencing, mass spectrometry, fluorescence in situ hybridization (FISH) and magnetic resonance [18]. It is difficult to predict that which diagnostic platform will overcome barriers of cost, sample preparation, multi-step technician dependance, and use of expensive instrumentation. Despite these challenges, molecular based fungal testing can be holy grail of diagnostic mycology and can replace standard culture-based methods.
None.
None.