Human beings propel themselves through their environment primarily by walking. This activity is a sensitive indicator of overall health and self-efficacy. Impairments in gait lead to loss of functional independence and are associated with increased fall risk.
This structured review examines the basic biology of gait in term of its kinematic properties and control. It describes the common gait disorders in advanced age and proposes a scheme for their recognition and evaluation in the regular ambulatory care setting.
The electronic databases, PubMed, Scopus and Web of Science, were searched in a 3-phase process for publications on gait disorders combined with older adults, clinical evaluation and no or minimal instrumentation during 2000-2020. Twenty-six articles were identified and pulled.
Step width increases with age while step length decreases. Since cadence hardly changes over time, gait velocity decreases by about 1% annually in old age. Central pattern generators in the cervical and lumbar segments of the spinal cord create the basic cyclic gait sequence. They are modulated by supraspinal networks as well as visual vestibular and proprioceptive peripheral inputs. Gait activity and cognitive processes, especially attention, are closely related and the neurological substrate of this nexus is the central cholinergic system. Dynamic stability during gait relies on predictive, proactive and reactive strategies. Predictive control enables the anticipation of displacements of the center of mass and adjustments to them. Proactive strategies make decisions about modifications to the locomotor pattern in advance of the encounter with a potential challenge. Reactive strategies are evoked after a perturbation has occurred. Gait disorders are classified on a phenomenological scheme and their defining clinical presentations are described. An approach to the older adult patient with a gait disorder comprising standard (history and physical examination) and specific gait evaluations, is presented. The specific gait assessment has qualitative and quantitative components. Not only is the gait disorder recognized, it enables its characterization in terms of severity and associated fall risk.
Gait is the most fundamental mobility task and a key requirement for independence. Its impairment and attendant consequences can be incapacitating to a very significant degree. The proper diagnosis of gait disorders is thus important for interventional and prognostic purposes. A compact, high-yield evaluative scheme that is appropriate for use in the ambulatory care setting is here proposed. Although inertial measurement units became available about two decades ago, a niche has still not been found for them in the clinic environment and careful hyphenate - as in history-taking along with physical examination by the clinician remain the linchpin of gait assessment.
Gait disorders, Older adults, Clinical assessment
Gait or walking is the predominant means whereby human beings propel themselves through their environment. It involves repetitive cyclical activities of the lower extremities which result in locomotion, a key in ingredient in activities of daily. Gait depends on the integrity of and complex interaction among multiple body systems - musculoskeletal, nervous, cardiopulmonary - and not surprisingly, has been shown to be a sensitive indicator of overall health and functional status, even in those who do not report any gait impairment [1,2]. For instance, self-selected gait speed is closely correlated with life expectancy [3] and has been recommended as the "6th vital sign" [4].
About two-thirds of body weight is about two-thirds of body height above a relatively narrow base of support. This, inherently, is a potentially unstable configuration. Also, periods of single- and double- limb support, with variable stability, alternate during walking. As such human bipedal gait is a veritable balance control challenge. Most falls in older adults occur during gait [5,6]. Falls are the most common causes of accidental injury and death in older adults [7].
Gait disorders have an estimated incidence of 168.6 per 1000 person-years [8]. At least a third of community-dwelling older adults either experience difficulties walking or require the assistance of another person or special equipment to walk [9]. The prevalence increases with age, from about 15% at age 60 to about 82% at age 85 [8]. Men have a higher incidence of neurologic gait disorders while impairments with a non-neurologic etiologic basis predominate in women [8], due in part to the female preponderance of lower-extremity degenerative joint disease which is the most common cause of walking-related disability among older adults in the United States [10]. It is estimated that in 2040, there will be 15.4 million older adults in the United States (about 19% of that population segment) unable to walk 2-3 blocks, a scenario which will add approximately $42 billion to the overall healthcare cost [11].
The enormity of the problem of gait disorders and their potential clinical consequences notwithstanding, the subject is not specifically covered in most textbooks and physicians are often insufficiently trained to assess gait and axial motor symptoms. As a result, these disorders are inadequately evaluated and largely under-diagnosed [2,12]. The purpose of the present structured review is to provide a synopsis of the basic biology of gait and its clinical classification. In addition, an approach to the patient with a gait disorder suited to the ambulatory care setting is proposed. Instrumented gait analysis and the management of gait disorders are beyond its scope.
Three electronic bibliographic databases: PubMed, Scopus and Web of Science, were searched for gait assessment tools (Figure 1). The searches reached back to 2000, using medical subject heading (MeSH) terms and keywords obtained from four sentinel articles as well as controlled vocabulary terms appropriate to each database. The first step was a high-sensitivity search of PubMed, using the following strategy: http://www-ncbi-nlm-nih-gov.proxy.lib.umich.edu/pubmed?term= (("gait disorders"[tiab] OR neurologic gait disorders[mh] OR neurologic gait disorders[tiab] OR non-neurologic gait disorders[tiab] AND (office OR clinic) AND (clinical assessment OR evaluation)). The filters "human", "English", "aged: 65+ years", "reviews", "randomized controlled trials" and "2000 - 2020" were applied and 175 titles were generated. The searches of SCOPUS and Web of Science yielded 424 and 31 articles respectively.
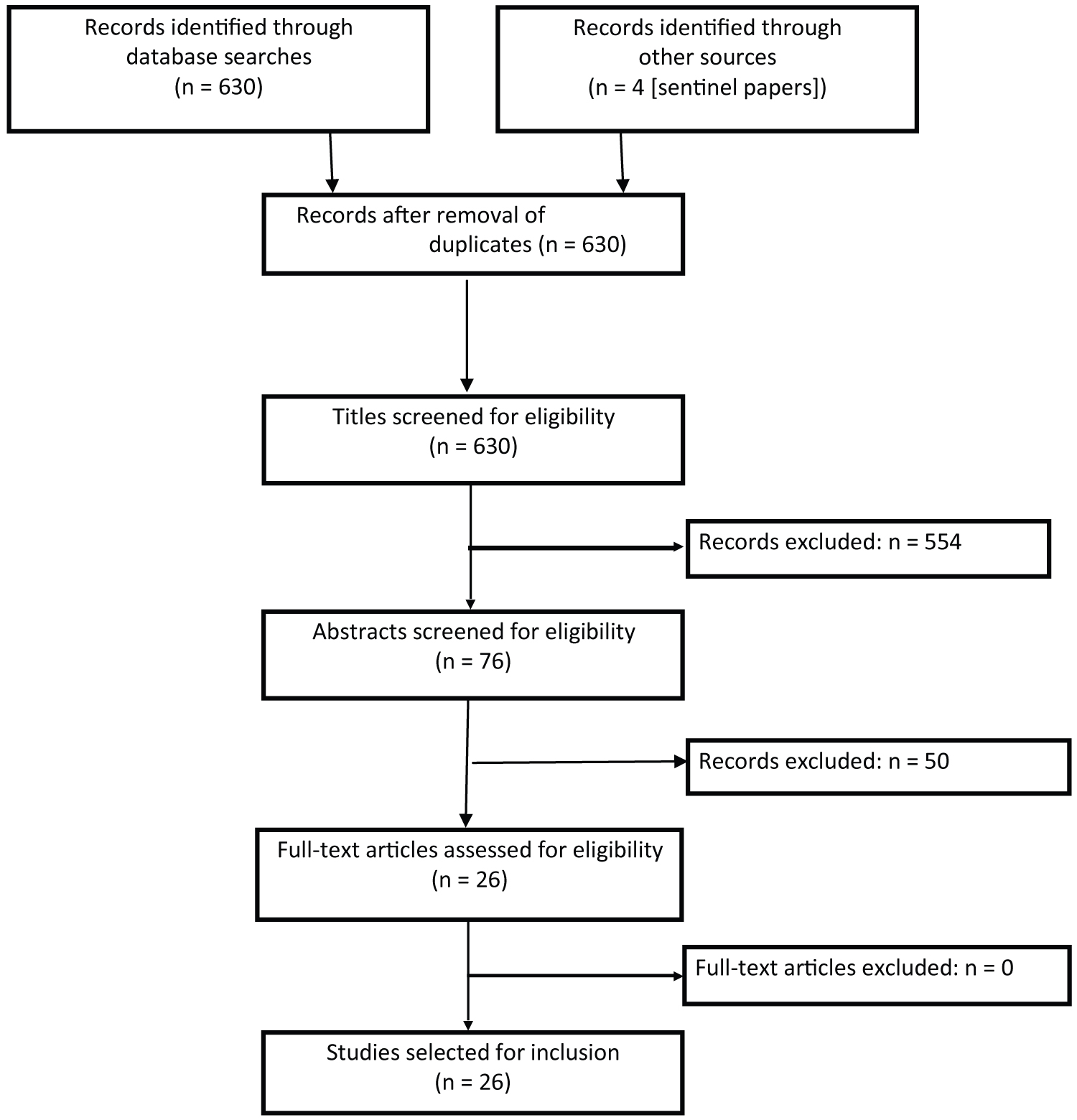 Figure 1: Literature search strategy.
View Figure 1
Figure 1: Literature search strategy.
View Figure 1
The titles were then reviewed for mention of gait or gait disorder, physical examination or gait performance and no mention of instrumentation (instrumented walkways, inertial measurement units, moving platforms, motion capture systems). A total of 76 articles met the criteria and their abstracts were retrieved. The settings and methods were checked for ecological appropriateness with regard to time investment in the ambulatory care environment. Twenty-six articles were found suitable and the present review is based on the information obtained from their perusal.
The unit of gait is the gait cycle or stride (Figure 2). It begins and ends with ground contact by the same foot as the body moves forward and is divided into two phases - swing and stance. The swing phase begins with toe-off and ends with heel-strike. It constitutes about 40% of the gait cycle and is further subdivided into initial swing, mid-swing and terminal swing. During the stance phase, body weight is transferred from the hind-foot to the forefoot. At the beginning and at the end, both feet are on the support surface and each of these double support periods makes up about 10-12% of the gait cycle [13]. The stance phase is subdivided into initial contact, loading response, mid-stance and pre-swing.
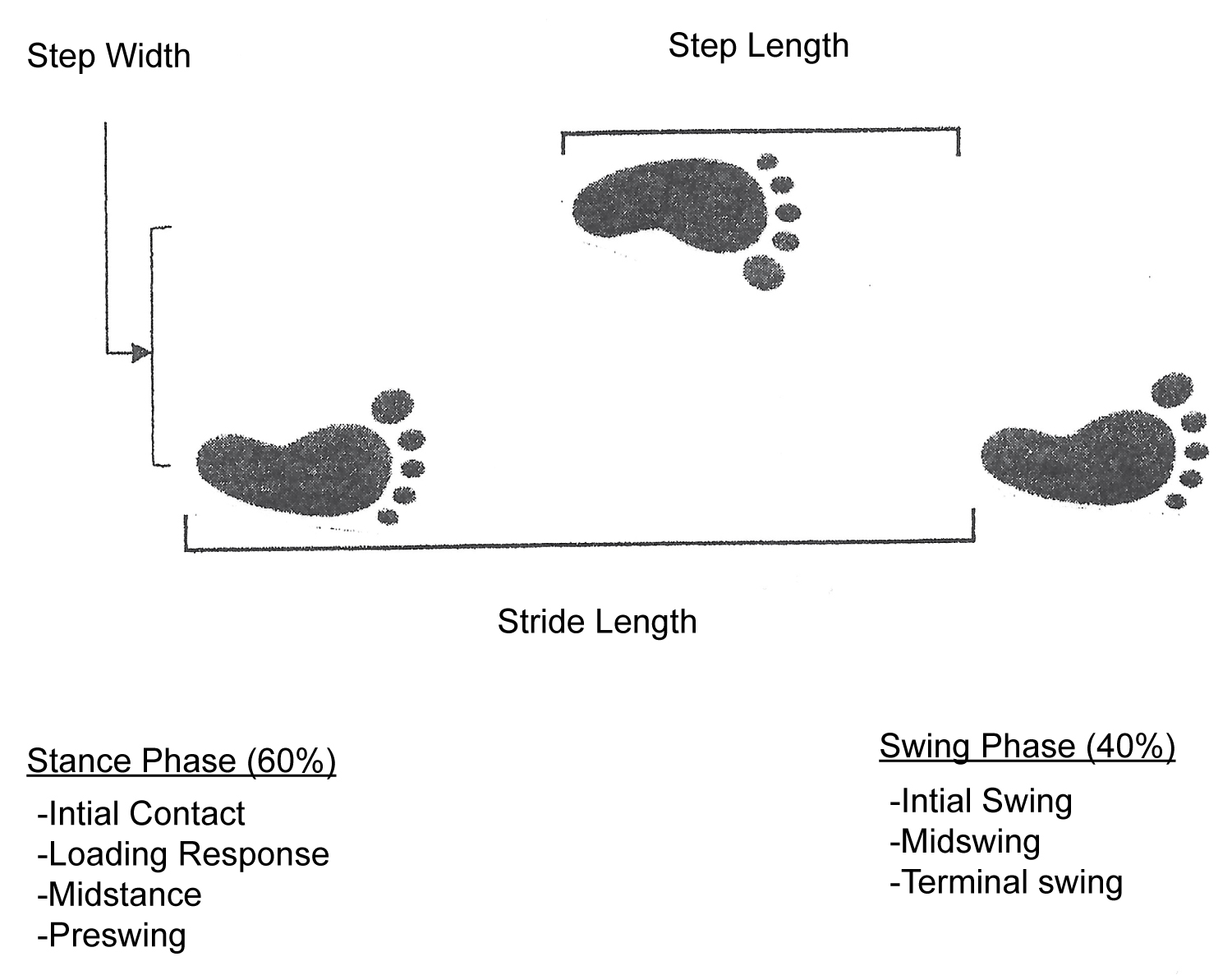 Figure 2: The gait cycle and basic gait measures.
View Figure 2
Figure 2: The gait cycle and basic gait measures.
View Figure 2
Important spatiotemporal gait measures, in the nomenclature of biomechanics, include stride length, step length, step width, gait speed and cadence. The stride length is the linear distance covered in one gait cycle. In healthy adults, it is 150-170 cm on average. Step length is measured from the point of contact of one foot to the next point of contact of the contralateral foot. It declines with advancing age. The step width is measured between the midpoints of both heels. It is about 40% greater in older adults, approximately 8 cm in women and 10 cm in men [14]. It is worth mentioning that neurologists commonly refer to step length as defined here, as stride length and step width as base.
Between ages 30-59 years, the preferred gait speed of healthy adults is about 1.43 m/s. Thereafter, it decreases by approximately 1% per year to about 0.95 m/s in the 9th decade [15]. Cadence is the number of steps taken per unit time. It is little affected by age.
Until about a quarter of a century ago, gait was considered to be a predominantly motor event of which gait speed alone was a satisfactorily representative metric [16]. Subsequent research has shown gait to be a highly complex skill requiring the integration of external sensory information within elaborate neural networks in the central nervous system. In one study [17], principal components analysis of gait kinematics disclosed five factors which explain about 80% of gait variance namely, pace, rhythm, variability, asymmetry and postural control. They define pace as the step velocity and rhythm as the step time. Variability is represented by the standard deviation of pace. Asymmetry describes the coordination between the limbs and is expressed as a swing time function while step length asymmetry is used to denote postural control.
Bipedal locomotion is a tenuous method of mobility because the upright body posture is constantly under threat. During the single support period of the stance phase of the gait cycle for instance, the line of force through the center of mass actually passes medial to the stance foot, creating a gravitational moment which could accelerate the center of mass toward the unsupported side. Thus, elaborate mechanisms involving multiple systems - nervous, peripheral sensory, musculoskeletal and cardiopulmonary - have evolved for the maintenance of stability and normal gait function.
The precise nature of the neural control of gait remains unclear but much has been learned in the past quarter century. Reconceptualization of the central nervous system as integrative neural networks rather than circumscribed topographical regions has contributed to this understanding in both a dynamic context and the overlap between gait and other functions such as cognition.
The basic cyclical pattern of human gait is believed to be created by coordinated groups of interneurons in the cervical and lumbar segments of the spinal cord called "central pattern generators" [18]. They regulate the activation of antigravity muscles and the alternate activation of agonist and antagonist muscle groups in the upper and lower extremities. Their activity is modulated by signals from supraspinal networks as well as afferents organs in the head (visual, vestibular) and the periphery (proprioceptive). The premotor and motor regions of the frontal lobe are involved in the initiation of gait behavior, in terms of its goal and purpose, as well as the adaptation of the locomotor pattern. Specific motor programs run via the basal ganglia and sensory feedback from the thalamus mediate postural control [19]. The interfastigial cerebellum has been implicated in pace regulation and the tegmentum of the midbrain also plays an important role in gait initiation.
Neuroimaging techniques including magnetic resonance imaging (MRI; regular and functional), MR spectroscopy and functional near-infra red spectroscopy (fNIRS), have been used in attempts to map various gait parameters to discrete areas of the brain [20-22]. In general, the "healthier" the brain in terms of grey matter volume and white matter integrity, the greater the quality of gait performance [23]. With regard to specific gait measures however, the results have been inconsistent across studies. Nevertheless, the most commonly evaluated metric, gait velocity, usually shows direct correlation with grey matter volume in the frontal lobe [24,25] and the hippocampus [26,27]. It is noteworthy that the cognitive domains of executive function and memory which are known to be associated with gait velocity [28,29], map respectively to the frontal lobe and the hippocampus.
It is often the case that an attention-demanding non-motor task and gait are performed simultaneously such as watching out for traffic while crossing a busy street. The safe and effective accomplishment of such concurrent tasks requires the efficient apportionment of cognitive resources. Accordingly, the defective management of these resources in states of cognitive impairment would be reflected in changes in the performances of the pertinent tasks. This has inspired the dual-task experimental paradigm and abundant evidence has accumulated showing that the interference in gait performance (dual task cost) is reflective of the degree of cognitive impairment [30].
The neurologic substrate at which gait and cognition converge is the central cholinergic system. Gait control depends on cholinergic inputs to cortical (from the nucleus basalis of Meynert) and subcortical (from the pedunculopontine nucleus) processing [31]. In numerous studies (reviewed [32]), a variety of techniques (physiological, pharmacological, lesions and genetic) have been used to establish cholinergic signaling in an important role in cognitive processes, especially attention and memory. In one evaluation of attentional performance in older adults with and without Parkinson disease, it was found that only cortical-cholinergic integrity (compared to thalamic-cholinergic and striatal-cholinergic) made a specific contribution to the ability to resist distraction from a highly salient but irrelevant environmental task [33].
The capacity to initiate and terminate gait is a requirement for the control of adaptive locomotion [34]. Gait is initiated by the inhibition of ankle plantarflexors, which causes the center of pressure to move posterior to the center of mass [35]. The center of pressure is also displaced laterally towards the limb about to swing [36]. These excursions of the center of pressure propel the center of mass forward as well as laterally toward the stance limb. Mirroring the mechanism for initiation, gait is terminated when the center of pressure is moved anterior to the center of mass so as to arrest its forward momentum. However, gait does not then stop instantaneously, but requires at least two steps: the first associated with increased extensor activity in the lead limb and the second by deceleration of the swing limb [37,38]. Therefore, in order to stop before colliding with an object while walking at comfortable speed, it is necessary to be able to anticipate events at least two steps into the future.
Dynamic stability is maintained by the use of predictive, reactive or proactive strategies [34]. Predictive control enables the body to anticipate and adjust to displacements of its center of mass as it moves. Belenkii (cited in [34]) showed that recruitment of postural muscles usually precedes that of prime movers. This point highlights the importance of these muscle groups with regard to fall prevention.
Reactive strategies are invoked after a perturbation has occurred. They involve reflexes modulated to provide phase- and task- dependent responses. In a study by Eng and colleagues [39], a tripping challenge early during the swing phase elicited a flexor response in the swing limb and an extensor response in the stance limb ("elevating strategy"). In contrast, perturbation during the late swing phase resulted in a different pattern of muscle activation (inhibition of the stance vastus lateralis and excitation of the biceps femoris) to prevent a potentially dangerous flexion of the swing limb ("lowering strategy") upon contact with the support surface. Both strategies ensure obstacle clearance. Pijnappels, et al. [40] further demonstrated in a similar protocol that older subjects, as compared to younger, showed a decreased magnitude and rate of development of plantar flexor activity. This relative impairment in plantar flexor function accounted for the falls the older subjects experienced during the research.
With proactive control, visual cues are obtained about a potential challenge before it is encountered, enabling a decision in advance regarding appropriate modifications to the locomotor pattern [41,42]. These modifications fall under two overlapping categories, namely avoidance and accommodation. Avoidance maneuvers include varying gait parameters in order to step over the obstacle, changing the direction of progression (steering), and stopping. Accommodation strategies take into account the physical properties of the support surface [42]. Some avoidance strategies can also be used for accommodation such as shortening step length while walking on icy or slippery terrain [43].
Accommodation and avoidance rely primarily on vision. Visual environmental sampling during gait need only be intermittent. For safe locomotion, a gaze pattern with less than 50% of travel time devoted to sampling has been shown to be sufficient, irrespective of terrain constraints on foot placement [44]. This ensures the availability of ample visual system resources for other purposes.
The vestibular and kinesthetic systems also play significant roles in the successful execution of proactive strategies. Vestibular input influences the direction of gait and persons with unilateral vestibular disease are known to veer to the affected side while walking [45]. A chance observation that a dog suffering from acute unilateral labyrinthitis deviated and staggered while walking [46] but maintained her chosen course while running, suggested that the effect of vestibular failure on locomotor control depended on gait velocity [47,48]. Brandt and colleagues replicated these findings in human beings and speculated that the inverse relationship between gait velocity and vestibular involvement was due to the suppression of vestibular sensory inflow at high gait speed by a spinal program through down-and-up channeling. Proactive strategies require accurate foot placement; however, a diffuse peripheral neuropathy has been found to increase foot placement variability in the frontal plane, particularly on an irregular surface [49]. This may explain the disproportionate decrease in gait speed such subjects demonstrate on irregular surfaces as compared to controls [50].
The frames of reference of the visual and kinesthetic systems predispose them to sensory error. The visual system is sensitive to relative motion between the body and the environment and so may confuse environmental movement with self-motion. Likewise, the kinesthetic system, which is referenced to the support surface, is apt to generate erroneous output when the surface moves. Nevertheless, all the systems collaborate to resolve any signaling conflict when one proves erroneous [51,52].
The motor control system operates on a feedback principle and its key drawback is sensitivity to time delays in the sensorimotor loop [53]. These delays occur during signal transduction and impulse conduction, central processing and in the motor output. They range between 30 ms for spinal reflexes and 300 ms for visually-guided responses and determine the stability of the system through their interaction with the controller function gain. Stable systems converge on a fixed value or oscillate in very close proximity to the reference input, whereas unstable systems diverge widely [54]. Time delays are an important reason why negative feedback control systems exhibit speed-accuracy trade-offs. Accordingly, older persons with clinical conditions that prolong feedback, such as peripheral neuropathy or dementia, demonstrate increased gait variability, particularly on irregular surfaces or under dual-task conditions [50,55].
A variety of schemes have been proposed for the classification of gait disorders. Each of them has its drawbacks as shown in Table 1. From a clinical standpoint, the phenomenological scheme would seem to be the most useful since it is based on features observable in the ambulatory care setting. The hierarchical model of Nutt and colleagues [56] includes a higher-level category of disorders which are due to impairments in the cortical control of gait. Although their phenotype is clinically recognizable, they do not share any dominant gait feature. In the hybrid model suggested by Snijders, et al. [57] the higher-level category has been retained in what is essentially a phenomenological classification. They however caution that further subdivision of that category is unwarranted in the current state of our knowledge. They discouraged the use of the term "frontal gait disorder" since its features are present in multiple unrelated phenotypes and so not clinically helpful. They did not include gait impairments that are secondary to disorders that are predominantly due to deficits in balance control such as the astasia-abasia syndrome of thalamic stroke.
Table 1: Gait classification schemes*. View Table 1
Since about 20% of older adults in the 9th decade walk normally, gait abnormality is not considered an inevitable accompaniment of normal aging. Nevertheless, many otherwise normal older adults walk with a so-called "senile gait" characterized by reduced push-off force, floor clearance and gait speed, increased step width and double-stance time, and a bent posture [58]. In these individuals however cross-sectional brain imaging has disclosed increased white matter hyperintensities. In addition, an increased risk of dementia and all-cause mortality has been documented [57,59]. As a result, "senile gait" is now considered to be subtle evidence of preclinical disease.
The key clinical features of specific gait syndromes are depicted and briefly described below in Table 2.
Table 2: Clinical gait syndromes. View Table 2
In this gait pattern, the stance time on the affected limb is kept as short as possible to reduce the load it bears. The patient walks with a limp as a result. Antalgic gait is seen in painful conditions of the lower extremities like osteoarthritis, sprains and stress fractures [13].
The upper trunk is inclined toward the affected side during ipsilateral stance (Duchenne sign). However, the pelvic abductors are intact and there is no declination of the unsupported contralateral hemipelvis. In this regard, coxalgia gait differs from waddling gait. Coxalgic gait is a feature of painful hip conditions.
Pain and weakness develop in the muscles of the lower extremities as well as paresthesias after walking a distance that is often characteristic for the individual. The symptoms resolve with rest, especially in a position of spinal flexion. Also, gait and related bipedal activity performed with the trunk flexed (e.g. going upstairs or walking uphill, pushing a shopping cart, riding a bicycle) are much less provocative. Vascular claudication differs from neurogenic claudication in that there are no sensory symptoms or postural influences. Neurogenic claudication is associated with lumbar spinal stenosis. Physical examination at rest may be entirely benign.
This is a paretic/hypotonic gait in which the leg needs to be lifted higher than usual for the toes of the foot to clear the gait surface during swing due to weakness of ankle dorsiflexors. The tip of the hallux may still scuff the gait surface and the swing phase may end with a slap of the entire sole on the gait surface ("foot drop") rather than heel-strike. The patients are incapable of heel-walk. The ankle dorsiflexors are supplied by the deep peroneal nerve, a branch of the common peroneal nerve which is relatively superficial at the neck of the fibula where it is very susceptible to compressive injury. However any lesions involving fibers from the fifth lumbar segment of the spinal cord (L5) will cause steppage gait with lower motor neuron features. Foot drop may also occur in cerebrovascular accident and systemic neuropathies but the clinical picture will be dominated by features other than gait deficit.
This gait disorder is characterized by a drop of the hemipelvis on the normal side during swing (Trendelenburg sign), usually accompanied by a compensatory lurch of the upper trunk to the affected stance side (Duchenne sign). It is due to weakness of the musculature of the pelvic girdle or upper thigh. The patients have difficulty with sit-to-stand transfer. Orthopedic conditions which result in closer approximation of the attachments of the gluteus medius muscle (e.g. post-traumatic elevation of the greater trochanter, pseudoarthrosis of the femoral neck) are also associated with a waddling gait. Waddling gait is common in myopathies, many of which affect proximal muscles preferentially.
Ataxia, by definition, is irregular movement. Ataxic gait syndromes are the result of the impaired coordination of the motor activities required for the maintenance of a selected path during gait. The patients may stagger from side to side (as in sensory and cerebellar ataxia) or veer to one side as in vestibular ataxia. In stance, they prefer to maintain a wide base because they feel insecure otherwise.
The gait in cerebellar ataxia is broad-based and wobbly. The degree of sway during narrow-based stance is not significantly different when observed with the patient's eyes open compared to eyes closed. Other corroborative cerebellar features (dysmetria, dysdiadochokinesia) might be present. The kinematic pathomechanism in cerebellar ataxia is stepping irregularity [60]. Among the causes of cerebellar disease in older adults are vascular events, toxic insult (alcohol use disorder), autoimmune processes and neoplasia.
The gait in sensory ataxia is slower than in cerebellar ataxia and may have a stomping quality since the feet tend to be lifted higher during swing. Patients are more symptomatic under conditions of suboptimal lighting and sway during narrow-based stance is markedly aggravated by eye closure (Romberg sign). Common causes of ataxic peripheral neuropathy in older adults are diabetes mellitus, cobalamin deficiency, toxic insult (chemotherapeutic exposure), paraproteinemia and autoimmune disease.
In vestibular ataxia, the gait deficit is also exacerbated by deprivation of visual input. However, walking fast or running attenuates the impairment (Brandt, et al. 2001). Taking 60 steps in place (at approximately 1 step/sec) with pronated outstretched upper extremities and eyes closed (Unterberger test) will result in an ipsilateral deviation of over 30 degrees. A smartphone application is available for correctly measuring the rotation in a clinic setting [61]. Other features of vestibular insufficiency (vertigo, nystagmus) may be present. There is also evidence of impaired proprioception which can be elicited by position sense testing as described by Richardson [62]. Hold is taken of the dominant hallux at its medial and lateral aspects with the thumb and index finger. It is then moved smoothly (up, down in random order) through an arc of about 1 cm over a period of about 1 second with the patient required to call out the direction of movement, first with eyes open and then with eyes closed. Proprioceptive acuity is impaired if more than 2 of 10 blind calls are incorrect.
The commonest vestibulopathy in old age is vestibular neuronitis. Less common disorders are Meniere disease, toxic exposure (e.g to amiodarone), neoplasia (vestibular schwannoma). These patients are more likely to present with "dizziness" and other symptoms than with a gait deficit per se. It is noteworthy that cerebellar stroke can mimic peripheral vestibular disease. The HINTS-plus battery [63] enables a distinction to be made between central (e.g. stroke) and peripheral (e.g. vestibular neuronitis) causes of acute vestibular syndrome. It will be discussed further in a subsequent section.
This is a stiff-legged gait due to enhanced anti-gravity muscle tone. The thigh adductors are in spasm and if unilateral, circumduction occurs during swing of the paretic limb. There is increased plantar flexion of the normal stance limb (vaulting) to facilitate clearance of the gait surface by the contralateral foot. In spastic paraparesis, the successive circumduction of both lower extremities during swing has the appearance of scissoring. The wear pattern of the soles of the patient's shoes is usually informative.
Strokes and demyelinating diseases are commonly associated with spastic gait. The myelopathic gait of cervical spondylosis is spastic. The osteophytes and hypertrophic ligaments can cause compression of the spinal cord with motor and sensory consequences [8].
Dystonia is a heterogeneous syndrome characterized by involuntary abnormal postures or movements resulting from intermittent or sustained muscle contractions. Dystonic gaits may be due to focal, segmental or generalized disorders and are often bizarre. They are also task-specific and may manifest during usual gait but not when walking backwards, for instance. Sometimes, sensory tricks, like placing a hand on the back of the neck, could improve or normalize a dystonic gait [2]. The most common focal dystonia involves ankle inversion and extension of the hallux. In some patients with Parkinson disease, dystonic thoracolumbar flexion (camptocormia) occurs when they stand up to walk. Such posturing is absent when the patient is sitting or supine. Damage to the basal ganglia, hypoxic or toxic, can cause dystonic gait. "Cock gait", characterized by strutting on the toes with flexed upper extremities and extended trunk, is described in manganese deposition in the basal ganglia. Levodopa and dopamine receptor blocking drugs are associated with dystonia.
Dyskinesias are characterized by excessive abnormal movements which are superimposed on gait which may or may not be abnormal. In either case, the gait pattern is altered by the adventitious movements.
In myoclonic gait, the additional movements may be involuntary jerks causing movement at a joint (positive myoclonus) or repetitive sudden loss of muscle tone (negative myoclonus). The occurrence of the latter in the proximal limb musculature of the lower extremities can result in a "bouncy gait". The common causes of myoclonic gait are cerebral ischemia or hypoxia. It is also a rare manifestation of refractory celiac disease [64].
Choreatic gait has an irregular, swaying or writhing, dance-like quality. Stepping and direction are very variable. To compensate, the step width is broad and speed is slow. Drug treatment-related chorea is common in Parkinson disease. Hypoxic damage to the basal ganglia as might occur during cardiopulmonary bypass (so-called "post-pump" chorea [2]) is another cause.
Cautious gait is considered to be an excessive degree of the so-called idiopathic senile gait seen in many otherwise healthy older adults [58]. Posture is slightly stooped, speed is slow, gait base is wide and arm swing is reduced. It is usually observed after the individual has fallen for the first time and if untreated, can progress to a disabling phobic gait disorder in which extreme fear of falling might completely inhibit walking activity. Cautious gait improves strikingly with relatively little external support and responds well to rehabilitative therapies.
A careless gait is one executed without appropriate cognizance of one's physical and other limitations in relation to the challenges in the gait environment. On irregular or slippery terrain for instance, it might be too fast when a slower speed would have been better suited to the circumstance. Careless gait may be due to lack of insight as in delirium and dementia as well as disinhibition in frontal lobe disorders.
Psychogenic or functional gait disorder used to be considered as a diagnosis of exclusion but more recently, clinical inconsistency has come to be accepted as its hallmark [2]. The patient may present with any other kind of gait disorder but frequently it is bizarre, with exaggerated effort ("huffing and puffing" sign [65]) and an uneconomic posture. Clinical findings may not be reproducible if the patient is re-examined when distracted or unaware. Okun et al. [66] have suggested that a "chair test" first used in 19th century is useful for demonstrating the ambulatory incongruity in psychogenic gait. Although the patients show impaired erect walking, they have no difficulty propelling a swivel chair on castors with their feet. Falls are rare and are usually non-injurious when they happen. Secondary gain may be involved.
The characteristics of this category of gait disorders are paucity of movement and increased tone in the extremities. The classic example is Parkinson disease which is due to neurodegeneration in the substantia nigra. The gait is slow and shuffling. In advanced disease however, festination i.e. the tendency to accelerate with gait progression, can occur. Step width is narrow and arm swing is both reduced and asymmetrical. The reduction in arm swing can precede other features by many years. In some of the atypical neurodegenerative Parkinsonism syndromes (Lewy body dementia, progressive supra nuclear palsy [PSP] and multiple system atrophy [MSA]), arm swing is more symmetric.
An unsteady gait with a broad step width, dementia and urinary incontinence are the classic triad of clinical features of normal-pressure hydrocephalus (NPH). Arm swing is largely preserved but freezing of gait occurs regularly. It is worth mentioning that many neurologists consider the idiopathic variant of NPH a controversial, if dubious, concept.
Freezing of gait is a brief episode of sudden arrest of locomotion when the feet appear glued to the gait surface. There may be an ineffectual effort at stepping with weigh shifting in the frontal plane but no forward engagement ("slipping clutch" effect). Freezing may occur at gait initiation ("start hesitation"), during turns or when an obstacle or door threshold is encountered. It may be overridden by sensory, especially visual, cues although cues may paradoxically precipitate freezing occasionally. Freezing of gait is common in moderate and severe Parkinson disease. At earlier stages of the disease, it is linked to periods of loss of response to Levodopa ("off" phases). Hypokinetic-rigid gait disorders with a vascular etiology usually have a preponderance of lower extremity features with minimal upper extremity involvement ("lower body parkinsonism"). Arm swing is intact and there are hardly any hand tremors.
Trunk alignment is often altered especially in the neurodegenerative disorders. A forward stoop (camptocormia) is common in Parkinson disease while a lateral lean (Pisa syndrome) is seen in MSA. Gait apraxia suggests frontal lobe involvement as in corticobasal degeneration. In these patients, gait may be severely impaired but the legs remain able to perform other motor activities reasonably well, like making cycling movements when non-weightbearing.
Many conditions present with hypokinetic-rigid gait disorder and differential diagnosis often relies substantially on non-gait clinical features and neuroimaging.
Patients with higher-level gait disorders often give the impression of having forgotten how to walk. When asked to initiate gait, they may struggle to assume a stable upright posture. They may sway and make exaggerated upper extremity movements. Gait is slow and shuffling, with very effortful initial swing as if the feet are stuck ("magnetic" gait). Even more variability in the stepping pattern is introduced by the greater susceptibility to emotional and external environmental influences. Inappropriate synergies like cross-stepping may occur and freezing is common. As in hypokinetic-rigid syndromes with frontal lobe involvement, gait apraxia may be present. Higher-level gait impairments are usually unimproved by use of assistive mobility devices. Neuroimaging in disease entities manifesting with gait disorders in this category show lesions in the frontal and parietal regions of the forebrain [67], interhemispheric communication [68] and the network linking them to the midbrain locomotor region [69].
The list of medications associated with gait impairment is long (Table 3). A variety of gait types is seen and the pathomechanisms are often complex. Ataxia is common and often due to oversedation (benzodiazepines, opiate analgesics, non-selective antihistamines, anticholinergics). Dopamine replacement therapy in Parkinson disease can cause "dizziness" in the short term but after prolonged use, dyskinesia may result. Neuroleptics are strongly linked to the development of tardive dyskinesia.
Table 3: Medications associated with gait impairment [57]. View Table 3
The evaluation of the patient with a gait disorder in the clinic setting is outlined in Table 4. It begins with the standard practice of history-taking and physical examination. The systems closely scrutinized during physical examination are suggested by the history obtained. This is followed by specific gait assessment which is implemented in the hallway. It has both qualitative and quantitative components. The choice made regarding laboratory testing and imaging for further refinement of diagnosis, if necessary, will depend on the findings.
Table 4: Approach to the Patient with a Gait Disorder. View Table 4
Patients with a gait disorder are likely to present with pain during walking and/or reduced ambulatory range. Other common symptoms are stiffness and numbness [70]. Their family members or caregivers may complain that they walk more slowly or fail to "pick up" their feet during gait. A sudden onset may indicate a vascular event while a slowly evolving course is more consistent with a degenerative process. Information about circumstances that affect symptom severity - terrain conditions (outdoors/irregular vs. regular), ambient lighting, multitasking - is important. A need for haptic enhancement (e.g. "cruising" furniture, walls or other fixtures) suggests a proprioceptive or dorsal column deficit.
Fall history is very significant. In some conditions like PSP, it is often the falls that bring the patient to clinical attention. They are early and frequent. If the falls are tonic ("like a log"), then protective reflexes are very defective as in higher-order and hypokinetic-rigid gait disorders [71].
The majority of gait disorders are connected with underlying disease. A feature of advanced age is multimorbidity and some of the diseases in patients' medical history (neurologic, musculoskeletal, endocrinologic) may be relevant to their gait concerns.
A review of the patient's medications is important. The etiologic potential of pharmacotherapeutic agents viz-a-viz gait disorders has been discussed. Polypharmacy per se is not a risk factor unless medications that have a known individual impact on gait and balance are included [72].
A family history of movement disorders and cognitive impairment adds perspective.
Socio-functionally, the prevalence of gait disorders among nursing home residents is much higher than among their community-dwelling counterparts (about 50% vs. about 30%; [9]). The usual gait of urban dwellers is significantly faster than that of rural dwellers [73]. In the English Longitudinal Study of Aging, a slower gait speed and greater decline in gait speed over a period of 4 years were associated with lower total wealth in adults aged ≥ 60 years (richest quintile 0.91 m/s to 0.82 m/s; poorest quintile 0.75 m/s to 0.71 m/s; [74]). Patients who successfully ameliorate their impairment by use of an assistive device such as cane or walker, are unlikely to have significant higher-order disorder.
Systems are reviewed for collateral non-gait symptoms that might increase the probability of particular gait diagnoses. Vertigo may be present in vestibular ataxia. A history of olfactory dysfunction is a modestly useful feature of Parkinson disease diagnostically. Patients with NPH usually have urinary incontinence. It is also common in Parkinson disease and almost universal in MSA. Questions exploring cognitive function (memory, attention, orientation and language) should be posed.
At the beginning of the physical examination, note is taken of the patient's posture, facies and adventitious movements, if present. Blood pressure and pulse are checked, with the patient sitting, then standing, to assess orthostatic tolerance if there is suspicion of dysautonomia. The general regional and systemic evaluations proceed along the same lines as described for falls elsewhere [75]. It includes the assessment of cognition using a standardized instrument, preferably one that captures executive function well (e.g. Montreal Cognitive Assessment [76], Executive Clock Drawing [77], Mini-Addenbrooke's Cognitive Examination [78]). There is also considerable overlap between gait and balance assessment. Hence, depending on the presentation, it may be necessary to perform vestibular tests and these have been extensively reviewed in the balance context [79]. The Unterberger test has already been described.
In patients presenting with acute vestibular syndrome (new-onset gait unsteadiness, persistent vertiginous or other dizziness, nystagmus, head motion intolerance, nausea or vomiting), a four-component battery, the so-called HINTS-plus (head impulse, nystagmus, test of skew and new hearing loss) has excellent operating characteristics for stroke diagnosis (99.2% sensitivity, 97.0% specificity; [63]). However, special examiner skills not commonly possessed by geriatricians and other physicians are required. Alternatively, a video-oculographic device can be used but it too is not commonly available and so HINTS-plus fails to meet the criteria of inclusion in the diagnostic toolbox of the regular ambulatory care setting. It is mentioned here only for completeness.
The specific gait assessment is conducted in a hall way 10-20 meters long with adjustable lighting. The qualitative component evaluates gait in three modes - usual, challenged and assisted. The patient is observed as s/he transfers out a standard armchair with arms folded across the chest (to assess proximal muscle strength) and initiate gait in well-lit ambience. Note is made of the stride in terms of the rhythm of stance and swing, floor clearance, step length, gait trajectory, arm swing, upper body alignment, adventitious movements and character of the turn (segmental vs. en bloc).
Gait is then challenged by having the patient walk in subdued lighting, especially if a vestibular source is suspected. It can also be stressed by having her perform a concurrent cognitive non-gait task like counting backwards from 50 or reciting the months of the year backwards. Interference might occur in such dual task scenarios with resultant performance cost to either or both tasks which can be quantified as explained in the next section. The patient should be spotted during challenged gait testing.
The patient is next asked to use a cane, walker or swivel chair during gait and observed for any improvement or otherwise to impairments noted in unassisted gait. Gait performance in higher-level disorders is unaffected by use of mobility assistive devices and may even deteriorate due to the inability of the patient to use them properly. In the chair test already described, a patient with psychogenic gait disorder should be able to make effective use of his lower extremities.
The quantitative components of the specific gait evaluation are two timed tests and a scalar test. The dynamic gait index [80] and its functional gait assessment derivative [81] might have been suitable but for their stair-climbing component. The regular office environment might not easily accommodate a modular staircase. Self-selected comfortable gait speed is measured over a distance of 10 meters ideally [4] but about half that length would suffice, with half the timed distance on either side for acceleration and deceleration respectively. Tables of normative values reflecting variation by age, gender and anthropometrics are available [82]. They show that the mean comfortable gait speed of healthy adults aged 20-79 years varies from 1.27 m/s for women in their seventies to 1.46 m/s for men in their forties. A gait speed under 0.6 m/s is consistent with dismobility characterized by dependence in basic and instrumental activities of daily living [11]. Measurement of gait speed under dual task conditions as already described will enable quantification of the performance cost of the distraction: 100 (single task performance - dual task performance)/single task performance.
The dual task cost is a marker of attention and executive function efficiency as well as fall risk [83].
The timed up-and-go (TUG) test is a global assessment of functional mobility [79,84]. The seated patient is timed as she gets up from a standard armchair, walks 3 meters as quickly (but safely) as possible, turns and returns to her chair and sits down. A cutoff point of 13.5 sec discriminates between fallers and non-fallers with a sensitivity and specificity of 87%.
The ordinal scale of choice is the Performance-Oriented Mobility Assessment (POMA; [85]). It consists of two sub scales, gait and balance. The former has 8 items, all but two of which are scored on a binary scale. Gait path and trunk sway are scored on a 3-point scale. The nine items on the balance subscale evaluate performance of maneuvers involved in activities of daily living. Scores in both subscales are interpreted in aggregate: ≤ 18 indicative of high fall risk and ≥ 25, of low fall risk. The POMA takes only about ten minutes to complete and so can fit into most clinic schedules.
The approach described above is ecologically appropriate to an ambulatory care setting and will greatly facilitate the identification of the clinical gait syndrome involved. The findings will also suggest what further testing might be necessary to elucidate the diagnosis in biochemical or anatomical terms. Peripheral neuropathy in a non-diabetic for example, should prompt the measurement of serum cyanocobalamin and methylmalonate levels and protein electrophoresis. Elevated levels of systemic inflammatory biomarkers have been associated with slow gait speed (Interleukin-6 in the Einstein Aging Study [86]; C-reactive protein in the International Mobility Aging Study [87]).
Basic radiological studies are warranted in antalgic, coxalgic, neurogenic claudicatory, myelopathic and waddling gait syndromes. Useful information can be obtained using electroneuromyography in cases of steppage and myopathic waddling gait syndromes.
Suspicion of a higher-level disorder should prompt structural magnetic resonance imaging and referral to a movement disorders neurology service for further evaluation and co-management. With the recognition of gait as a complex function dependent on significant cognitive engagement for its effective performance, reference is consistently made to it in the context of the advanced functional and other neuroimaging techniques (fluorodeoxyglucose-positron emission tomography, diffusion tensor imaging) used to study cognition. The findings correlate slow gait speed with beta amyloid burden, white matter hyperintensities, lacunar infarcts, regional changes in cortical volume and rates of glucose metabolism [88]. However, these technologies remain in the realm of research and have limited if any clinical applications at the present time.
Since the turn of the century, the use of body-worn inertial sensors to assess gait (so-called inertialocography) has grown tremendously [89,90]. The methodology however remains plagued by numerous problems among which are the wide variety of protocols, absence of normative values and the basic fact that the local coordinates of the devices are not aligned with any physiologically meaningful axis. They collect a lot of data but it is unproven that the extra information adds significant value to careful clinical observation [57].
There is rarely a need to refer patients to a gait laboratory where optoelectronic camera systems and instrumented platforms are used to capture kinematic, kinetic and goniometric data. Occasionally however, such data may be required for surgical treatment planning or the customization of special segmental orthoses.
All activities of daily living require mobility, segmental or whole-body. As such, mobility is a key requirement for independence and, of all mobility tasks, gait is the most fundamental [91]. Its apparent simplicity is deceptive because in reality, walking is a complex motor behavior requiring elaborate active control for its safe and effective execution.
All activities of daily living require mobility, segmental or whole-body. As such, mobility is a key requirement for independence and, of all mobility tasks, gait is the most fundamental [91]. Its apparent simplicity is deceptive because in reality, walking is a complex motor behavior requiring elaborate active control for its safe and effective execution.
In the present article, the physiology and pathophysiology of gait have been reviewed. A clinically useful phenomenological characterization of gait abnormalities was provided and a compact high-yield approach to the correct diagnosis of these gait syndromes in the ambulatory care setting has been laid out. It is ecologically sound, not requiring any piece of equipment not available in the regular physician office. The findings on history and physical examination guide further work-up. Ancillary testing and interventions were not covered.
Wearable inertial measurement units became available about two decades ago. They enable continuous monitoring but are not standardized, among other problems. A role has yet to be found for them in the clinic setting. For now, careful history-taking and physical examination remain the linchpin of gait assessment in the ambulatory care environment.
We are grateful to Drs. Roger L. Albin, Department of Neurology, University of Michigan Health System and James Ashton-Miller, Department of Biomechanics and Mechanical Engineering, University of Michigan, for their very helpful comments on the manuscript. Our thanks are due also to Ms. Carol Shannon, Taubman Health Sciences Library, University of Michigan, for assistance with the literature search.
Conceptualization: JON; Methodology: JON, INO; Validation: JON, INO; Formal analysis: INO; Data Curation: INO, CVO; Writing - Original Draft Preparation: JON; Writing - Review & Editing: CVN, INO; Supervision: JON.
The authors declare no conflict of interest.