Infection to Helicobacter pylori has been associated to many gastrointestinal diseases including gastritis, peptic ulcer disease, gastroesophageal reflux disease, atrophic gastritis and gastric cancer. Chronic infections are frequent and severe in patients with diabetes mellitus (DMT2), probably due to the impairment of their immune status. The link between H. pylori infection and diabetes mellitus (DM) remains controversial. This study aimed at detection and comparison of anti H. pylori antibodies (IgG) in serum of diabetes mellitus type 2 (DMT2) and non-diabetic dyspepsia subjects and also to find if there exists any significant correlation between H. pylori infection and DMT2.
This case control study of 82 patients (51 diabetics and 31 non-diabetic subjects) was carried out in Yaounde Cameroon during the period January-April 2017. Clinical and sociodemographic information of both groups were recorded 5 ml of blood was aseptically collected for H. pylori IgG antibodies. Assay parameters were analysed using a software application GastroSoft (www.GastroPanel.com). Data was analysed using Epi info 7.0. All statistics were realized at 95% CI. Authorizations were obtained at the Yaounde Central Hospital, the Cite Verte District Hospital. Ethical clearance was also obtained from the National Ethics Committee.
Significantly raised Anti H. pylori antibodies (IgG) were found in diabetics (88.2%) than in the non-diabetic control group (67.7%), (P = 0.015) showing strong correlation between the association of H. pylori and DMT2. Dyspepsia was very common in anti H. pylori positive cases (83.6%). The most common diabetic complications observed in H. pylori positive diabetic subjects were retinopathy (81.0%), neuropathy (33.3%), diabetic foot (19.0%) and nephropathy (9.5%). The major diabetic risk factors in H. pylori positive subjects were obesity (39.5%), overweight (31.6%) and hypertension (31.7%). Significantly raised anti H. pylori antibodies were observed in almost all age groups in the diabetic groups.
The result showed that diabetics are more prone to H. pylori infection and need continuous monitoring.
Helicobacter pylori, Diabetes mellitus, Biomarker, Case control
Infection to spiral rod-shaped gram-negative bacterium, Helicobacter pylori (H. pylori) is associated to many gastrointestinal diseases including gastritis, peptic ulcer disease, gastroesophageal reflux disease, atrophic gastritis and gastric cancer [1,2]. H. pylori infection has been estimated in up to half of the world's population and mainly acquired in childhood by the feco-oral, oro-oral or gastro-oral routes [3]. H. pylori infection has also been implicated in other non-gastrointestinal diseases like diabetes mellitus, ischemic heart disease, neurological disorder, hypertension, thyroiditis, dermatological, rheumatological and cerebrovascular disease [4]. Diabetes mellitus (DM) is a common, chronic and progressive disease characterized by chronic hyperglycemia with disturbances in carbohydrate, fat and protein metabolism resulting from either absolute or a relative deficiency of insulin and/or action [5]. The chronic hyperglycemia in DM patients is associated with long-term damage, dysfunction, and failure of various organs including the eyes, kidneys, nerves, heart as well as blood vessels. H. pylori has been implicated on cardiovascular conditions, insulin resistance, and metabolic syndrome potentially mediated by elevations in inflammatory markers such as C-reactive protein (CRP) and Interlukin-6 (IL-6) [6]. Several studies all over the world have reported high H. pylori prevalence amongst DM patients although the link between H. pylori infection and diabetes remains controversial. Impairment of cellular and humoral immunity, reduction of gastrointestinal motility and acid secretion, altered glucose metabolism and increased susceptibility of pathogens as they regularly attend hospital settings have been implicated in increased susceptibility to infection in diabetic patient [1]. H. pylori IgG antibodies and pepsinogen II are well known markers for Helicobacter pylori infection and gastric inflammation respectively. Thus, the aim of this study was to evaluate the prevalence of H. pylori infection in DMT2 patients in Yaounde and also to find if there exists any significant correlation between H. pylori infection and DMT2.
A total of 51 type 2 diabetic patients undergoing follow up at Yaounde Central Hospital, the Cite Verte District Hospital and the Etouge-Ebe Baptist Hospital were included during the three-month period January-April 2017. The control group consisted of 31 healthy students and health personnel with no history of diabetes and hypertension, none of the controls included was on antimicrobial agents, H2 receptor antagonist or proton-pump inhibitor for at least last 4 weeks. Age, sex, height, weight, and body mass index of all the participants were recorded. History of dyspepsia was recorded. The diabetic subjects were also enquired for intake of non-steroidal anti-inflammatory drugs, history of treatment of H. pylori infection, use of proton pump inhibitors, H2 receptor antagonist and antacids. 5 ml of venous blood was aseptically collected in dry tubes, which was kept undisturbed for 1 hour for clot formation. The serum was transferred into sterile cryotubes and stored at -20 ℃, for estimation of specific IgG anti H. pylori antibodies and pepsinogen II levels using the HPIgG Elisa and PGII Elisa respectively of the GastroPanel (Biohit plc Finland, www.biohithealthcare.com). All patients with H. pylori IgG ≥ 30 EIU and PGII > 15 μg/l were considered positive for active Helicobacter pylori infection. All the diabetic subjects in study group were observed for various diabetes mellitus related complications like diabetes retinopathy, nephropathy, neuropathy, and diabetic foot. Assay parameters were analysed using a software application GastroSoft (www.GastroPanel.com). Data was interpreted using Epi Info software 7.0. Chi-square test was applied to test the difference between two proportions; student (t) test was applied to test the difference between two means; Fisher (F) test was applied to test the difference of more than two means and P value less than 0.05 was regarded as significant (P < 0.05). Ethical clearance was obtained from the National Ethics committee. All patients signed an informed consent form.
In total, 82 subjects were recruited aged 19-77 yrs, mean 50.0 ± 13.7 yrs. 52 (63.4%) females (aged 19-77, mean 48.7 ± 14.7 yrs) and 30 (36.6%) males aged 29-72, mean 52.3 ± 11.6 yrs. The study included 51 (62.2%) diabetic patients aged 19-72 years (mean age 55.9 ± 9.8 yrs) and 31 (37.2%) healthy controls aged 32-77 years (mean 40.3 ± 13.8 years). The Female/Male Sex ratio was 35:16 in diabetics and 17:14 in control group.
Anti H. pylori antibodies (IgG ≥ 30 EIU) were detected in 45/51 (88.2%) diabetics and in 21/31 (67.7%) in control group. Higher seroprevalence of anti H. pylori antibodies were noticed in age groups of the diabetics than the control groups (Table 1). Anti H. pylori antibodies positivity rate was 100.0%, 66.7%; 86.7% and 100.0% in diabetics group and 83.3%, 55.6%, 75.0% and 0.0% in control group in age group 30-40 & 51-61, 52-62 and 63-73 years respectively. There was statistical significant difference in the mean IgG levels in the diabetic (P = 0.001) than controls (p = 0.09). H. pylori IgG seropositivity was associated with females (86.5%) than males (70%), and especially in the control cases (X2 = 3.7, p = 0.04).
Table 1: Patient information. View Table 1
H. pylori seropositivity (IgG ≥ 30 EIU) was higher in participants presenting with dyspepsia complaints (86.4%) than non-dyspepsia subjects (71.4%) Table 1. However, H. pylori prevalence was not associated with dyspepsia in both the diabetic and control groups (p > 0.05). Various risk factors and complications were observed in both the groups of diabetic patients (seropositive, seronegative). The common factors and complications observed in H. pylori antibody positive diabetic patients were retinopathy (81.0%) followed by obesity (39.5%), neuropathy (33.3%), hypertension (31.7%), overweight (31.6%), diabetic foot (19.0%), nephropathy (9.5%). Statistically significant negative association was observed in neuropathy complication between the seropositive and seronegative diabetic group (P = 0.009) (Table 1).
Significantly raised pepsinogen II levels (PGII > 15 μg/l) were observed in all aged groups of the diabetics (F = 3.1; p = 0.02) than in the non-diabetic group (F = 2.2; p = 0.09). Pepsinogen II levels did not differ statistically between dyspepsia (24.6 ± 16.4 µg/l) and non-dyspepsia subjects (19.9 ± 9.7 µg/l), (t = 1.0; p = 0.31). Raised Pepsinogen II levels were observed in the diabetics presenting with dyspepsia (29.0 ± 17.0 µg/l) than non-diabetics dyspepsia subjects (14.0 ± 8.1 µg/l). No significant difference in the mean Pepsinogen II levels was observed amongst the diabetic complications: Diabetic foot (t = 1.3; p = 0.18); neuropathy (t = 1.43; p = 0.16); nephropathy (t = 1.1; p = 0.27); retinopathy (t = 0.8; p = 0.43). Significantly raised pepsinogen II levels were observed in H. pylori positive (25.2 ± 15.2 μg/l) than negative subjects (15.5 ± 17.2 μg/l), t = 2.1; p = 0.03. A strong positive correlation was obtained between IgG and PGII levels r = 0.98, p = 0.001, Figure 1.
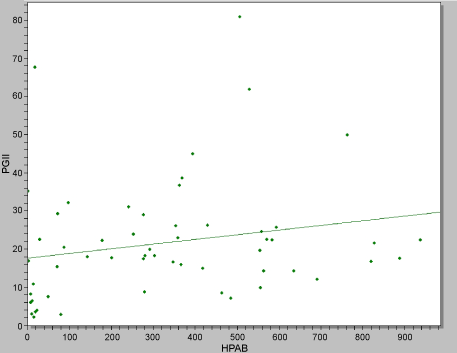 Figure 1: Correlation between HP IgG antibodies and Pepsinogen II levels. View Figure 1
Figure 1: Correlation between HP IgG antibodies and Pepsinogen II levels. View Figure 1
In this study, the prevalence of H. pylori seropositivity was 66/82 (80.5%). This prevalence is similar with what had been reported earlier amongst dyspeptic patients in Cameroon by Ebule, et al. [7] (78.7%) and 79.80% respectively in 2017 and 2013 and by Noah, et al. [8] (81.40%) and in other countries including Benberin, et al. [3] (76.5%). Highest H. pylori antibody seropositivity was observed in subjects above 70 years. There was however no association of H. pylori positivity with age in both the diabetic (p = 0.32) and control group (p = 0.18). Infection to H. pylori has been associated to age [9,10]. Most of the H. pylori related diseases are associated with male gender [9,11]. However, we observed an association of H. pylori seropositivity with females 45/52 (86.5%) than males 21/30 (70%), X2 = 3.3; p = 0.04). Similar observations have been reported by Kanbay, et al. [12] and Ali and Sarkis [1].
Patients with diabetes mellitus are often affected by chronic infections [1]. Many studies have evaluated the prevalence of H. pylori infection in diabetic patients and the possible role of this condition in their metabolic control. The present study found that diabetic patients are more prone to acquire H. pylori infection (88.2%) than the control group (67.7%, p = 0.001) (statistically significant). Jeon, et al. [13], in a prospective cohort showed that infection of H. pylori leads to an increased incidence of diabetes. Sherwal, et al. [4] and Taher, et al. [14], also observed higher prevalence of H. pylori in diabetic dyspeptic patients than in non-diabetic. Many other authors across the world including Ali and Sarkis [1], Kimiaki, et al. [15], Oldenburg, et al. [9] Quadri, et al. [16] Gulcelik, et al. [17] have also observed statistically significant association of H. pylori with diabetes mellitus. Several reasons have been advanced as to the increased susceptibility to infection in diabetic patients. These include, diabetes-induced impairment of cellular and humeral immunity, diabetes-induced reduction of gastrointestinal motility and acid secretion may promote pathogen colonization and infection rate in the gut, altered glucose metabolism may produce chemical changes in the gastric mucosa that promote H. pylori colonization and frequently exposed to pathogens by diabetics than their healthy counterparts as they regularly attend hospital settings [1,4].
The common manifestations and complications observed in H. pylori antibody positive diabetic patients were dyspepsia (86.4%), retinopathy (81.0%), obessity (39.5%), neuropathy (33.3%), hypertension (31.7%), overweight (31.6%), diabetic foot (19.0%), nephropathy (9.5%). These manifestations and complications were also observed among H. pylori seronegative diabetic patients. Gasbarrini, et al. [18] and Sherwal, et al. [4] had also reported similar results in a group diabetic patients and healthy controls.
Significantly raised pepsinogen II levels were observed in H. pylori positive (25.2 ± 15.2 µg/l) t = 2.1; p = 0.03 and a strong positive correlation was obtained between IgG and PGII levels r = 0.98, p = 0.001. Pepsinogen II levels are known to be strongly associated to Helicobacter pylori antibodies [19,20] (www.biohithealthcare.com). Raised pepsinogen II levels (PGII > 15 μg/l) were observed in all aged groups of the diabetics (F = 3.1; p = 0.02) than in the non-diabetic group (F = 2.2; p = 0.09) and in the diabetics presenting with dyspepsia (29.0 ± 17.0 μg/l) than non-diabetics dyspepsia subjects (14.0 ± 8.1 μg/l). This difference is as a result the increased susceptibility of H. pylori infection in diabetic patients. Pepsinogen II is a known marker of gastric inflammation due to Helicobacter pylori and prolonged use of anti-inflammatory drugs [2,20] (www.biohithealthcare.com).
The study indicates that diabetics are more prone to H. pylori infection and the growing need for continuous monitoring of H. pylori infection in diabetic patients. However more extensive well-designed cohort studies are needed to definitely conclude the relationship of H. pylori infection with diabetes mellitus and its complications.