HIV remains a major global public health threat with one million HIV-related deaths, two million new infections and over 1 million HIV-associated TB cases each year. However, population-based studies suggest marked declines in incidence, prevalence and deaths in the countries in East and Southern Africa that have expanded antiretroviral treatment (ART) at the fastest rate in the world. Previous research has indicated that most of the positive epidemiologic developments are indeed due to the rapid expansion of ART. However, many modeling groups' estimates of the impact of ART on these trends vary widely. This calls into question the ART efficacy, effectiveness and coverage parameters that modelers use to project HIV incidence and prevalence.
We reviewed 2015-2016 global and national mathematical modeling studies regarding ART's impact on new HIV infections. We extracted ART and HIV transmission parameters (i.e., proportion diagnosed, proportion on ART, proportion on ART and virally suppressed, proportion on ART and not virally suppressed, percentage reduction in transmission for those on ART and virally suppressed, percentage reduction in transmission for those on ART but not suppressed, and retention). We then derived a model-specific ART effectiveness percentage that captured the aggregate impact of ART on transmission by 2020. We describe the available parameters and 2020 ART effectiveness calculations for 9 models and compared the two with the lowest and highest ART effectiveness. The ART effectiveness expressed as a percentage reduction in HIV transmission by 2020 ranged from 20% to 86%. ART effectiveness disparities between the highest (SACEMA) and lowest (GOALS) models for Mozambique are highlighted in Figure 1. The GOALS Mozambique model limits eligibility for ART initiation to 80% coverage of people living with HIV and with a CD4 + cell count below 350 cells/μL, assumes that ART reduces transmission by 80%, and that 70% of patients are retained, which yields a derived ART effectiveness figure of 20%. The SACEMA model, on the other hand, assumes 90-90-90 by 2020 (i.e., 73% viral suppression of estimated PLHIV), that ART reduces transmission by 96% in those on ART and virally suppressed, and by 88% in those on ART but not virally suppressed, yielding a derived ART effectiveness value of 86%. ART parameter selection and assumptions dominate, and low ART effectiveness translates into lower impact. The disparity between the models is striking, and the implications are significant: The more realistic parameters that yield higher ART effectiveness valuation suggest that the continued expansion of ART and support for sustainable viral suppression will make it possible to significantly reduce transmission and eliminate HIV in many settings.
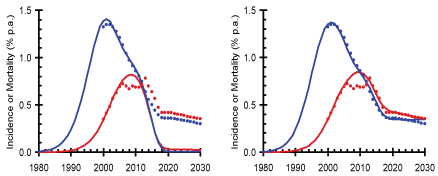 Figure 1: Annual HIV incidence (blue) and mortality (red) for Mozambique.
Figure 1: Annual HIV incidence (blue) and mortality (red) for Mozambique.
Legends: Dots: GOALS model [51]; Lines: SACEMA model to assume only 65% of those infected [47]; Left: GOALS projections compared to SACEMA projections; Right: Adjustment of SACEMA model to match GOALS model requires 1) reduction of people living with HIV on ART to only 65% and 2) reduction of ART transmission efficacy to only 65%. With these significant reductions in the coverage and effectiveness parameters the SACEMA model gives results that are very close to the GOALS model.
View Figure 1
Despite progress, HIV is still a major public health threat with over 35 million deaths and counting, and in 2016, there were still over two million infections and one million HIV-associated TB cases [1,2]. The impact of the HIV epidemic prompted an unprecedented response and we now know more about HIV than any other pathogen in history. The discovery of effective treatment with antiretroviral therapy (ART) in 1996 and subsequent evidence regarding the prevention of illness, death and transmission transformed the epidemic from an unending, unmitigated disaster into something that could be prevented and even ended [3-10]. Evidence from the early 2000s showed that viral suppression prevented transmission, that earlier treatment had health benefits, and that people who did not have access to treatment had increased morbidity and mortality even at higher CD4 cell counts [11-13]. Additionally, as the number of people diagnosed and placed on treatment increases it serves to normalize HIV while encouraging others to seek testing and treatment. Although the preventive benefits of the virtuous cycle engendered by expanding understanding regarding treatment as prevention has not been quantified, the recent Undetectable = Untransmittable campaign provides proof of concept regarding its potential significance. Proponents of traditional prevention interventions (e.g., condoms, male circumcision, and PrEP) are increasingly aware that increasing the number of people living in the community who are virally suppressed has a prevention benefit for everyone at risk of infection. For example, uninfected sex workers in a community that has achieved high ART coverage will be far safer regardless of which other prevention strategies they may be able to use. The concept of expanded access to ART to curb the epidemic and treatment as prevention (TasP) was introduced in 2006 [5] and formalized by the World Health Organization (WHO) in 2012 [14]. Over the past decade, the global and local HIV strategy has shifted from "test-and-wait" to "test-and-treat" combined with the 2020 90-90-90 target (73% of people on ART and virally suppressed) to control the HIV epidemic and end AIDS (defined as universal treatment with less than one AIDS case and one AIDS-related death per 1,000 population) [15-17].
Policy discussions around treatment as prevention of HIV illness, death and transmission have focused on how to increase access to testing and how quickly after diagnosis to provide treatment. Global HIV leaders and stakeholders turned to modeling to explore the impact of possible ART expansion scenarios [18-22]. While a few models explored expansion of ART access beyond existing WHO guidelines, most models limited testing and treatment to WHO recommendations to treat only those who were severely immunocompromised [22]. Similarly, traditional costing efforts could be classified as "doomsday costing" insofar as they took a health sector perspective and only looked at the costs of providing earlier treatment while ignoring the potential prevention benefits and cost savings of earlier treatment [23,24]. More modern "second generation" approaches to economic modeling took into account the prevention impact of scaling up treatment along with other interventions [21,25-27]. These models explored treatment as prevention of illness, death, and transmission. In some cases, the models were combined with a costing framework to examine the costs, cost benefits, and cost savings of various scale-up scenarios [25-28]. The dominant model (GOALS) used by the Joint United Nations Programme on HIV/AIDS (UNAIDS), the Global Fund, the United States government, and many other governments now includes the prevention impact of ART and is used to determine the health and transmission impact, costs and cost-savings for various HIV response scenarios [26]. The UNAIDS GOALS model incorporates data from available surveys and other surveillance information and makes forward projections of incidence, prevalence and resource needs according to their financial framework categories [26]. In the 2016 UNAIDS HIV Update and in the UNAIDS resource needs projections, incidence and prevalence were reported as being roughly stable from 2010 to 2015 in all regions of the world except for Eastern Europe, where the rates reportedly increased [1,26]. During this time the world spent an estimated US$100 Bn on attempting to control HIV and one conclusion that can be drawn from the report is that the significant investment has had little or no impact on incidence or mortality. If true, this would have major implications for future resource needs and global HIV control strategies since the impact of treatment would appear to be far less than expected. However, there are reasons to question the flat-line UNAIDS estimates of new infections and prevalence, as these results contrast with other models and scientific evidence regarding the potential impact of ART and other prevention interventions. Recent population-based studies from a number of countries in East and Southern Africa recorded marked declines in incidence, prevalence and deaths further support the notion that they are mostly likely due to treatment expansion [29].
The marked contrast in model outcomes, where one predicts flat-line incidence and prevalence in four of five regions [1,26], while others project a steady decline to elimination, prompted us to explore the importance of ART efficacy, effectiveness and coverage parameters. Specifically, for 2015 and 2016, we reviewed global and Africa-specific mathematical modeling studies that looked at the impact of ART (with or without other HIV prevention interventions) or 90-90-90 on new HIV infections and/or required future investments. We reviewed these HIV epidemiologic and costing modeling studies for their structure and parameterization around ART. For each modeling study, we extracted the parameters that pertained to ART and HIV transmission (i.e., proportion diagnosed, proportion on ART, proportion on ART and virally suppressed, proportion on ART and not virally suppressed, percentage reduction in transmission for those on ART and virally suppressed, percentage reduction in transmission for those on ART but not suppressed, and retention). We then used them to derive a model-specific ART effectiveness percentage that captured the aggregate impact of ART on transmission. Modeling studies projected varying program implementation rates and we calibrated the derived ART effectiveness to 2020, expressing effectiveness as a percent reduction in HIV transmission (Figure 2). Not all of the ART effectiveness equations were equivalent since the parameters varied by modeling study and we only used the available parameters for the model-specific ART effectiveness calculation. Proportion of people on ART coverage by 2020 was used as part of the calculations since coverage has a direct impact on the epidemic. While ART efficacy is a measure that assesses the degree to which ART produces its intended biomedical effects, ART effectiveness does not imply that ART works better but rather reflects the aggregate parameters and assumptions used by each model to determine overall reduction in transmission. Consequently, when a model includes assumptions for higher ART coverage, higher retention, or higher efficacy in reducing transmission, its ART effectiveness figure is proportionally higher.
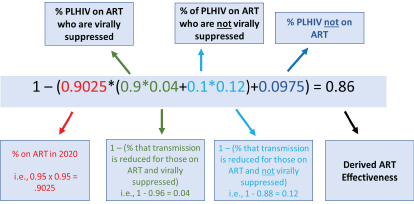 Figure 2: Annotated ART effectiveness formula for SACEMA model. This formula was applied using the model-specific parameters to derive the ART effectiveness for each of the for each of the models (see Table 1 for the results).
View Figure 2
Figure 2: Annotated ART effectiveness formula for SACEMA model. This formula was applied using the model-specific parameters to derive the ART effectiveness for each of the for each of the models (see Table 1 for the results).
View Figure 2
Examining the available parameters and derived ART effectiveness outcomes for the nine modeling studies is instructive (Figure 2, Table 1). The modeling parameters for ART effectiveness by 2020 ranged from 20% to 86%. We highlighted the disparity in ART effectiveness by comparing the lowest GOALS modeling study and highest SACEMA model for Mozambique (Figure 1). In the graph on the left, the GOALS model, which forms the basis for UNAIDS estimates, gives a more pessimistic prediction of the trends in incidence and mortality when compared with the SACEMA model. The study using the GOALS model limits eligibility for ART initiation and assumes that "full coverage" is equivalent to just 80% of those infected with HIV and with a CD4+ cell count below 350 cells/µL. With these assumptions, about half of all those infected with HIV will be on ART which, when coupled with the lower estimate of transmission impact, yields a derived ART effectiveness of only 20%. The SACEMA model, on the other hand, assumes that we reach 90% ART coverage by 2020 and then continue to roll-out ART at the same rate while the reduction in transmission for those who are on ART and virally suppressed is 96%; this results in a derived ART effectiveness of 86% and a much more optimistic-yet still plausible-forecast. The underlying models are similar. To match the GOALS model results for Mozambique, the SACEMA model would need to adjust the parameters downward to assume that only 65% of those infected with HIV are on ART at full coverage (instead of 90%) and ART reduces transmission by 65% (instead of 96%). Put more starkly, this level of effort would leave around 40% of people living with HIV not on ART and not suppressed and at immediate risk of illness, death and transmission. Clearly, these two parameters are the critical determinants of the impact of treatment on incidence and mortality, since the forecast trends are nearly identical after these factors are controlled for (illustrated in the graph on the right of Figure 1). Comparisons with other modeling studies show similar results with the decisions around key ART effectiveness parameters driving the outcomes in terms of prevention impact.
Table 1: Description of models with calculated ART effectiveness. View Table 1
Modeling plays an important role in exploring potential outcomes while illustrating what data are needed to make more accurate predictions. Parameterization relies on studies and other surrogate information and our comparison demonstrates why it is so important to be transparent about the judgments involved in selecting critical values for interventions such as ART coverage and effectiveness. While there is general agreement about the model structure, the highly influential HIV models we reviewed varied significantly in their parameterization. Parameters such as 'ART coverage' and 'on ART and virally suppressed' have a major impact on outcomes, and models that predict lower coverage or lower ART efficacy yield correspondingly lower derived ART effectiveness values. In many of the papers we reviewed, the parameterization is less than obvious for even the mathematically inclined with time on their hands. The ART effectiveness calculation allows the reader to see sometimes hidden assumptions in models that may otherwise be missed. For example, a lower ART effectiveness percentage usually reflects assumptions that there will be poor program performance (e.g., lower ability to keep people in care, on treatment and suppressed) or lower ART expansion targets (e.g., higher proportions of people off ART and unsuppressed).
Arguments that parameters should be conservative and reflect "realistic" local program performance or limited resources for treatment expansion may appear sound on first glance. However, using pessimistic assumptions about the potential for successful expansion of ART programs tends to underestimate what is possible and simply does not make sense. For example, when the global response to the epidemic got underway in the early 2000s, if one assigned "realistic" parameters informed by the fact that only 30,000 people in Africa were on ART or relied on what most experts believed was possible at the time, models never would have yielded the projected impact or supported the planning necessary to reach 3 million by 2005 or the 21.5 million people reported to be on ART in 2017. Pessimistic assumptions around ART access were influenced by debates around resource needs, cultural stereotypes, and allocation that pitted earlier testing and treatment against other prevention interventions [30,31]. For example, to bolster their position against offering treatment, officials articulated unspoken and unproven assumptions around adherence among Africans [30]. The "prevention first" proponents used the traditional mantras that "there is no silver bullet" [32] and "we cannot treat ourselves out of the epidemic" to support their arguments for prioritizing traditional prevention interventions at the expense of expanded access to treatment as prevention of illness, death and transmission" [33]. Likewise, this restricted vision around the availability of resources, feasibility, and the potential of treatment to curb the epidemic contributed to the "public health approach" that could also be characterized as a "scarcity and hoarding strategy". The approach envisioned basic treatment access in low and middle-income settings for only those people with severe immunocompromise (e.g., < 350 CD4 cell count) and over the years served to block incorporation of earlier treatment into guidelines, models, and planning [34]. Another example of the negative advocacy and framing around expanding access to treatment was when some officials relied too heavily on early studies that suggested retention among African programs was poor [35]. However, selectively using traditional retention parameters from early, poorly performing research cohorts did not reflect the retention that was seen in many other programs and would have caused the model to predict higher ART effectiveness [36].
To take our argument to the extreme, if the 1960s Kennedy administration would have only used pessimistic scenarios regarding the potential of manned-space flight to the moon, it would have never made the decision in favor of going. In our efforts to model the potential impact of ART expansion, assumptions around program performance and the efficacy of interventions should be explicit; whether modelers choose to be "realistic" or "optimistic", the perspective should be applied similarly across interventions and readers should have a chance to understand the subjectivity behind the modeling assumptions. Models also need to frame the scenarios with the assumption that treatment will be offered to most or all people living with HIV. With shifts in resources, capacity and policy, past models that pitted prevention vs. treatment interventions without offering treatment to most people living with HIV are no longer ethical and should not be employed [22]. Models are meant to explore potential scenarios, and if a model uses optimal program performance and targets such as 95-95-95 [37], then the model can be used to evaluate what would be required for the program to reach its targets. As with any major international public health endeavor, these sorts of ambitious projections are vital for conceptualizing, planning, budgeting, and working together to reach targets and realize the projected gains in health outcomes.
The good news is that program performance has improved and 90-90-90 is not only feasible, but has already been achieved in some settings [38-42]. Studies show that around 90% or more of people will agree to an HIV test if it is offered, that people started on ART adhere to a very high level, that ART suppresses the virus and reduces transmission by close to 100%, and that retention is probably much higher than predicted [11,43,44] . Additionally, ART costs have fallen significantly, and high levels of ART and viral suppression can be achieved. Given the good performance of many programs in these areas, our brief review suggests that many of the current models have likely underestimated the potential impact of ART. The lack of clarity regarding methods and assumptions may extend to current global UNAIDS projections since they also incorporate unpublished ART effectiveness and other assumptions into the model [45]. Such overly conservative assessments could account for past recommendations to under-invest in expanding access to treatment while using remaining resources for less cost-effective interventions [22,46]. Additionally, assumptions around poor program performance and ART effectiveness may have also contributed to projections with slower declines in incidence and consequent inflation of needs assessments. Using optimal ART parameters based on actual program performance and the available scientific data suggests that as we expand access and support the achievement of sustainable viral suppression, it will be possible to significantly reduce transmission, and indeed, control or eliminate the HIV epidemic and end AIDS in many settings.