In situ right and left lobe cadaveric split liver transplantation for two adult recipients is a challenging and infrequently performed procedure to increase the liver donor pool. It should be considered for small sized adult recipients who do not have living donors and coming from regions with few donors. In situ split gives the additional advantage of reduced cold ischemia time, better delineation of hilar structures during warm dissection, improved assessment of viability of liver lobes, reduced perioperative bleeding during transplantation and possibly reduced biliary complication rates as compared to ex vivo split.
We will share with you this uncommon procedure which enabled our two small sized adult recipients, who did not have living donors, to receive timely liver transplantation within a short waiting time. The cadaveric donor's age, body mass index, pressor requirements were all within acceptable limits for this surgery. After laparotomy and satisfactory visualization of the liver we split the liver in to full right and left lobe using techniques used for living donor hepatectomy. The two full hemi livers were then transplanted to two adult recipients who required liver transplantation. After six months follow up, both patients have good graft function with no evidence of small for size, biliary or vascular complications. One recipient is following up as outpatient while the other required readmission for pulmonary complications and is currently recovering.
Availability of an ideal cadaveric donor, effective communication between the various teams, appropriate use of radiological tools, meticulous planning and incorporating techniques used in living donor liver transplantation helped us to overcome the various technical and logistic challenges. Ethical dilemma of a shorter waiting time versus smaller graft size needs to be resolved.
In situ cadaveric split, Two adult recipients, Waiting time, Graft size
In situ splitting of a cadaveric liver into full right and left hemi livers and transplanting it to two adults is an alternative method of increasing donor pool, especially in a country like Saudi Arabia where there is severe dearth of deceased donor organs [1]. Traditionally split liver transplantation (SLT) is performed for an adult and pediatric recipient. The impact on reducing the waitlist for adults can be enhanced only if the SLT can be done for two adults. In situ SLT has gained popularity due to the emerging experience with living donor liver transplantation (LDLT) [2,3]. In situ SLT can be particularly advantageous for adult recipients having low body weight, without living donor and in a region of depleted cadaveric donor pool. Here we share the experience of the first successful full right and left lobe In situ split liver transplantation for 2 adult recipients performed in Saudi Arabia.
The donor was a 41 year of lady, weighing 57 kgs, 160 cm tall and body mass index (BMI) 22. She was declared brain dead at her local hospital after massive stroke. Brain death was confirmed after two separate neurological examinations and an electroencephalogram. Total duration of intensive care unit (ICU) stay was 3 days. Laboratory tests for liver and renal function were normal with preoperative values of sodium 141meq/L, total bilirubin 0.17 mg/dl, serum alanine amino transferase (ALT) 18 U/L and international normalized ratio (INR) 1. She did not require vasopressor drugs. The donor was promptly transferred to our ICU for proper optimization and further investigations. Pre donation Computed Tomography (CT) volumetry showed adequately sized right and left lobe of liver (Figure 1). Total liver volume was 1362 cc, right lobe measured 962 cc and left lobe 400 cc. CT angiography showed standard vascular anatomy favorable for in situ full right and left lobe splitting of liver.
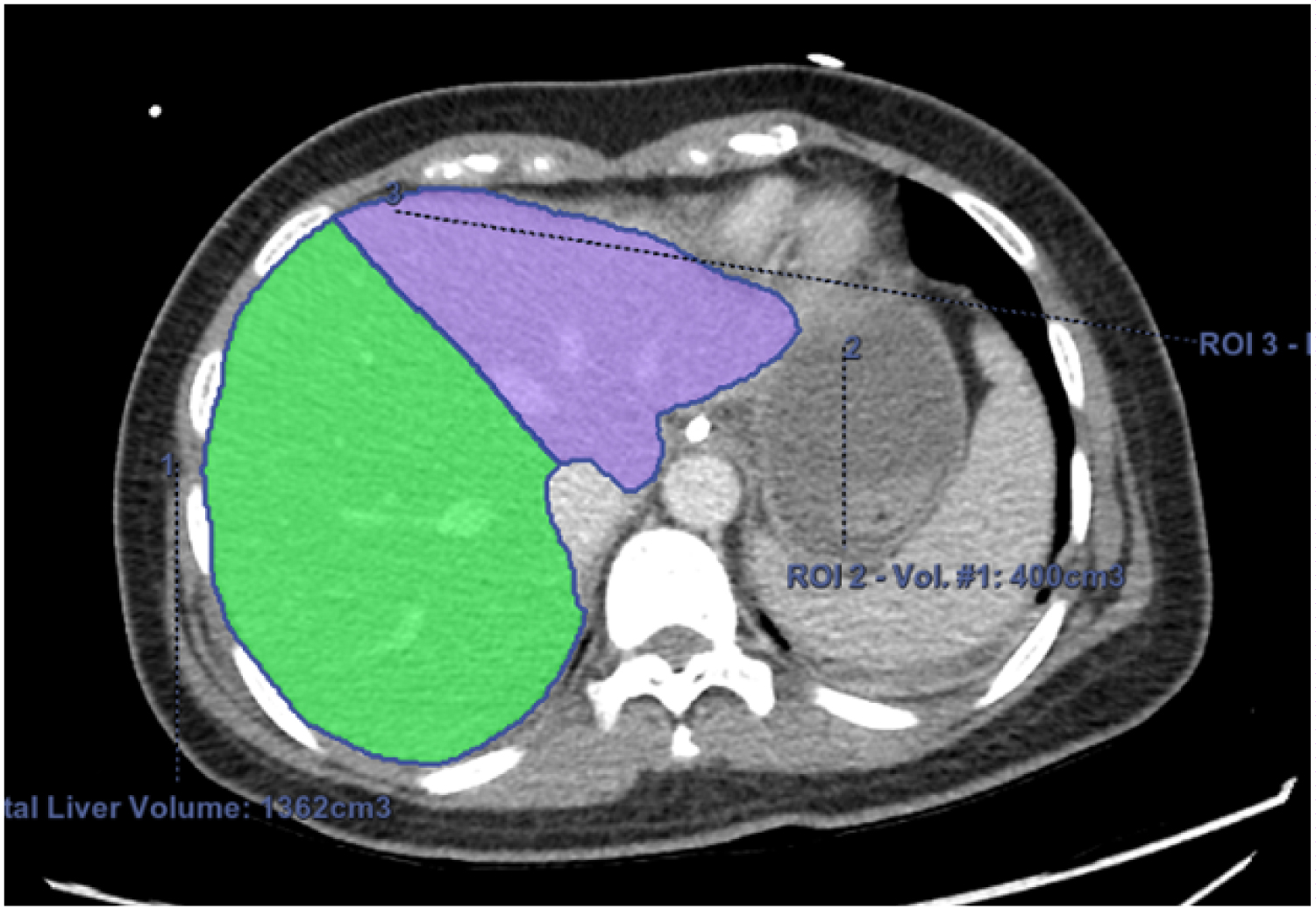 Figure 1: Preoperative CT volumetry of the liver showing the total liver volume of 1362 cc, right lobe volume 962 cc and left lobe volume 400 cc.
View Figure 1
Figure 1: Preoperative CT volumetry of the liver showing the total liver volume of 1362 cc, right lobe volume 962 cc and left lobe volume 400 cc.
View Figure 1
After laparotomy the liver size, color and consistency were assessed. Cholecystectomy was done and on table cholangiogram was performed to assess the biliary anatomy. The liver was mobilized off the inferior vena cava by dividing the short hepatic veins. The right hepatic vein was dissected and isolated. Hilar structures were dissected and isolated using vessel loops (Figure 2). Parenchymal transection was done through Cantlie's line with the help of hanging maneuver and using Erbe hydrojet dissector (Erbe Elektromedizin GmbH, Tuebingen, Germany). After completing the transaction, the next steps for procurement of the liver lobes were done after cross clamping and cold perfusion with University of Wisconsin solution. Total time for surgery for the in situ split liver procurement was 5 hrs 30 mins.
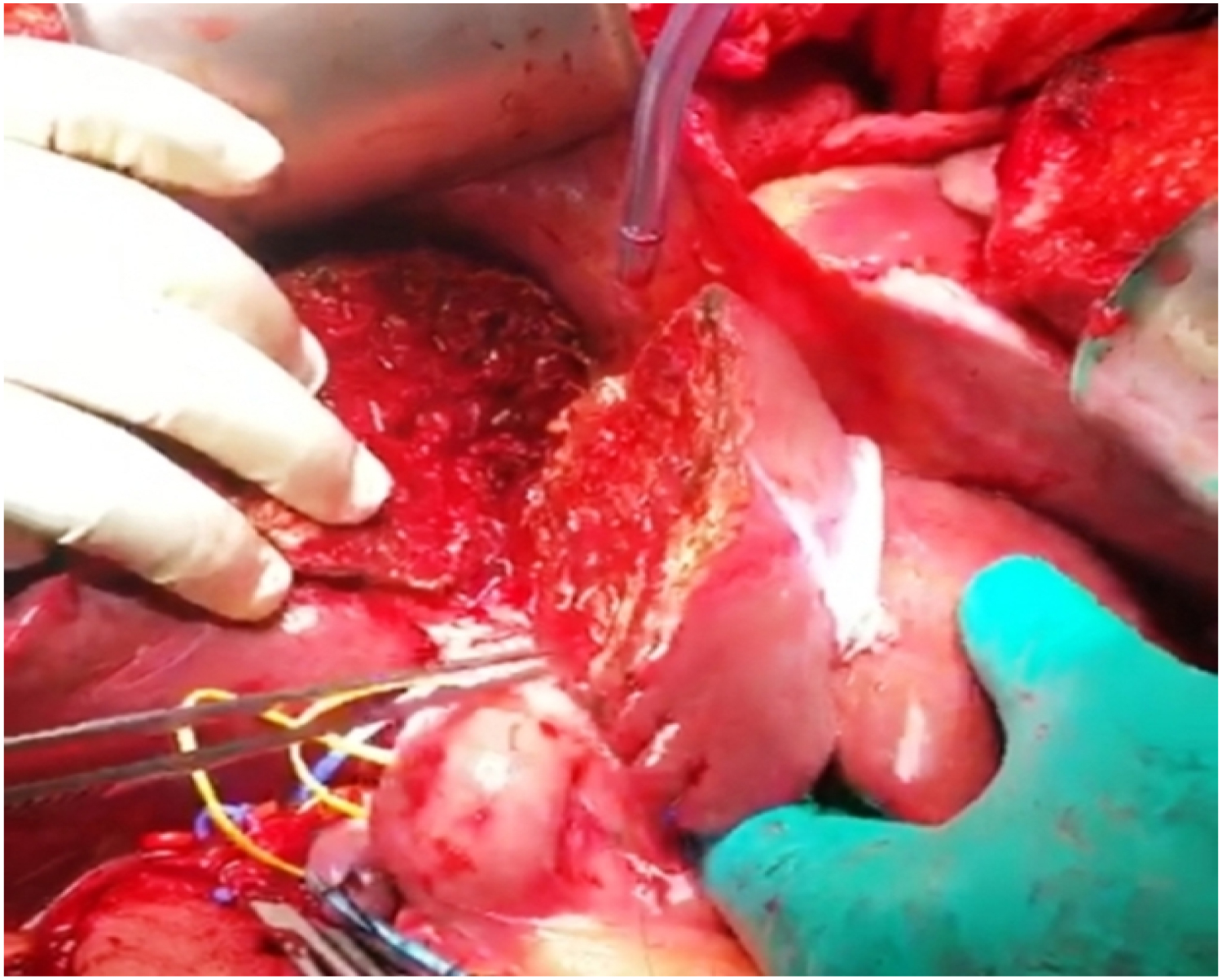 Figure 2: Intraoperative image of parenchymal division of the donor liver into full right and left hemi liver grafts. The structures of the hilum have been dissected and isolated with vessel loops.
View Figure 2
Figure 2: Intraoperative image of parenchymal division of the donor liver into full right and left hemi liver grafts. The structures of the hilum have been dissected and isolated with vessel loops.
View Figure 2
Full right liver graft was without the middle hepatic vein and consisted of segments V to VIII with right hepatic duct, right portal vein, right hepatic artery and right hepatic vein. The full left liver graft included segments I to IV, common bile duct, main portal vein, celiac axis and the retro hepatic vena cava. Two significant veins (> 5mm) draining the segment V and VIII of the right lobe were reconstructed in the back table using an iliac artery Y graft. Heart, lungs, kidneys and the pancreas were also procured from the donor and were transplanted.
The recipients were informed about the indication, advantages and possible complications of split liver transplantation. Both recipients were petite with low body weight and low model for end-stage liver disease (MELD) scores. Recipient 1 was a 47 years female, 149 cm tall, weighing 47 kgs and BMI of 21. She was diagnosed as non-alcoholic steatohepatitis (NASH) and CT imaging showed a single enhancing lesion in segment III measuring 1.8cmX1.2cm which was diagnosed as hepatocellular carcinoma (HCC). Her MELD score was 11. The patient had under gone selective hepatic artery chemoembolization for the liver lesion and subsequent imaging ruled out any evidence of recurrence. She received the full right hemiliver with adequate graft recipient weight ratio (GRWR) of 2.04. Operating time was 6 hours, cold ischemia time (CIT) 3 hours and warm ischemia time (WIT) 30 minutes. There was moderate blood loss requiring transfusion of blood products including 4 units of packed red cells (Table 1). Recipient 2 was a 31 years lady, weighing 38 kgs, 148 cm tall and BMI 17. She had history of kidney stones and renal failure and underwent living unrelated kidney transplant in another country one year earlier. She was subsequently diagnosed to have primary hyper oxaluria during work up for ongoing kidney allograft dysfunction. She was transplanted the full left hemiliver with sufficient GRWR of 0.95.Operating time for her for 4 hours, CIT was 7.5 hours and WIT 39 minutes. There was minimal blood loss and did not require transfusion of any blood products (Table 1). For both, the biliary continuity was established using duct to duct anastomosis.
Table 1: Recipient details. View Table 1
Initial post-operative course for both the recipients were smooth and both were extubated and off vasopressors in the following day. They were sent to the floor within 48 hours of surgery. Both patients were discharged within 3 weeks of surgery with improving graft functions. There was no evidence of vascular, biliary or small for size complications. There was also no suspicion of liver allograft rejection. Laboratory examination at the time of discharge for recipient 1 showed bilirubin 4.2 mg/dl, ALT 119U/L and INR 0.9. The laboratory values for recipient 2 on discharge were bilirubin 2.04 mg/dl, ALT 39U/L and INR 1.1 (Table 1). As per our hospital protocol, induction of immune suppression for both recipients was done with methyl prednisolone bolus at the time of transplantation. Post operatively they received tapering dose of steroid and oral mycophenolate. Tacrolimus was started for recipient 1 on second post-operative day but was withheld for recipient 2 due to underlying renal impairment.
The explant liver histology of the first recipient showed cirrhosis in the back ground of NASH with embolic material in segment III without any evidence of residual HCC. As expected, the biopsy for the explanted liver for the second recipient looked normal.
Recipient 2 was readmitted with pneumonia and sepsis. She was intubated and ventilated, started on broad spectrum antibiotics and required drainage of empyema by insertion of chest tube. She was later diagnosed to have pulmonary aspergillosis and was started anti-fungal therapy with intravenous voriconazole. Both steroids and mycophenolate were stopped at that time and liver function remained normal. During this time she was also started on tube feeding secondary to her poor nutritional status. Patient responded to treatment and is currently recovering on the floor. She is tolerating oral diet, walking with support and getting physical therapy to help her regain strength. Latest CT shows near complete resolution of the pulmonary cavitations and was started on tacrolimus for immune suppression. Recipient 1 is being followed as an outpatient and did not require any readmission.
During the 6 months follow up, both the recipients have good liver function. Latest laboratory results for Recipient 1, shows bilirubin 0.4 mg/dl, ALT 29U/L and INR 1 while that for Recipient 2 is bilirubin 1 mg/dl, ALT 19 U/L and INR 1.1 (Table 1). They did not develop biliary or vascular complications. There was no evidence to suggest small for size complications or allograft rejection.
This is the first in situ full right and left lobe split cadaveric liver for two adults successfully performed in Saudi Arabia. This is a novel way of increasing the number of liver allograft. Literature on in situ SLT exists mostly in the form of center specific case reports and case series [3]. Classical SLT was first described by Pichlmayr in 1988 [4]. This resulted in transplanting the smaller left lateral segment to a pediatric recipient and the extended right lobe to an adult. Bismuth pioneered full right and left split of the liver enabling the grafts to be utilized for two adult recipients [5]. Most of the splits performed around the world are classical and are typically done ex vivo [6]. Often these are done at centers where the left lateral segments are utilized for the pediatric recipient and the extended right lobe is shipped to centers accepting it for adult recipient resulting in longer CIT.
Vast majority of the patients in liver transplant waiting list are adults. Conventional splitting of liver for a pediatric and adult recipient does not positively impact the adult waiting time [7]. Both our recipients had no living donor, had low MELD scores and were in urgent need for transplantation. They were both small in size and getting a small or pediatric deceased donor is extremely difficult in our region. Full in situ split of a cadaveric liver was the best option to help them get transplanted.
With increasing experience of living donor liver transplants, there has been a global increase in the number of reported cases of in situ splits for full right and left lobe hemilivers for adult recipients [8]. Although another center in Saudi Arabia has shared experience in SLT, it has largely been for adult and pediatric ex vivo split [9]. Advantages of in situ splitting over ex vivo split includes reduced CIT spent in the back table during ex vivo split, better identification of structures during warm dissection for in situ SLT, reduced blood loss during the transplantation, better assessment of viability of the segments IV and I during the split and also reduced rates of biliary complications [10].
Cadaveric donor characteristics for a successful split include age of donor below 45 years, ICU stay preferably within 5 days, near normal liver function test and low BMI to avoid fatty liver (macrosteatosis < 10%) [7,8,11].
There were quite a few logistic and technical challenges we had to overcome with the intention of having the best results. We curtailed donor ICU stay by prompt diagnosis of brain death. To keep the CIT short we transferred her to the recipient hospital, opened separate operating rooms and mobilized alternative teams for the two recipients. Preoperative CT for measurement of liver volumes, assessment of vascular anatomy and intra operative cholangiogram could be promptly performed due to radiology support.
In situ SLT can cause added load on the donor hospital and the other donor teams [11] due to the increase in the OR time by about 2 hours. We established clear communication with the other procuring teams regarding the plan and ensured their agreement. Transection of the liver was challenging as the CVP was not kept low so that perfusion to the other organs was not compromised. We used meticulous surgical techniques learnt from our LDLT experience to ensure proper retrieval and subsequent uneventful transplantation of the two full right and left lobes to the two recipients.
Biliary and vascular complication rates are higher after SLT due to the smaller caliber of the structures [12]. Biliary complications of leak or obstruction can go up from 20 to 40% while hepatic artery thrombosis rates has been reported between 5 to 10% [2,8,13]. Preoperative CT scan, intra operative cholangiogram, in situ warm dissection at the time of split and experience with LDLT have potentially contributed to the absence of these complications in our two recipients. Small for size syndrome can be significant problem after SLT for two adults [13]. CT liver volumetry of donor liver was helpful to ensure adequate GRWR for our two adult recipients.
Increasing experience with LDLT makes in situ two adult SLT an attractive means to increase the available liver grafts and will positively influence waiting list time and mortality. It can be particularly useful for small sized recipients without living donors, especially in regions where cadaveric donors are sparse. However a balance between the ethical issue of exposing the recipient to higher rates of complications and shorter waitlist time has to be realized. Logistic challenges to keep the CIT and WIT low with establishment of excellent team dynamics between the donor and recipient teams is necessary to achieve the best results.
We would like to thank our colleagues at the transplantation center for their technical support and patient care which resulted in this successful outcome.
There is no financial support or grant from any source in writing this paper.
The authors declare no conflicts of interest.
The authors have all contributed equally in this study.