Single lung transplantation is a valuable treatment option for end stage lung disease cases of which bilateral lung transplantation is not necessarily indicated. The main disadvantage of single lung transplantation is complications related to the native lung. The most common complications are infection and malignancy which tend to behave aggressively and often require surgical intervention. Safety and feasibility of surgery in lung transplant recipients has been shown in several case reports, however there is very little data regarding native lung pneumonectomy (NPL) and we found no data to date, describing allograft function post-procedure.
We retrospectively extracted data on lung transplant recipients at the Rabin Medical Center, Israel between May, 1997 and June, 2017. We collected and analyzed demographic information, indication for lung transplant and pneumonectomy, postoperative complications and allograft function.
We identified 12 patients who required native lung pneumonectomy. By the end of the study, five of 12 patients (42%) were alive. Seven patients were deceased: 3 (25%) did not survive the post-operative period. One patient died from bronchopleural fistula complications eight months after NLP; two died from progressive malignant disease. One patient died from chronic lung allograft dysfunction. Of nine patients who survived NLP to hospital discharge two patients with underlying emphysema had increase in FEV1; six developed significant allograft dysfunction with an average drop in forced expiratory volume in the first second (FEV1) of 29% (range 22-41%) at 3 month post NLP, one patient had a < 10% decline.
In conclusion, native lung pneumonectomy is feasible in carefully selected group of patients. It can lead to significant lung allograft dysfunction and we recommend case-by-case decision, where the risk of post-procedure complications, including the possibility of allograft loss, is weighed against the primary disease burden and expected survival without surgery.
Lung transplant, Pneumonectomy, Lung allograft dysfunction, Lung
Lung transplantation has become an established treatment option for patients with end stage lung diseases; it prolongs survival and improves quality of life. Patients with Cystic Fibrosis and Bronchiectasis require bilateral lung transplantation due to the septic nature of the primary disease and concerns about contaminating the allograft with prior infection [1]. Aside from this population, both single and bilateral lung transplantation has been successfully performed for other common indications such as COPD and IPF [2,3]. Despite the slightly lower overall survival, single lung transplantation has several clear advantages. It is a shorter and technically easier procedure which significantly extends the donor supply [4,5]. The main disadvantage of single lung transplantation is related to the possibility of native lung complications. Among these are infection and malignancy, which tend to behave aggressively in the remaining native lung and may require surgical intervention [6]. Although the safety and feasibility of surgery in lung transplant recipients has been described in several case reports, there is little data regarding outcomes following native lung pneumonectomy (NLP). Furthermore, to our knowledge, there is no published data to date regarding allograft function post NLP. A number of NLP procedures performed at our institution led us to collect all data on these patients in an attempt to better assess safety, short and long-term complications, and the impact on allograft function and survival.
We performed a retrospective analysis of all patients who underwent single lung transplantation at our program from May 1, 1997 to June 30, 2017. We identified 12 patients who required NLP. The decision to perform a pneumonectomy was based on the etiology, severity and extension of the disease in the native lung, and the risk of progression or recurrence without pneumonectomy alongside estimated lung reserve after the procedure.
We collected demographic information including age, gender, lung transplant indication, and pneumonectomy indication. Detailed medical histories, physical examinations, surgical procedure, hospitalization and long-term follow up were available for all patients as part of our center's routine clinical follow up for post transplanted patients. For all patients, pre-surgical assessment of lung function was available, including forced expiratory volume in the first second (FEV1) and quantitative lung perfusion scan. Data following the NLP was collected where applicable.
During the study period, a total of 379 patients underwent single lung transplantation. Of these, 12 patients developed native lung complications requiring pneumonectomy. Patient demographics, lung transplant and NLP indication, and time from transplant to pneumonectomy are presented in (Table 1). There were 6 (50%) males, with median age of 66 years. Median time from transplant to pneumonectomy was 51.5 months, with a range of 8-108 months. A majority of patients (8/12) underwent right pneumonectomy.
Table 1: Patient demographics, transplant indication and time to pneumonectomy. View Table 1
The indication for NLP was non-small cell lung cancer (NSCLC) in 83% (10 of 12 patients), with 66.7% (8 of 12) having histological features of squamous cell carcinoma and the other two having features of the adenocarcinoma type. Two patients underwent NLP due to severe uncontrolled native lung infections. (Table 2) summarizes our clinical pre- and post-pneumonectomy data.
Table 2: Pre and post pneumonectomy clinical data. View Table 2
Six patients had early post-operative complications. Three died within 8 days of surgery: 2 from severe cardiovascular complications and 1 from pneumonia-induced sepsis. Other early complications included post-surgical atelectasis (1) that responded to conservative therapy, atrial fibrillation (1) that converted with amiodarone to sinus rhythm, and hypercapnic respiratory failure (1) requiring a temporary tracheostomy for ventilator support. Of 9 patients who survived to discharge, 6 developed a significant decline in lung function, with an average FEV1 drop of 29% (range 22-41%) at 3 month post NLP. One patient had a < 10% decline, and 2 had a substantial increase in FEV1. Patients with an increase in FEV1 post-NLP were transplanted for COPD and had native lung emphysema. (Figure 1) demonstrates FEV1 pre and post NLP for those patients alive to date, and (Figure 2 ) for those who are deceased.
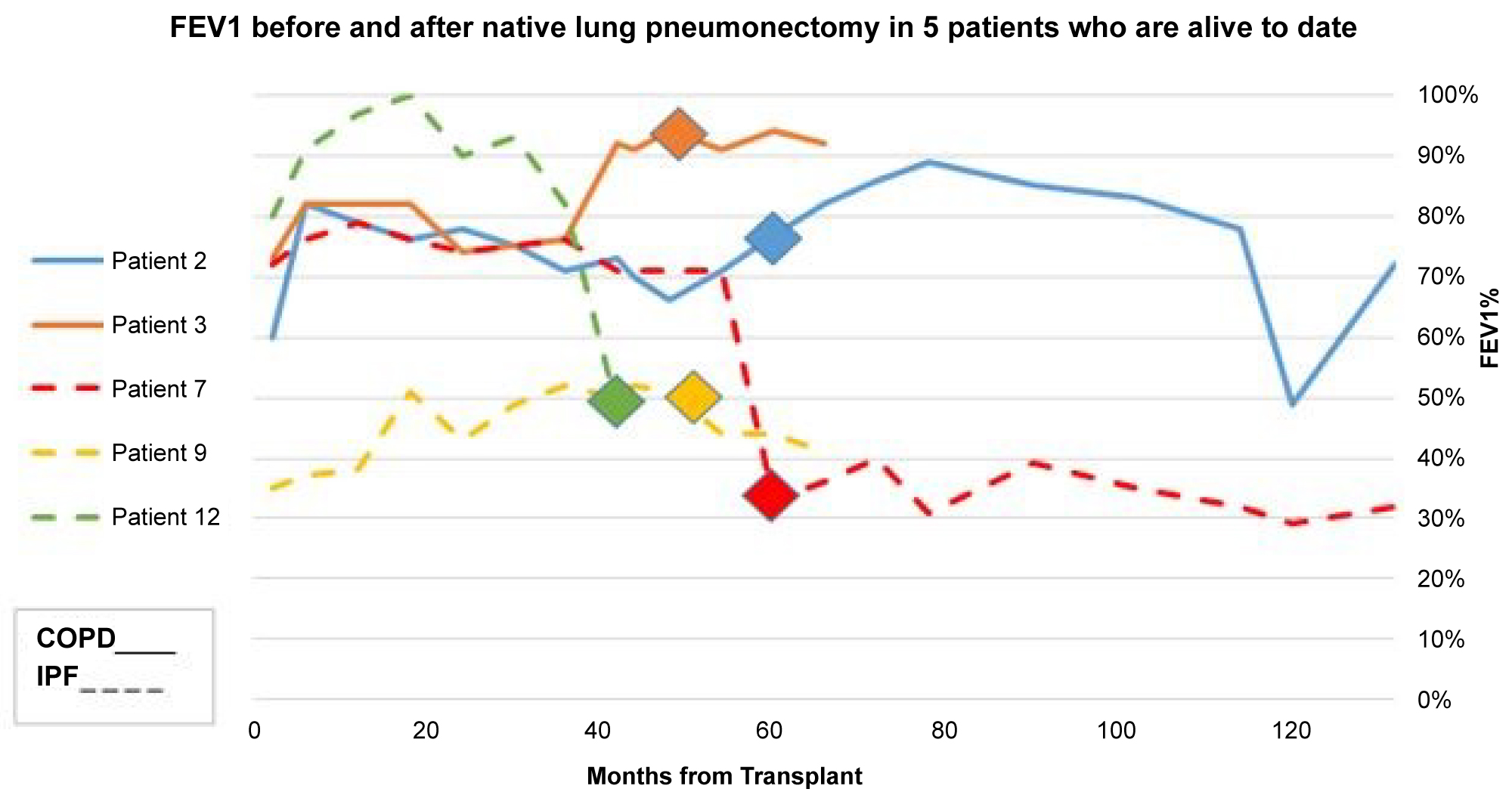 Figure 1: FEV1% is plotted on the Y-axis vs. months transplant on the X-axis. The rhombus represents the first FEV1 measured post-pneumonectomy.
Figure 1: FEV1% is plotted on the Y-axis vs. months transplant on the X-axis. The rhombus represents the first FEV1 measured post-pneumonectomy.
*Note: The solid line demonstrates COPD patients with improved lung functions post-pneumonectomy, while the dotted lines demonstrate IPF patients.
View Figure 1
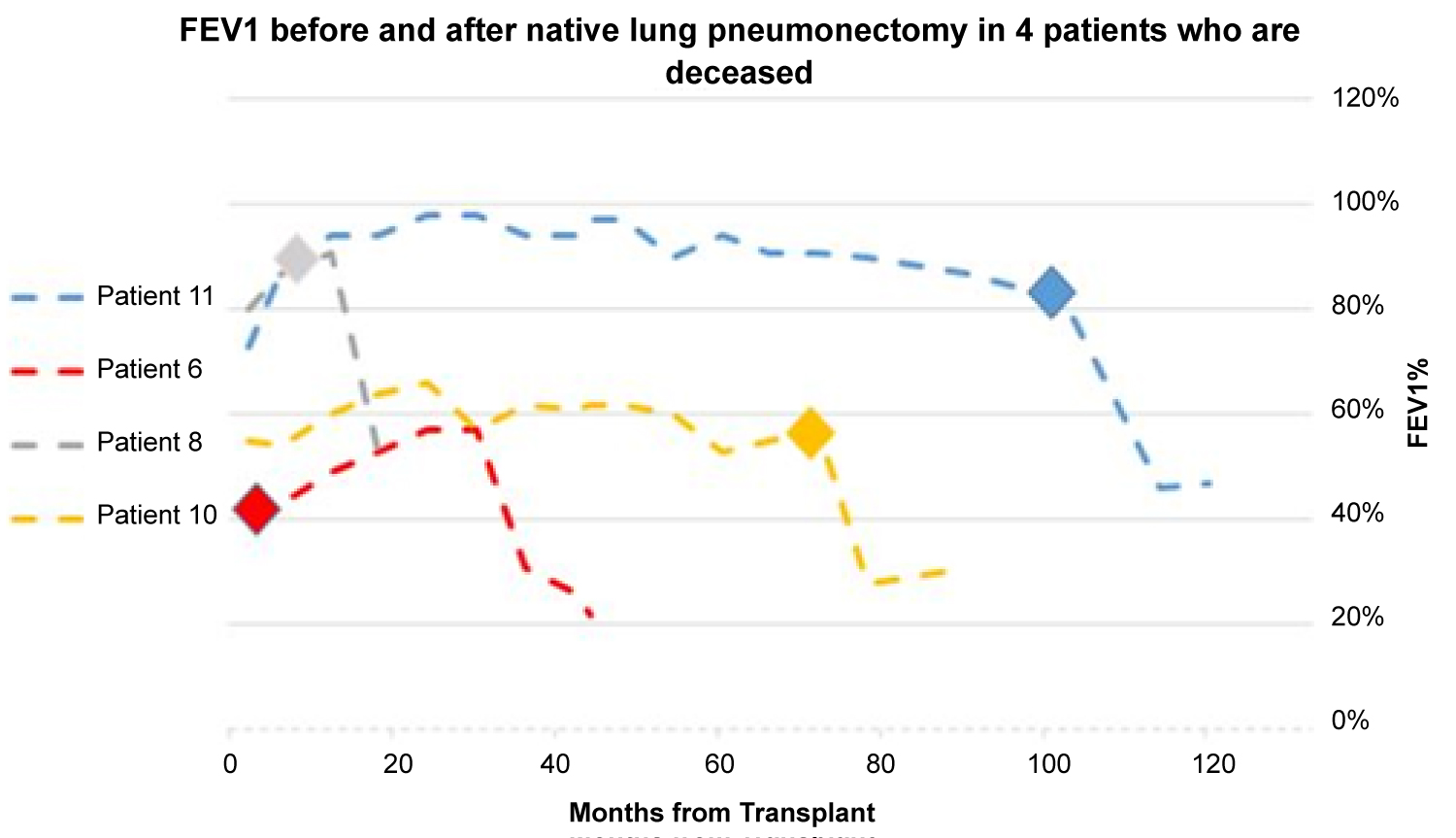 Figure 2: FEV1 is plotted on the Y-axis vs. months from transplant on the X-axis. The rhombus represents the first FEV1 measured post-pneumonectomy.
Figure 2: FEV1 is plotted on the Y-axis vs. months from transplant on the X-axis. The rhombus represents the first FEV1 measured post-pneumonectomy.
*Note: All patients are IPF patients.
View Figure 2
All 6 patients who developed allograft dysfunction underwent an extensive workup to exclude active infection, airway complications, and acute rejection. Four patients eventually died; 2 from cancer progression at 10.5 and 15 months after NLP, 1 from complications following a bronchopleural fistula at 8 months after NLP, and 1 from chronic allograft rejection at 30 months after NLP. Five of 12 patients (42%) are alive by the end of the study period with stable allograft function.
For immunocompetent patients, pneumonectomy is associated with a higher morbidity and mortality due to a variety of causes, including anatomic changes, significant decline in lung function, and other potential complications [7]. Post-pneumonectomy 30-day mortality rates range from 2-11% [8-10]. Right-sided pneumonectomy is associated with a higher mortality rate at 1-3 months post-procedure, as compared to left-sided pneumonectomy (8-16% versus 4-9%, respectively). Long-term survival is also affected by laterality: 1- and 3-year survival after right pneumonectomy is 63% and 39%, compared to 70% and 41%, respectively, after left pneumonectomy [11].
While there is limited data regarding the safety and feasibility of pulmonary resection after lung transplantation, most studies report acceptable safety results. In 2003, Fitton, et al. published their encouraging experience with lung resection/lobectomy in 11 lung transplant recipients [12]. Their series included both native and allograft lung resections. In 2011, Souilamas, et al. reported the feasibility of lung resection in cystic fibrosis lung transplant recipients [13]. They described acceptable outcomes and surgical risk for partial pulmonary resection, while pneumonectomy was associated with a high early mortality.
Thus far, the largest series of native lung complications came from the Inova Fairfax Lung transplantation program in 2009 [14]. From 180 SLT recipients, 25 (13.8%) developed significant native lung complications, and 11 required NLP. Four of 11 patients (36.4%) had postoperative complications, including mechanical ventilation, pneumonia, and atrial fibrillation. All patients survived to hospital discharge. The authors concluded that NLP can be performed with an acceptable morbidity and mortality; however, this study did not describe allograft function or long term survival following NLP.
Our study is the largest series to date of NLP patients following SLT, and the only one addressing lung allograft function after surgery. Despite the high early mortality, 9 patients survived the post-operative period, 6 were alive at 1 year after NLP, and 1 remains alive has not yet reached 1 year mark (78% conditional survival). (Figure 3) demonstrates our 12 patients' outcome.
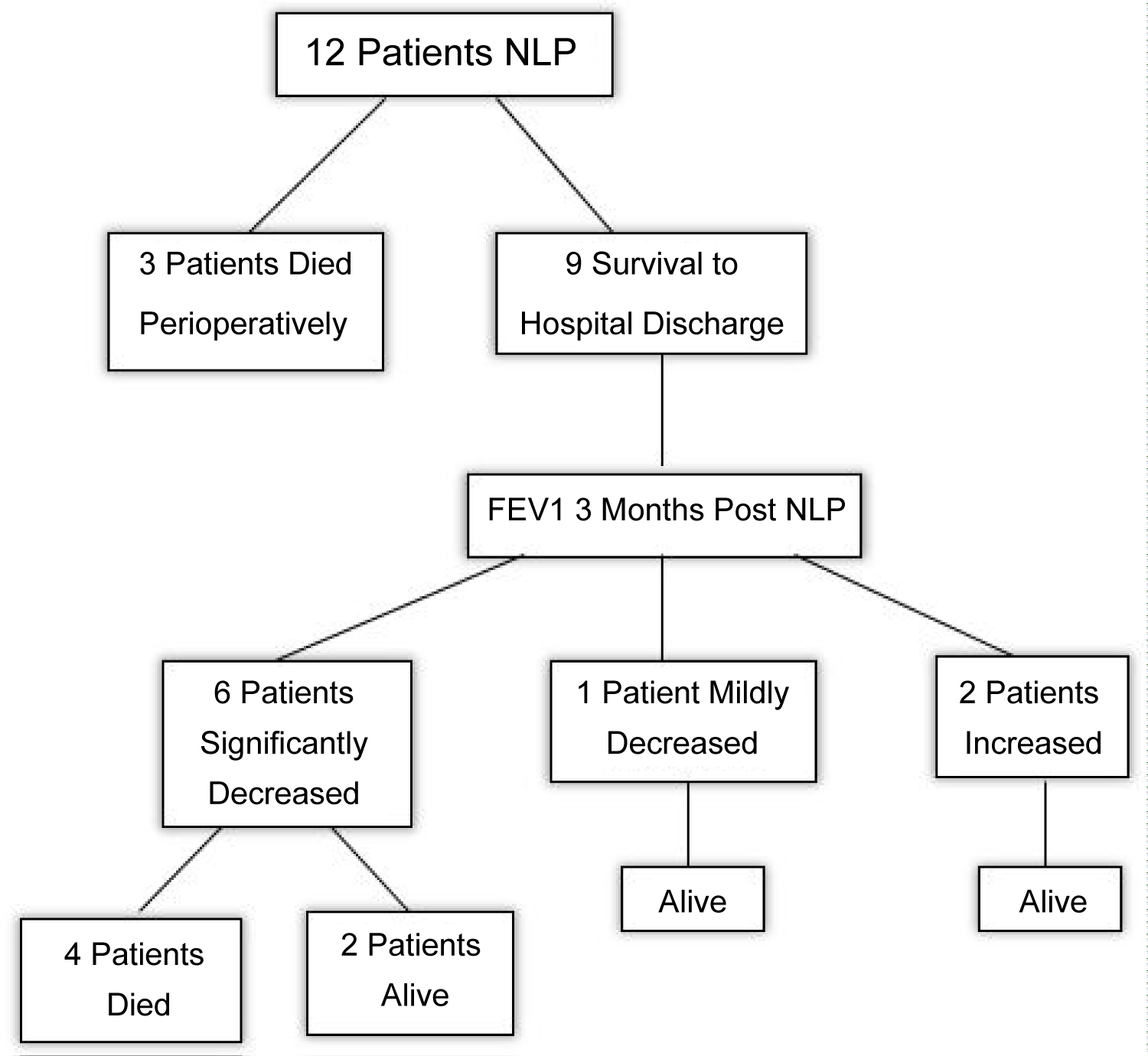 Figure 3: Native Lung pneumonectomy, outcome of 12 patients.
View Figure 3
Figure 3: Native Lung pneumonectomy, outcome of 12 patients.
View Figure 3
Data is accumulating that NLP may be feasible and safe to perform, with mortality rates approaching those for non-transplant patients. However, post-NLP allograft function is an issue of great relevance, in terms of long term recipient survival and quality of life. This is especially true for our patients, as many of the complications requiring NLP presented later after transplant, when the risk of developing chronic lung allograft dysfunction is also high.
Six of 9 patients who survived to discharge developed significant allograft dysfunction, with an average FEV1 drop of 29% (range 22-41%) at 3 month post NLP, and without clear evidence of infection, airway complications, or acute rejection. The underlying mechanism leading to post-pneumonectomy allograft dysfunction is not completely understood. It has been proposed that several events, including mechanical ventilation and sedation medications, may activate an immunological response [14]. Alongside this, alterations in the immunosuppressive regimen, such as holding or decreasing the calcinueurin-inhibitors (sometimes inevitable in the context of aggressive malignancy or infection), potentially contribute to lung allograft function decline. Subclinical infection and deconditioning frequently accompany the hospital stay and further worsen the situation. However, it is important to note that the median time from transplant to pneumonectomy in our study was 54 months (4.5 years). As the risk for chronic lung allograft rejection increases with time from transplant and affects 50% of recipients beyond 5 years, it is unclear whether allograft function decline is related to NLP or rather just the natural history of lung transplantation. Presurgical lung function or % perfusion to the native lung was not predictive of lung function outcomes in our series.
Of the 12 patients in our study, 3 underwent transplant for COPD. One died peri-operatively, while the surviving 2 demonstrated allograft function improvement after NLP. Improvement in lung transplant mechanics after volume reduction from NLP can explain the increased allograft function in this patient population. Previous case reports on lung volume reduction (LVR) in single lung transplant recipients have shown inconsistent survival results, and data on lung function, such as FEV1, after the procedure is lacking. Kroshus, et al. described 3 patients with marked hyper-expansion of the native lung, with mediastinal shifting and compression of the transplanted lung, who underwent successful LVR with multiple wedge resections. Although all patients survived surgery, FEV1 improvement was only observed in 1 patient [15]. Fitton, et al. published their experience with lung resection on 4 single lung transplant recipients. Unfortunately, 2 patients died shortly after the procedure, and no data regarding lung function was provided for the others [12]. Current data regarding the morbidity and mortality of pneumonectomy do not support NLP for the purpose of LVR in single lung transplant recipients with native lung hyperinflation.
Evidence is emerging for the utility and safety of thoracoscopic pneumonectomy in SLT recipients [16]. This procedure is less invasive; however, it is not yet known whether thoracoscopy is better tolerated by SLT recipients, leads to better short term post-operative results, or preserves allograft function.
While our patient cohort is relatively small, this series does shed some light on post-procedure allograft function and outcomes following NLP in transplant recipients. Although NLP has survival benefit for majority of patients there is a possibility of surgery precipitating allograft dysfunction especially for recipients with restrictive lung disease. NLP serves as volume reduction surgery and is beneficial for single lung recipients with underlying emphysema. In our experience, proceeding with NLP remains a case-by-case decision, where the risk of post-procedure complications, including the possibility of allograft loss, is carefully weighed against the primary disease burden and expected survival without surgery.
Native lung pneumonectomy is feasible following single lung transplantation. Despite of increased early mortality it has survival benefit for carefully selected recipients. The impact of NLP on post-surgery allograft function is different. Patients with underlying native lung emphysema benefit from volume reduction. However single lung recipients with restrictive native lung may experience significant decline in allograft function. In our experience, proceeding with NLP remains a case-by-case decision, where the risk of post-procedure complications, including the possibility of allograft loss, is carefully weighed against the primary disease burden and expected survival without surgery.
Victoria Rusanov and Osnat Shtraichman contributed equally to the manuscript.
None.
None.
There are no conflict of interest.