A tissue engineered liver graft will provide a sustainable source of transplantable organs. A porcine liver scaffold was utilized to develop a re-endothelialized revascularized liver capable of sustained in vivo perfusion in a large animal model.
Liver transplantation remains the only definitive treatment for end-stage liver disease. With only 6000 donor livers available each year nationally, the need far exceeds the supply. Innovative cell-based therapies, such as hepatic tissue engineering, offer alternatives to supplement the limited supply of quality organs for transplantation.
Whole livers harvested from 6-10 kg pigs underwent rapid perfusion decellularization resulting in an acellular scaffold with an intact vascular network and capsule. Scaffolds were re-endothelialized with Human or Porcine Endothelial Cells derived from Human or Pig Umbilical Vein (HUVEC or PUVEC cells) at physiologic perfusion pressures. PUVEC seeded grafts were implanted as auxiliary liver transplants in recipient pigs.
Non-endothelialized grafts demonstrated thrombosis at 30 minutes post perfusion with absent vascular inflow. Re-endothelialized grafts demonstrated patent vascular inflow, absence of gross thrombus, and patent post-explant portal venogram after 72 hours of in vivo hemoperfusion. Histologic review demonstrated viable endothelial cells following graft explant.
Vascular patency of a re-endothelialized porcine whole liver graft was achieved in vivo in the absence of systemic anticoagulation. This implantable bioengineered re-endothelialized liver offers early proof of concept of a future unlimited source of patient specific livers for transplantation.
End-Stage Liver Disease (ESLD) is the 8th most frequent cause of death in the United States, accounting for roughly 40,000 deaths; worldwide the impact is even greater with 383,000 deaths in China alone [1]. ESLD has its greatest impact on (45-64)-year-old adults during the peak economic period of their life, with its estimated direct annual cost exceeding $10 billion and 186,000 hospital admissions annually in the United States alone [2]. The only curative therapy for ESLD is currently liver transplantation. Unfortunately, the success of liver transplantation has led to its greatest challenge, the scarcity of transplantable donor organs. In the United States, about 16,000 patients are waiting for liver transplantation, but only about 6,000 of these patients will benefit from transplantation annually. Liver tissue engineering offers a unique potential for an unlimited supply of compatible organs for transplantation.
Alternative strategies for organ procurement have been trialed, including xenogeneic transplant and cell-based transplantation. Application of xenogeneic organs has been limited due to immune rejection and graft thrombosis. Cell-based hepatocyte transplantation offers another alternative to whole organ transplantation; however, limitations in cell supply, cell quality, and poor engraftment, especially under cirrhotic conditions, have limited the clinical efficacy of cell-based therapy. The use of whole organ tissue engineering overcomes these barriers to engraftment and offers potential for an unlimited supply of transplantable organs.
Whole liver tissue engineering utilizes a scaffold or Extracellular Matrix (ECM) to allow for growth and expansion of patient specific cellular material on a background of native organ structure [3]. The use of a porcine decellularized organ matrix preserves the natural ECM proteins and growth factors which allow for cell attachment and proliferation in a native configuration [4]. The presence of an ECM has been demonstrated to support cell mitogenesis and chemotaxis, direct cell differentiation, and support cell remodeling [5]. Prior to the report of perfusion decellularization, decellularized tissues were limited to relatively thin samples (small intestine submucosa, bladder, skin, etc.) [6]. In contrast, whole organ perfusion decellularization has achieved a form of whole organ decellularization that preserves the native three-dimensional structure, mechanical properties, architecture, and vascular network [7,8]. The result is an ideal liver specific scaffold for liver engineering that can be seeded with patient specific endothelial cells, hepatocytes, cholangiocytes, or progenitor cells. This will allow for engineering of a transplantable organ specific to the recipient, circumventing the need for immunosuppression [9]. A critical step in tissue engineering a transplantable liver is the creation of a functional vasculature capable of long-term perfusion following anastomosis. Without an appropriate endothelial lining of the vessels, continuous blood perfusion of the graft in the absence of anticoagulation quickly results in thrombosis. In this study, we report a method for endothelial seeding of a decellularized porcine liver graft and evaluation of seeded grafts under hemoperfused in vivo conditions. The juvenile porcine liver was selected for its anatomical structure similar to that of a human liver. Grafts were seeded in vitro using allogeneic (porcine) umbilical cord derived endothelial cells. The cells provided single layer coverage of vascular structures and the hepatic sinusoids within the graft to reduce the risk of thrombogenesis. We observed no evidence of graft thrombosis in recipient animals after 60 minutes and limited thrombosis at the study endpoint of 72 hours in the absence of chronic anticoagulation.
This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. All animal care was conducted in accordance with the Mayo Clinic Institutional Animal Care and Use Committee Standards. This specific study was approved under protocols A58912 and A49315. All animals were housed, fed, cared for, and euthanized by study personnel and animal husbandry staff of the Mayo Clinic Department of Comparative Medicine.
Donor livers were harvested immediately post-mortem from 10 kg donor pigs obtained from a slaughterhouse, Midwest Porcine Recovery. Animals were sedated and euthanized via lethal direct intracardiac injection of sodium pentobarbital by study personnel. A midline laparotomy was performed immediately following euthanasia. The portal vein was isolated and cannulated with 10 French tubing. The liver underwent in situ perfusion with 1 L of chilled phosphate buffered saline. The hepatic artery and common bile duct were suture ligated. The infrahepatic and suprahepatic IVC were dissected to allow for ample length for post-explant cannulation. The liver was explanted and further perfused with 1 L of phosphate buffered saline and stored at 4 ℃.
The explanted livers were cannulated and continuously perfused at 12 mmHg throughout the decellularization process. The livers were actively decellularized with 0.6% SDS 4-12 liter baths for up to 48 hours, followed by serial washes in sterile water, 0.1% Peracetic Acid (PAA), and Phosphate Buffered Saline (PBS). To reduce the bioburden risk, the decellularization process was done in a class 10,000 cleanroom using aseptic techniques. Isolation of PUVECs Porcine Umbilical Cord Vein Endothelial Cells (PUVECs) was isolated from unrelated donor animals. Only umbilici from type O animals were used to isolate endothelial cells. To isolate PUVECs, an umbilicus was washed with a solution of 4% penicillin streptomycin in PBS until all visible red blood cells were removed. Vessel branches were ligated, and the vessel lumen filled with a digestion solution consisting of 10,000 U of Type 2 collagenase (Invitrogen) and 0.5 g of BSA (Sigma) suspended in 50 mL of PBS. The vessel was incubated for 15 min at 37 ℃. The vessel was massaged to enhance cell release and the cells collected by centrifugation. The digestion was repeated and cultures started independently to isolate those cultures consisting of endothelial cells. Cells were cultured in complete Endothelial Growth Media (EGM-2, Lonza) and passaged at approximately 90% confluence. Endothelial cells were confirmed by cell surface markers (CD-31, manufacture), LDL uptake, and morphology. Cells were grown up in large lots and cryopreserved for re-endothelialization. Prior to re-endothelialization, cells were placed into culture and allowed to fully recover.
Whole decellularized livers were seeded with PUVEC or Human Umbilical Vein Endothelial Cells (HUVEC, Lonza). Whole decellularized livers were cannulated via the hepatic vein and mounted into a sterile bioreactor. The bioreactors were filled with ~750 ml of EGM-2 media and the livers were continuously perfused at 12-14 mmHg over the course of the culture; peristaltic flow rates were adjusted to maintain continuous perfusion pressure. The bioreactors were oxygenated and pO2 levels were monitored to confirm continuos oxygenation. 100 × 106 to 120 × 106 PUVECs between passage 8 and 12 were isolated. The cells were concentrated to 5 × 106 cells/ml and introduced into the hepatic perfusion. 24-48 hours later, the liver was cannulated via the portal vein and seeded with an additional 100 × 106 to 120 × 106 PUVECs between passage 8 and 12 at 5 × 106 cells/ml. Following administration, perfusion was stopped for 30 minutes to enable initial cellular engraftment followed by continuous perfusion at 12 mmHg. 500 ml of media was changed daily for the duration of the experiment to ensure that the cells obtained proper nutrition. Metabolic analysis was performed on the media to ensure glucose remained in the media (Flex-Nova Biomedical). If glucose consumption exceeded 15 mg/hr, 800 ml of media was changed daily. The livers were cultured for 10-21 days with daily media changes.
Recipient pigs were identified as physiologically healthy 30-40 kg animals. These animals underwent midline laparotomy with exposure of the retro peritoneum. All re-endothelialized PUVEC grafts were implanted as auxiliary liver grafts with inflow via end to end anastomosis with the left renal vein and outflow via end to side anastomosis from the donor suprahepatic inferior vena cava to the recipient infrahepatic inferior vena cava (see Figure 1, see supplemental video). A nephrectomy was not performed; inflow to the hepatic graft occurs via the left renal vein. Intravenous heparin was infused via a single dose of 150 units/kg just prior to vascular anastomosis. No additional anticoagulation was administered.
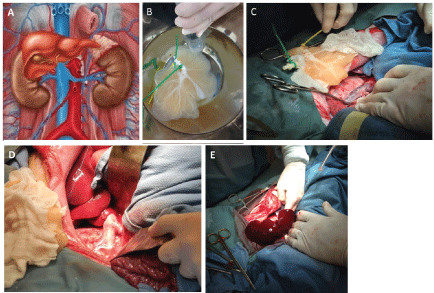 Figure 1: A) Liver grafts were implanted as auxiliary liver transplants with inflow via end to end anastomosis with the left renal vein; B) Endothelialized liver being flushed with saline prior to transplantation; C,D) Recipient inferior vena cava and left renal vein prepared for transplant. The outflow was completed via an end to side anastomosis of the donor suprahepatic inferior vena cava to the recipient infrahepatic inferior vena cava; E) Liver graft following perfusion with the recipient's blood. View Figure 1
Figure 1: A) Liver grafts were implanted as auxiliary liver transplants with inflow via end to end anastomosis with the left renal vein; B) Endothelialized liver being flushed with saline prior to transplantation; C,D) Recipient inferior vena cava and left renal vein prepared for transplant. The outflow was completed via an end to side anastomosis of the donor suprahepatic inferior vena cava to the recipient infrahepatic inferior vena cava; E) Liver graft following perfusion with the recipient's blood. View Figure 1
Vascular patency was assessed intraoperatively with vascular flow probes (FSB-series, Transonic.com) for inflow and outflow venous monitoring. 2-dimension and Doppler ultrasound (S-series, Sonosite.com) was employed to assess graft patency both intraoperatively as well as daily postoperatively.
At the conclusion of the study, recipient animals were euthanized by study personnel and the re-endothelialized graft was explanted. Post explant portal vein was cannulated and CT angiogram was completed. All grafts were fixed in formalin and sectioned for histological analysis with Hematoxylin and Eosin (H&E) as well as immunohistochemical staining with an anti-CD-31 antibody against vascular endothelial cadherin. Histology services were provided by the Mayo Core Histology Laboratory, Rochester MN.
Twelve animals were included in the acute phase study. Three animals received decellularized liver grafts which were not reseeded with endothelial cells (acellular); these animals made up the control group. Six recipient animals underwent auxiliary transplantation of a re-endothelialized liver graft in the acute study arm (60 minute graft implant); three animals underwent auxiliary transplantation of a re-endothelialized liver graft in the chronic study arm (72 hour graft implant).
Perfusion decellularization facilitates rapid access to the whole organ by cannulating the artery and running a mild detergent solution through the native vasculature. This results in rapid decellularization of the organ from the inside-out as illustrated in Figure 2A and Figure 2D, where a whole porcine liver was decellularized in ~24 hours. Histological examination demonstrated the lack of cellular material and nuclei within the decellularized liver with preservation of the overall structure including the lobules and central veins (Figure 2B and Figure 2E). Immunohistochemical staining demonstrates paucity of galactosyl-alpha (1,3) galactose (alpha-Gal) following perfusion decellularization (Figure 2C and Figure 2F). Furthermore, DNA quantification via Picogreen demonstrated a reduction of DNA from 1,477 ± 142 ng DNA/mg tissue (mean ± SEM) in native liver to 19.4 ± 2.9 ng DNA/mg tissue following decellularization.
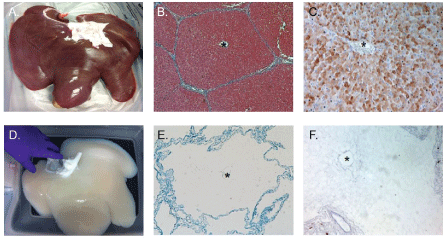 Figure 2: Perfusion decellularization allows for decellularization through the native vasculature, which ensures the rapid decellularization of large organs while maintaining the vascular conduits critical to re-engineering of tissue. A) Isolated porcine liver and D) perfusion decellularized porcine liver containing an intact vasculature and organ capsule. Masson's trichrome staining; B) before and E) after perfusion decellularization demonstrates the lack of cellular material and retention of the native architecture including the (*) central vein within the lobule. Immunohistochemical staining of galactosyl-alpha (1,3) galactose (alpha-Gal); C) before and F) after perfusion decellularization demonstrates the high concentration of alpha-Gal associated with cellular material and its lack of detection within the matrix following perfusion decellularization. View Figure 2
Figure 2: Perfusion decellularization allows for decellularization through the native vasculature, which ensures the rapid decellularization of large organs while maintaining the vascular conduits critical to re-engineering of tissue. A) Isolated porcine liver and D) perfusion decellularized porcine liver containing an intact vasculature and organ capsule. Masson's trichrome staining; B) before and E) after perfusion decellularization demonstrates the lack of cellular material and retention of the native architecture including the (*) central vein within the lobule. Immunohistochemical staining of galactosyl-alpha (1,3) galactose (alpha-Gal); C) before and F) after perfusion decellularization demonstrates the high concentration of alpha-Gal associated with cellular material and its lack of detection within the matrix following perfusion decellularization. View Figure 2
Whole decellularized livers were mounted into bioreactors and continuously perfused at 12-14 mmHg over the course of the culture (Figure 3). During re-endothelialization, HUVEC seeded livers were closely monitored for viability. Glucose consumption increased over the 10-14 culture period with similar kinetics (Figure 3) and served as a nondestructive analytical tool to determine when a graft achieved a desirable re-endothelial level. Grafts consuming a minimum of 10 mg glucose/hr were characterized by H&E staining, fluorescence in situ hybridization, and scanning electron microscopy (Figure 3) and consistently demonstrated a high percent of endothelialization. Further confirmation of vascular patency was accomplished through corrosion casting and CT angiography (Figure 4). The bare matrices pooled contrast primarily in the medial portion of the graft and failed to achieve perfusion at the capillary level. In contrast, the matrices endothelialized with either HUVECS or PUVECs demonstrated uniform filling of contrast within the matrix to the level of the capillaries. HUVEC seeded matrices demonstrated improved coverage and kinetics when compared to PUVEC seeded grafts. HUVEC seeded grafts, however, were not functionally evaluated within the pig model.
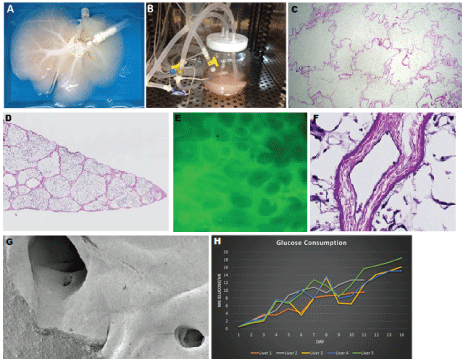 Figure 3: A) Perfusion decellularized whole liver with the portal and hepatic veins cannulated for mounting into the; B) Bioreactor system where the liver is continuously perfused and seeded with endothelial cells; C) Hematoxylin and eosin staining of decellularized liver graft prior to loading with endotheliali cells; D) Hematoxylin and eosin staining of 14 day HUVEC re-endothelialized liver graft demonstrates gross hepatic architecture; E) Fluorescence in situ hybridization demonstrates completely re-endothelialized liver; F) Hematoxylin and eosin stain represents a single monolayer of endothelial cells in the re-endothelialized liver graft (see black arrow); G) Scanning electron microscopy reveals a single flat monolayer of endothelial cells; H) Characterization of glucose consumption rates (mg glucose/hr) of liver grafts seeded with HUVECs. View Figure 3
Figure 3: A) Perfusion decellularized whole liver with the portal and hepatic veins cannulated for mounting into the; B) Bioreactor system where the liver is continuously perfused and seeded with endothelial cells; C) Hematoxylin and eosin staining of decellularized liver graft prior to loading with endotheliali cells; D) Hematoxylin and eosin staining of 14 day HUVEC re-endothelialized liver graft demonstrates gross hepatic architecture; E) Fluorescence in situ hybridization demonstrates completely re-endothelialized liver; F) Hematoxylin and eosin stain represents a single monolayer of endothelial cells in the re-endothelialized liver graft (see black arrow); G) Scanning electron microscopy reveals a single flat monolayer of endothelial cells; H) Characterization of glucose consumption rates (mg glucose/hr) of liver grafts seeded with HUVECs. View Figure 3
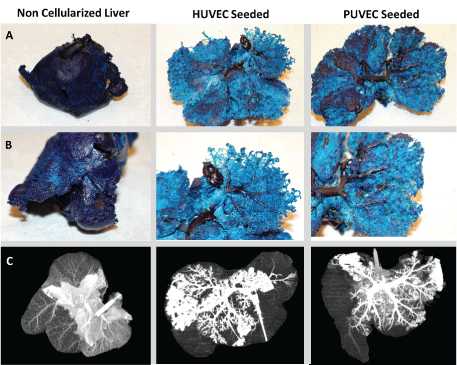 Figure 4: Complete endothelialization to the capillary level is confirmed through casting (Rows A and B) and CT angiography (row C). The non-endothelialized grafts primary pooling of the contrast within the central parenchyma with minimal contrast reaching the outer lobules. The HUVEC and PUVEC seeded grafts both demonstrate improved perfusion throughout the majority of the graft with minimal pooling of contrast. View Figure 4
Figure 4: Complete endothelialization to the capillary level is confirmed through casting (Rows A and B) and CT angiography (row C). The non-endothelialized grafts primary pooling of the contrast within the central parenchyma with minimal contrast reaching the outer lobules. The HUVEC and PUVEC seeded grafts both demonstrate improved perfusion throughout the majority of the graft with minimal pooling of contrast. View Figure 4
All animals in the control (acellular graft) arm of the study thrombosed their grafts within 30 minutes of reperfusion. These animals were immediately euthanized. In contrast, all animals in the acute and chronic study arms with PUVEC seeded grafts demonstrated patent graft flow at the conclusion of the study period with blood flow of 35-40 mL/min across the portal vein inflow and hepatic vein outflow anastomoses. Three animals in the acute study developed intraoperative bleeding complications while investigators developed the surgical implant technique. Two of these intraoperative complications were capsular tears resulting in bleeding from the graft capsule. Capsular tears were overcome by enclosing the graft in moistened sterile gauze while completing the anastomosis to prevent inadvertent injury. The third animal had uncontrolled bleeding from a coronary vein which was not identified at the time of donor hepatectomy and therefore not controlled prior to re-endothelialization.
All animals undergoing chronic (72 hour) perfusion studies were allowed to recover from general anesthesia and resume normal activities. These animals demonstrated no adverse consequences from the transplanted liver graft. Liver allograft perfusion was monitored with Doppler ultrasound on a daily basis to confirm patency.
At explantation, all acute liver grafts were studied with a portal venogram to confirm vascular patency (Figure 5). Non-endothelialized grafts demonstrated extravasation of contrast and loss of hepatic vascular structure. In contrast, endothelialized grafts demonstrated normal vascular structure to the level of the capillaries without contrast extravasation. At 72 hours, the endothelialized grafts remained patent with vascular patency and hemoperfusion confirmed by both Doppler ultrasound prior to explantation and by portal venogram post explantation (Figure 6). Histologically, endothelial cells were demonstrated lining the vasculature via H&E and CD-31 staining (Figure 6) with localization of Red Blood Cells (RBCs) within the parenchymal space, given the absence of hepatocytes within the graft.
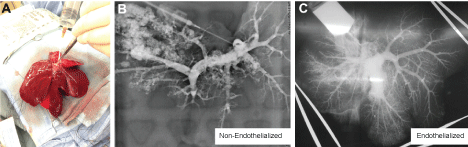 Figure 5: A) Explanted liver graft from acute transplantation; B) Resulting venogram of non-endothelialized liver graft that demonstrates limited perfusion and significant leakage of contrast when compared to; C) the endothelialized liver graft, which demonstrated global perfusion with continued capillary bed resolution following acute transplantation. View Figure 5
Figure 5: A) Explanted liver graft from acute transplantation; B) Resulting venogram of non-endothelialized liver graft that demonstrates limited perfusion and significant leakage of contrast when compared to; C) the endothelialized liver graft, which demonstrated global perfusion with continued capillary bed resolution following acute transplantation. View Figure 5
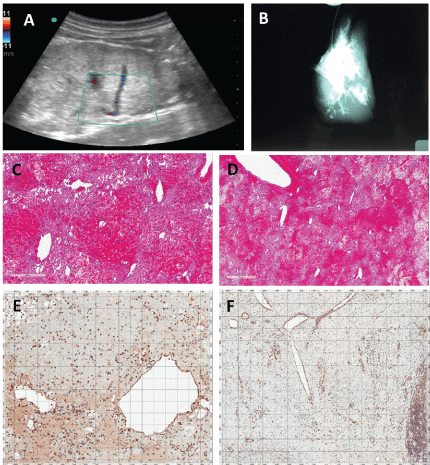 Figure 6: Images of Graft after 3 days of in Vivo Hemoperfusion. A) Doppler ultrasound demonstrates patency of the portal vein inflow and inferior vena cava (outflow) at 72 hours post implantation; B) Venogram of explanted graft at 72 hours confirming ultrasound data through the visualization of the perfusion bed; C,D) Histologic examination by H&E staining demonstrated the filling of the parenchyma niche with RBCs, likely leading to the reduced overall perfusion. However, a large perfusion bed still exists at 72 hours demonstrating the ability to maintain continuous hemoperfusion, and allowing for the addition of hepatocytes in future grafts; E,F) Histologic examination by CD31 (Dako - JC70A clone) staining demonstrated endothelial cell coverage of vascular structures after continuous hemoperfusion at 72 hours. Panels C and E are high power (bar = 300 microns). Panels D and F are low power (bar = 1 mm). View Figure 6
Figure 6: Images of Graft after 3 days of in Vivo Hemoperfusion. A) Doppler ultrasound demonstrates patency of the portal vein inflow and inferior vena cava (outflow) at 72 hours post implantation; B) Venogram of explanted graft at 72 hours confirming ultrasound data through the visualization of the perfusion bed; C,D) Histologic examination by H&E staining demonstrated the filling of the parenchyma niche with RBCs, likely leading to the reduced overall perfusion. However, a large perfusion bed still exists at 72 hours demonstrating the ability to maintain continuous hemoperfusion, and allowing for the addition of hepatocytes in future grafts; E,F) Histologic examination by CD31 (Dako - JC70A clone) staining demonstrated endothelial cell coverage of vascular structures after continuous hemoperfusion at 72 hours. Panels C and E are high power (bar = 300 microns). Panels D and F are low power (bar = 1 mm). View Figure 6
After 72 hours the patient was readmitted due to severe abdominal pain, constant, fever and malaise. Laboratory tests with 800 leukocytes, hemoglobin of 10 g/dl, platelets 235,000 and preserved renal function (creatinine 1.0 mg/dl). CT scan was updated finding a pelvic tumor adjacent to the renal graft of 5 × 8 × 6 cm, with enhancement in the arterial phase, establishing the diagnosis of renal graft aneurysm, which involved the right external iliac artery (Figure 2 and Figure 3).
Advances in the field of liver transplantation have made it a highly successful treatment for ESLD, but one that is limited by the scarcity of transplantable donor organs: yearly, only 38% of the 16,000 waitlisted patients in the United States will receive a liver. With demand for transplantable organs projected to rise by 23% in the next 20 years [10], the gap between waitlist needs and organs available for transplantation will only increase. For this reason, alternative paradigms to traditional whole organ liver transplantation have been envisaged, including tissue engineering of transplantable liver grafts.
Tissue engineering not only provides an opportunity to bypass the transplantable organ deficit, but foreseeably offers some advantages to traditional whole organ transplantation, as well. For example, the possibility of reseeding the graft with autologous cells through stem cell technology eliminated the need for immunosuppression, with the added clinical and economic benefits that this entails. Immunogenicity of the actual decellularized scaffold has also been investigated, showing no increased immune response with neither allogeneic nor xenogeneic transplants [11].
Several groups, including ours, have been successful in generating large-scale organ scaffolds though SDS-based whole-organ perfusion decellularization techniques [11,12]. This method preserves essential ECM proteins for cell engraftment and function, while retaining the organ's native vascular structures that can be anastomosed to the recipient circulation [13], and is therefore superior to other techniques, such as the implantation of hepatic tissue sheets in a vascularized subcutaneous cavity [14]. Preservation of the native vascular and microvascular network in the liver is especially important due to the high oxygen demands of functioning primary hepatocytes and their consequent susceptibility to ischemic damage [15].
A major challenge of tissue engineering a transplantable liver is developing a vasculature that perfuses the graft adequately and without thrombosis. This has limited most attempts at transplantation of whole-liver scaffolds to a few hours [16]. An encouraging alternative to anticoagulation, which is associated with post-surgical hemorrhagic complications, is the seeding of the scaffold's vascular bed with endothelial cells, or re-endothelialization. If 100% coverage is achieved, and contact between blood cells and the exposed collagen in the scaffold is avoided, thrombosis should theoretically be prevented [17]. This strategy has been tested in bioengineered kidney grafts with promising results [18]. Other methods, such as layer-by-layer heparinization of decellularized liver matrices, have not yielded results as successful [19].
The current study addresses a novel approach to tissue engineering a transplantable liver in a large animal model. It represents the first large animal study of a re-endothelialized liver matrix demonstrating vascular patency at 72 hours post implantation without use of chronic anticoagulation. Previous implantation attempts have utilized rodent models which cannot be directly translated to human applications [20], relied on chronic anticoagulation, or achieved shorter in vivo graft survival times. Ko, et al. re-endothelialized livers utilizing immobilization of CD-31 antibodies which can ultimately lead to adhesion of endothelial cells within non-vascular structures, thus altering the ECM. These re-endothelialized livers were transplant into pig models through portal vein to renal artery anastomosis following nephrectomy [21]. Which would not be suitable for clinical application, as it requires a nephrectomy and the long term consequences of arteria flow in the liver's portal system are unknown. Even so, this study only able to demonstrated patency for 24 hours. Furthermore, there is no mention of anticoagulation in this study, although their previous work with renal scaffolds involved the use of salicylic acid and clopidogrel [22]. Our post explant studies have demonstrated preservation of the liver architecture with functional hepatic vasculature at 72 hours post perfusion after auxiliary liver transplant with inflow via end to end anastomosis with the left renal vein.
We have demonstrated that re-endothelialization with Porcine Umbilical Cord Derived Endothelial Cells (PUVECs) results in a single monolayer on H&E and SEM which is capable of in vivo vascular perfusion to the capillary level on portal venogram. We postulate that this will provide for adequate oxygen and nutrient delivery to support hepatocyte seeding and development.
This study represents the foundational study for the development of a large animal transplantable liver graft. Our ongoing work is directed at optimizing hepatocyte seeding and biliary reconstruction. We postulate that with completely re-endothelialized liver vasculature, the graft will be capable of providing for the intense oxygenation needs of functioning hepatocytes.
In summary, this study represents a novel large animal model of a tissue engineered liver capable of sustained perfusion within a large animal recipient. This study will provide the foundation for future graft implants seeded with hepatocyte and liver parenchymal cells.
We thank Scott Thompson, MD, PhD and Gregory Michalak (Mayo Clinic, Rochester) for imaging support and the Mayo Clinic Rochester Core Histology laboratory.
Supplemental video: Liver graft.