Lipoid pneumonia is an uncommon non-infectious condition that results from the accumulation of lipids of animal, vegetable or mineral origin, in the lungs [1,2]. Most often it is a result of aspiration from altered swallowing or cough mechanism. Children with palatal abnormalities debilitated older adults or adults with neurological or gastrointestinal disorders are commonly affected [1,3,4]. Lipoid pneumonia should be considered as a differential for a lung cavity lesion, especially in patients at risk for aspiration. Such lesions can persist on long-term follow up. However, infection and malignancy should be ruled out.
Here, we present an atypical and rare radiographic presentation of lipoid pneumonia, where in patient presented with complaints of shortness of breath and productive cough. Chest imaging showed air space disease in the RUL with a central cavity lesion. Transbronchial biopsy of the lesion demonstrated features of lipoid pneumonia and was felt to be secondary to his chronic aspiration. Infectious work up was negative. Despite being on strict aspiration precautions, follow up CT thorax remonstrated the cavity in the right upper lobe. Repeat biopsy of the lesion showed the features of lipoid pneumonia.
Lipoid pneumonia is believed to be secondary to aspiration given his history of mental retardation and altered mental status. Patient was deemed to be a high-risk candidate for lung resection surgery given the altered mental status and contractures and is being clinically monitored.
Lipoid, Pneumonia, Aspiration
BUN: Blood Urea Nitrogen; CT: Computed Tomography; RUL: Right Upper Lobe; RML: Right Middle Lobe; AFB: Acid Fast Bacilli; PEG: Percutaneous Endoscopic Gastrostomy; HU: Hounsfield Units; NTM: Non-Tuberculous Mycobacteria; NPO: Nothing Per Oral
Lipoid pneumonia is an uncommon non-infectious condition that results from the accumulation of lipids of animal, vegetable or mineral origin, in the lungs [1,2].
Here we present an atypical and rare radiographic presentation of lipoid pneumonia and its long term follow up over 5 years.
A 44-year-old male with medical history significant for hiatal hernia, gastroesophageal reflux disease, and mental retardation and diffuse body contractures, a resident of group home, presented to the hospital with complaints of shortness of breath and productive cough. Patient did not have any fever, chills or night sweats. Patient never smoked, no history of alcohol use or any recreational drug use. He lives in a group home, given his altered mental status and contractures. On presentation, patient was afebrile, blood pressure was 144/86, respiratory rate of 20 and oxygen saturation 95% on room air. Patient was alert, not oriented and not in distress. Physical exam was significant for rhonchi on right side. Diagnostic studies revealed sodium 140, potassium 3.7, chloride 103, bicarbonate 24, BUN 23 and creatinine 0.9, white blood cell count 6.7, hemoglobin 12.8, hematocrit of 37.9 and platelets of 144. Blood cultures and sputum cultures were negative. Chest X ray demonstrated focus of airspace disease within the right mid-lung. CT Thorax showed air space disease in the RUL with a central cavitary lesion measuring up to 2 centimeters with patchy densities in the inferior part of RUL and RML (Figure 1). Mediastinal nodes measuring up to 1.1 centimeters in right para tracheal and sub-carinal station were noted.
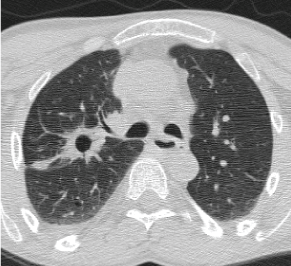 Figure 1: 2011 CT Thorax showing a thick walled cavitary lesion with a 2.4 × 1.5 cm in the RUL posterior segment. View Figure 1
Figure 1: 2011 CT Thorax showing a thick walled cavitary lesion with a 2.4 × 1.5 cm in the RUL posterior segment. View Figure 1
He underwent bronchoscopy with RUL posterior segment transbronchial biopsies. Histopathology demonstrated alveolated parenchyma with lipid-laden histiocytes raising suspicion for lipoid pneumonia and was felt to be secondary to his chronic aspiration (Figure 2). Bronchial washings, brushings and transbronchial biopsies were negative for AFB, bacterial and viral cultures. He was started on strict aspiration precautions and eventually had a PEG tube placed. He was followed up closely for several years with repeat imaging stable, with no progression of previously identified lesions. However, after five years, he presented to pulmonary clinic with complaints of persistent and progressively worsening cough. CT thorax remonstrated the cavity lesion in the right upper lobe. He was treated with a prolonged course of antibiotics. Repeat CT thorax demonstrated increasing size of the lung cavity (Figure 3). Therefore, repeat bronchoscopy and RUL transbronchial biopsies were performed and pathology once again showed large collections of vacuolated histiocytes and multinucleated giant cells associated with chronic inflammation and some fibrosis consistent with exogenous lipoid pneumonia (Figure 4). This was again felt to be secondary to his chronic aspiration given his mental retardation. His gastric feeding tube was advanced to his jejunum and aspiration precautions were again emphasized as treatment. Patient was referred to thoracic surgery for resection of lung cavity. However, patient was deemed to be a high-risk candidate for surgery given the mental retardation and body contractures. Patient is currently being monitored clinically along with radiographic surveillance.
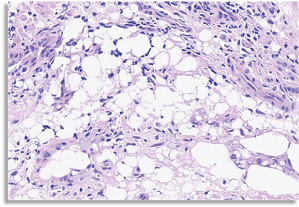 Figure 2: 2011 RUL Posterior segment transbronchial biopsies showing alveolated parenchyma with large collections of vacuolated histiocytes and multinucleated giant cells associated with chronic inflammation. View Figure 2
Figure 2: 2011 RUL Posterior segment transbronchial biopsies showing alveolated parenchyma with large collections of vacuolated histiocytes and multinucleated giant cells associated with chronic inflammation. View Figure 2
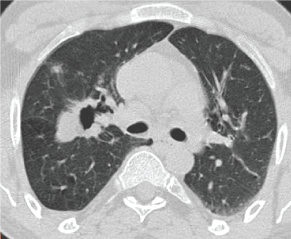 Figure 3: 2016 CT Thorax showing a persisting thick walled cavitary lesion with a 2.4 × 1.5 cm in the RUL posterior segment. View Figure 3
Figure 3: 2016 CT Thorax showing a persisting thick walled cavitary lesion with a 2.4 × 1.5 cm in the RUL posterior segment. View Figure 3
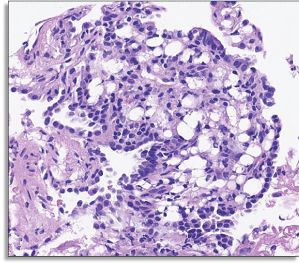 Figure 4: RUL Posterior segment transbronchial biopsies showing alveolated parenchyma with large collections of vacuolated histiocytes and multinucleated giant cells associated with chronic inflammation and some fibrosis. View Figure 4
Figure 4: RUL Posterior segment transbronchial biopsies showing alveolated parenchyma with large collections of vacuolated histiocytes and multinucleated giant cells associated with chronic inflammation and some fibrosis. View Figure 4
Lipoid pneumonia is an uncommon non-infectious condition that results from the accumulation of lipids of animal, vegetable or mineral origin, in the lungs [1,2]. Most often it is a result of aspiration from altered swallowing or cough mechanism. Children with palatal abnormalities debilitated older adults or adults with neurological or gastrointestinal disorders are commonly affected [1,3,4].
Depending on the source of the fat like compound, lipoid pneumonia can be classified into endogenous or exogenous forms. Endogenous form of lipoid pneumonia is a result of accumulation of endogenous lipids derived from the lung parenchyma [5,6]. It is seen in conditions involving alveolar cell wall destruction. Exogenous lipoid pneumonia is the result of inhalation of oils present in food, radiographic contrast media, or oil-based medications such as laxatives [1,7,8].
These substances upon aspiration into the lung elicit a foreign body reaction, inflammation and subsequent fibrosis [9]. Based on the duration of exposure, exogenous lipoid pneumonia can be classified into acute and chronic forms. Clinical presentation can range from asymptomatic to severe, life-threatening respiratory failure [10]. Most commonly affected patients present with chronic cough and dyspnea [11]. Physical examination is usually non-specific, may be normal or may present dullness to percussion, crackles, wheezes, or rhonchi [12].
Lipoid pneumonia commonly involves the lower and middle lobes [1,13,14]. The radiographic findings are highly variable, nonspecific and can present as focal or diffuse air space process, mass-like lesions, ground glass opacities, reticulonodular pattern, mixed alveolar and interstitial opacities, crazy paving pattern or nodular lesions or thin-walled cysts (pneumatoceles) and pleural effusions may also be seen [15-17]. However, when present, areas of fat attenuations with values between -150 HU and -30 HU in consolidations/air space process is a characteristic feature of lipoid pneumonia [2,15,18-21]. Cavitation is occasional [15].
Magnetic resonance imaging shows a high-intensity signal on T1-weighted images and decreased signal on T2-weighted images, suggestive blood or fat [22,23].
Routine laboratory test results are usually normal or can be non-specific with leukocytosis and elevated erythrocyte sedimentation rate, especially when the lipoid pneumonia is associated with super infection [24]. Patients may be hypoxemic depending on the extent of involvement [16,25]. Pulmonary function test may be normal, or may have a restrictive pattern [9,16,26-28].
Definite diagnosis of lipoid pneumonia can be established based on history and presence of lipid-laden macrophages on BAL and transbronchial biopsy in the appropriate clinical setting [5,7,29].
Histopathologic changes include lymphocytic infiltration, lipid-containing macrophages, granulomas and fibrosis. BAL is usually turbid with fat globules floating on the surface [12,13,20].
Cytological demonstration of extracellular oily droplets is more specific for exogenous lipoid pneumonia. It's the size of the cytoplasmic vacuoles in the foamy macrophages that differentiate the exogenous and endogenous forms of lipoid pneumonia. Intracytoplasmic vacuoles are small in endogenous form while larger in the exogenous form [12,30].
Various fat stains are used to identify the intracytoplasmic vacuoles in the foamy macrophages and include Sudan III, Sudan IV, and Oil Red O.
Histopathologic findings include presence of lipid-laden macrophages in the alveoli and interstitium in association with presence of lipid material, inflammatory cells, and variable amount of fibrosis [1,7].
Presence of lipid enhances the growth of certain microorganisms by impeding the phagocytic process by host macrophages. Though rare, subjects diagnosed with lipoid pneumonia are at risk for non-tuberculous mycobacteria, aspergillus and nocardia infections [11,31].
Almost all cases reported in literature with cavitary lipoid pneumonia are associated with infection. Hadjiliadis D, et al. reported fourteen patients with M.fortuitum infection, the six patients with M. chelonae infection and are much more common in patients with esophageal disorders [32,33]. There was only one reported case of NTM infection and Aspergillus fumigatus infection [34].
These patients can also develop hypercalcemia given the granulomatous nature of the disease and the propensity for calcitriol production by the inflammatory cells as seen in other granulomatous disorders [35,36].
There is a theoretical risk for lung cancer development in patients with lipoid pneumonia however the study that evaluated 100 patients with lipoid pneumonia over 20 years did not reveal any lung cancer in those patients [37].
The natural history and outcomes are variable, and patients can develop fibrosis eventually or can have complete resolution of clinical symptoms and radiological findings. Severe respiratory failure requiring mechanical ventilation is seldom required unless it is associated with massive aspiration. Most of the current evidence and literature is based up on the case reports.
The treatment of lipoid pneumonia is mainly supportive and comprises discontinuing exposure to the offending agent and implementing measures to avoid aspiration, oxygen supplementation, treating superimposed infection [12,38]. Role of steroids in the treatment of lipoid pneumonia is controversial and is usually reserved for cases with severe inflammation [39].
Our case is unique in that the lipoid pneumonia persisted and progressed despite keeping patient NPO without any evidence of malignancy or super imposed infection, in contrast, with the so far reported such lipoid pneumonia cases.
Lipoid pneumonia should be considered as a differential for a lung cavity lesion, especially in patients at risk for aspiration. Such lesions can persist on long-term follow up. However, infection and malignancy should be ruled out. Thoracic surgery evaluation is recommended for possible resection of such lesions to avoid further super imposed infections. Our case is unique in its presentation as a lung cavity lesion without any co existent infection; in contrast, with the so far reported such lipoid pneumonia cases are associated with either malignancy or infection.