Osteoporosis means "Porous bone" is a disease characterized by progressive bone thinning. The deterioration of bone tissue can lead to bone fragility and fracture, especially of the hip, spine, shoulder and wrist. Osteoporosis is characterized by decreasing bone mineral density (BMD). Bisphosphonates are the most commonly prescribed drugs for the treatment of osteoporosis in the US and many other countries including India. Alendronate (Aln) is a widely used anti-osteoporosis drug, exhibits strong inhibitory effect on bone resorption performed by osteoclast cells. Alendronate-sodium is a BCS class III bisphosphonate, used in the treatment of osteoporosis, acts as a potent, specific inhibitor of osteoclast-mediated bone resorption. Alendronate was the first bisphosphonate to be approved for osteoporosis in the US in 1995.
The objective of the present study is to develop, optimize, and evaluate Alendronate-loaded chitosan nanoparticles (NPs) for the treatment of osteoporosis. NPs were prepared by the Ionic gelation method and optimized for various parameters. The prepared nanoparticles were characterized using particle size analyser (DLS), transmission electron microscopy (TEM), scanning electron microscopy (SEM) and fourier-transform infrared spectroscopy (FTIR).
Formulated NPs were obtained in the average size ranging from 60 nm to 220 nm in TEM, SEM and DLS studies. The release profile was depended on the dissolution medium. The proposed nanoparticles offer an interesting alternative for alendronate delivery via the oral route. Our results indicated that alendronate-loaded chitosan nanoparticles provide an effective medication for the treatment of osteoporosis.
Bone mineral density (BMD), Drug loaded nanoparticles, Ion gelation, Osteoporosis, Release kinetics
Osteoporosis is the second most common bone disease with low bone mineral density (BMD) and structural deterioration of bone. In this disease bones become more fragile and porous, it results the reduced bone strength and increased fracture risk. The most effected bones are the hip, spine, shoulder and wrist bones [1]. Decreasing bone density is the main cause of Osteoporosis. The hip and spinal fractures are the most dangerous aspect of osteoporosis. It is commonly seen in old age but women have the high risk after menopause called postmenopausal osteoporosis (PMO). It causes Stooped posture, loss of height and chronic pain with resultant reduction in mobility [2].
There are many risk factors of osteoporosis on the basis of age group and different backgrounds. But these factors may be reason of osteoporosis. Some factors can be controlled and some cannot be controlled are following summarized in Table 1 mentioned by S. Misner, et al. [3,4].
Table 1: Risk factors of osteoporosis. View Table 1
Bisphosphonates are the most commonly used drugs for the treatment of osteoporosis; the efficacy of this drug class for reducing the risks of osteoporosis has been well established in large clinical trials. The strong affinity of bisphosphonates to hydroxyapatite, their relative low toxicity, and their regulatory approval make them agents of choice for bone targeting [3]. Bisphosphonates are used to treat osteoporosis in the US and many other countries including India. Alendronate was the first FDA approved bisphosphonate drug for treatment of osteoporosis in the US in 1995. The osteoclast cells are responsible for bone resorption and caused osteoporosis, so the bisphophonate drugs target to these cells and inhibits the activity of bone resorption. Alendronate is the most potent and recommended drug for the treatment of osteoporosis in medical trials [5]. Alendronate is highly efficient but presents low absorption after oral administration, due to high water solubility fast release occurs. Low systemic bioavailability and burst release of the drug inside the body are main challenges in Alendronate drug delivery systems. Drug nanocarriers help in reducing toxicity, improving solubility and bioavailability, enhancing release and provide better formulation opportunities for drugs [6,7].
Nanotechnology-based drug delivery systems, i.e. polymeric nanoparticles (NPs), solid lipid NPs, liposomes, nanoemulsions, nanosuspension, and micelles etc. have been proved a promising approach for controlled, sustained, and targeted drug delivery in treatment of many diseases. Chitosan (CS) exhibits excellent biocompatibility and biodegradability, and has been extensively used as a carrier for drug delivery systems [8-10].
There are a number of drugs whose clinical development failed due to low bioavailability and other poor biopharmaceutical properties, so presently research work is focused on resolving these problems. The major task in the development of these drugs is the improvement in solubility, thereby enhancing oral bioavailability. The most frequently applied nanotechnology-based strategy in the development of drug delivery systems is polymeric nanoparticles (NPs) based formulations, which present a significant approach for enhancing solubility and oral bioavailability of drugs [11-13].
Chitosan is an important polymeric carrier for many drugs because of its specific properties such as polycationic nature, biodegradability, biocompatibility and non-toxic nature. Chitosan is a natural polysaccharide derived by the process of deacetylation of chitin. Ion gelation is the commonly used method for the formulation of CS NPs. The electrostatic interaction between the amine group of chitosan and a negatively charged group of polyanion such as sodium tripolyphosphate (TPP) is the basic mechanism of this method. In many previous studies, it is reported that different drugs loaded CS nanoparticulate formulations are stable, permeable and therapeutically active [14,15].
In the present study the authors have developed Alendronate loaded CS NPs for better bioavailability and acceptability to the biological systems. Drug delivery systems are designed for promoting the therapeutic effect of a drug and minimizing its toxic side effects, which is achieved by different process variables. The NPs were prepared by Ionotropic gelation method and were characterized for average particle size, surface charge, size distribution, drug entrapment efficiency and in vitro drug release. The effect of CS concentration, TPP concentration and stirring speed on the drug entrapment efficiency and particle size was evaluated. Drug delivery systems are designed for promoting the therapeutic effect of Alendronate drug and minimizing its toxic side effects, which is achieved by different process variables. Different mathematical models were used for drug release kinetics study of formulated drug loaded NP [16-19].
The drug Alendronate-sodium (M.W. of 249.09 g/moL) was procured from Sigma Aldrich. (Mumbai, India). CS with medium molecular weight (M.W. = 750,000 Da) was purchased from Himedia (India). Dialysis membrane (Mol. wt. cut-off: 12,000 Dalton, flat with 25 mm, diameter of 16 mm) was purchased from Himedia (India). High purity water was used for all experiments, prepared by using (Millipore). All other chemicals and reagents were of analytical grade.
Alendronate loaded CS NPs were prepared by the ionic gelation method [20-24]. In this method, CS-NPs were obtained upon the addition of aqueous sodium-tripolyphosphate (TPP) solution to acidic CS solution stirred at room temperature. NPs formation was the result of ionic interaction between positively charged amino groups of CS and negatively charged TPP. In this method, CS was dissolved in (2% w/v) acetic acid solution and stirred to complete dissolution. This acidic chitosan solution was filtered with 0.2 µ filter paper and pH was adjusted up to 4.8-5.0 by adding 0.1M NaOH solution. A fix amount of drug (20 mg) was added in CS solution after adjusting the pH and Pluronic F-68 was added as surfactant The CS-NPs were prepared by the drop wise addition of TPP solution to chitosan solution at room temperature on magnetic stirring for 3-4 hours. The process variables such as CS concentration, TPP concentration, stirring speed and pH for nanoformulations were optimized. The prepared NPs suspension was analysed by transmission electron microscopy (TEM) and Dynamic light scattering (DLS) for particle size. The optimized NPs suspension was centrifuged at 15000 rpm for 30 min using cooling centrifuge (C24, Remi Centrifuge, and Mumbai (India). The pellets were freeze-dried and stored at 5 ± 3 ℃. The weights of freeze-dried nanoparticles were also measured. The entrapment efficiency (%) was analysed by UV spectrometer for the supernatant. Centrifuged NPs were lyophilized by using lyophilizer (LABCONCO, GNCIIM) for 36 h.
The Entrapment efficiency (EE) of Alendronate loaded CS NPs was determined by the indirect method. The nanoparticles were centrifuged at 15,000 rpm for 30 min and the pellet of NPs was collected and the supernatant was separated. The amount of unentrapped drug in the supernatant was determined by using the method developed by Ostovic, et al. and used by (Cohen-Sela, et al. [25,26]. EE% was determined at 240 nm wavelength following the addition of copper (II) reagent (5 mM copper sulfate in 1.5 × 10-3 M HNO3) by complexation between Alendronate and copper ions. Encapsulated drug amount was obtained by using UV-Visible spectrophotometer (UV-1800 Shimadzu, Japan)) after proper dilution. The percentage entrapment efficiency (% EE) was calculated by using the following formulae:-
Particle size, Poly dispersive index (PDI) and zeta potential (ZP) of formulated Alendronate loaded CS NPs were determined through dynamic light scattering analysis (DLS) with Malvern Zetasizer Nano S (Malvern, UK).
Surface morphology of the best formulation was carried out using scanning electron microscope (SEM) using (Nanosem, Quantum 200E Instrument). Formulated NPs were also confirmed using Transmission Electron Microscope (TEM) for surface morphology. The prepared sample was examined by TEM (Morgagni 268D TEM, Boston, MA).
The interaction between drug and polymer was identified from the Fourier transform-infrared, Attenuated total reflection FTIR (ATR-FTIR, Bruker Tensor-37) studies. The FTIR spectrum of pure drug Alendronate, polymer (chitosan), and Alendronate loaded CS NPs were obtained. The samples were prepared by grinding with anhydrous KBr powder and compressed into pellets The FTIR spectra of drug and drug-loaded NPs were measured over the range of 4000-400 cm-1.
The drug release of NPs was studied using dialysis bag method [27,28]. The membrane with a pore size of 2.4 nm and molecular weight cut-off between 12,000 and 14,000 in phosphate buffer saline (PBS) pH 6.8 at 37 ± 2 ℃ was used. The drug-loaded NPs were placed into a dialysis membrane, tied at both the ends and placed in a beaker containing 100 mL of diffusion medium (PBS pH 6.8). Temperature and speed were maintained at 37 ± 2 ℃ and 100 rpm, respectively, using magnetic stirrer. Aliquot samples were withdrawn at predetermined time intervals, and the same volume was replaced with fresh buffer to maintain the sink condition. The amount of drug released was analyzed spectrophotometrically at 240 nm (complex formation method) for Alendronate drug. Cumulative percentage release was calculated from the amount of drug release. The release kinetics were determined by some mathematical kinetic equations such as zero order, first order, Higuchi's model and Korsmeyer-Peppas model. Values of R2 and K were calculated from the linear curve obtained by regression analysis of the plots.
In the current study, Alendronate loaded CS NPs were prepared by Ionic gelation method using polymer (Chitosan), a cross-linking agent sodium tri polyphosphate (TPP) and surfactant (Pluronic F-68). Different formulations were prepared to optimize the process variables i.e. CS concentration, TPP concentration and pH etc. The experimental results from analysis of above formulations, process variance are found to be significant and the optimized formulation is obtained. The results of average particle size and analysis reveal that changing the concentration of CS from 0.5 mg/ml to 2 mg/ml have shown slight increase in the particle size, due to the increasing viscosity of chitosan solution. The most significant change in the particle size was observed due to increasing concentration of TPP, it was increased the cross-linking density of polymer and drug - polymer electrostatic interaction.
The optimized formulation of Alendronate loaded CS NPs was selected on the basis of the minimum value of particle size and the maximum value of entrapment efficiency. The Optimized formulation has shown average particle size of 214.6 nm and maximum drug entrapment 66.82%. These results showed the best condition for preparing the optimized formulation of Alendronate loaded CS NPs. The above optimized formulation was considered for further studies i.e. characterization and in-vitro drug release kinetic studies.
Fourier transform infra red (FTIR): The FTIR spectra of pure drug Alendronate, polymer Chitosan and drug loaded NPs were obtained by using FTIR to characterize the chemical structure of biopolymer and nanoparticle and obtained spectra are shown in Figure 1a, Figure 1b and Figure 1c.
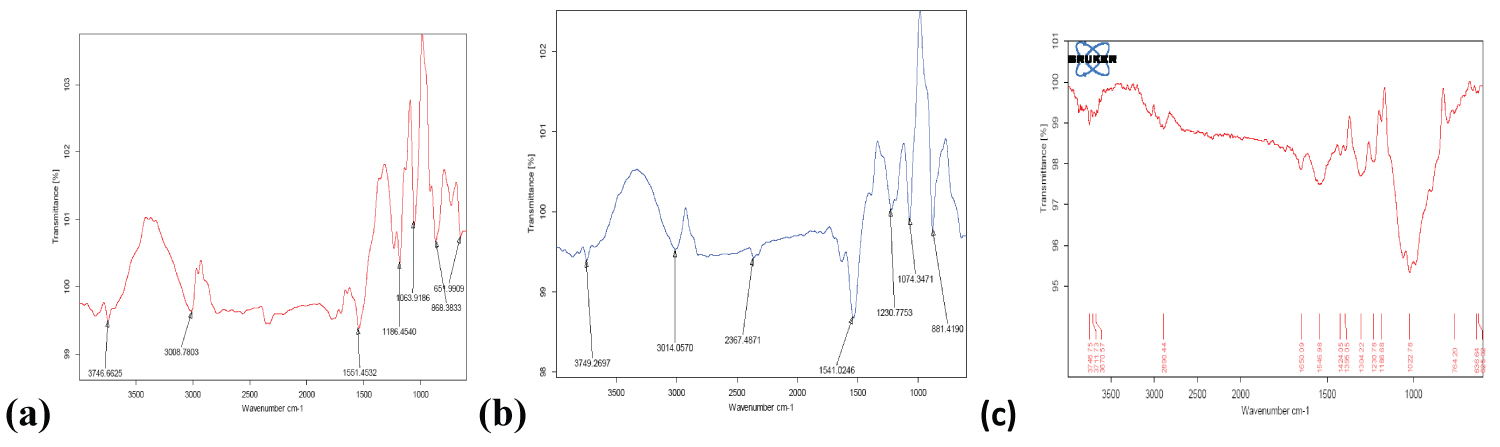 Figure 1: FTIR spectra of (a) Alendronate (API); (b) Alendronate CS NPs; (c) Chitosan.
View Figure 1
Figure 1: FTIR spectra of (a) Alendronate (API); (b) Alendronate CS NPs; (c) Chitosan.
View Figure 1
The main characteristic peaks of Alendronate- sodium shows at 1186.45 cm-1 and 1063.91 cm-1 (in the region 1200-900 cm-1) correspond to (C=O stretching), and (P=O stretching) respectively. FTIR spectra of Chitosan shows the characteristic peaks at 3370.57 cm-1 (OH and NH2 stretching) and 2890.44 cm-1 due to C-H stretching. The spectrum of drug loaded NPs shows new characteristic absorption peak at 1230.77 cm-1 for (C-O-C stretching) and 1541.02 cm-1 for (amide II) which indicated the electrostatic interaction between amine groups of chitosan and phosphoric groups of sodium tri poly phosphate (TPP). A new peak at 881.41 cm-1 confirmed the encapsulation of drug Alendronate inside the NPs [29-31].
Nanoparticle size: Alendronate loaded CS NPs were prepared by the ion gelation method. This method was used to prepare the desired NPs and considered to be adequate nanocarriers for the encapsulation of drug due to its easy methodology, cost-effectiveness and non-toxic nature. The prepared Alendronate loaded CS NPs were found in nano range and their spherical shapes were confirmed using TEM and SEM in Figure 2a and Figure 2b. The particle size and Zeta potential were analyzed by Malvern Nano Zetasizer as shown in Figure 3a and Figure 3b.
 Figure 2: Images (a & b) elaborating SEM and TEM analysis of synthesized Alendronate- CS NPs.
View Figure 2
Figure 2: Images (a & b) elaborating SEM and TEM analysis of synthesized Alendronate- CS NPs.
View Figure 2
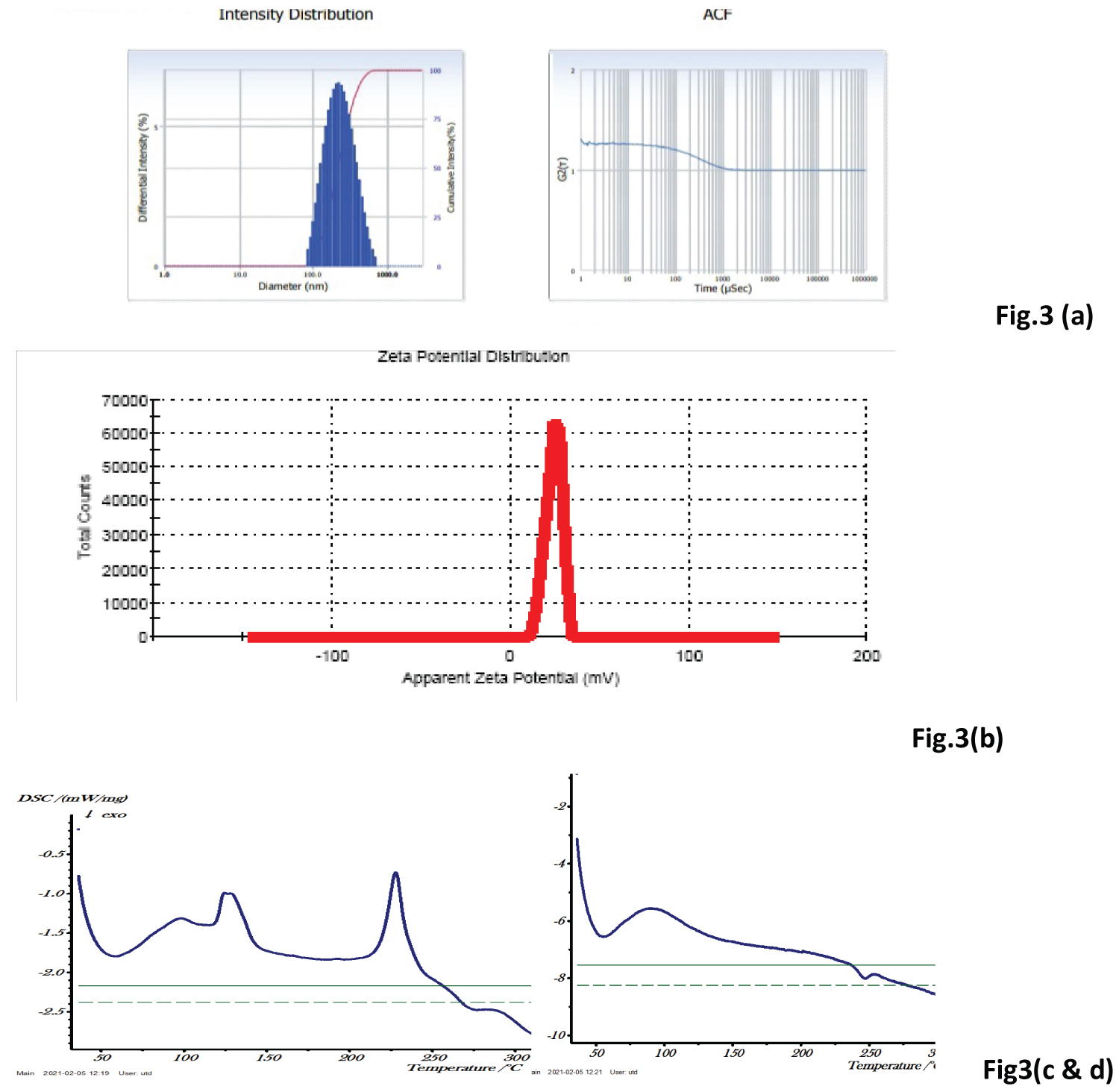 Figure 3: (a) Average Particle size of Alendronate- CS NPs; (b) Zeta Potential of Alendronate CS NPs; (c) DSC curve of Alendronate (API); (d) DSC curve of Alendronate.
Figure 3: (a) Average Particle size of Alendronate- CS NPs; (b) Zeta Potential of Alendronate CS NPs; (c) DSC curve of Alendronate (API); (d) DSC curve of Alendronate.
Images 3 (a & b) showing zeta sizer analysis of synthesized Alendronate- CS NPs (Mean ± SD, n = 3), (c & d) Elaborating the DSC curve of Alendronate (API) and NPs.
View Figure 3
The morphology of nanoparticles was analysed by using to SEM, the scanning Electron Microscopic studies shows the particle size in the range of 200 nm. The particle size of optimized NPs formulation was also characterized with TEM and the result was found to be in the range below 200 nm. TEM image showed completely spherical and symmetrical nanoparticles were formed in the optimized formulation.
Based on the results of particle size and entrapment efficiency, the optimized formulation showed the lesser particle size (214.6 nm) than the other batches with higher entrapment efficiency (66.82%) and poly dispersive index (PDI) was 0.156. The zeta potential of the optimized formulation was found to be +24.1 mV. On the basis of results found in the optimization studies, the best ratio between polymer (CS) and crosslinking agent (TPP), (2:0.5) mg/ml was found optimum in which the particle size was found in the range below 200 nm.
Differential scanning calorimetry (DSC): DSC analysis is important to identify possible interactions between the components of nano-formulation i.e. drug compatibility study. The DSC thermograms of pure drug Alendronate and Drug loaded polymeric nanoformulation are shown in Figure 3c and Figure 3d. Thermal analysis of pure drug Alendronate showed a sharp peak at 234 ℃, indicating its sharp crystalline nature. This temperature corresponding to its melting point. The melting peak drug was not detected in the NP formulation, indicating that the drug was encapsulated into the NPs and decreased crystallinity indicates the change in solid state structure of chitosan due to cross-linking by TPP. The amorphous form of the drug circulates freely in the systemic circulation.
The compatibility study of drug and excipients was identifying by DSC and shows the drug and excipients are compatible with each other and can be used for further development of formulation.
In drug release study, ADME process is involved, in which drug molecules migrate from the initial position and available for the biological action in to the systemic circulation. In vitro drug release of formulated Alendronate loaded CS NPs was compared with pure drug solution shown in Figure 4. Results showed that release of pure drug was fast, about 90% drug release within 8-10 hours, while CS NPs formulation showed the sustained release of drug up to 30 h. Initially, CS NPs formulation showed the burst release (45% drug releases in 10 h), followed by sustained releases up to 30h. Initial fast release may be due to the presence of the adsorbed drug on the surface of CS NPs.
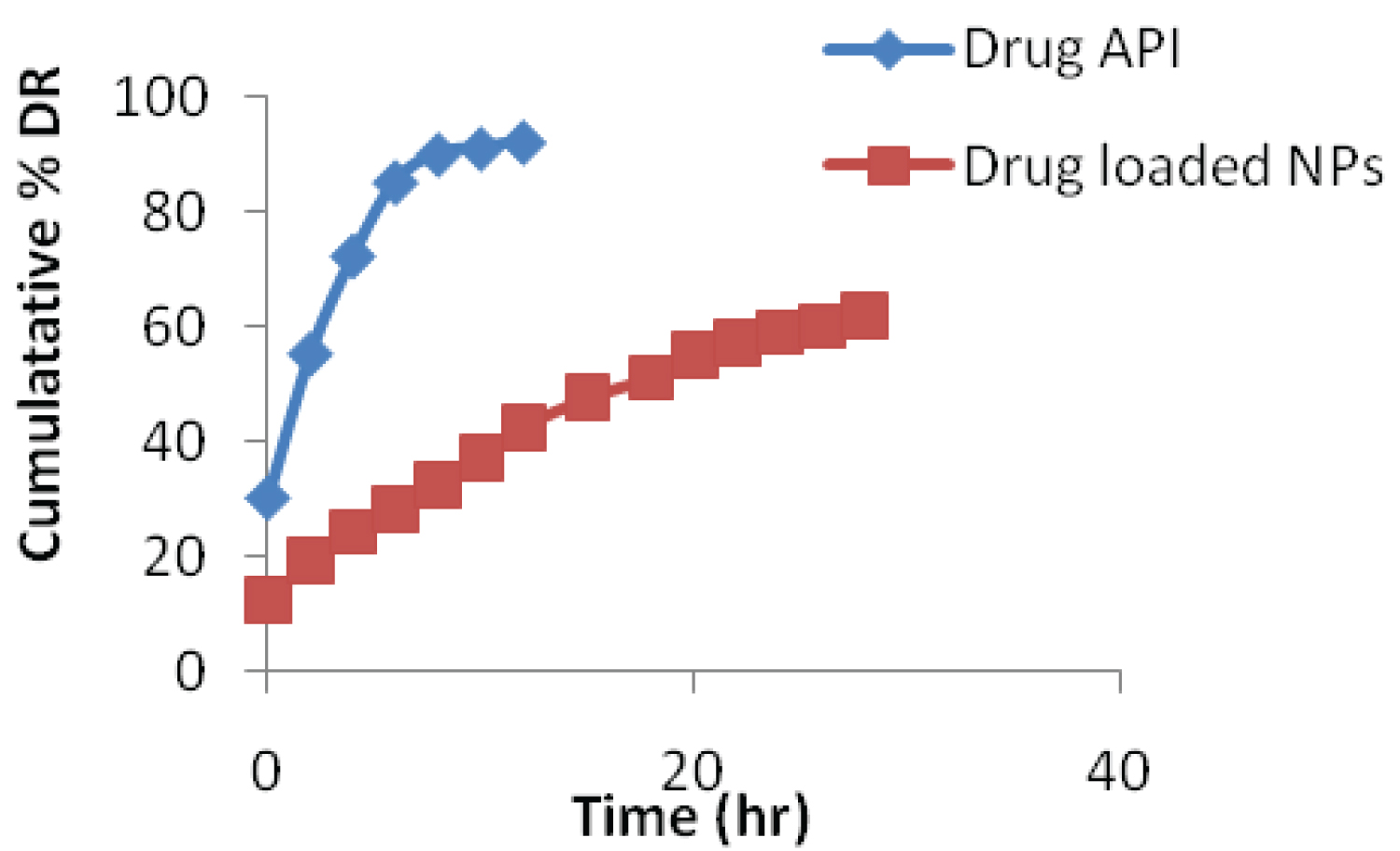 Figure 4: In-vitro drug release profile of Alendronate loaded CS NPs and Pure drug in PBS (pH 6.8).
View Figure 4
Figure 4: In-vitro drug release profile of Alendronate loaded CS NPs and Pure drug in PBS (pH 6.8).
View Figure 4
In-vitro drug release studies were carried out using dialysis bag method for different mathematical kinetic models. For kinetic study of formulated drug NPs, the plots were made for Zero order kinetic model (cumulative % drug release vs. time), First order kinetic model (log of cumulative % drug remaining vs. time), Higuchi model cumulative (% drug release vs. square root of time) and Korsmeyer-Peppas model (log cumulative % drug release vs. log time).
Plots of above mentioned models are shown in Figure 5 and results are summarized in Table 2. In the above table "R2" is correlation value, "K" is rate constant and "n" is release exponent.
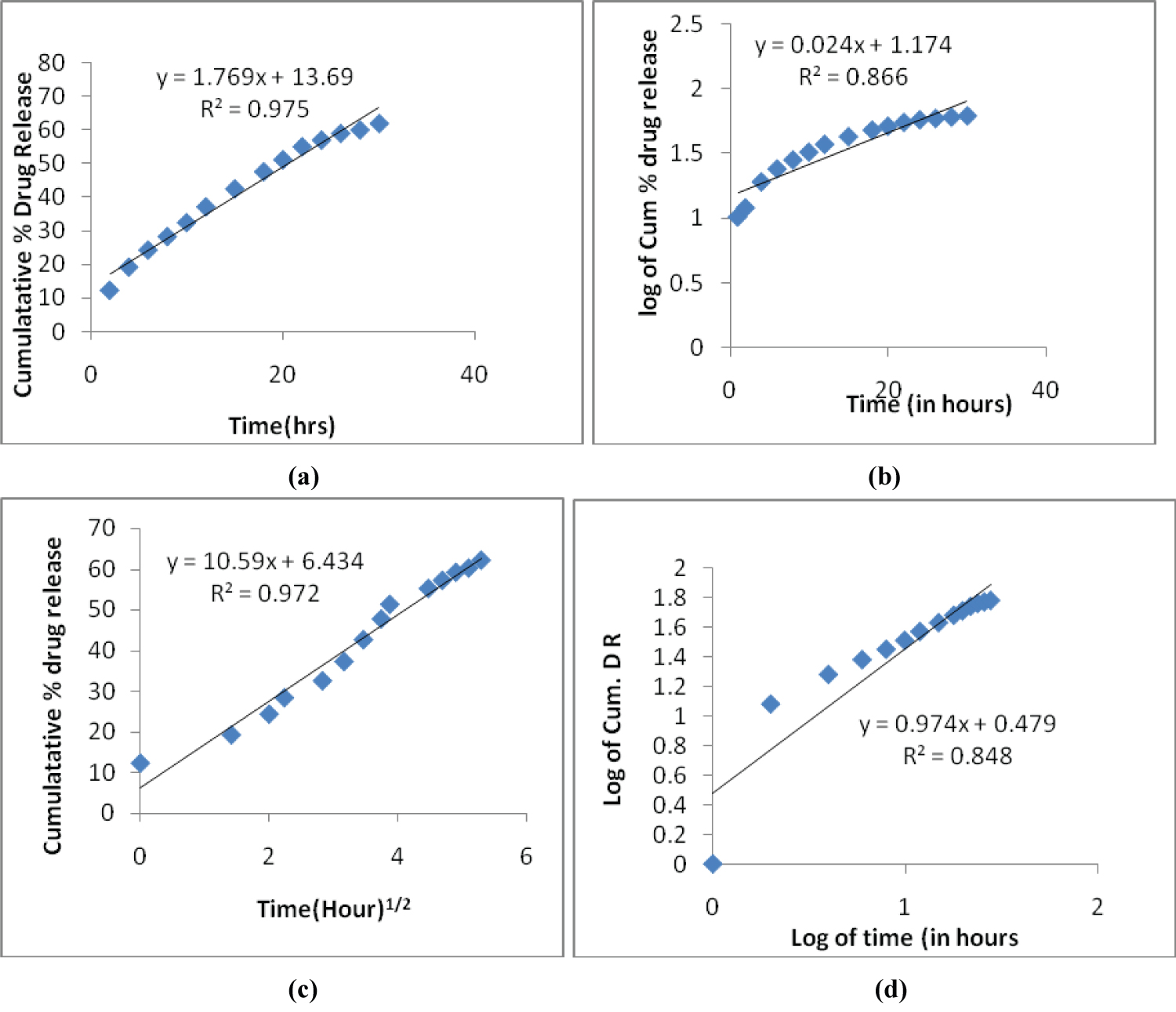 Figure 5: (a) Zero order plot; (b) First order plot; (c) Higuchi model plot; (d) Korsemeyer - Peppas model.
View Figure 5
Figure 5: (a) Zero order plot; (b) First order plot; (c) Higuchi model plot; (d) Korsemeyer - Peppas model.
View Figure 5
Table 2: Interpretation of R2 values and rate constants (K) of release kinetics of NPs. View Table 2
On the basis of above values of (R2), the best fit kinetic model with the highest correlation value is Zero order model. It is concluded that in the optimized formulated NPs follow Zero order model kinetics. In the Korsmeyer-Peppas model, release exponent value "n" is 0.47. The magnitude is in the range (0.45 < n < 0.89) indicates the release mechanism is non-Fickian diffusion.
Alendronate is the most efficient bisphosphonate for the treatment of Osteoporosis, but there is increasing concern about their long-term safety. Medications with novel mechanisms and novel formulation of drug loaded polymeric NPs can be expected to treat osteoporosis in future. The results of the current study would help us to find a new approach for drug discovery and drug delivery by preparing the anti-osteoporotic drug Alendronate in the Nano-range. Alendronate loaded CS NPs prepared via ionic gelation method presented the significant results of drug release profile and increased the therapeutic efficacy of Alendronate drug for the effective treatment of osteoporosis.
The authors are grateful to Electron Microscope Unit AIIMS, New Delhi and IISER (Bhopal) for DLS studies. We acknowledge Sophisticated Instrumentation Center (SIC), Dr. Harisingh Gour Vishwavidyalaya (A Central University), Sagar for providing us the SEM, TEM, FTIR and DSC facilities.