Immunomodulatory drugs are widely used for the treatment of inflammatory and immune-mediated conditions; however, their prolonged use is usually associated with the appearance of side effects and many immune disorders continuous without a suitable treatment. Natural products are an important source of therapies to a variety of illnesses. The present work investigated the immunomodulatory activity of a hexane extract and isolated compounds from Annona vepretorum Mart. Roots of A. vepretorum were used to produce a hexane extract, which was fractionated to obtain three diterpenes belonging to ent-kaurane series. The cytotoxicity of samples was determined using J774 macrophages. The immunomodulatory activity of natural products on macrophages activated with LPS and IFN-γ was determined by evaluation of nitric oxide (NO) and cytokine production. The effects of samples on lymphocyte proliferation were evaluated using splenocyte cultures stimulated with concanavalin A or in mixed lymphocyte reaction (MLR), by assessment of 3H-thymidine incorporation. The production of IL-2 and IFN-γ by lymphocytes was evaluated by ELISA. The effects of extract on the cell cycle progression of lymphocytes were evaluated by flow cytometry. The extract had low cytotoxicity and was able to modulate macrophages and lymphocytes in atoxic concentrations. AVHR (A. vepretorum hexane extract) significantly decreased NO, TNF-α, IL-1β and IL-6 production by activated macrophages. The extract also inhibited lymphocyte proliferation and IL-2 and IFN-γ production by activated splenocytes. In a concentration-dependent manner, the hexane extract arrested cell cycle in G0/G1 phase, accompanied by an increase of cells in PreG1 phase. Isolated compounds also were capable for modulates macrophage activation and lymphocyte function. The present findings demonstrate a potent immunomodulatory activity of hexane extract from roots of A. vepretorum.
Annona vepretorum, Immunomodulatory activity, Macrophage activation, Lymphocyte function, Annona vepretorum hexane extract
Abnormal immune responses are involved in the initiation and development of a large number of diseases, including autoimmune diseases, allergies, cancer, immunodeficiency syndromes and neurodegenerative diseases [1,2]. Abnormalities on lymphocyte and macrophages functions are the key factors that trigger to the development and maintenance of immune-mediated diseases [3,4]. A variety of immunosuppressive drugs are currently available; however, due to side effects associated to a prolonged use and the inefficacy of conventional treatments in certain conditions, the search for new immunosuppressive drugs is needed.
Natural products constitute an important source of therapies to a large number of illnesses [5]. A number of studies have demonstrated the immunomodulatory effects of crude extracts of traditional herbal medicines and natural compounds [6-8]. Annona L. (Annonaceae) includes approximately 175 species of trees and shrubs distributed in lowland tropical regions [9]. The genus is well recognized by the nutritional use of their fruits and the use of different parts of the plant in folk medicine [10,11]. Previous chemical and pharmacological studies on some species of the genus have identified bioactive compounds (alkaloids, steroids, flavonoids, terpenoids) in roots, leaves, bark, seeds and fruits [12,13]. Isolated compounds, as well as crude extracts from different Annona species show biological effects such as cytotoxicity against tumor cell lines, antimicrobial, antiprotozoal and immunomodulatory properties [11-15].
A. vepretorum Mart. (Annonaceae), popularly known as "bruteira", is a shrub or tree native from Brazilian biome Caatinga and is widely used in the human nutrition. In folk medicine, it has been used on bath to treat allergies, bites of bees and snakes and other inflammatory conditions [16]. Previous studies of this specie revealed a potent cytotoxicity activity of the essential oil from leaves of A. vepretorum in different cancer cell lines and an ethanolic extract of A. vepretorum showed antinociceptive and anti-inflammatory properties [9,16,17].
The popular usage of this specie against inflammatory conditions and previous studies showing a suppressive effect on immune system suggest an immunomodulatory potential from A. vepretorum. In the present work, the immunomodulatory activity of a hexane extract and isolated compounds from roots of A. vepretorum Mart. on macrophage activation and lymphocyte function was investigated.
The roots of A. vepretorum were collected in the proximity of the city of Poço Redondo [coordinates: S 09° 47' 44.6" W 37° 40' 35.3''], Sergipe State, Brazil, in March 2013. The identity of the plant was confirmed by Dr. Ana Paula do Nascimento Prata, a plant taxonomist of Department of Biology from Federal University of Sergipe (UFS), Brazil and a voucher specimen (#23158) has been deposited in the Herbarium of UFS. The authors have authorization from the Chico Mendes Institute for Biodiversity Conservation, and from Brazilian Ministry of the Environment for plant collection (#25637-1).
The dried and powdered of A. vepretorum (839.0 g) was successively extracted with hexane to yield hexane extract (AVHR; 34.90 g). A part (5.0 g) of this extract was initially subjected to silica gel column chromatography (CC; 2.5 × 50.0 cm) eluted with increasing concentrations of CH2Cl2 in n-hexane (100:0 to 10:90, v/v), followed by EtOAc in CH2Cl2 (100:0 at 20:80, v/v), and MeOH in EtOAc (100:0 to 50:50, v/v), affording 185 fractions (30 mL each). The eluted fractions were evaluated and pooled according to TLC analysis, to afford 33 groups (GF1 to GF33). Group GF16 (1650.5 mg) from n-hexane-CH2Cl2 (20:80 and 100:0, v/v) was submitted to a new silica gel CC (1.5 × 40.0 cm) eluted with increasing concentrations of CH2Cl2 in n-hexane (100:0; 80:20; 60:40; 20:80 and 40:60, v/v), followed by EtOAc in CH2Cl2 (100:0 to 80:20, v/v), affording 75 subfractions (30 mL each), that were evaluated and pooled according to TLC analysis, to afford 4 groups (GF16.1 to GF16.4). Group GF16.3 resulted in the isolation compound 1 (1154.3 mg). Group GF19 (271.6 mg) from CH2Cl2-EtOAc (90:10 and 80:20, v/v) was submitted to a new silica gel CC ( 1.5 × 40.0 cm) eluted with increasing concentrations of CH2Cl2 in n-hexane (100:0 to 0:100, v/v), affording 62 subfractions (30 mL each), that were subsequently pooled to afford 4 groups (GF19.1 to GF19.4). Group GF19.3 afforded compound 2 (128.9 mg). Group GF20 (287.1 mg) from CH2Cl2-EtOAc (80:20, v/v) was submitted to a new silica gel CC ( 1.5 × 40.0 cm) eluted with increasing concentrations of CH2Cl2 in n-hexane (100:0; 80:20; 60:40 and 40:60, v/v), followed by EtOAc in CH2Cl2 (100:0 and 90:10, v/v), affording 31 subfractions (30 mL each), that were subsequently pooled to afford 5 groups (GF20.1 to GF20.5). The group GF20.4 furnished compound 3 (55.2 mg).
ent-kaur-16-en-19-oic acid (1): White needles (n-hexane-EtOAc 8:2); mp 162-163°C (lit. 161-162°C); [17]; [α]D22 -105.7° (c 0.7, CHCl3); IR (KBr) νmax 3479, 2924, 2868, 1695, 1470, 1450, 1262, 1055, 1035, 1013, 873 cm-1; 1H and 13C NMR data according to [18]; EI-MS m/z 302 [M]+..
ent-3β-hydroxykaur-16-ene (2): White solid (n-hexane-EtOAc 8:2); mp 162-164.0°C (lit. 161-163.0°C); [16]; [α]D20 -151.5° (c 0.7, CHCl3); IR(KBr) νmax 3345 (OH), 3070, 2924, 2854, 1651 (C = C), 1487, 1427, 1101, 1041, 989, 869 cm-1; 1H and 13C NMR data according to [16]; EI-MS m/z 288 [M]+..
ent-16β-hydro-kauran-17-oic acid (3): White needles (Hexane-CHCl3 7:3); mp 193-194.0°C (lit. 192.0-194.0 °C) [19]; [α]D20 -63.2° (c 0.7, CHCl3); IR, and 1H and 13C NMR data, according to [19]; EI-MS m/z 304 [M]+..
Melting points (m.p.) were measured on a Microquímica MQAPF 301 apparatus (Palhoça, Santa Catarina, Brazil). IR spectra were acquired in KBr pellets on a Shimadzu IR Prestige-21 spectrophotometer (Kyoto, Kyoto, Japan). Optical rotations were recorded in CHCl3 on a Jasco P-2000 polarimeter (Hachioji, Tokyo, Japan). GC-MS analyses were performed on a Shimadzu QP5050A GC-MS system (Kyoto, Kyoto, Japan) equipped with an AOC-20i auto-injector. The separation of the compounds was achieved employing on RTx®-5SilMS fused capillary chromatography column (30 m × 0.25 mm × 0.25 μm film thickness) coated with 5%-diphenyl-95%-dimethylpolysiloxane (Restek, Belleh1e, PA). The column temperature program was 200°C/5 min, a rate of 10°C/min to 320°C, and then 320°C/10 min (27 min total time analysis); carrier gas, He (99.999%; 1.2 mL/min); split ratio, 1:20; injection volume, 0.5 mL of the compound in CH2Cl2 (5.0 mg/mL). MS were taken at 70 eV with a scan interval of 0.5 s and fragments from 40-500 Da. Low Resolution Mass Spectra (LRMS) were determined using an ultra-high performance chromatography-mass spectrometry system (Acquity UHPLC-TQD - Waters) with an ESI and APCI source in the positive and negative ion mode (Milford, Massachusetts). 1D and 2D NMR data were recorded at 303 K in CDCl3 on a Bruker Avance III 600 NMR spectrometer (Karlsruhe, Baden-Württemberg, Germany), operating at 14.1 Tesla, observing 1H and 13C at 600 and 150 MHz, respectively. The spectrometer was equipped with a 5 mm multinuclear inverse detection probe (1D and 2D NMR experiments) with z-gradient. One-bond and long-range 1H - 13C correlation from HSQC and HMBC NMR experiments were optimized for an average coupling constant 1J(C,H) and LRJ(C,H) of 140 and 8 Hz, respectively. All 1H and 13C NMR chemical shifts (δ) are given in ppm related to the TMS signal at 0.00 ppm as an internal reference, and the coupling constants (J) in Hz. Silica gel 60 (70-230 mesh) was used for column chromatography (Fluka Analytical, Schnelldorf, Bavaria, Germany), while silica gel 60 F254 was used for analytical (0.25 mm), and preparative (1.00 mm) TLC (Merck, Darmstadt, Germany). Compounds were visualized by exposure under UV254/365 light and spraying of anisaldehyde reagent (Neon, São Paulo, SP, Brazil) followed by heating on a hot plate.
BALB/c and C57BL/6 female mice (6 to 10 weeks old) were bred and maintained at the Gonçalo Moniz Research Center (Oswaldo Cruz Foundation, Bahia, Brazil) in sterilized cages, under a controlled environment and receiving a balanced rodent diet and water ad libitum. All animal experiments and procedures were approved by the institution’s committee on the ethical handling of laboratory animals (Approved number: 011/2011).
Gentian violet (Synth, São Paulo, SP, Brazil) was used as positive control in the cytotoxicity assays. Dexamethasone (Sigma-Aldrich, St. Louis, MO), a synthetic glucocorticoid, was used as positive control in immunomodulatory assays. All compounds were dissolved in DMSO (Sigma-Aldrich) and diluted in cell culture medium for use in the assays. The final concentration of DMSO was less than 1% in all experiments.
To determine the cytotoxicity of the hexane extract and isolated compounds, the murine macrophage-like cell line J774 were seeded into 96-well plates at a cell density of 1 × 104 cells/well in Dulbecco’s modified Eagle medium (DMEM; Life Technologies, GIBCO-BRL, Gaithersburg, MD) supplemented with 10% fetal bovine serum (FBS; GIBCO), and 50 μg/mL of gentamycin (Novafarma, Anápolis, GO, Brazil) and incubated for 24 h at 37°C and 5% CO2. After that time each test inhibitors were added at least in five concentrations in triplicate and incubated for 72 h. Twenty μL/well of Alamar Blue (Invitrogen, Carlsbad, CA) was added to the plates during 6 h. Colorimetric readings were performed at 570 and 600 nm. CC50 values (Cytotoxicity concentration at 50%) were calculated using data-points gathered from three independent experiments.
For cytokine and nitric oxide determinations, J774 cells were plated in 96-well tissue culture plates at 2 × 105 cells/well in DMEM medium supplemented with 10% of FBS and 50 μg/mL of gentamycin for 2 h at 37°C and 5% CO2. Cells were then stimulated with LPS (500 ng/mL, Sigma-Aldrich) plus IFN-γ (5 ng/mL; Sigma-Aldrich) in the absence or presence of natural products in testing or dexamethasone at different concentrations, and incubated at 37°C. Cell-free supernatants were collected 4 h (for TNF-α measurement) and 24 h (for IL-1β, IL-6 and IL-10 quantification) and kept at -80°C. Cytokine concentrations in supernatants from J774 cultures were determined by enzyme-linked immunosorbent assay (ELISA), using the DuoSet kit from R&D Systems (Minneapolis, MN), according to the manufacturer’s instructions. Quantification of nitric oxide was done using the Griess method [20].
BALB/c splenocyte suspensions were prepared in DMEM medium supplemented with 10% of FBS and 50 μg/mL of gentamycin. Splenocytes were cultured in 96-well plates at 1 × 106 cells/well, in triplicate, in the presence of concanavalin A (Con A; 2 μg/mL, Sigma-Aldrich), alone or in various concentrations of natural products in testing, as described in figure legends. After 48 h of incubation, 1 μCi of 3H- thymidine (Perkin Elmer, Waltham, MA) was added to each well, and the plate was returned to the incubator for more 18 h. The plate was then transferred to a β-plate counter (Multilabel Reader, Finland) and the 3H-thymidine uptake was measured. Dexamethasone was used as a positive control. Cell-free supernatants were collected after 24 h of incubation and stored at -20°C for cytokine analysis. IL-2 and IFN-γ concentrations in supernatants from splenocyte cultures were determined by ELISA, as described above.
BALB/c mice were weekly immunized with intraperitoneal injection of 107 splenocytes obtained from C57BL/6 as previously described [21]. After 3 weeks of immunization, mice were euthanized for spleen cell preparation in DMEM medium supplemented with 10% of FBS and 50 μg/mL of gentamycin. Spleen cells were plated in 96 well plates at a cell density of 1 × 106 cells/well in the absence or in the presence of irradiated C57BL/6 splenocytes at 106 cells/well (dose of 3 Gy in a 137Cs source irradiator purchased from CisBio International, Cordolet, France) and different concentrations of AVHR, in triplicates. After 72 h of incubation, 3H-thymidine was added to each well, and the plate was returned to the incubator for more 18 h. The plate was then transferred to a β-plate counter and the 3H-thymidine uptake was measured.
Splenocytes from BALB/c mice were plated at a cell density of 5 × 106 cells/well in 1 mL of complete medium containing 5 μg/mL of Con A in the absence or presence of different concentrations of AVHR (5, 10 and 20 μg/mL) for 48 h. Cells were centrifuged and then pellet was washed twice with cold PBS and resuspended in a solution of PBS with propidium iodide (2 μg/mL) and 0.1 % of Triton X-100 at 37°C for 30 min. The cells were analyzed for cell cycle using flow cytometry (FACS Calibur, BD Biosciences, San Jose, CA). Cell debris and clumps were gated out, and PreG1, G0/G1, S and G2/M populations were quantified using Cell Quest software.
Statiscal analysis were performed by one-way analysis of variance and Newman-Keuls multiple comparison tests using Graph Pad Prism version 5.01 (Graph Pad Software, San Diego, CA). Differences were considered significant when the values of P were < 0.05.
The plant extract was evaluated against mammalian cells to determine the cytotoxicity. The hexane extract showed a CC50 value of 39.3 μg/mL and non cytotoxic in concentrations lower than 25 μg/mL (Table 1). Therefore, concentrations lower than 25 μg/mL were used as the concentrations of the extract in the subsequent experiments.
Table 1: Cytotoxicity against J774 macrophages of the natural products of A. vepretorum. View Table 1
To assess the effects of AVHR on macrophages, proinflammatory mediators production was evaluated. The treatment with the hexane extract (5, 10 or 20 μg/mL) significantly decreased the production of nitric oxide, TNF-α, IL-1β and IL-6, as well as the treatment with dexamethasone (20 μg/mL) compared to untreated and stimulated cultures (Figure 1A, Figure 1B, Figure 1C and Figure1D). The cell-free supernatant was also used to quantify IL-10, however, the treatment with AVHR didn´t increase the levels of this cytokine (data not shown).
 Figure 1: Assessment of nitric oxide and cytokine production by J774 macrophages treated with AVHR. Effect of AVHR (20, 10 or 5 μg/mL) or dexamethasone (20 μg/mL) on nitric oxide (A); TNF-α (B); IL-1β (C) and IL-6 (D) production. Values represent the means ± SEM of four determinations obtained in one of two experiments performed. ***P < 0.001; **P < 0.01; *P < 0.05 compared to untreated and stimulated cultures.
View Figure 1
Figure 1: Assessment of nitric oxide and cytokine production by J774 macrophages treated with AVHR. Effect of AVHR (20, 10 or 5 μg/mL) or dexamethasone (20 μg/mL) on nitric oxide (A); TNF-α (B); IL-1β (C) and IL-6 (D) production. Values represent the means ± SEM of four determinations obtained in one of two experiments performed. ***P < 0.001; **P < 0.01; *P < 0.05 compared to untreated and stimulated cultures.
View Figure 1
The action of the hexane extract on lymphocyte activation was also analyzed. Addition of plant extract in Con A-induced lymphocyte proliferation assays and MLR cultures of spleen cells of BALB/c mice stimulated with C57BL/6 cells also caused a significant inhibition of proliferation greater than 90% (Figure 2A and Figure 2B). The cell-free supernatant of cultures of splenocytes stimulated with Con A and treated with hexane extract or dexamethasone was also investigated to understand the effect of compounds on cytokines production by lymphocytes. As shown in figure 2C and figure 2D, the treatment with various concentrations of AVHR significantly decreases the secretion of IL-2 and IFN-γ compared with untreated and stimulated cultures. Under the same conditions, dexamethasone also promotes a high decrease in cytokines production.
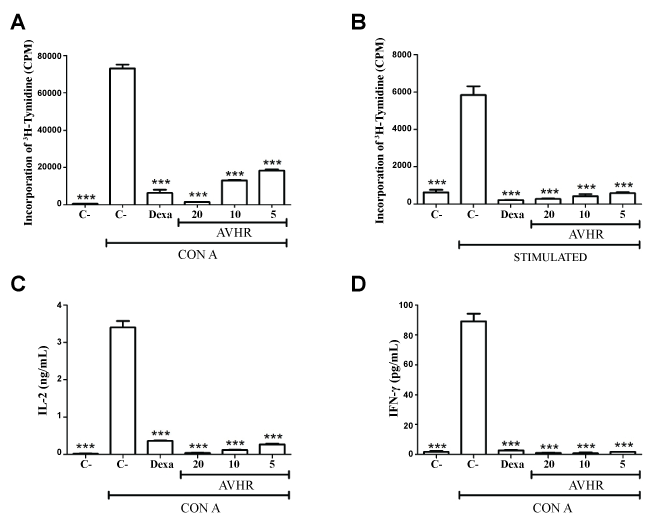 Figure 2: Inhibition of lymphocyte proliferation and cytokine production by AVHR. (A) Effect of treatment with AVHR (20, 10 or 5 μg/mL) or dexamethasone (20 μg/mL) for 48 h on Con A-induced lymphoproliferation; (B) Splenocytes from BALB/c mice sensitized with C57BL/6 cells were cultured in the absence (unstimulated) or presence (stimulated) of irradiated C57BL/6 spleen cells, without or with AVHR (20, 10 or 5 μg/mL) or dexamethasone (20 μg/mL) for 72 h. Concentrations of IFN-γ; (C) IL-2; (D) were determined in splenocytes treated or not with AVHR or dexamethasone after 24 h of treatment. Values represent the means ± S.E.M. of 4 determinations. ***P < 0.001 compared to untreated and stimulated cultures.
View Figure 2
Figure 2: Inhibition of lymphocyte proliferation and cytokine production by AVHR. (A) Effect of treatment with AVHR (20, 10 or 5 μg/mL) or dexamethasone (20 μg/mL) for 48 h on Con A-induced lymphoproliferation; (B) Splenocytes from BALB/c mice sensitized with C57BL/6 cells were cultured in the absence (unstimulated) or presence (stimulated) of irradiated C57BL/6 spleen cells, without or with AVHR (20, 10 or 5 μg/mL) or dexamethasone (20 μg/mL) for 72 h. Concentrations of IFN-γ; (C) IL-2; (D) were determined in splenocytes treated or not with AVHR or dexamethasone after 24 h of treatment. Values represent the means ± S.E.M. of 4 determinations. ***P < 0.001 compared to untreated and stimulated cultures.
View Figure 2
The effect of AVHR on cell-cycle progression of splenocytes revealed arrested splenocytes on G0/G1 phase, which was accompanied by an increase in PreG1 phase. Importantly, these effects were in a concentration-dependent manner (Figure 3).
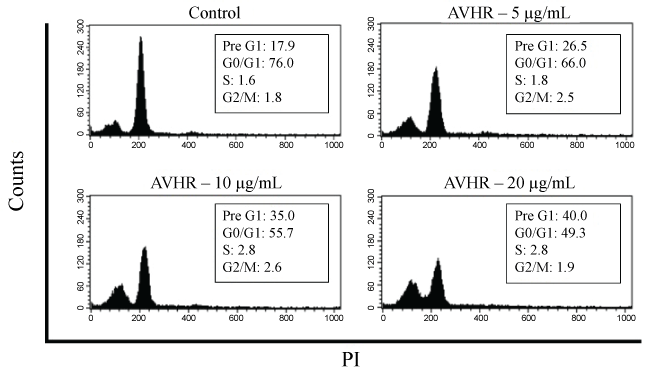 Figure 3: Analysis of cell cycle progression after treatment with AVHR. Splenocytes were untreated or treated with AVHR (20, 10 or 5 μg/mL) for 48 h. The distribution and percentage of cells in pre-phase, G0/G1, S and G2/M phase of the cell cycle are indicated.
View Figure 3
Figure 3: Analysis of cell cycle progression after treatment with AVHR. Splenocytes were untreated or treated with AVHR (20, 10 or 5 μg/mL) for 48 h. The distribution and percentage of cells in pre-phase, G0/G1, S and G2/M phase of the cell cycle are indicated.
View Figure 3
Finally, phytochemical investigation of AVHR led to the isolation and characterization of three diterpenes belonging to ent-kaurane series, identified as ent-kaur-16-en-19-oic acid (1), ent-3β-hydroxykaur-16-ene (2) and ent-16β-hydro-kauran-17-oic acid (3) (Figure 4).
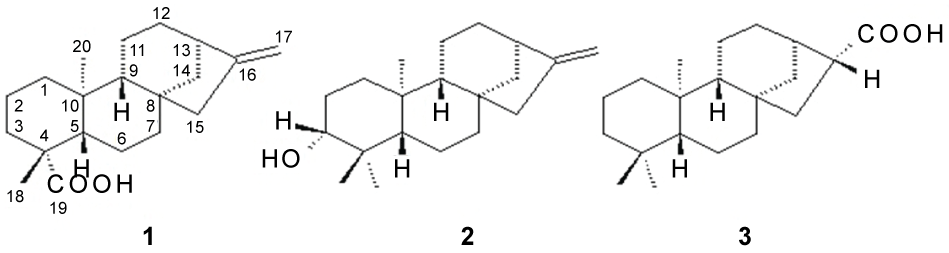 Figure 4: Chemical structures of ent-kaurane diterpenes isolated from the roots of A. vepretorum.
View Figure 4
Figure 4: Chemical structures of ent-kaurane diterpenes isolated from the roots of A. vepretorum.
View Figure 4
All isolated compounds showed low cytotoxicity to J774 macrophages with values of CC50 above 90 μM (Table 1). When tested in cultures of J774 macrophages stimulated with LPS and IFN-γ, the compounds were able to reduce the production of nitric oxide, especially compounds 1 and 2 in highest concentration (50 μM) (Figure 5A). Regarding the immunosuppressive effect on lymphocyte proliferation, compound 2 showed the best profile with a significantly inhibition of lymphoproliferation at the concentrations of 50 and 25 μM (Figure 5B).
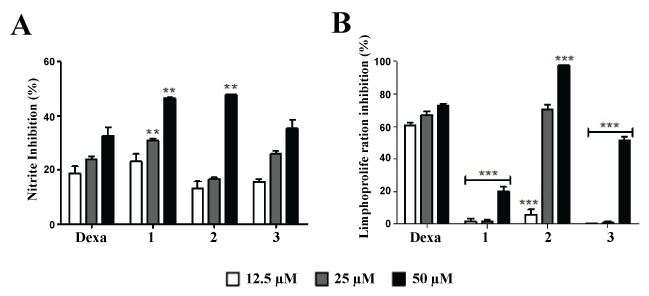 Figure 5: Effect of ent-kaurane diterpenes isolated from the roots of A. vepretorum on NO production and lymphoproliferation. (A) Comparison of nitric oxide inhibition by different isolated compounds from roots of A. vepretorum; (B) Comparison of Con A-induced lymphoproliferation inhibition by different isolated compounds from roots of A. vepretorum Values represent the means ± S.E.M. of 4 determinations obtained in one of two experiments performed. ***P < 0.001; **P < 0.01; *P < 0.05 compared to cultures treated with dexamethasone in the same concentration.
View Figure 5
Figure 5: Effect of ent-kaurane diterpenes isolated from the roots of A. vepretorum on NO production and lymphoproliferation. (A) Comparison of nitric oxide inhibition by different isolated compounds from roots of A. vepretorum; (B) Comparison of Con A-induced lymphoproliferation inhibition by different isolated compounds from roots of A. vepretorum Values represent the means ± S.E.M. of 4 determinations obtained in one of two experiments performed. ***P < 0.001; **P < 0.01; *P < 0.05 compared to cultures treated with dexamethasone in the same concentration.
View Figure 5
Despite its use in folk medicine, little is known about the immunomodulatory properties of Annona vepretorum. In this report, the immunomodulatory activiy of a hexane extract and isolated compounds from roots of A. vepretorum was demonstrated.
Macrophages are a population of immune cells that have a range of roles in both the induction and resolution of inflammation mainly through the secretion of cytokines [22]. Pro-inflammatory mediators such as TNF-α, IL-1β, IL-6 and nitric oxide are well-recognized by participate in various inflammatory diseases [22]. TNF- α is a key mediator on inflammation establishment by recruiting and activating macrophages and T cells and favoring the secretion of other pro-inflammatory cytokines [23]. IL-1β has similar effects to TNF-α and on high concentrations leads to cell death or tissue injury [24]. IL-6 has a majority contribution on the production of most acute-inflammatory proteins. Nitric oxide due to over production on inflammatory conditions promotes vasodilatation, vascular permeability and edema, signs of acute inflammation [25]. Through Griess method and cytokine measurement by ELISA was shown a potent effect of hexane extract from roots of A. vepretorum on inhibition of nitric oxide production and pro-inflammatory cytokines on macrophages stimulated with LPS plus IFN-γ. Moreover, these results are in agreement with previous studies with an ethanolic extract of A. vepretorum, that showed the ability of extract in inhibit leukocyte migration on peritoneal cavity induced by injection of carrageenan and reduce paw edema induced by carrageenan or histamine [17]. Both models are well recognized to evaluate anti-inflammatory effects and involved participation of chemical mediators and cytokines such as nitric oxide, IL-1β, IL-6 and TNF-α [17,26]. These data reinforce the potential of AVHR on inflammatory conditions, once the extract inhibits these chemical mediators and cytokines production on activated macrophages.
A promising immunosuppressive activity on lymphocytes was also observed on AVHR. The plant extract was able to inhibit lymphocyte proliferation induced by Con A and allospecific lymphocyte proliferation, indicating that inhibition was not a nonspecific phenomenon due to interference on the interaction of ConA with surface molecules on spleen cells [21]. Flow cytometry analysis of cell cycle by propidium iodide staining reinforced the effect of the hexane extract on lymphoproliferation, since it was observed that extract treatment blocked cell-cycle progression by arresting cells at G0/G1 phase. Moreover, AVHR reduced IL-2 and IFN-γ production, key cytokines on lymphocyte proliferation and activation [27]. In fact, the antiproliferative activity of this specie is well recognized in different lineages of tumor cells, especially in leukemia lineages such as HL-60 and K562 cells [16,28]. Furthermore, the in vivo efficacy of an essential oil from A. vepretorum was demonstrated in mice inoculated with B16-F10 mouse melanoma [28].
Finally, phytochemical investigations of AVHR culminate with the isolation and characterization of three diterpenes belonging to ent-kaurane series. These compounds have been described for the first time in the roots of A. vepretorum, however compounds 1 and 2 recently were described in the stem bark of this specie, indicating that the plant accumulates these compounds in different part, such as stem bark and root [16]. These compounds are well represented in family Annonaceae found in different species, particularly in the species of the genus Annona and Xylopia and considered as chemotaxonomic markers [18,29]. Previous biological investigations described the cytotoxicity effects of 1 and 2 on different tumor cell lines [16]. Here, it was demonstrated the immunomodulatory effects of these diterpenes, especially the compound 2, on the inhibition of nitric oxide production and lymphoproliferation. Previous reports, also demonstrated the immunomodulatory effects of others ent-kaurane on the inhibition of nitric oxide production in RAW264.7 macrophages [30,31]. Moreover, the anti-inflammatory activity of 1 on an in vivo model of paw edema induced by carrageenan was also reported, reinforcing this class of compounds as a promising source for obtaining immunomodulatory drugs [32].
However, the isolated diterpenes showed a weak activity when compared with AVHR, suggesting that the higher potency of extract may be attributed to other compounds present in its composition or a synergistic effect of some of its components.
The present work suggests a potent immunomodulatory activity of hexane extract from roots of A. vepretorum and isolated diterpenes belonging to ent-kaurane series. The extract and isolated compounds inhibited cytokine production and cell cycle development. Therefore, the investigation of other members of this family is important.
This work was supported by CNPq, PRONEX and FASPEB.
None of the authors have any financial or personal relationships that could inappropriately influence or bias the content of the paper.