The anti-tumor immune response includes both acquired and innate immunity, with T cells playing a central role. Recent advances in immunotherapy have shown great promise, but only a small percentage of patients benefit. The low efficacy of immunotherapy is often linked to the tumor microenvironment (TME), which hampers immune responses. Tumor cells undergo metabolic reprogramming, significantly affecting the TME and immune cell function. Tumor cells not only deplete nutrients from the TME, limiting immune cell activation, but also accelerate the efflux of metabolites, further suppressing immune responses. Additionally, the poor vascularization and low material exchange in the TME result in metabolite accumulation, which inhibits immune cell activation. Tumor-specific metabolic pathways can also evade immune surveillance by reducing the immune system's constraints on tumor tissue. This article focuses on the roles of key metabolic pathways, such as aerobic glycolysis, lipid metabolism, and amino acid metabolism, in tumor immunity. Understanding the relationship between tumor metabolism and anti-tumor immunity is crucial for developing more effective therapeutic strategies, including improving immune response efficiency by modulating nutrient flow and reducing the adverse effects of metabolites.
Anti-tumor immunity, Tumor microenvironment, Metabolic reprogramming, Immunotherapy, Immune evasion
Tumor metabolic reprogramming alters the microenvironment and suppresses immunity, presenting new therapeutic targets for enhancing immunotherapy.
In normal tissues, a variety of metabolic enzymes convert the three major nutrients, sugar, fat and protein into complex metabolic intermediates, which form a network of biochemical reactions. Studies had shown that these intermediate metabolites are important regulators of various cellular activities such as intercellular signal transduction and intracellular signal transduction, and play a very central role in maintaining cell stability and adaptation in response to intracellular and extracellular stimuli.
Since the "Warburg effect" (also known as aerobic glycolysis), the unique metabolic characteristics of tumor tissues have been gradually discovered, and with the deepening of research, metabolic reprogramming has been established as one of the characteristics of malignant tumors [1,2]. The study found that, although no metabolic activity was limited to tumor tissues, there were still significant differences in the metabolic level between tumor tissues and normal tissues [3,4]. This difference in metabolic levels provides tumor cells with abundant nutritional basis, and promotes tumor progression from many aspects such as tumor cell dormancy, proliferation, metastasis and invasion, and drug resistance. Now, researchers have developed a series of targeted drugs or small molecule compounds in view of the difference in the metabolic level between tumor tissues and normal tissues, which gradually becomes a new and effective anti-tumor strategy. However, recent studies have shown that tumor-specific metabolism also reduces the restriction effect of the immune system on tumor tissue in various ways, thereby evading immune surveillance and gaining growth advantages. Existing treatments targeting tumor metabolism can also inhibit tumor growth by stimulating the body's immune system. Therefore, this paper summarizes the latest progress of tumor metabolism-related research and anti-tumor immunity-related research, and reviews the existing relationship between them, in order to provide some ideas for related work.
There are multi-type immune effector cells and immune effector molecules in the body, which interweave with each other to form a powerful immune response system. Immune system can be roughly divided into two types: acquired immune response or innate immune response. Acquired immune response means that the immune system's immune response to some antigens depends on whether the same antigen has been encountered before: if the immune system encounters and recognizes these new antigens for the first time, the immune system is trained and produce memory. When acquired immune response encountering the same antigen again, the immune system will produce a strong immune response. Innate immune response means that the cells of the immune system instinctively participate in the immune response without prior antigen recognition and training. The innate immune response and the acquired immune response are intertwined and constitute the multi-layered anti-tumor immune system of the body, which has obvious restraining effects on the proliferation of tumor cells and the formation of tumors. Even through, the immune system hardly inhibits tumors occurrence and development totally. Research reports that, in the early stage of tumor occurrence, immune system limited and killed the tumors cells by recognizing specific tumor antigens. However, in the stage of advance tumor, the immune effector cells, such as T cells, NK cells etc., become fatigued, unable to be activated, and even tend to apoptosis. Some effector cells, such as macrophage, even make tumor progress. But, anti-tumor immune response, seemly useless in advance tumor, always play an important role in prolonged survival and improve the prognosis of tumor patient. Research reported that the tumor patient whose high ratio of immune cells infiltrating tumors often have a significantly positive prognosis significance [5,6]. On the contrary, patients with tumors lacking immune cells infiltration often have poor disease prognosis [7]. In addition to T cells, a variety of immune cells play an important role in anti-tumor immunity [8-10]. Most tumors have a hidden onset and have entered an advanced stage when they are found clinically. There are two reason why immune system hardly restrict tumors occurrence: Firstly, tumor cells have low immunogenicity and cannot be recognized by the immune system when tumors are formed. These cells grow vigorously and quickly form clinical tumors. The second is that the tumor is immunogenic at the beginning. While the immune system is constantly attacking, the tumor tissue has been adapting to the host, and finally find a way to escape from immune surveillance and as a result, led to the occurrence of tumors. So once discovered, tumors may have great immune resistance (or extremely low immunogenicity). In recent years, immunotherapy based on immune checkpoint inhibitors (ICIs) has made great achievements in the treatment of tumors. Immunotherapy based on antibody to CTLA-4, programmed cell death 1 (PD-1) and its ligand PD-L1 has changed the landscape of tumor treatment [11]. However, only about 10-30% of patients benefit from treatment with ICIs, and most patients do not respond to ICIs or achieve durable responses [12,13].
The reason why low effective rate of ICIs on tumor patient may be closely related to the low response of immune effector cells and the complex tumor microenvironment (TME) in clinical. The main structure of TME is composed of a cell population dominated by tumor cells, cancer associated fibroblasts (CAFs), vascular endothelial cells, immune cells and other cells. In addition, a variety of cellular components and a large number of cellular metabolites also participate in the formation of TME. The complex TME composition makes the tumor tissue completely different from the normal tissue structure: highly chaotic the internal structure of the tumor tissue and highly poorly differentiated the blood vessels results in the difficulty of oxygen and nutrient transportation and low metabolic waste removal efficiency. With the influence of TME, how to eliminate the immunosuppression in TME, enhance the immunogenicity of tumor cells, induce tumor cell death in an immune death manner, activate and promote the survival immune effector cells has become the four key questions of promotion of anti-tumor response [14]. Especially, activating immune effector cells and promoting the survival of effector cells, have become the hotspots and difficulties of current anti-tumor immune response research.
In fact, not only tumor cells undergo metabolic reprogramming, but immune cells also undergo metabolic reprogramming. According to research reports: CD8+ T cells obtain energy through oxidative phosphorylation (OXPHOS) when they are dormant; once T cells are activated, the way T cells obtain energy changes from OXPHOS to glycolysis. However, when tumor cells and immune cells are in the same region, the high metabolic level of tumor cells inhibits the activation of immune cells from two aspects: on the one hand, tumor cells upregulate various types of transporters (such as glucose transporters, amino acid transporters, etc.) to absorb nutrients predatory in the microenvironment, resulting in the state that lack of nutrients and the difficulty in activating immune cells; on the other hand, tumor cells upregulate some transporters, such as monocarboxylate transporter1/Monocarboxylate transporter 4(MCT1/MCT4), etc., efflux lots of metabolites who have negative effect on immune effector cells. Additionally, because of the poor vascular differentiation in the TME, material exchange efficiency is quite low and resulting in the highly enriched metabolites in the TME. The accumulation of some metabolites is further inhibited the activation of immune cells. How to relieve the growth advantage brought by the high metabolic level of tumor tissue to improve the response ability of the body's immune system, and enhance the restraint effect of the body's immune system on the tumor is a major problem in current research.
Aerobic glycolysis: In tumor tissues,various cancer genes, such as c-MYC, KRAS and YAP,drives the overexpression of multiple types of glucose transporters and enzymes related to glycolytic metabolism [15]. GLUT1, as a representative of glucose transporter, its high expression in tumors is highly correlated with poor prognosis of patients [16-20]. In addition, many metabolic enzymes related to the glycolysis process, including HK2 [19-21], PFKFB3 [20,22], PKM2 [20,23] and LDHA [20,24,25], also are involved in the occurrence and development of tumor. Ultra-high glucose intake rate and specific glucose metabolism mode - aerobic glycolysis (Aerobic glycolysis is the process by which tumor cells still convert glucose into lactate in the presence of oxygen,also named as “Warburg effect”) provides significant growth advantages to tumor cells.
It is worth noting that, as the main effector cells for tumor clearance, T cells not only need to obtain strong antigen stimulation, but also need to obtain sufficient nutrients. Therefore, after receiving immune stimulation, the metabolism of T cells changes from OXPHOS to aerobic glycolysis, the purpose of which is to obtain energy quickly [26]. Research reports that the same metabolic model between activated T cell and tumor cells would bring fierce nutrition competition to them. Inhibition to the glycolysis process of tumor cells can stimulate T cells to produce more IFN-γ. However, the reality is that the tumor inhibits the function of T cells by competing with glucose, resulting in low T cell reactivity and killing activity, although T cells recognize powerful tumor antigens [27,28]. In addition to the adverse effects of nutrient competition, the metabolites secreted by tumor cells can also adversely affect the body's anti-tumor immune response. Example with lactic acid, a metabolite produced by tumor cell glycolysis,is exported to the extracellular environment to form an acidic microenvironment by MCT1/MCT4. As alternative nutrient sources for TCA cycle of other tumor cells, lactic acid not only inhibits the restraining effect of the immune system on tumors in various ways to achieve immune escape of tumor cells, but also has a wide range of positive effects on the biological behavior of tumors such as angiogenesis and metastasis [29-31]. As important effector cells of the innate immune response, macrophages migrate to various tissues through the circulatory system and pass through the M1 (pro-inflammatory phenotype, favoring phagocytosis and killing) and M2 (anti-inflammatory phenotype, favoring tissue repair) phenotypic switch to play important biological roles at different sites. However, in tumor tissues, it has become a consensus that M2 type macrophages promote tumor progression [32]. In fact, both acidic environment of TME and lactate induce the polarization of macrophages to M2 type, prompting tumors to show better viability and adaptability, which are directly related to the resistance to radiotherapy and chemotherapy [30,33]. In contrast, the expression of genes associated with M1-type macrophages was continuously reduced under the stimulation of an acidic environment, severely inhibiting the activation of anti-tumor immune responses [34]. And recent studies have shown that, with the progression of tumors, the level of PD-1 on M2 macrophages is significantly increased compared with M1 macrophages, and the phagocytosis and antigen presenting ability to tumor cells of macrophage are weaken at the same time [35]. Similarly, as an important part of the innate immune response effector cells, NK cells mainly target and kill tumor cells that express stress proteins (such as MICA, MICB, ULBP4, etc.) or lack MHC class I expression specifically. Lactic acid can limit the activity of NK cells by inhibiting the secretion of IFN-γ and TNF-α. Knockdown of MCT4 expression on tumor cells can reduce lactate efflux and increase NK cell cytotoxicity [36]. In the adaptive immune response, cytotoxic T cells (also known as cytotoxic T lymphocytes, CTL, CD8+ cells) play a key role in anti-tumor immunity, which specifically recognizes MHC I-tumor oligopeptide antigen complexes targeted killing. Toxic proteins secreted by CTLs are endocytosed into the cytoplasm of target cells in the form of multiprotein complexes (including granzymes and perforin), which subsequently activate the pro-apoptotic protein caspase, thereby activating the apoptotic pathway. CTL can also activate the apoptosis pathway by expressing FasL, the ligand of Fas death receptor of target cells. We know that aerobic glycolysis is critical for T cell activation [37,38]. However, the high concentration of lactic acid in TME inhibits the output of lactate in T cells, thereby inhibiting the glycolysis process of CTL, resulting in a significantly decrease in the proliferative capacity and cytotoxic activity of CTL [39]. And more important, high concentrations of lactate inhibited the recruitment process of intratumoral CTL and decreased the expression of IFN-γ in CD8+ T cells [40]. In addition to CD8+ T cells, CD4+ T cells also target tumor cells in various ways, and at the same time promote the activation of CTL. As a result, CD4+ T cells largely ensures the quality of the CTL response during initiation, which is an important part of the anti-tumor immune response, significantly optimized CTL antitumor response [41]. However, lactate promotes the entry of lactic acid into CD4+ T cells by up-regulating the transporter SLC5A12 in CD4+ T cells surface. Lactic acid and inhibits CD4+ T cells through the PKM2/STAT3 pathway resulting in the ineffectiveness of CD4+ T cells, thereby further inhibiting the anti-tumor immune response activation [42] (Figure 1).
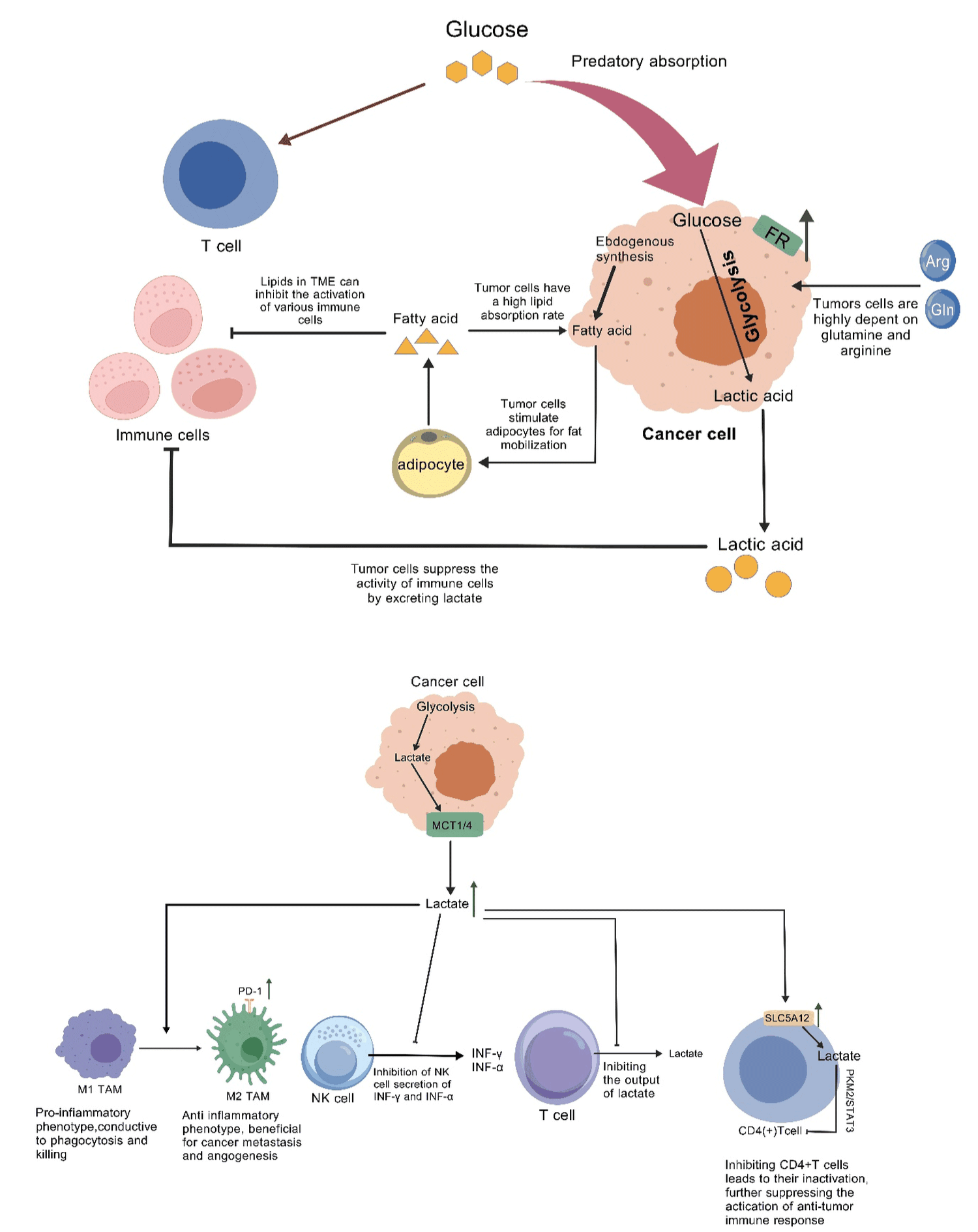 Figure 1: Lactic acid is a metabolic product produced by glycolysis of tumor cells, which is exported to the extracellular environment under the action of MCT1/MCT4, forming an acidic microenvironment. Acidic TME and lactate can induce macrophages to polarize towards M2 type, promoting better survival and adaptability of tumors, which is directly related to resistance to radiotherapy and chemotherapy; On the contrary, genes associated with M1 macrophages continue to decrease in expression under acidic stimulation, severely inhibiting the activation of anti-tumor immune responses; Moreover, recent studies have shown that the level of PD-1 on M2 macrophages is significantly elevated compared to M1 macrophages; And as the tumor progresses, the expression level of PD-1 on tumor associated macrophages gradually increases, while the phagocytic and antigen presentation abilities of tumor cells weaken . Lactic acid can reduce the activity of NK cells by inhibiting their secretion of IFN - γ and TNF - α. The high concentration of lactate in TME inhibits the output of lactate in T cells, thereby suppressing the glycolysis process of CTLs, leading to a decrease in their proliferation ability and a significant reduction in their cytotoxic activity. Lactic acid promotes the entry of lactate into CD4+T cells by upregulating the transport protein SLC5A12, and inhibits CD4+T cells through the PKM2/STAT3 pathway, leading to the inactivation of CD4+T cells and further suppressing the activation of anti-tumor immune responses.
View Figure 1
Figure 1: Lactic acid is a metabolic product produced by glycolysis of tumor cells, which is exported to the extracellular environment under the action of MCT1/MCT4, forming an acidic microenvironment. Acidic TME and lactate can induce macrophages to polarize towards M2 type, promoting better survival and adaptability of tumors, which is directly related to resistance to radiotherapy and chemotherapy; On the contrary, genes associated with M1 macrophages continue to decrease in expression under acidic stimulation, severely inhibiting the activation of anti-tumor immune responses; Moreover, recent studies have shown that the level of PD-1 on M2 macrophages is significantly elevated compared to M1 macrophages; And as the tumor progresses, the expression level of PD-1 on tumor associated macrophages gradually increases, while the phagocytic and antigen presentation abilities of tumor cells weaken . Lactic acid can reduce the activity of NK cells by inhibiting their secretion of IFN - γ and TNF - α. The high concentration of lactate in TME inhibits the output of lactate in T cells, thereby suppressing the glycolysis process of CTLs, leading to a decrease in their proliferation ability and a significant reduction in their cytotoxic activity. Lactic acid promotes the entry of lactate into CD4+T cells by upregulating the transport protein SLC5A12, and inhibits CD4+T cells through the PKM2/STAT3 pathway, leading to the inactivation of CD4+T cells and further suppressing the activation of anti-tumor immune responses.
View Figure 1
In summary, we can conclude that tumor tissues not only gain growth advantage by competing for glucose and inhibit the anti-tumor immune response, but also further inhibit the anti-tumor immune response by effluxing metabolite. Current studies have shown that removing the glucose competition of tumors and reducing the glycolysis rate of tumor tissue can significantly improve the efficiency of ICIs and significantly enhance the effect of anti-tumor immunity. In tumors with lower rates of glycolysis, CTLA-4 blockade showed better efficacy, resulting in more immune infiltration within the tumor tissue [43]. Treatment targeting lactate metabolism and lactate transport also significantly enhanced the efficacy of anti-PD-1 immunotherapy [44].
Lipid metabolism: Abnormal lipid metabolism is common in tumors. In some tumors such as renal cancer and prostate cancer, abnormal lipid metabolism significantly promotes the growth and metastasis of these tumors, and is closely related to drug resistance and radiotherapy resistance [45,46]. Tumors accumulate a large amount of lipids through endogenous synthesis and exogenous absorption, which provides raw materials for the synthesis of tumor cell membrane and organelle membrane. As a core exogenous lipid reserve, adipocytes promoted the proliferation and metastasis of melanoma [47], gastric cancer [47] and other cancers [48]. In breast cancer, tumor cells stimulate adipocytes for fat mobilization and fatty acids release. The released fatty acids are subsequently transferred to breast cancer cells that manifests as a marked increase in the rate of glucose oxidation, proliferation and increased ability to metastasis [49]. FABP4, a lipid transport protein in adipocytes, is highly regulated in various tumor [50]. Recently research reported that FABP4 could transfer fatty acids from adipocytes to cancer cells, which is associated with poor prognosis of the patients whose FABP4 is overexpression in tumor tissues. However, it is worth noting that compared with exogenous uptake, the main way for tumor cells to obtain lipids is endogenous lipid synthesis [51,52]. The inhibitions to enzymes involved in fatty acid synthesis,such as ATP-citrate lyase (ACLY) [53], Acetyl-CoA carboxylase 1(ACC1) [54], Fatty acid synthase (FAS) [55] and etc. could restrict tumor proliferation and metastasis significantly [56]. The high lipid synthesis rate and high lipid absorption rate of tumor cells make TME rich in lipids [57,58]. Studies have shown that lipids in these TMEs inhibit the activation of various immune cells. Su and Luo, et al. found that TAMs in human and mouse tumor tissues were rich in lipids. TAMs with high lipid levels exhibited an M2-type polarized phenotype while reducing the phagocytic ability and increasing PD-L1 expression. By inhibiting lipid uptake (blocking CD36), or selectively inhibiting PI3K-γ by IPI549, the functional activity of macrophages can be restored, promoting CTL-mediated tumor killing, and inhibiting tumor growth and progression in vivo [57,59]. In the lymphoma setting, fatty acid levels were significantly elevated, directly inhibiting the glycolytic process of NK cells, which ultimately manifested as low IFN-γ production rates and low killing activity. Inhibition of PPAR α/δ or blocking lipid transport reversed NK cells and restored cytotoxicity [60,61]. As one of the main effector cells of anti-tumor immunity, the activity of CTL is also closely related to the lipid concentration inside the TME. High free fatty acid concentrations can reduce the killing activity of CD8+ T cells [56,62]. In addition, the study found that after CD8+ T cells homing to the tumor, the high concentration of total cholesterol in T cells was associated with the characteristics of T cell exhaustion, low cytotoxicity, and obvious apoptosis in the TEM. The expression level of PD-1 in TME is significantly increased. Reducing TME or cholesterol in T cells can also enhance CD8+ T cells Antitumor activity of cells [48,63].
Amino acid metabolism : Amino acids, as basic metabolites, are essential for the synthesis of biological macromolecules that sustain life activities. According to the degree of dependence of the body, they can be divided into essential amino acids and non-essential amino acids. Essential amino acids refer to the amino acids that the human body cannot synthesize or synthesize very slowly and cannot meet the needs of the body and can only be ingested through food. Non-essential amino acids refer to amino acids that can be obtained by the human body through self-synthesis or conversion from other amino acids. Amino acids play a very important role in maintaining the balance of redox reactions, regulating energy metabolism, supporting the synthesis of biological macromolecules, and maintaining environmental homeostasis. However, tumor tissue proliferates rapidly and ingests a large amount of amino acids for nutritional maintenance in various ways, so that some of these non-essential amino acids have become essential amino acids in tumors [64,65].
Glutamine metabolism. As an important carbon and nitrogen source, glutamine is highly dependent on by most tumor cells [66]. Tumor glutamine metabolism involves a variety of tumor biological pathways, such as biomacromolecule synthesis (synthesis of proteins and nucleic acids), maintenance of REDOX balance (Glutamine can be deaminated to synthesize glutathione with cystine and glycine), energy supply (Continuous deamination to generate alpha ketone,which enters the tricarboxylic acid cycle) etc. In tumor cells, highly expressed glutaminase (GLS) catalyzes the hydrolysis of glutamine to glutamate and ammonia. Subsequently, glutamate dehydrogenase (GDH) catalyzes further deamination of glutamate to produce alpha-ketoglutaric acid and ammonia. Glutamic acid and α-ketoglutaric acid enter the glutathione synthesis pathway and tricarboxylic acid cycle, respectively, to promote the growth of tumor tissue [67]. At the same time, under the action of GLS and GDH, a large amount of ammonia is released, and then ammonia is absorbed by nearby tumor-associated fibrocytes (CAF), which promotes autophagy of CAF cells and releases glutamine, making glutamine return to tumor cells, and then supporting tumor cell growth. The over-dependence of tumor cells on glutamine has led to extensive metabolic inhibition of tumor cells by anti-tumor therapy targeting glutamine metabolism, resulting in interruption of NADP (H) homeostasis and growth obstruction in tumor cells. Studies have shown that inhibition of glutamine-related metabolic enzymes can effectively reduce the proliferation rate of tumor cells. Therefore, according to the glutamine-dependent metabolic characteristics of tumor cells, a variety of glutamine-targeting compounds have been developed and synthesized [68], and in vitro experiments, the proliferation and invasion ability of a variety of tumor cells have been significantly inhibited. It is worth mentioning that, unlike tumor cells, for normal cells, glycolysis, OXPHOS and glutamine metabolism are easily converted and have strong plasticity, and inhibiting glutamine metabolism in normal cells has little effect on the function of normal cells.Therefore, anti-glutamine therapy targeting tumor cells can significantly reduce the effect on normal cells, especially T cells. Recent studies have found that the use of anti-tumor therapy of anti-glutamine can promote the adaptive metabolic reprogramming of T cells, reduce T cell dependence on glutamine, and enhance the survival, proliferation and effect function of T cells [69]. Glutamine antagonist combined with anti-PD-1 therapy can effectively enhance the anti-tumor effect of immune cells, which is manifested as a significant increase in the permeability of high proliferation rate and highly active CD8+T cells, and an increase in the long-term memory-like T cell phenotype [70-72]. Anti-glutamine therapy plays an important role in the innate immune response against tumors. The application of anti-glutamine synthesis therapy can reprogram the phenotype of macrophages to differentiate into macrophages with M1-like phenotype, and resist immunosuppression and tumor blood vessel budding [73,74].
Arginine : Arginine is a conditionally essential amino acid that detoxifies ammonia in the body through the urea cycle. In addition to exogenous uptake of arginine, the body syntheses arginine through two key enzymes, arginine succinate synthetase (AS) and arginine succinate lyase (ASL), from the "enterorenal axis" or "citrulline-nitric oxide cycle". Under normal circumstances, the body is self-sufficient; During periods of stress or rapid growth, the supply of arginine becomes very limited. In tumor tissues, AS is almost absent or less expressed, so tumors are heavily dependent on exogenous arginine. So the tumor is heavily dependent on exogenous arginine. Studies have shown that arginine is necessary for the growth of tumor cells in vitro , and arginine deprivation leads to the death of most malignant proliferating cells [75-77]. After ingestion of arginine by tumor cells, arginase Arg1/Arg2 rapidly transforms arginine, and high expression of Arg1/Arg2 is associated with poor survival of tumor patients [78-80]. In addition, arginine is also the only substrate for the production of NO in vivo , which plays an important role in promoting tumor angiogenesis and regulating the DNA repair damage of tumor cells [81,82]. In fact, TAM and CAF also contain highly active arginase. Studies have shown that these highly expressed arginases are closely related to the low survival rate of solid tumors [83-86]. In recent years, more and more evidence shows that the role of arginine in immune regulation has been gradually discovered, especially in the activation of macrophages, dendritic cells and T cells. Slight changes in arginine determine different immune results [87]. Activated T cells need to consume large amounts of arginine and induce metabolic reprogramming of T cells from glycolysis to OXPHOS while converting it to downstream metabolites. Arginine can significantly improve the survival rate of CD4+ and CD8+T cells. However, the inhibition of arginase in T cells will not lead to the proliferation of T cells, and even can significantly improve the survival ability of active CD4+T cells and the anti-tumor activity of CD8+T cells in vivo [88,89]. CB-1158, an oral arginase inhibitor, can significantly increase the infiltration of CD8+ T cells and NK cells in tumor tissues, and induce the expression of inflammatory cytokines and interferon-inducible genes, which turns the anti-inflammatory environment in TME into a pro-inflammatory environment. At present, the related therapy targeting the metabolic pathway of arginine combined with immunotherapy is considered to be an effective anticancer strategy, which not only inhibits the proliferation of tumor cells and the cancer-promoting effect of related stromal cells, but also changes the environment of TME and promotes immune infiltration [90-92].
Tryptophan : Tryptophan is an essential amino acid that can only be obtained from food and plays an important role in the body as a signal transduction molecule. In recent years, the role of tryptophan in immune regulation has been gradually discovered, and the disorder of tryptophan metabolism can lead to the apoptosis of immune cells and related dysfunction. Although tryptophan is one of the essential amino acids in the human body, in fact, only a small amount of tryptophan is used for protein synthesis. More than 95% of free tryptophan is metabolized through Kyn pathway, and indoleamine 2, 3-dioxygenase 1/2 (IDO1/IDO2) and tryptophan 2, 3-dioxygenase (TDO) are key enzymes in this pathway. In various tumor tissues such as lung cancer, colon cancer, skin cancer, pancreatic cancer and breast cancer, IDO1 and TDO are often abnormally activated and are significantly associated with poor prognosis. Kynuridine produced by IDO1 degradation of tryptophan promotes β-catenin activation through PI3K/AKT/ GSK-3β pathway, leading to the proliferation of bowel cancer cells. In addition to tumor cells, stromal cells and macrophages in TME have high expression of IDO1, resulting in tryptophan deficiency and kynuridine accumulation in TME. On the one hand, tryptophan depletion in TME can inhibit the function of antigen-presenting cells, and lead to the apoptosis of major tumor killer cells: CD8+ T cells and NK cells. On the other hand, kynuridine can enter CD8+ T cells through the transporter SLC7A5 and then up-regulate the expression of PD-1 in CD8+ cells, ultimately leading to significant inhibition of anti-tumor immunity [93-97]. The obvious immunosuppressive effect of kynuridine has attracted a large number of anti-tumor drugs, especially small molecule inhibitors for Kynuridine synthesis, which is the focus and hot spot of anti-tumor drugs at present. Various types of inhibitors targeting tryptophan metabolic pathways are also being developed, but the final effect of clinical single drug application is not ideal [98]. However, it has been found that the treatment of anti-tryptophan metabolism can improve the efficacy of ICIs, and its related results have been achieved in the treatment of melanoma, kidney cancer and lung cancer [99-102] (Figure 2).
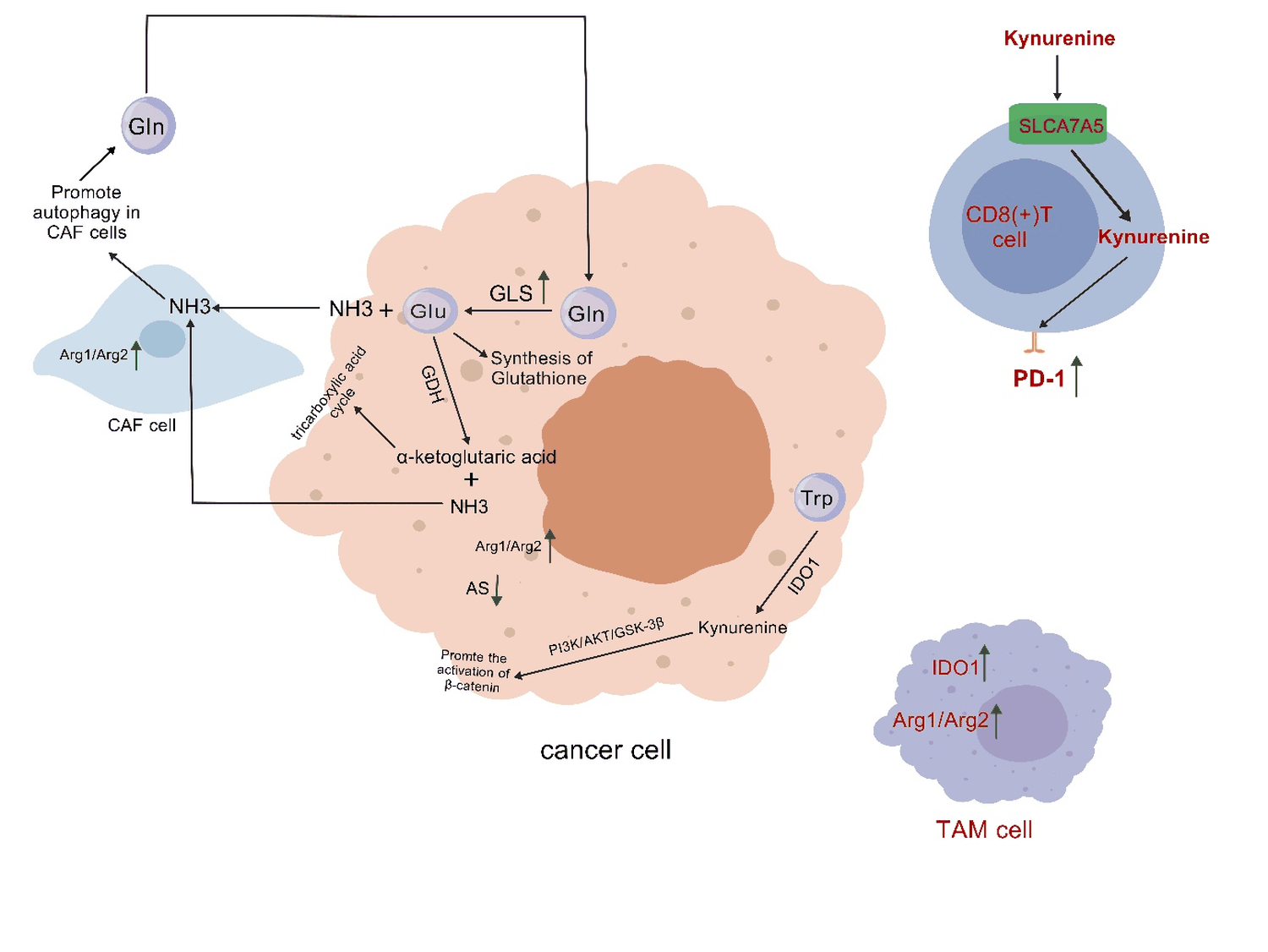 Figure 2: In tumor cells, highly expressed glutaminase (GLS) catalyzes the hydrolysis of glutamine into glutamate and ammonia. Subsequently, glutamate dehydrogenase (GDH) catalyzes the further deamination of glutamate, producing alpha ketoglutarate and ammonia. Glutamic acid and α - ketoglutarate enter the glutathione synthesis pathway and tricarboxylic acid cycle, respectively, promoting the growth of tumor tissue. At the same time, under the action of GLS and GDH enzymes, a large amount of ammonia is released, which is then absorbed by adjacent tumor associated fibroblasts (CAFs), promoting autophagy and releasing glutamine in CAF cells, causing glutamine to reflux back to tumor cells, thereby supporting tumor cell growth. In tumor tissues, AS is rarely expressed or expressed at low levels, so tumors heavily rely on exogenous arginine. Research has shown that arginine is essential for the growth of tumor cells in vitro, and arginine deprivation leads to the death of most malignant proliferating cells. After tumor cells ingest arginine, arginase Arg1/Arg2 rapidly converts it, and high expression of Arg1/Arg2 is associated with poorer survival rates in tumor patients. In addition, arginine is also the only substrate for the generation of NO in the body, playing an important role in promoting tumor angiogenesis and regulating the process of DNA repair and damage in tumor cells. In fact, TAM and CAF also contain highly active arginase. In a variety of tumor tissues, such as lung cancer, colon cancer, skin cancer, pancreatic cancer and breast cancer, IDO1 and TDO are often abnormally activated, and are significantly associated with poor prognosis. IDO1 degrades tryptophan to produce canine urea, which promotes the activation of β - catenin through the PI3K/AKT/GSK-3 β pathway, leading to the proliferation of colon cancer cells. In addition to tumor cells, stromal cells and macrophages within the TME also highly express IDO1, leading to tryptophan deficiency and accumulation of kynurenine in the TME. On the one hand, depletion of tryptophan in TME inhibits the function of antigen-presenting cells and leads to apoptosis of major tumor killing cells: CD8+T cells and NK cells; On the other hand, canine uric acid enters CD8+T cells through the transporter SLC7A5, upregulating the expression of PD-1 in CD8+cells, ultimately leading to significant inhibition of anti-tumor immunity.
View Figure 2
Figure 2: In tumor cells, highly expressed glutaminase (GLS) catalyzes the hydrolysis of glutamine into glutamate and ammonia. Subsequently, glutamate dehydrogenase (GDH) catalyzes the further deamination of glutamate, producing alpha ketoglutarate and ammonia. Glutamic acid and α - ketoglutarate enter the glutathione synthesis pathway and tricarboxylic acid cycle, respectively, promoting the growth of tumor tissue. At the same time, under the action of GLS and GDH enzymes, a large amount of ammonia is released, which is then absorbed by adjacent tumor associated fibroblasts (CAFs), promoting autophagy and releasing glutamine in CAF cells, causing glutamine to reflux back to tumor cells, thereby supporting tumor cell growth. In tumor tissues, AS is rarely expressed or expressed at low levels, so tumors heavily rely on exogenous arginine. Research has shown that arginine is essential for the growth of tumor cells in vitro, and arginine deprivation leads to the death of most malignant proliferating cells. After tumor cells ingest arginine, arginase Arg1/Arg2 rapidly converts it, and high expression of Arg1/Arg2 is associated with poorer survival rates in tumor patients. In addition, arginine is also the only substrate for the generation of NO in the body, playing an important role in promoting tumor angiogenesis and regulating the process of DNA repair and damage in tumor cells. In fact, TAM and CAF also contain highly active arginase. In a variety of tumor tissues, such as lung cancer, colon cancer, skin cancer, pancreatic cancer and breast cancer, IDO1 and TDO are often abnormally activated, and are significantly associated with poor prognosis. IDO1 degrades tryptophan to produce canine urea, which promotes the activation of β - catenin through the PI3K/AKT/GSK-3 β pathway, leading to the proliferation of colon cancer cells. In addition to tumor cells, stromal cells and macrophages within the TME also highly express IDO1, leading to tryptophan deficiency and accumulation of kynurenine in the TME. On the one hand, depletion of tryptophan in TME inhibits the function of antigen-presenting cells and leads to apoptosis of major tumor killing cells: CD8+T cells and NK cells; On the other hand, canine uric acid enters CD8+T cells through the transporter SLC7A5, upregulating the expression of PD-1 in CD8+cells, ultimately leading to significant inhibition of anti-tumor immunity.
View Figure 2
Nucleotide metabolism : Nucleotides, as the basic components of DNA and RNA, are widely distributed in the body and play a variety of important biological functions. Intracellular nucleotides are mainly synthesized by the organism's cells themselves,and their anabolic pathways play an important role in the occurrence and development of organisms, which are mainly divided into two types: de novo synthesis pathway and salvage pathway. In particular, rapidly growing tumor cells have significantly increased metabolic requirements such as DNA synthesis and RNA synthesis. However, the nucleotides provided by the remedial synthesis pathway are very limited, so the de novo synthesis pathway of nucleotides is particularly important [103]. Different from lipid synthesis and protein synthesis, nucleotide anabolism is a relatively complex and comprehensive metabolic process, requiring a variety of amino acids, CO2, carbon unit and other raw materials [104,105]. It has long been found that antimetabolic therapy targeting related nucleotide metabolism can significantly inhibit the proliferation of tumor cells and promote the death of tumor cells. As the basic materials of nucleotide synthesis, one carbon unit mainly comes from the serine, glycine, tryptophan and histidine catabolism, including methyl (CH3, methyl), a alkenyl (=CH2, methylene), methenyl (-CH=, methenyl), formyl (-CHO, formyl) and iminomethyl (-CH=NH, formimino). As a carbon unit transferor, folic acid closely links the metabolism of one carbon unit with the metabolism of nucleotides. Folic acid cycle is one of the core reactions of nucleotide metabolism and participates in the synthesis and transformation of many biological macromolecules. The use of the antifolate chemotherapeutic drug methotrexate became a milestone in antitumor therapy. Methotrexate inhibits the production of tetrahydrofolate by inhibiting dihydrofolate reductase, which interrupts folate circulation, and significantly inhibits tumor proliferation. In addition, a variety of tumor cells express high folate receptors on their membrane surfaces with the purpose of converting folate and taking in large amounts of folate. Therefore, a variety of drugs will be conjugated with folic acid to improve the affinity of drugs and tumor cells, and the relevant has entered the clinical trial stage. Some results have been achieved [103]. In addition to the high expression of folate in tumor cells, it was found that there are also high expression of folate receptors on TAM, and folate receptor beta (FRβ) positive TAM subpopulations exhibit an immunosuppressive M2-like phenotype. CAT-T mediated FRβ+TAM clearance in selective TME,which can increase the recruitment of CD8+ T cells in tumor tissue, limit tumor growth, and prolong survival. Folate receptor (FR4) is also highly expressed in CD25+ CD4+ Treg cells, and antagonizing FR4 with specific antibodies can enhance the anti-tumor immune process both in vivo and in vitro [106]. Methotrexate, a folate analogue, also promotes the maturation of antigen-presenting cells, increases the proliferation and activation of CD8+ T cells, and releases IFN-γ [107] (Figure 3). In pancreatic cancer, the use of mFOLFIRINOX, which includes antinucleotide metabolites, can increase the probability of surgery and prolong survival, and may be associated with the patient's T cells and monocytes [108]. 5-fluorouracil can enhance the anti-PD-1 effect and promote the infiltration of CD8+ T cells in tumor tissues [109-111]. In conclusion, targeting the nucleotide anabolic pathway in tumor tissue can enhance the effect of anti-tumor immune response.
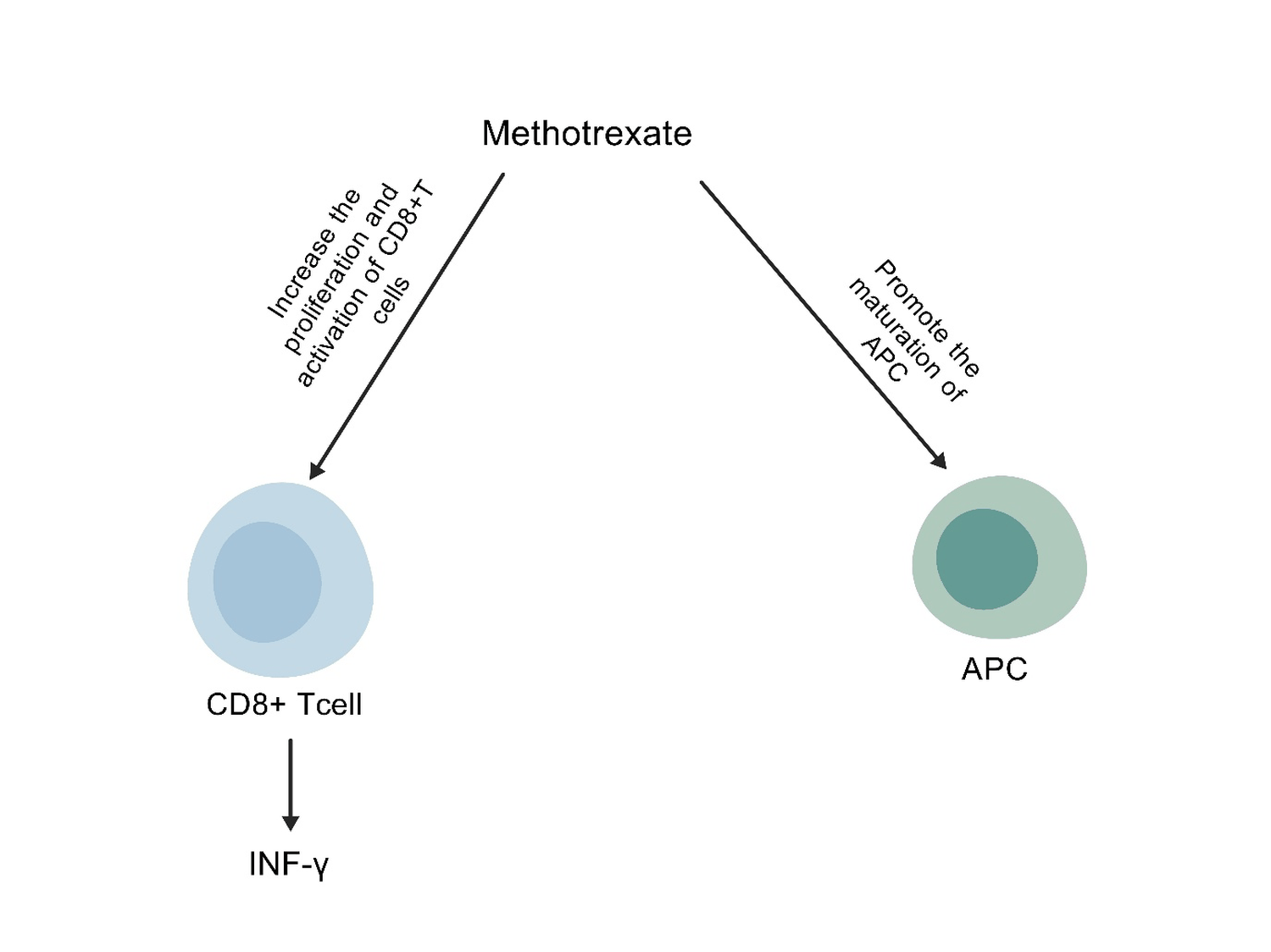 Figure 3: In addition to tumor cells highly expressing folate receptors, studies have found that there are also highly expressed folate receptors on TAMs, and the folate receptor beta (FR β) - positive TAMs exhibit an M2 like phenotype with immune suppression. Folic acid receptor 4 (FR4) is also highly expressed in CD25+CD4+Treg cells, and antagonizing FR4 with specific antibodies can enhance the anti-tumor immune process both in vivo and in vitro [104]; Methotrexate, an analog of folate, can also promote the maturation of antigen-presenting cells, increase the proliferation and activation of CD8+T cells, and release IFN -γ.
View Figure 3
Figure 3: In addition to tumor cells highly expressing folate receptors, studies have found that there are also highly expressed folate receptors on TAMs, and the folate receptor beta (FR β) - positive TAMs exhibit an M2 like phenotype with immune suppression. Folic acid receptor 4 (FR4) is also highly expressed in CD25+CD4+Treg cells, and antagonizing FR4 with specific antibodies can enhance the anti-tumor immune process both in vivo and in vitro [104]; Methotrexate, an analog of folate, can also promote the maturation of antigen-presenting cells, increase the proliferation and activation of CD8+T cells, and release IFN -γ.
View Figure 3
Combined with relevant literature reports, we can find that the internal microenvironment of tumor tissues is completely different from the environment of other parts of the body: the chaotic structure and malformed blood vessels in tumor tissues give tumor tissues a strong barrier. Within this barrier, tumor cells frantically plunder nutrients and expel metabolites (tumor cells can even reprogram these metabolites to produce energy again). On the one hand, the effector cells of anti-tumor immunity are deprived of nutrition, on the other hand, they are affected by metabolites, which eventually leads to the state of "more than willing but less capable" of effector cells, so that the anti-tumor immune effector cells are exhausted or apoptotic. What's more, some immune effector cells become accomplices for tumor cells and are used by tumor cells (such as macrophages), promoting the occurrence and development of tumors. The entry of nutrients into tumor cells is one of the fundamental causes of this situation. How to change the destination of nutrients while avoiding the adverse effects of metabolites of nutrients is a major research direction to efficiently kill tumors and improve anti-tumor immune response, which is worthy of in-depth exploration and research.
This review explores the complex relationship between tumor metabolism and immune evasion and evaluates the potential of combining metabolic-targeted therapies with immunotherapy. Although existing studies highlight the critical role of metabolic pathways in tumor immune evasion, their applicability across diverse patient populations remains insufficiently validated. Tumor metabolic characteristics vary significantly across different cancer types and stages, making it difficult to achieve consistent efficacy with metabolic-targeted therapies in a broad patient population. Current research is largely based on small-scale animal studies and short-term clinical trials, lacking sufficient long-term data to fully assess therapeutic efficacy and side effects. Furthermore, how to precisely regulate tumor metabolism without harming normal cells, as well as overcoming resistance due to metabolic adaptation, remains a major challenge for future research.
Future research should focus on several key areas: First, developing personalized treatment strategies through the precise selection of metabolic biomarkers to optimize immunotherapy outcomes and ensure more targeted treatments. Second, large-scale clinical trials should further evaluate the combined effects of metabolic modulation and immune checkpoint inhibitors (e.g., PD-1/PD-L1 inhibitors), particularly in patients with refractory and metastatic cancers. Additionally, there is a need to explore the regulation of the metabolic microenvironment, especially the accumulation of key metabolites (such as lactate and fatty acids), to enhance immune cell activity and anti-tumor responses.
In terms of clinical application, the combination of metabolic-targeted therapy and immunotherapy shows significant potential in tumor types with prominent metabolic reprogramming. Future clinical trials should assess the efficacy and safety of these combined therapies, while utilizing metabolic biomarker screening systems to provide personalized treatment options for patients. Despite the challenges in this field, with continued research, the integration of metabolic-targeted therapy and immunotherapy could become a breakthrough strategy, significantly improving patient outcomes and survival.
In conclusion, research on tumor metabolism and immune evasion provides a solid theoretical foundation for new therapeutic strategies. However, to translate these findings into clinical practice, several technical and clinical challenges must be overcome. Future research should focus on the applicability of metabolic-targeted therapies, the issue of resistance, and the clinical validation of combined strategies with immunotherapy to drive significant progress in this field.
Not applicable.
D.J., B.Y., and B.C. conceived and designed the study and coordinated the project. B.Z. collected the data. R.L. and Q.Z. reviewed the manuscript and provided critical comments. All authors participated in the revision of the manuscript and approved the submitted version.
N/A.
N/A.
None declared.
National Natural Science Foundation of China (grant number 82303192 to Doudou Jing).
No new data were generated or analysed in support of this research.