In cancer and inflammation, the haemoreological response is altered and remains in flux. The study of hemorheology may provide valuable information on the disease progression and therapeutic response.
Full blood profile, fibrinogen level, C-reactive protein (CRP) and whole blood viscosity (WBV) was measured in 100 randomly selected oral cancer patients enrolled to receive palliative chemotherapy.
Whole blood viscosity (WBV) was raised in all the cancer patients. A fall in the whole blood viscosity was observed during the course of the therapy. Incidentally, some changes in the fibrinogen and CRP levels was also observed in them.
Hyper-viscosity of blood was documented in cancer. A marginal fall in the blood viscosity was noted during treatment. In some patients, a significant fall in the viscosity, hematocrit and plasma protein level was observed. We noted no post-treatment survival benefit in them. The fall in viscosity was possibly linked to lowering of immunological factors, anti-inflammatory response, reduction in cancer cell density and also serum proteins and hematocrit. We need to conduct well designed studies in the field of hemorheology, cancer and inflammation to understand such complex underlying phenomenon.
Cancer, Chemotherapy, Whole blood viscosity
Haemoreological study in health and disease includes analysis of flow properties of blood, blood viscosity, and resistance to flow, tissue perfusion, and endothelial function. In cancer and inflammation, an altered haemoreological behavior and a state of hypercoagulability is often observed [1,2]. Whole blood viscosity is known to be influenced by circulating immunoglobulins, hematocrit, fibrinogen and proteins. We analyzed whole blood viscosity (WBV), Hematocrit, C-reactive protein (CRP) and Fibrinogen in terminally ill oral cancer patients. Unlike conventional chemotherapy, the patients received a low dose continuous regime of methotrexate and COX-2 inhibitor, also known as palliative metronomic chemotherapy [3]. The scientific basis of its therapeutic action was believed to be through vascular & immune modulation.
Full blood profile, CRP, serum proteins and blood viscosity at low shear rate (5-15 Sec-1) on Day 0 (pre-therapy), Day 30 and Day 180 (end of 6 month survival point) was done in 100 randomly selected oral cancer patients enrolled to receive low dose metronomic palliative chemotherapy. The patients belonged to diverse background having diverse food or diet habits, but were mostly vegetarians. The experimental test run was initiated with a venipuncture on the patient using a 19-gauge stainless steel needle. Fresh blood was first directed from the first to the second stopcock to collect blood into the syringe. About 5 ml of blood was collected in the syringe for tests with a cone-and-plate viscometer (Brookfield DV-III) and hematocrit measurements, and the syringe was then removed from the system. Approximately 0.5 ml of this fresh blood from the syringe was immediately transferred to the sample cup of the Brookfield rotating viscometer that was maintained at a constant temperature of 37 ℃ by a water bath connected to the cup. The viscosity (cP) measurements at different low shear rate (5-15 Sec-1) with the rotating viscometer were completed within approximately 1 min from the time when the blood left the human body. Ethics approval: Research project (including collection of blood or tissues samples) was started after obtaining approval of the TMC- ACTREC IRB on dated, 06-06-2013, No-94. It was conducted in accordance with the ICH-GCP and ICMR ethical guidelines, 2006 involving human subjects.
Mean age of the patients under treatment was 47.66 Yrs; Male 86 % and female 14%; Mean Hemoglobin (Hb) was 11.9 gm % ± 1.92 gm %; Average value of WBV was higher in cancer patients (7.10 ± 2.96) compared to normal healthy population (5.5 ± 3.5). Baseline blood fibrinogen level was also generally high (> 400 mg/dl) in most of the cancer patients. Incidentally, we also noted a direct co-relationship between fibrinogen level and viscosity (Figure 1). A fall in the WBV on day 30 of receiving chemotherapy was noted in all the cancer patients (Table 1).
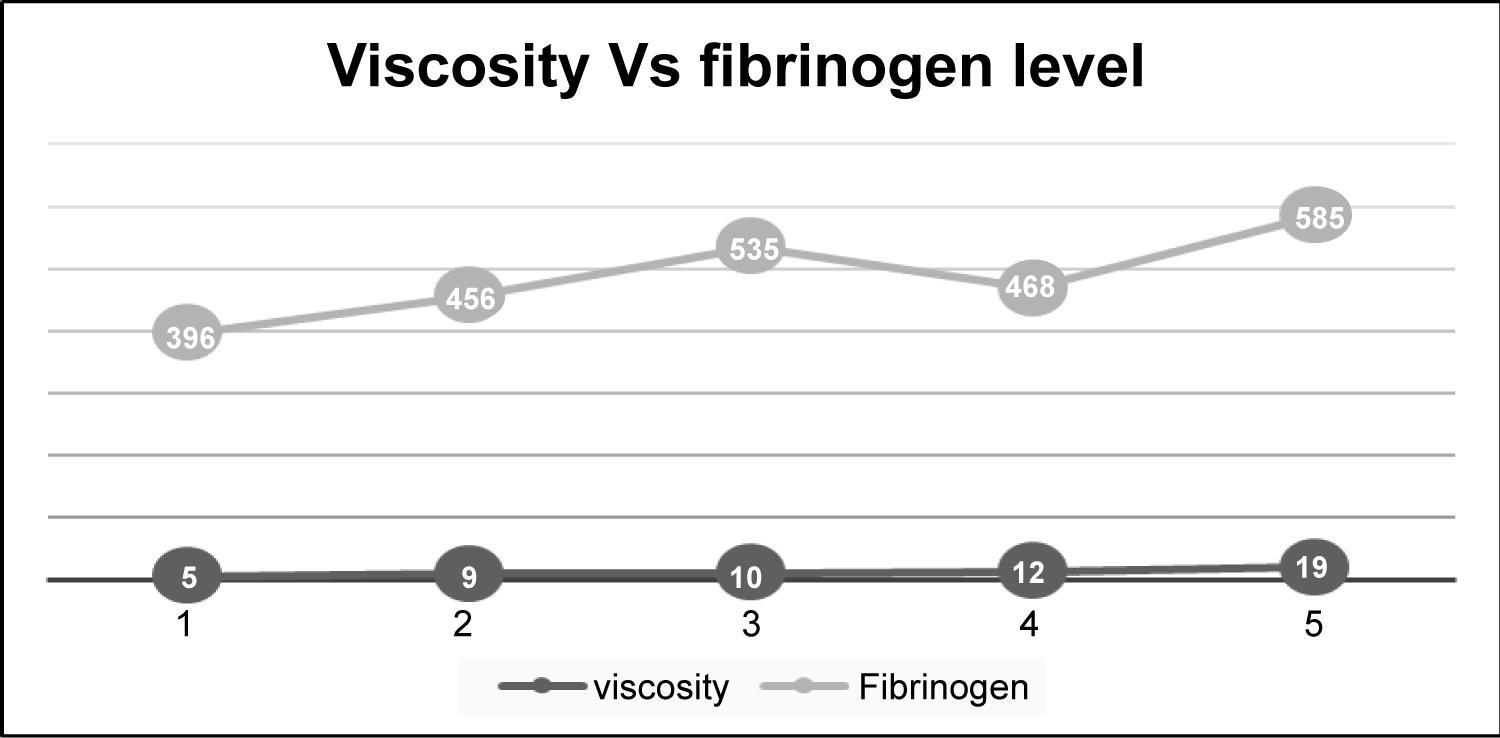 Figure 1: A positive correlation between blood viscosity (cP) and fibrinogen level (mg/dl).
View Figure 1
Figure 1: A positive correlation between blood viscosity (cP) and fibrinogen level (mg/dl).
View Figure 1
Table 1: Whole blood viscosity value during palliative chemotherapy on day 0, day 30 and day 180. View Table 1
A marginal fall in WBV was noted in all the cancer patients on day 30 of treatment. However in 8 patients, so called responders, who survived for more than 180 days, no further fall in WBV (6.15 cP @ D30 and 6.18 cP @ D180) was observed. Hematocrit level did not show any significant fall in them suggesting absence of anemia. Blood fibrinogen level remained high. On the other hand, a significant fall in viscosity (< 4.5 cP) on day 30 of therapy was observed in poor responders (N = 9). A low hemoglobin (< 10 gm %) and serum protein level (Mean value < 3.0 gm/dl) was observed in them during treatment. All the enrolled patients were routinely assessed for nutritional status and advised balanced diet. The compliance was poor in majority of them due to both socio-economic and health reasons. Most of them suffered from varying degree of anorexia, nausea and vomiting during treatment. A fall or lowering of viscosity is known to improve tissue perfusion and drug delivery but we did not find any significant survival benefit in patients who had significant fall in WBV during early days of therapy. Therefore, we can assume that the fall in viscosity following intake of NSAID and low dose methotrexate (cytotoxic drug) in most cancer patients was possibly linked to lowering of immunological factors, anti-inflammatory response and reduction in cancer cell density. Whereas in some non-responders, a greater fall in viscosity was both as a consequence of anti-inflammatory (NSAID) and cytotoxic drug (methotrexate) action as well as anemia and hypoproteinemia. Anorexia, nausea and bone marrow suppression resulting from medicine side effects and disease progression led to anemia and hypoproteinemia. Blood coagulation factors Fibrinogen level directly influences blood viscosity. Incidentally, we also noted a fall in the C-reactive proteins (CRP) in all the treatment responders on day 30 and day 180 of therapy. We reported variation in the microvascular flux during tumor progression and regressions [4]. The scientists working in the field of hemorheology and inflammation have documented differential action of anti-inflammatory agents such as NSAIDS on vascular remodeling, systemic and local haemoreological & immunological response [5-7]. However, a clear understanding of the key underlying interactions and causal relationship between all the above mentioned factors is lacking in cancer. Cancer is known to be an inflammatory disease. Therefore, large well designed studies are needed to improve our understanding on the role different immunoglobulins, cytokines, other immune & inflammatory markers, cytotoxic and anti-inflammatory agents play in relation to haemoreological status in cancer and inflammation.
Cancer is a hyper-viscous condition. Effective treatment may cause a fall in blood viscosity but the specific mechanism remained largely unanswered as of now. Future studies should focus on the role played by immunoglobulins, cytokines, cancer cell density and other anti-inflammatory markers on cancer hemorheology and therapeutics.
The author sincerely thanks Dr. S. V Chiplunkar, Director, ACTREC-TMC for releasing the fund to procure viscometer. We are grateful to Dr. Kumar Prabhash, Dr. Amit Joshi, Medical oncologist and their team for meticulous clinical follow up of the patient. We also thank all the patients and staff who cooperated in executing this study.
None.