Long-term infection with human papillomavirus (HPV) is the cause of cervical cancer and its precursor - cervical intraepithelial neoplasia (CIN). The presence of HPV infection can be presumed in more than 99% of cases of cervical cancer worldwide. The introduction of DNA testing for the presence of HPV has increased the effectiveness of screening programs for the detection of this cancer. This article contains the latest reports on the structure, function and role of HPV in the development of cervical cancer. It was also underlined the importance of performing molecular tests for the presence of HPV DNA as a more sensitive and the most accurate method of detecting the threat posed by infection with this oncovirus. The review also highlights the most beneficial prophylactic algorithm, which should be guided by gynecologists during the diagnosis, individual clinical cases.
HPV, Cervical cancer, Virus, Carcinogenesis, Oncovirus
In December 2018, it will pass exactly 10 years from the day Professor Harald zur Hausen, a German researcher, virologist and physician received the Nobel Prize in physiology and medicine, for a breakthrough discovery of showing the irrefutable correlation between human HPV (Human Papillomavirus) and the occurrence of neoplastic lesions in normal cells, primarily the cervix [1]. Describing this phenomenon seems even more important, given that until the late 1970s, Papillomaviruses (PVs) were treated in a marginal way, as they were only attributed to the development of benign skin lesions, most commonly in the form of human warts and some mammals. The development of techniques of genetic engineering and molecular biology that took place in the 80's undoubtedly contributed to the expansion of knowledge about HPV. Thanks to the current polymerase chain reaction (PCR) technique (Polymerase Chain Reaction), it has been possible to discover and describe over 200 types of this virus, of which in about 150 cases the isolation and complete sequencing of viral genomes has been made. Moreover, it has been proven that HPV types are molecularly quite divergent, and individual viral genomes evolve at a similarly slow pace as in the genomes of their hosts [2-4].
The modern HPV taxonomy is based primarily on the assessment of changes that occur in the evolution of viruses, and not on their characteristic phenotypic traits. This direction of research is influenced by the fact that various, often very phylogenetically distant HPV types may be associated with the induction of similar symptoms. A perfect example of this phenomenon are HPV16 and HPV18, both highly oncogenic types (occurring in pair or individually), having an indisputable effect on the development of cervical cancer. They are definitely less related to each other than to the types of virus whose presence or impact on the carcinogenesis of this organ has never been confirmed [3].
The unprotected HPV virion is a circular, covalently closed dsDNA molecule (double-stranded DNA) of approximately 8 kbp, with only one coding thread. Capsid is a form consisting of the following structures:
• L, so-called Late Genes - L1 and L2, which are also assigned a function related to the virus's entry into epithelial cells, the assembly and release of posterior virions and the infectivity of the molecule,
• E, so-called Early Genes - E1, E2, E4, E5, E6, E7, which encode regulatory proteins, thus corresponding to the survival of the virus in the host cell, initiation of viral genome replication, or activation of the lytic cycle,
• LCR, so-called long regulatory region - taking part, among others, in virus replication (Figure 1) [3,5,6].
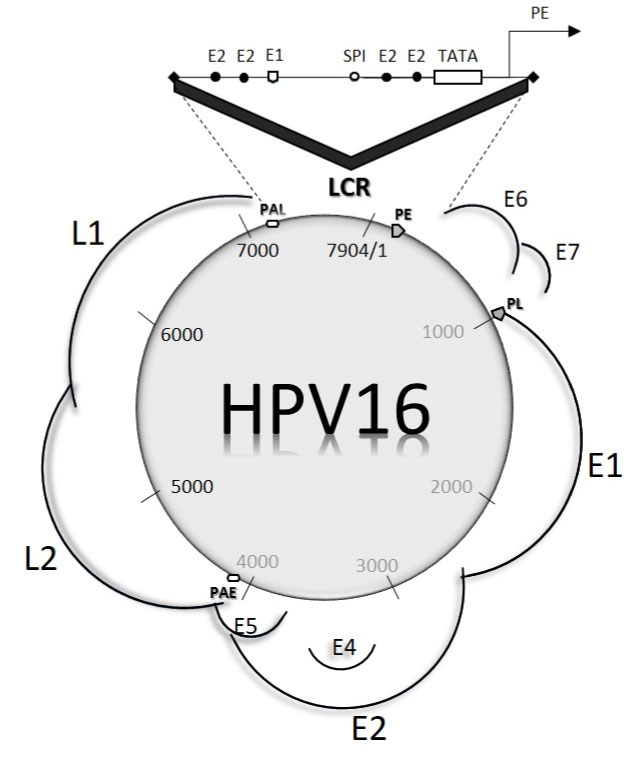 Figure 1: HPV 16 genome, as an example of a typical molecular structure for high-oncogenic viruses. Genes building a molecule encode a large number of protein products resulting from mRNA splicing. The LCR section contains binding sites for cellular transcription factors (SPI). Also distinguished are characteristic promoter structures (PE - Early Promtor, P - Late Promotor), responsible for regulating the expression of mRNA molecules, differentiated as a result of splicing, during epithelial cell division. PAE (Proximal Early Polyadenylation Signal) and PAL (distal Late Poliadenylation Signal) responsible for "pointing" space in the early and late polyadenylation site in the genome, i.e. the modification of the mRNA in eukaryotic cells. View Figure 1
Figure 1: HPV 16 genome, as an example of a typical molecular structure for high-oncogenic viruses. Genes building a molecule encode a large number of protein products resulting from mRNA splicing. The LCR section contains binding sites for cellular transcription factors (SPI). Also distinguished are characteristic promoter structures (PE - Early Promtor, P - Late Promotor), responsible for regulating the expression of mRNA molecules, differentiated as a result of splicing, during epithelial cell division. PAE (Proximal Early Polyadenylation Signal) and PAL (distal Late Poliadenylation Signal) responsible for "pointing" space in the early and late polyadenylation site in the genome, i.e. the modification of the mRNA in eukaryotic cells. View Figure 1
By sequencing the individual types of Papillomaviruses that scientists isolated from human cells (i.e. HPVs), the viruses were divided into five groups (α, β, γ, μ, ν), differing mainly from the life cycle and diseases caused by them. Most of them cause low-invasive infections in people with an optimal level of immunity, which is certainly related to the fact that the interactions between host cells and individual types of PVs have been co-motivating for hundreds of years. This led to the phenomenon in which the virus adapting to the host, can carry out and end its life cycle, without causing visible disease and at the same time remaining in the population [4].
Clinically the most important types of virus belong to the alpha-Papillomaviruses group (α-PVs). It is said then about viruses that are imperative of changes in the mucous membrane or genital organs. They were also divided into subgroups related to the level of risk and oncogenic potential that is associated with infection with these viruses. The viruses were divided into high-risk (hrHPV) and low-risk viruses (lr-HPV) [3,7,8].
The gateway to HPV infection is the formation of a minute of the basal layer of squamous epithelial cells and mucous membranes into which the virus is tropism. After binding to the appropriate receptor on the epithelial cell surface (some data suggest that the receptors may be heparin sulfate or α6β4 integrins), for which the L1 structural protein corresponds, the virus performs a productive replication cycle [9]. Its course is conditioned by the efficiency of the epithelial maturation process, specific systemic and cellular factors as well as viral proteins [9,10]. The early structural proteins of the virus play a key role in the infection. E5 oncoprotein induces the activity of an enzyme inducing the process of cellular apoptosis - COX-2. The E6 protein binds to the p53 protein, which in the host cell is one of the key elements regulating the cell cycle, and the resulting E6-p53 complex excludes p53-dependent regulation of cell division processes, resulting in a state of instability at the chromosomal level, thus increasing the degree of cellular mutation, having a direct impact on its proliferation and the development of cancerous transformation. In addition, E6 forms complex structures with other proteins that are involved, for example, in DNA repair mechanisms or cell cycle control mechanisms (e.g., TNFR-1, called Tumor Necrosisi Factor Receptor 1, MGMT, or O6-Methylguainine-DNA Methyltransferase). In turn, E7 oncoprotein binds to the Rb (Retinoblastoma protein) suppressor protein (in addition to the p107 and p130 proteins), which is responsible for maintaining the cell in the G1 phase of the cycle. The resulting complex implies the release of E2F, which is responsible for above-average cell division and transcription, directing the cell to the S phase of the cycle, which leads to its immortalization [10,11]. Interestingly, HPV has also developed mechanisms that help avoid immune responses from the host. This is due to the activity of structural early proteins E6 and E7, which implicating the excitation of translocations of the IRF-2 and IRF-3 (Interferon Regulatory Factor) genes, reduce the production of IFRα and IFNβ, which are directly involved in the reduction of virus multiplication. In addition, these oncoproteins disrupt the expression of TLRs (Toll-like Receptors) responsible for the detection of pathogens in the host organism and activation of the above-mentioned interferons [12].
As mentioned, many HPV types are distinguished, whose presence has been confirmed both in the presence of clinically harmless symptoms (warts), and in cases in which HPV has been assigned the effect on the development of cancer among men and women (Table 1) [1]. It has been demonstrated that HPV is primarily a precursor to the development of cervical carcinogenesis (about 70% of all cancers are caused by the most severe HPV16 and HPV18, with 99.7% of cancer cases being detected) [6]. In addition, its oncogenic effect on squamous cells was confirmed correctly in the vulva, vagina and penis (in about 40% of cases, the presence of a hrHPV type associated with these conditions was detected), but also in the throat, larynx, oral cavity (mainly the tongue root) palatine tonsils, upper part of the esophagus, conjunctivitis, paranasal sinuses and lungs [11]. It is worth noting that in the case of cancer last of the above mentioned organs, detection of hrHPV estimates in the range of 0 to 78% [13]. The development of a patient's malignant disease is also influenced by environmental factors, mainly the place of residence, lack of a healthy lifestyle, including drinking excessive amounts of alcohol and smoking, but also the type of cancer being analyzed or genetic determinants of the patient. Similar conclusions were obtained when analyzing cases of head and neck cancer, and it was also proven that tumors of those parts of the body that HPV contributed to (mainly 16 and 18) respond better to treatment than those on which the virus had no effect on the virus [14]. What is also interesting, the presence of the virus (including 5 and 8) was also confirmed in people with rare genetic (recessive autosomal) skin disease - Epidermodysplasia verruciformis (EV) [15].
Table 1: Table showing the division of HPV due to the level of their oncogenicity and the type of diseases caused by these viruses. View Table 1
Cervical cancer is the fourth most common cancer among women in the world, causing annually about 275,000 deaths [16,17]. There are many factors affecting the development of this life-threatening disease, such as the socio-economic status, the moment of sexual intercourse, alcohol consumption or smoking, as well as genetic load, immunosuppression and a large number of pregnancies and births (especially for young women) [18]. However, the most important factor that has a huge impact on the development of cervical cancer is primarily persistent infection with hrHPV (mainly types 16 and 18), which can lead to uncontrolled course of infection (Figure 2). In the data analysis of a multicentre comparative case study of the International Agency for Research on Cancer (IARC), the odds ratio (OR) for squamous cell carcinoma due to HPV infection was 158.2, with the analysis limited to studies using approved HPV detection techniques [19]. In this study, the odds ratio for cervical cancer ranged from 109 to 276 in studies from different parts of the world [19].
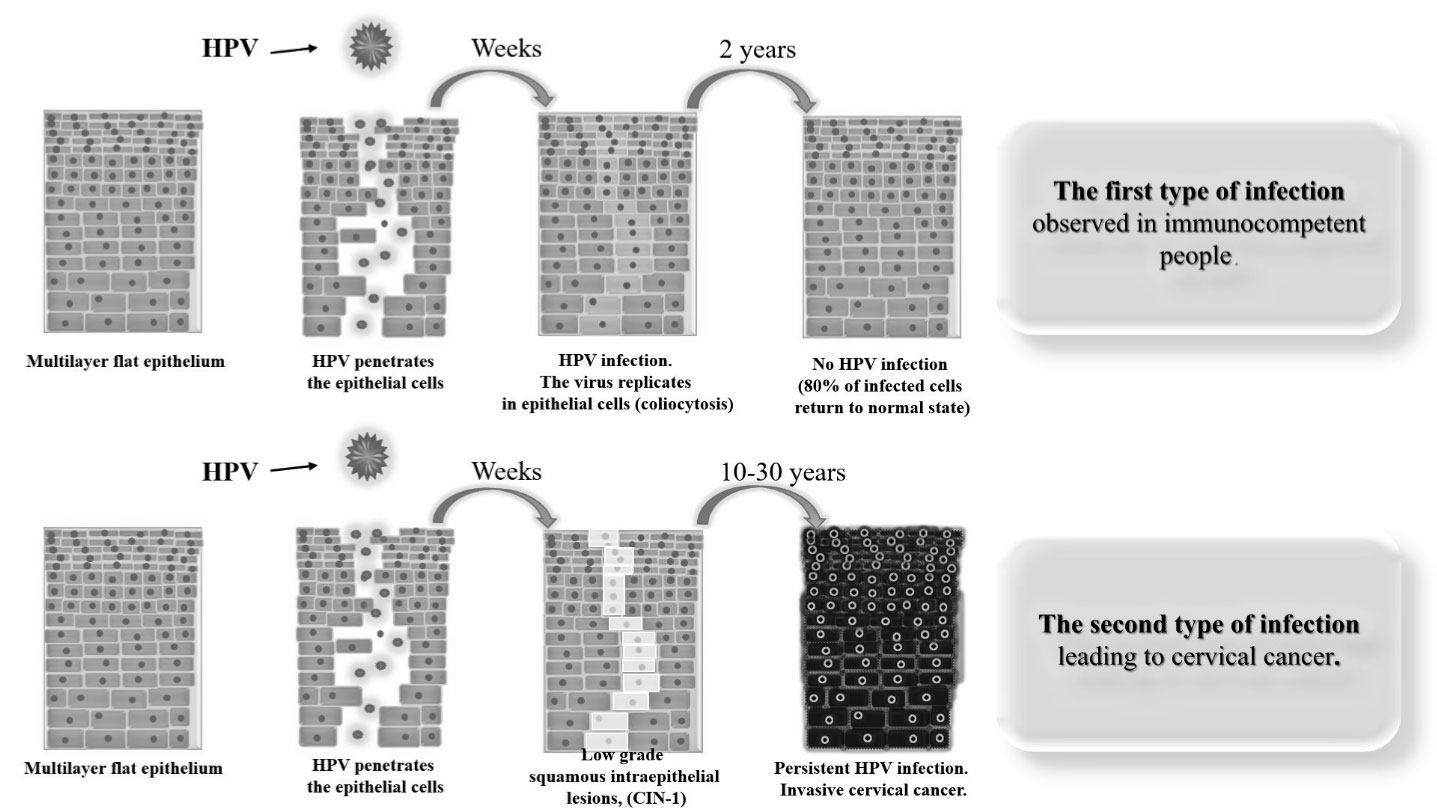 Figure 2: Schematic representation of the mechanism of HPV infection in the case of immunocompetent persons (top) and in the case of persistent infection leading to cervical cancer (down). View Figure 2
Figure 2: Schematic representation of the mechanism of HPV infection in the case of immunocompetent persons (top) and in the case of persistent infection leading to cervical cancer (down). View Figure 2
HPV is transmitted most often through sex, infecting 6.2 million new people each year, which makes it one of the most frequently transmitted viruses in the world. Regardless of gender, and depending on the degree of sexual activity, the risk of HPV infection is around 50% throughout life (both in women and in men) [1]. Women most often get infected with a virus between 15 and 25 years of age. Most infections among immunocompetent women pass asymptomatic after about 24 months. It is worth noting that the lack of symptoms is a hallmark of HPV-induced infection, which undoubtedly contributes to ignoring the patient's regular tests confirming the presence or absence of pathogen threat, but also allowing the virus to initiate a persistent infection [8]. If the infection does not subside, within 10-30 years of viral replication will be accompanied by neoplastic changes, concerning the endothelium of the cervix, so-called CIN (Cervical Intraephitelial Neoplasia). In histopathological findings, they are divided into the degree of advanced neoplasia to low-grade dysplasia, referred to as CIN-1, medium-sized neoplasia is CIN-2 and precancerous stage referred to as CIN-3. In addition, cancer stages are also distinguished, which are classified depending on the stage of cancer disease, i.e. from I to IV [1,20].
However, taking into account the current state of knowledge and the current view on the etiology of cervical cancer, it was found that cytological examinations and their evaluation system (used since the 1950s, referred to as the "golden standard", the Papanicolau scale - Pap Smear), do not constitute the entire diagnostic set that the patient can use. Currently, it is also recommended to perform diagnostic tests extended with a molecular algorithm. Thanks to it, it is possible not only to detect the HPV virus inthe cytological material, but also to determine what specific types of virus are present in it, highlighting above all the most dangerous from the point of view of the implications of the carcinogenesis process. It is also worth paying attention to the fact, which is related to the limitations of the test, which is cytology. This test is characterized by relatively low sensitivity and repeatability, which leads to variable accuracy of the obtained results. In addition, repeating the cytological examination, a greater number of false diagnoses were noted, increasing in a unit of time. What's more, cytology works mainly in the detection of cervical squamous cell carcinoma, in addition to which cervical adenocarcinoma is also present, which unfortunately does not detect this. It is not surprising then that for many years attempts have been made to "support" prophylactic activities concerning cervical research, based mainly on cytology, adding to this standard characterized by significantly higher quality and sensitivity of molecular tests (Figure 3) [8,16,17].
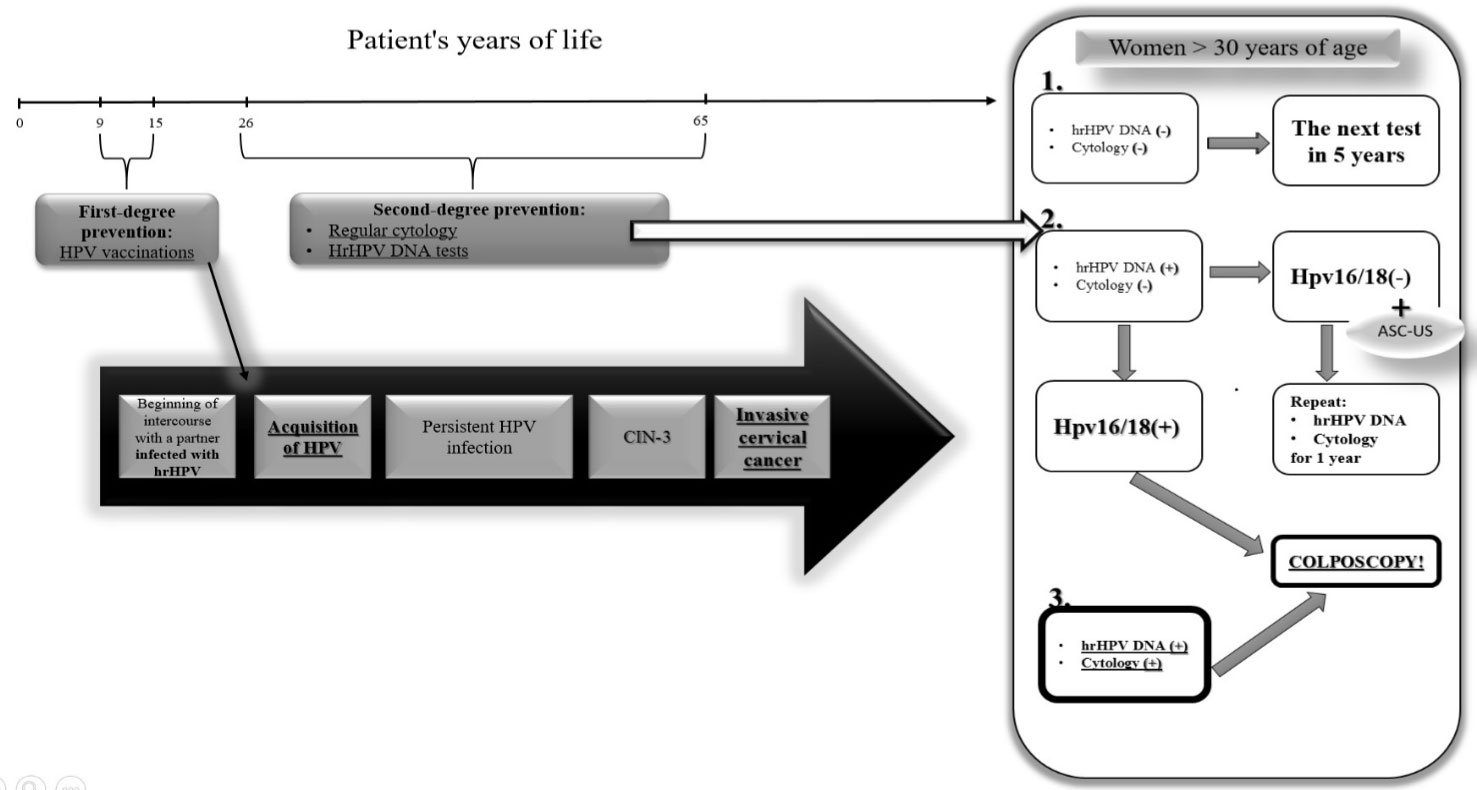 Figure 3: The diagram presents the possibility of developing cancer caused by persistent hrHPV infection, in a hypothetical situation when the patient was not vaccinated at the right time (before sexual intercourse) and a basic pattern of gynecological procedures, providing for several variants. Variant number 1 assumes negative results of both cytological examination and hrHPV DNA tests. In this situation, the patient is referred for further tests in the standard course of action, i.e. for 5 years. In variant number 2 the hrHPV test is negative, but additionally there was a situation when the cytology did not show the presence of cancerous cells, however, it revealed the ASC-US (Atypical Squamous Cells of Undetermined Significante), indicative of the presence of inflammation in the cervix. In this situation, the patient is referred for further tests for the year. If the cytology was correct, but the results of the hrHPV molecular tests are adding, the standard procedure says that an experienced gynecologist must immediately refer the patient to a colposcopic examination to detect pre-invasive cervical cancer. Variant 3 is the worst situation in which both cytology showed disturbing neoplastic changes and what additionally confirmed molecular tests for the presence of hrHPV DNA. It is necessary to immediately perform a colposcopy [20]. View Figure 3
Figure 3: The diagram presents the possibility of developing cancer caused by persistent hrHPV infection, in a hypothetical situation when the patient was not vaccinated at the right time (before sexual intercourse) and a basic pattern of gynecological procedures, providing for several variants. Variant number 1 assumes negative results of both cytological examination and hrHPV DNA tests. In this situation, the patient is referred for further tests in the standard course of action, i.e. for 5 years. In variant number 2 the hrHPV test is negative, but additionally there was a situation when the cytology did not show the presence of cancerous cells, however, it revealed the ASC-US (Atypical Squamous Cells of Undetermined Significante), indicative of the presence of inflammation in the cervix. In this situation, the patient is referred for further tests for the year. If the cytology was correct, but the results of the hrHPV molecular tests are adding, the standard procedure says that an experienced gynecologist must immediately refer the patient to a colposcopic examination to detect pre-invasive cervical cancer. Variant 3 is the worst situation in which both cytology showed disturbing neoplastic changes and what additionally confirmed molecular tests for the presence of hrHPV DNA. It is necessary to immediately perform a colposcopy [20]. View Figure 3
It is impossible not to notice more clearly the disproportion characterizing the global incidence rate of cervical cancer. It is abnormally higher among women living in undeveloped areas (85% of cases come from these areas), above all Sub-Saharan Africa, Central America and South-East Asia. A clear difference between countries (areas) considered to be developed, and those undeveloped or developing, is explained, among others differences in the risk of hrHPV infection, the level of consciousness of patients and the system of prevention and prevention of this disease. The lowest rates of both morbidity and, above all, mortality are observed in countries where an efficiently operating screening program is available, which results in the morbidity and mortality from cervical cancer falling for years [16,17].
In both developing and developed countries, a prevention program against hrHPV with vaccines is also being carried out. Using data that confirmed that in 60% of cases of cervical cancer HPV16 was detected, 18% HPV18, 8% HPV45 and 5% HPV31 developed vaccines containing purified virus-like particles of VLP (Virus-like Particles), for example the above-mentioned types of high-risk viruses [1,8]. The first approved vaccine was a quadrivalent formulation called Gardasil, now replaced with the newest generation Gardasil9 vaccine (nine-valent vaccine, against HPV infections 16, 18, 6, 11, 31, 33, 45, 52, 58), whose efficacy in preventing cervical cancer is comparable with the second Cervarix vaccine on the market (bivalent vaccine against HPV 16 and 18 infections). It is recommended to vaccinate adolescent girls (9-15 years), before starting sexual intercourse, giving them the preparation in two doses, preferably every six months (above 15 years of age, WHO recommends the administration of 3 doses of the vaccine). It is assumed that the vaccine will protect the patient from HPV infection until she is ready to begin routine cytological tests, preferably in conjunction with HPV molecular tests, proving at least the persistence of high HPV antibodies for 10 years after the injection of Cervarix [21]. It is true that 80% of cancers caused by persistent infection hrHPV cancer causes changes in the cervix in women, so it is also worth noting that it also recommends vaccination of young boys (11-12 years). Then it takes into account the transfer of viral particles, occurring mainly through sexual contact, and even statistics on head and neck cancer, or penis in men affected by the oncogenic HPV infection [22]. 12 years after the introduction of HPV vaccines recommended by WHO (the World Health Organization), the vaccine prophylaxis program functions in 74 countries, without confirming the occurrence of any side effects of the preparation [23]. In Poland, vaccinations against HPV are recommended, but they are not financed from the budget of the Ministry of Health. However, it is worth checking the current Local Preventive Programs, in which the schedule may include vaccinations against HPV (in 2018, 9 provinces joined the vaccination program) [24].
In the prevention of cervical cancer, it is not only important to detect changes early, but also to identify factors that have the most probable etiopathogenetic relationship to the carcinogenesis process within this organ. By detecting and then diagnosing HPV infected people, a high-oncology risk group can be identified, which can then be subjected to tighter control. Attempts to estimate the prevalence of HPV infection among women with subclinical or latent disease results in different results, depending on the population studied and the method used to detect the virus. Human viruses from the Papilloma family do not multiply in cell cultures, so far no model of infection of animals and breeding of viruses has been developed. Due to the difficulty of obtaining a viral antigen, standard serological methods cannot be used in in vitro cultures. Detection of HPV infection has become possible only after the introduction of methods used in molecular biology. The highest percentage of infections is diagnosed using a PCR reaction that is characterized by the highest sensitivity among all currently known molecular biology techniques. It allows you to demonstrate the presence of one copy of HPV on 105-106 cells. PCR is now becoming a common diagnostic technique that is used in many laboratories. The results obtained on this basis are comparable and allow to some extent to avoid their false interpretation. The introduction of DNA testing to detect the presence of HPV virus has increased the effectiveness of screening programs for detection of cervical cancer by prior detection of high-risk changes in women over 30 years of age with the NILM (Negative for Intaepithelial Lesion or Malignancy) cytology test and reducing the need for unnecessary colposcopy and treatment in patients after 21 years with ASC-US result of cytological examination [19,25-28]. In addition, the sensitivity of the test for the presence of HPV DNA as compared to PAP-smear in the detection of a high-grade disease in the population undergoing screening has been repeatedly confirmed. DNA testing for the presence of HPV virus with proven higher sensitivity was proposed and used as the primary first-line screening test in some screening programs.