Uveal Melanoma is a rare tumour that displays different clinical behavior and molecular features compared with cutaneous melanoma. It is generally resistant to systemic therapy and there is no current standard effective therapy to treat patients with advanced disease.
We searched Medline, PubMed, EMBASE and major oncology conference abstracts from the past 5 years to identify relevant studies evaluating ipilimumab monotherapy in uveal melanoma. Data were extracted on ipilimumab dose, sample size, Objective Response Rate (ORR), Progression Free Survival (PFS), median Overall Survival (mOS), Disease Control Rate (DCR), 1 year Overall Survival (1yrOS) and 2 year Overall Survival (2yrOS).
Nine studies were included in this study including Phase II clinical trials (n = 2), Expanded Access Programs (EAP) (n = 4) and retrospective studies (n = 3). Cases were a mix of pre-treated and treatment-naïve patients. Reported mOS ranged from 5.2-28 months (median: 9.3 months), and reported 1yrOS ranged between 4.5-65%. Calculated compound ORR was 3.4%, and compound DCR was 36%.
Ipilimumab has limited clinical activity in advanced uveal melanoma. Further research is needed to identify more effective systemic therapies for management of these patients.
Ipilimumab, Uveal melanoma, Anti-CTLA4
Uveal Melanoma (UM) is a rare tumour, with an estimated annual global incidence between 5.3-10.9 cases per million people [1]. It accounts for less than 5% of all melanoma cases [2]. UM arises from the vascular layers of the eye (iris, ciliary body, and choroid). It is a highly aggressive malignancy and about 50% of patients will develop metastatic disease at some stage after prior diagnosis of primary uveal melanoma [3]. Liver is the dominant site of metastases (60-90%) followed by lungs (25%) [4,5]. Prognosis in advanced disease is poor, with median overall survival ranging between 2-15 months [5-7].
UM has a distinct molecular profile, and displays different clinical behavior to cutaneous melanoma. In contrast to BRAF and NRAS mutations commonly seen in cutaneous melanoma, mutations in heterotrimeric G protein-α (GNA) subunits GNAQ and GNA11 have been reported in up to 80% of uveal melanomas [8].
Treatment options for UM are particularly limited and efficacy data is scarce [9]. There are a few small studies that describe options such as chemotherapy, metastasectomy, and hepatic intra-arterial chemoembolization [10]. Response rates are dismal [9], with one retrospective review reporting response rates to systemic treatment being as low as 1% [3].
Immunotherapy has revolutionized the treatment landscape for metastatic melanoma in the last 5 years. Ipilimumab is a fully humanized monoclonal antibody that blocks the Cytotoxic T-Lymphocyte-Associated Antigen 4 (CTLA-4), promoting anti-tumour immunity. It is routinely used in metastatic melanoma, and was one of the first treatment to ever demonstrate a survival benefit in large randomized trial in advanced melanoma [11,12]. However, most of the studies examining activity of ipilimumab in advanced melanoma have traditionally excluded patients with uveal melanoma [11,12].
The aim of this paper was to conduct pool analysis of published literature, evaluating the efficacy of ipilimumab monotherapy in the management of advanced uveal melanoma.
This review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [13].
MEDLINE, PubMed and EMBASE search was conducted to identify relevant articles, with no restrictions placed on the search. All published prospective and retrospective studies, examining ipilimumimab activity in advanced uveal melanoma, amongst both pre-treated and treatment-naïve patients were included. Relevant abstracts from the American Society of Clinical Oncology (ASCO) and European Society of Medical Oncology (ESMO) annual scientific meetings presented in the last 5 years were also evaluated. We used the following search terms: "Uveal melanoma" and "ipilimumab", in any combination in the titles of published trials or conference abstracts. Initial search results were screened for duplication and relevance. The full articles and abstracts were then reviewed in detail. Data were manually extracted on study design, ipilimumab dose, sample size, Objective Response Rate (ORR), Progression Free Survival (PFS), Overall Survival (OS), Disease Control Rate (DCR), 1 year Overall Survival (1yrOS) and 2 year Overall Survival (2yrOS). Note was made of any limitations and bias present in individual studies.
Our primary endpoints were median Overall Survival (mOS) and percentage 1 year Overall Survival (1yrOS). Secondary endpoints included Objective Response Rate (ORR), Disease Control Rate (DCR) and median Progression Free Survival (mPFS).
Due to wide variation in study design and reported outcomes, key efficacy endpoints from each individual trial were summarized. Forest plots were constructed to depict and compare PFS and OS, along with 95% Confidence Intervals (95% CI) for each trial where relevant data was available. Pooled data was used to calculate compound ORR and DCR using a random distribution model.
The identification and screening process as per PRIMSA guidelines is depicted in Figure 1. Nine articles were included in this study; including Phase II clinical trials (n = 2), Expanded Access Programs (EAP) (n = 4) and retrospective studies (n = 3) [14-22]. The key features of each trial are summarized in Table 1.
 Figure 1: Study Identification, Screening and Inclusion process (PRISMA flow chart). View Figure 1
Figure 1: Study Identification, Screening and Inclusion process (PRISMA flow chart). View Figure 1
Table 1: Summary of trials included in meta-analysis. View Table 1
The reported median patient age ranged from 42-67 years. Ipilimumab was prescribed at either a high induction dose (10 mg/kg) or standard dose (3 mg/kg). Reported mOS ranged from 5.2-28 months, with an overall median of 9.3 months (mean of 9.1 months) (Figure 2). Progression free survival data was limited and ranged from 2.8-3.6 (Figure 3). Median PFS could not be calculated for the reported studies as relevant data was not published or not provided. The 1yr OS ranged between 4.5-65% (Figure 4). Calculated compound ORR was 3.4% and compound DCR was 36%.
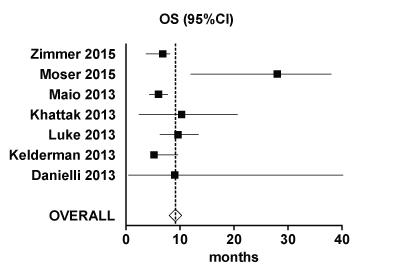 Figure 2: Forest plot of median Overall Survival (OS) from included trials. View Figure 2
Figure 2: Forest plot of median Overall Survival (OS) from included trials. View Figure 2
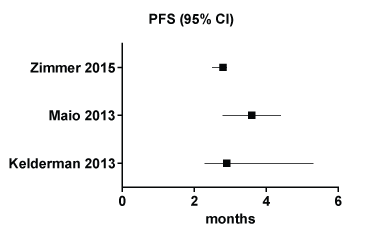 Figure 3: Forest plot of progression free survival from included trials. View Figure 3
Figure 3: Forest plot of progression free survival from included trials. View Figure 3
 Figure 4: Survival % at 1 and 2 years for reported trials.
View Figure 4
Figure 4: Survival % at 1 and 2 years for reported trials.
View Figure 4
Eight studies included a toxicity analysis, with immune related adverse effects (irAEs) graded according to common toxicity criteria. Common irAEs included rash, itch, diarrhea/colitis, hepatitis, and hypophysitis, in addition to fatigue and fever. Grade 3 or 4 irAEs occurred in 10-33% of cases. IrAE's were generally reversible when managed as per protocol-specific guidelines. No new safety signals were seen compared to original ipilimumab phase 3 studies.
Uveal Melanoma is a rare and highly aggressive tumour, with limited treatment options. Data on the activity of ipilimumab amongst patients with advanced UM is scarce. This pooled analysis contributes to the existing literature by summarizing the key efficacy endpoints in published trials and abstracts.
In terms of our activity endpoints, the reported efficacy of ipilimumab in UM is modest at best. However, median overall survival of 9.3 months amongst patients with UM treated with ipilimumab (range 5.2-28 months) is similar to the published mOS of cutaneous melanoma patients treated with ipilimumab [11,12]. The calculated compound ORR was disappointingly low (3.4%), but the compound DCR was potentially more promising (36%).
Most of the published data assessing the efficacy of ipilimumab focuses on response rate and median overall survival as primary endpoints. At least one abstract suggests that survival at 1-2 years is more relevant to assess the efficacy of ipilimumab, given the highly aggressive nature of the disease [14]. The long term benefit with ipilimumab in metastatic melanoma is noted by a consistent absolute increase of approximately 10% of patients alive in the ipilimumab-containing arms compared to the control therapy in the two pivotal clinical trials [11,12]. The late plateau or tail represents the long term responders. Therefore survival at certain time points like 1 and 2 yr OS% are more meaningful in the context of advanced UM. The 1 year OS% with ipilimumab in advanced UM in our analysis ranged between 4.5-65%.
Importantly, the toxicity profile described by these studies was comparable to the description and frequency of irAEs among patients with cutaneous melanoma treated with equivalent doses of ipilimumab. Whilst serious adverse events (grade 3/4) were common, they were easily managed and usually reversible when treated as per study protocol with steroids.
More recently, there have been studies with anti-programmed cell death protein 1 antibody (anti-PD1) monotherapy and targeted therapy with MEK inhibitors that have demonstrated some activity [23-25]. Although at least one small series has observed activity with pembrolizumab, more extensive experience suggests that responses and clinical benefit are much more limited than with advanced cutaneous melanoma [23,24]. The role of the newer agents in the treatment landscape for advanced UM needs to be determined and clinically justified bearing in mind the toxicity seen especially with targeted therapy [25].
One major limitation to this analysis is the heterogeneity of the studies selected. There was a lack of consistency between studies and the wide variation in study design, inclusion criteria and response assessment criteria is reflected in the wide variation in reported outcomes. There was limited consistency in choice of primary and secondary efficacy outcomes, with confidence intervals not reported in several studies. We were unable to retrieve more complete data by contacting the primary investigators despite our efforts to do so. Sample size in all these studies is relatively small and most are retrospective. It is possible that the inclusion of results from EAPs predisposes investigators to a positive publication bias.
Despite the limitations of the published data, we think there is reasonable evidence to suggest that ipilimumab has modest clinical activity and manageable toxicity when used in the treatment of advanced UM in the absence of any other suitable systemic therapy options. Future studies should explore newer immunotherapies and targeted therapies either as monotherapy or in combination.