Allergic rhinitis is a major chronic respiratory disease that affects between 10% and 30% of adults and up to 40% of children globally. The purpose of this study was to understand the impact of allergic rhinitis and subsequent GRAZAX® grass allergy immunotherapy (AIT) treatment on adults' and adolescents' quality of life.
Clinicians treating allergic rhinitis in the Netherlands and Germany recruited adults and adolescents who had received at least 3 years of GRAZAX® treatment. Telephone interviews were conducted with patients to explore their experience of allergic rhinitis and AIT. Interview transcripts were analyzed using grounded theory methods. Saturation analysis was conducted for impact-related concepts. 3 allergists were also interviewed to explore their experience of treating allergic rhinitis.
15 adults (aged 19-61) and 18 adolescents (aged 11-17) were interviewed. Clinicians and patients reported that before treatment with GRAZAX®, the most common allergic rhinitis symptoms were nose- and eye-related. Together with coping strategies, these had considerable negative impacts on patients' quality of life. Adults and adolescents reported notable improvement or disappearance of symptoms during treatment with GRAZAX®. Symptom reduction during treatment led to positive impacts on social, work/school, and physical functioning, with less need for impactful coping strategies. Some patients experienced oral side effects in the first months of AIT, though these resolved with time. Saturation of impact-related concepts was achieved.
GRAZAX® treatment considerably improved allergic rhinitis symptoms and therefore impacted on daily life for patients during and after at least 3 years of treatment. Despite long recall periods, the consistency of reports between doctors, adolescents and adults, and achieving saturation suggest the robustness of the results; a three year course of treatment with GRAZAX® positively impacts quality of life and supports the existing clinical efficacy data on GRAZAX® treatment.
Allergy, Immunotherapy, Burden of illness, Quality of life, Symptoms, Patient reported outcomes
AIT: Allergy Immunotherapy; ARIA: Allergic Rhinitis and its Impact on Asthma
Allergic rhinitis is a major chronic respiratory disease that affects between 10% and 30% of adults and up to 40% of children globally [1]. The European Community Health Respiratory Survey included 15,160 adults with allergic rhinitis from 15 countries, aged 20 to 44 years. Between 7.8% (Spain) and 26.3% (Switzerland) of this sample was found to have grass pollen allergy [2]. The condition negatively impacts quality of life [3] and represents an economic burden due to the reduction of work or school performance and loss of productivity [4-6]. Allergic rhinitis is also often linked to asthma, sinusitis and conjunctivitis [7-9].
In order to relieve the symptoms of allergic rhinitis, allergen avoidance is advised. A number of pharmacotherapies are also available: Antihistamines (oral and intranasal), intranasal corticosteroids, and leukotriene modifiers, although they are not effective for all patients. Allergy Immunotherapy (AIT), is also prescribed for patients with allergic rhinitis. In children and adults with allergic rhinitis due to pollen, sublingual and subcutaneous AIT is recommended by Allergic Rhinitis and its Impact on Asthma (ARIA) guidelines [10].
GRAZAX®, an AIT formulated oral lyophilisate containing a standardised allergen extract of grass pollen from Timothy grass (Phleum pratense) in 75,000 SQ-T per tablet [11] is available in a fast-dissolving tablet formulation [12] and has been available on most markets in Europe for over 10 years. It treats the underlying cause of grass pollen allergy and clinical trials have shown that it provides a sustained effect on symptoms such as runny nose, blocked nose, sneezing, itchy nose, gritty feeling/red/itchy eyes and watery eyes after treatment completion [11,13].
The aim of this study was to explore in greater depth the burden of disease and quality of life of patients before, during and after at least three years of treatment with GRAZAX® for allergic rhinitis in the Netherlands and Germany.
Telephone interviews were conducted with one clinician in Germany and two clinicians in the Netherlands who prescribe GRAZAX® treatment. Clinicians' reports of experience with allergic rhinitis and GRAZAX® treatment were summarized and used to develop the interview guide for subsequent patient interviews. Participating clinicians received financial compensation.
Fifteen adults and eighteen adolescent patients took part in sixty-minute telephone interviews. Before beginning recruitment, the study was submitted to and approved by ethics committees in Germany (Nordrhein, Baden Württemberg, Hessen and Brandenburg) and the Netherlands (Medisch Ethische ToetsingsCommissie). Patients were recruited through clinicians in Germany and the Netherlands according to pre-defined inclusion and exclusion criteria. To be included, patients had to present with an established diagnosis of allergic rhinitis. Adolescents had to have completed at least three years of treatment with GRAZAX® no more than eighteen months ago, and adults no more than two years ago. All patients were required to speak German/Dutch well enough to take part in telephone interviews and to sign an informed consent form. For patients under the age of eighteen, their parents signed the informed consent form. Patients with psychiatric or neuro-psychiatric diseases or disorders and patients suffering from drug dependence or drug abuse in the last year were excluded from the study. For each patient, the clinician completed and signed a screener to report information on the above criteria.
An interview guide was developed, covering the following subjects: Daily life, symptoms and disease burden before, during and after GRAZAX® treatment, and previous treatment history. Native Dutch/German trained interviewers with over ten years of interviewing experience conducted the interviews over the phone. Interviews were audio-recorded and transcribed word-for-word in the source language. All respondents were assigned a respondent identifier composed of two letters indicating their country (NL or DE), a letter indicating whether they are adults (A) or adolescents (C) and a two-figure number.
The analysis of the transcripts was carried out using Atlas ti.version 7, software specifically designed for qualitative analysis. Analysis followed grounded theory methods [14,15], by which codes were created based on the concepts reported by patients and not by imposing pre-defined codes on the data. The codes were organized into domains and concepts. Analysis was carried out by adult and adolescent sub-populations as it was hypothesised that the greatest differences in disease burden, treatment burden and quality of life would be seen according to age group and not between countries, given the cultural and climatic proximity of the Netherlands and Germany. Conceptual models illustrating symptoms and impact of allergic rhinitis before, during and after treatment with GRAZAX® were developed. In order to ensure that the sample size was sufficient and that all relevant concepts had been captured, saturation analysis was conducted. Saturation is defined as the point at which no new information arises from the data and allows researchers to determine whether the sample size included is sufficient. Saturation was evaluated by dividing interviews into sets of three; the concepts in the first set were compared to those in the second set, then those in the first two sets to those in the third, and so on, noting in which set a concept was reported for the first time. Saturation was said to be achieved if no new concepts arose in the final data set. Saturation analyses was conducted only for concepts relating to impact during and after treatment with GRAZAX® as this was the primary focus of our study. The impact of allergic rhinitis itself is already well documented.
 Figure 1: Overview of project steps. View Figure 1
Figure 1: Overview of project steps. View Figure 1
Thirty-three participants with allergic rhinitis were included in the study. As described in Table 1 nine adults and eleven adolescents from the Netherlands and six adults and seven adolescents from Germany were interviewed between December 2016 and March 2017. All patients had taken GRAZAX® treatment for at least three years, and four adults and one adolescent were still taking GRAZAX® treatment after three years. Patients were often unable to report exact names of previous medication, simply citing the mode of administration.
Table 1: Patient characteristics. View Table 1
Numerous adults and adolescents reported other allergies (Table 2). The most common other allergies were house dust mites for adults (n = 7, 47%) and other plants and flowers for adolescents (n = 9, 50%). Eight of eighteen adolescents also reported allergies to house dust mites and to animal hair (Figure 2).
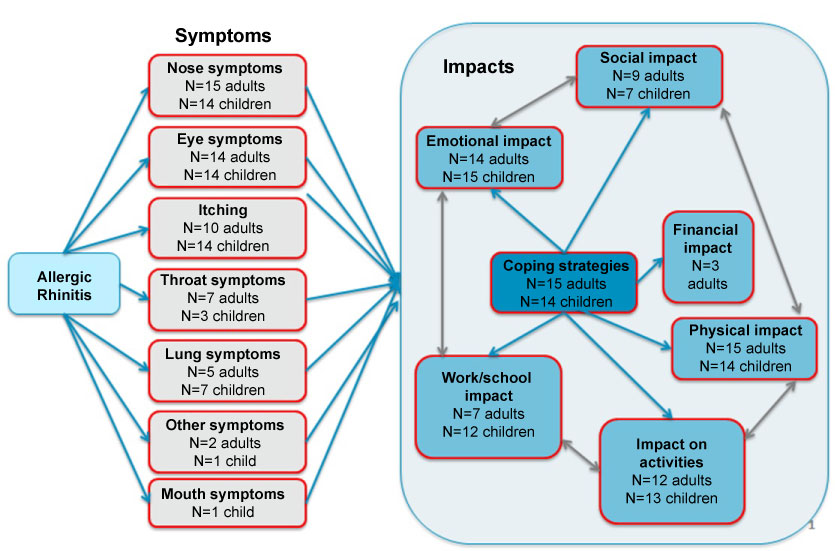 Figure 2: Allergic rhinitis symptoms and quality of life impact before treatment with Grazax®. View Figure 2
Figure 2: Allergic rhinitis symptoms and quality of life impact before treatment with Grazax®. View Figure 2
Table 2: Other patient-reported allergies. View Table 2
As shown in Figure 2, nose- and eye-related symptoms were the most commonly reported: "I used to wake up every morning with a stuffy nose" (NL-A4); "What was especially bad was that I had itchy eyes, I couldn't leave them alone and they swelled up" (NL-A7). Many patients took medication to manage extremely bothersome eye symptoms and irritating and burdensome itching: "Definitely eye drops. They were my survival drops. I had tissues in my car, and my eye drops." (DE-A2). Nose symptoms were considered as irritating, and sneezing was perceived as having the greatest social and emotional impact as it drew attention to the patient: "It was completely quiet in class and I had to sneeze all the time. I found that very embarrassing" (NL-A2). Lung-related symptoms such as asthma, coughing or wheezing were often reported in terms of the impact they had on the ability to play sports, and enjoy playing sport. Furthermore, they were often reported as worrying and anxiety-inducing. "I was really scared that something bad would happen" (DE-A2).
Patients in both Germany and the Netherlands reported allergy symptoms in the spring and summer (March/April - July/August). Some patients also reported tendencies to become ill more frequently throughout the year. Some adolescents (n = 4, 22%) reported having a persistent cold, whereas no adults reported this. This persistent cold may also be related to the perennial allergies they reported: "I always got red eyes very quickly, had a cold very often [...] I was very often sick.." (NL-C6). Almost half of adults (n = 7, 46%) were bothered by symptoms at night, whereas only a minority (n = 2, 11%) of adolescents reported this: "I couldn't sleep properly. My gums were itchy." (DE-A2); "That was what the trigger [for seeking treatment] was for me, that my sleep was so disturbed and that I was impacted by this sleep deficit." (DE-A5). Some patients reported that symptoms did not vary, but were constant over the grass pollen season. "My nose [symptoms] and cold. They were constant" (DE-C1).
The emotional impact and the physical impact of allergic rhinitis were the most frequently reported: "It was really unpleasant. I really just didn't feel very well" (DE-C3); "The symptoms were really bad and I felt terrible. You rub your eyes all the time and just feel like crying" (DE-A3). Patients also reported being unable or unwilling to take part in outdoor activities in order to limit their exposure to grass pollen. As many of these activities were usually undertaken with friends or family, this, in turn, had a negative impact on their social life: "When my friends say, on the weekend, it's 35 degrees outside, let's go to the lake. Walking with the dog or swimming with friends, then I just said no, go without me" (DE-A2). Work and school life was also affected for a majority of adolescents and just under half of adults. Symptoms and coping behaviours were disruptive and influenced concentration and performance: "Well you're going to perform less well, because you can concentrate less because you are tired. Sneezing or messing around all day" (NL-A11); "When as a teacher you're constantly blowing your nose [...] you're not very efficient" (DE-A5).
A minority of adults reported a financial impact such as out of pocket costs for over-the-counter medication: "Then social security stopped reimbursing it so I had to pay for it out of my own pocket. I spent years paying for it myself." (DE-A5).
As is shown in Figure 2, these impacts are interrelated, and the coping strategies put into place by patients have impacts that can be negative also. Coping strategies can be conscious or unconscious and can take many different forms. While some are concrete measures, such as taking medication or putting water on irritated eyes, others can be behavioural, such as avoidance and prioritising, or emotional, such as a positive attitude or taking time to adjust: "You set priorities for yourself. What is more important?" (DE-A3) "I always said, well, I'm not going to let it get me down" (DE-C7). Limiting activities is both coping mechanism for and an impact of symptoms. In turn this has a detrimental social and emotional impact.
Medication was the most common coping strategy for adults (n = 12, 80%) but only reported by just over half of adolescents (n = 10, 56%). A large number of patients were not able to identify the type of treatment they had received, beyond the mode of administration. Some coping strategies had a negative effect on social and emotional functioning, as well as on school, work and activities: "I can't keep my wife inside [...] so when we were outside in the garden [...] I had to go and hide away inside" (DE-A2); "Because you're inside all day, it makes you sad that you're constantly inactive" (NL-C13). Moreover, changing habits and behavioural strategies were also reported as having a negative impact on others; patients report that families had to adapt to changes in holiday destination and in social activities for example: "Going out to the lake wasn't something I would do. That was the kind of thing I would go out of my way to avoid" (DE-A3). This concept was reported spontaneously and was not part of the interview guide (Figure 2).
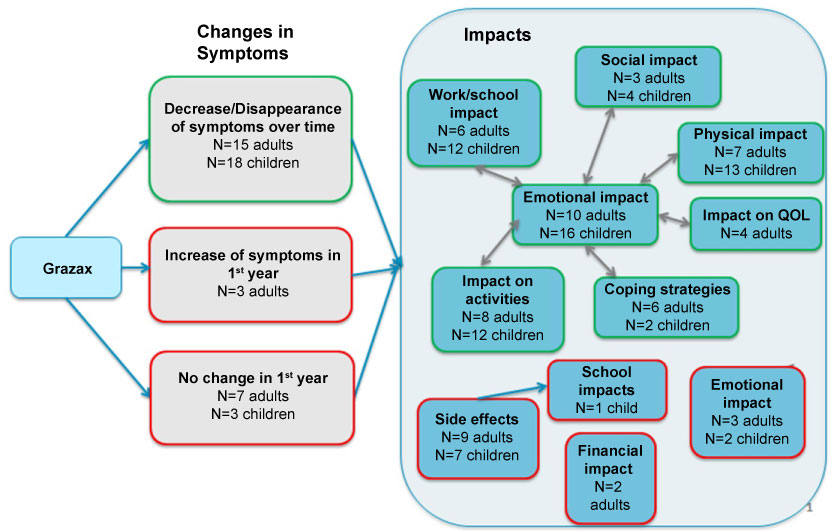 Figure 3: Allergic rhinitis symptoms and quality of life impact during treatment with Grazax®. View Figure 3
Figure 3: Allergic rhinitis symptoms and quality of life impact during treatment with Grazax®. View Figure 3
Figure 3 shows that, overall, all patients saw a decrease or disappearance in allergic rhinitis symptoms with the treatment over three years: "There was a real difference actually. I didn't have a cold so often anymore. Not as often allergic reactions anymore" (NL-C6). Nose-related and eye-related symptoms decreased the most: "My nose isn't blocked anymore, so I don't get a headache anymore" (NL-C3). Throat and lung symptoms also improved: "[My sore throat] didn't happen very often and wasn't very intense" (DE-A1); "It got less and less and then I could hardly feel it [asthma] at all [...] and then after two years I didn't feel any scratching in my throat at all anymore" (DE-C2). The majority of respondents reported what they perceived as a significant improvement within the first year of treatment. Those who did not report a significant improvement in the first year experienced gradual relief of symptoms throughout the treatment period. A small number of adults (n = 3, 20%) reported a flare-up in the first year, whereas no adolescents did. More adults than adolescents experienced side effects within in the first weeks or months of starting treatment (n = 9, 60% vs. n = 7, 39%), which were for the large majority mild oral side effects lasting a few weeks.
The improvement in allergic rhinitis symptoms during GRAZAX® treatment had a positive impact on patients. The emotional impact was the most commonly reported, particularly positive feelings and absence of burden. "You can just face your everyday life with more positivity and with more energy" (DE-A5). A majority of patients explained that they were able to do more activities outside and that there was less need for planning. The positive physical impact (increased fitness/energy, better overall health) was reported by a majority of adolescents (n = 13, 72%) but less than half of the adults (n = 7, 47%): "Riding my bike, rollerblading. I started jogging again shortly after I started taking the tablets. I used to jog, but [with the tablets] it got a lot easier" (DE-C4). Some patients also reported a positive social impact with an improved social life or a more positive attitude to others: "I did more things with friends. During the summertime there is also a lot of swimming. Being outside" (NL-C13). An improvement in terms of concentration and/or performance at work or school resulting in less absenteeism was experienced by 67% (n = 12) of adolescents and 40% (n = 6) of adults. A few patients also reported less need for coping strategies: "In the end I didn't need to take the antihistamine pills all the time" (NL-A3); "I didn't need to take so many tissues with me anymore" (DE-C5).
As was the case before treatment with GRAZAX®, there is an interaction between these different domains, where a decrease in symptoms led to lower physical impact, ability to do more activities, better social functioning and both directly and indirectly, better emotional functioning: "But I also felt better, because the symptoms weren't so strong. Emotionally and physically better, because you feel so ill with the symptoms" (DE-C3). Improved symptoms led to an improvement in activities, social life, physical functioning, school and emotions. These improvements in activities, social life, physical functioning and school also caused a positive impact on emotions: "Yes, it's just a nice feeling that you can just go after having a game like ohhh, I have not had any trouble. It just gives you a good feeling that you're not limited" (NL-A6) (Figure 3).
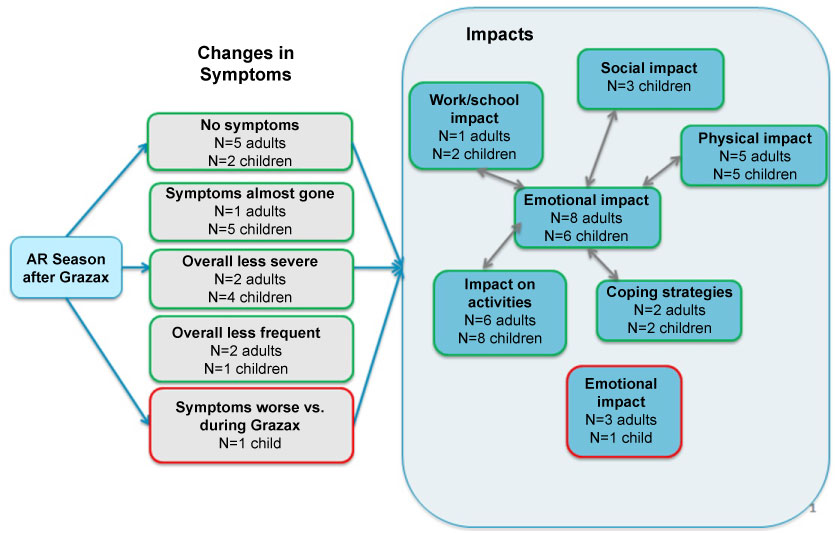 Figure 4: Allergic rhinitis symptoms and quality of life impact after treatment with Grazax®. View Figure 4
Figure 4: Allergic rhinitis symptoms and quality of life impact after treatment with Grazax®. View Figure 4
Of the thirty-three patients interviewed, nine adults and twelve adolescents experienced at least one allergy season since completing treatment with GRAZAX® and were able to report on this experience (Figure 4): All adults and the large majority of adolescents (n = 11, 92%) reported that symptoms had either decreased or disappeared in the allergy season since completing GRAZAX® treatment: "And now I am almost completely freed from this allergy" (DE-C5); "I didn't know when the hay fever season began. It was a real relief!" (NL-A4). One adolescent reported that some symptoms returned (in a milder form) after completing GRAZAX® treatment.
Patients who had experienced an allergy season since stopping treatment generally reported no difference between the final season of GRAZAX® treatment and the first season without GRAZAX® treatment. The large majority of adults (n = 8, 89%) and half of adolescents (n = 6, 50%) reported an increased positive emotional impact, including the absence of burden, positive feelings and a positive sense of self: "I'm not 'a sick child' anymore" (NL-C1). Most patients also reported a positive physical impact, notably with increased energy/fitness, better overall health and improved sleep: "Yes, because I do not have to wake up at night because my nose is blocked or because of problems, sneezing and tearing eyes, those kinds of things" (NL-A4). The majority of patients reported being able to do and enjoy more activities outside: "I can just do more things. Going out for trips, to amusement parks..." (NL-C4).
A few adolescents (n = 3, 25%) reported an improved social life: "I have become socialized. I also do much more with others than I did before. I had previously been a little on my own. And now no more" (NL-C4), where as no adults did. A few patients also reported an increased positive impact at school or work, in better concentration/performance: "Moderator: 'And what's changed?' Patient: 'Oh, my grades.'" (NL-C4). Small numbers of patients expressed some doubts or fears about future efficacy: "You're 12 when it starts and now you're 45 and you've had this problem for so long, and so you think: 'OK, the hay fever season is about to start, what will happen this year? Will it be worse or will it remain stable? So I still have a feeling of uncertainty about it all.' (NL-A1). Very few coping strategies were required after GRAZAX® treatment: "I used eye drops just maybe three or four times" (DE-A2).
When asked about the most important impact of GRAZAX® treatment on their lives, adults declared that the most important impact GRAZAX® treatment had was giving them the ability to enjoy the outdoors with a freedom that was not there when symptoms were present. Reduction or lack of symptoms was the next most important, though it was noted that enjoyment of the outdoors would not occur without reduction in symptoms, which suggests that these go hand in hand. Finally, adults reported that they had increased physical fitness, which came in third.
As far as adolescents were concerned, fewer/lack of symptoms was what mattered most. Doing activities with family/friends was the second most important impact for adolescents and enjoying the outdoors/freedom came in third position. Other important impacts for adolescents were increased physical fitness/freedom in sports and improvements at school.
Saturation analysis was conducted for all impact-related concepts to determine the point at which no new concepts appear, and therefore confirm adequate sample size. Regarding the impact reported by adults, all concepts during treatment were reported within the first twelve interviews and all concepts reported after treatment were reported within the first six interviews; saturation was reached for all concepts reported by adults. For adolescents, thirty out of thirty-one concepts on impact during treatment were reported within the first eleven interviews and sixteen out of seventeen concepts on impact after treatment were reported within the first nine interviews; improved sleep problems and being able to wear contact lenses arose as concepts in the final sets of adolescent interviews.
The clinicians interviewed see a majority of patients who suffer from moderate to severe allergy with polysensitisation. All clinicians agreed that the most bothersome symptoms are runny nose, sneezing, congested nose, ocular itching, redness, tearing and asthma. Clinicians unanimously reported that allergic rhinitis has a large impact on patients' lives; most patients cannot engage in many outdoor activities and refuse invitations to social activities during the grass pollen allergy season, and patients suffer from headaches and insomnia which lead to tiredness and lack of concentration at work/school.
All clinicians reported following ARIA guidelines in terms of the treatment pathway. Most underline that patients need immunotherapy throughout the whole allergy season as they otherwise experience severe symptoms or because they have poor symptom control due to inadequate treatment. There are, however, barriers to prescribing immunotherapy/GRAZAX® treatment in adolescents specifically as they are expected to have issues with treatment adherence and their parents will often express concerns about possible side effects.
It was observed that treatments given prior to immunotherapy only allowed poor or short-term control of the symptoms. Clinicians also mentioned that patients, and especially adolescents, are very bothered by the sensations caused by nose sprays and eye drops. The fact they have to take different medications to deal with the various symptoms and experience side effects was also part of the problem often seen by clinicians.
Clinicians report that the majority (70-80%) of patients are very satisfied with the treatment benefit of GRAZAX®. They see the benefits from the first season on. Dissatisfaction may, however, occur in the first few weeks due to the oral side effects, the bother of taking a tablet each day, lack of treatment adherence or simply because the wrong allergen was identified as being the cause of the patient's allergy. As a result, some patients will stop their treatment during this period.
No consensus was reached on estimated adherence rates although clinicians reported that patients who asked for treatment with GRAZAX® themselves are motivated, well informed, and are the most adherent.
Allergic rhinitis is a condition that has a considerable negative impact on individuals' quality of life. In this study, clinicians and patients reported that the impact of allergic rhinitis on adults and adolescents is profound, with notable restrictions in outdoor activities leading to social isolation and reduced physical fitness [16,17], which corroborates previous research in adults. Symptoms also make concentration at school and work more difficult.
Randomized controlled single-season trials of GRAZAX® treatment have demonstrated that treatment initiated before the grass pollen season began led to reduced symptoms, less use of rescue medication and improved quality of life (measured with the Rhinoconjunctivitis Quality of Life Questionnaire) compared to patients treated with placebo [18]. Improvement in symptoms and quality of life was confirmed in a follow-up study investigating outcomes two years after completing three years of treatment with GRAZAX® [19]. Our study confirms the short- and long-term improvement in quality of life, and provides insights into how and why patients' quality of life was impacted. It also allows greater understanding of what the changes mean to patients.
In this study, patients having taken at least three years of treatment with GRAZAX® were interviewed in depth in order to understand their symptoms and the impact of these symptoms before, during and after treatment with GRAZAX®. These interviews showed that treatment with GRAZAX® reduced symptoms and had a positive impact on the daily lives of all the patients interviewed. By the end of the treatment with GRAZAX® all adults and adolescents interviewed reported improvement or disappearance of grass allergy symptoms. Patients experienced improvements in their quality of life due to GRAZAX® treatment that led to improvements in a combination of emotional, physical and social functioning, work and school and activities.
The interviews with patients confirmed the assertions of the clinicians interviewed with regard to the most bothersome symptoms, the impact of allergic rhinitis on patients' quality of life, particularly in sports, leisure and social activities, and the efficacy of GRAZAX® treatment. Our results further confirm previous published literature that side effects are primarily mild or moderate local reactions to be expected during the treatment period. Clinicians reported that antihistamine treatment prior to GRAZAX® treatment reduced side effects; further research in this area may be warranted in order to improve adherence and patients' experience.
Adolescents reported being less impacted in their daily lives before GRAZAX® treatment, yet experienced greater improvements with GRAZAX® treatment, notably in school impact, physical impact, emotional impact and impact on activities. However, the positive emotional impact reported after GRAZAX® treatment was more frequently reported by adults than by adolescents (Adults 89% Vs. adolescents 50%) and the positive social impact was more frequently reported by adolescents (25%) than by adults (0%). Given the limited sample size inherent in this kind of qualitative research and the possibility of recall bias, these differences would warrant additional quantitative studies in order to obtain further insight into the impacts experienced. Care must be taken when comparing percentages in this study due to the small absolute number of patients included in this analysis.
In our conceptual models, the causal relationship for coping strategies before GRAZAX® treatment does not include possible alleviation of symptoms through these strategies. This topic was not explored in detail during the interviews, as discussions with patients on the time before GRAZAX® treatment were focused on the burden of illness.
Saturation was not reached for two concepts reported by adolescents: improved sleep during treatment with GRAZAX®, and being able to wear contact lenses after treatment with GRAZAX®, both reported as negative impacts (disturbed sleep, and not being able to wear contact lenses) before treatment. However, large numbers of patients suffer from other allergies, such as house dust mite allergies and take treatment for these. These other allergies as well as side effects of their treatment may negatively impact sleep and may be a confounding factor in reports on this concept; we therefore believe that not reaching saturation on this concept is not indicative of inadequate sample size. Saturating on the concept regarding contact lenses is not considered necessary since by its very nature this cannot be a concept that applies to all respondents - only those who need corrective lenses.
As per the inclusion criteria, all of the patients interviewed successfully completed at least three years of treatment with GRAZAX®. Our sample therefore did not include patients who stopped taking GRAZAX® treatment after less than three years. As with many long-term medications, adherence can be a challenge [20, 21]. Some of the patients interviewed declared being non-adherent at some point during the treatment; this was occasional and rarely deliberate; "It was just difficult to remember to take it at the beginning." (DE-C3). The clinicians interviewed also reported adherence to be an issue with adolescents particularly. Managing patient expectations about onset of effects of GRAZAX® treatment and possible treatment of side effects as described above may improve adherence.
Patients were asked to recall symptoms and impacts going back over a number of years, back to the period before they started taking GRAZAX® treatment. It is therefore likely that patients misremembered or forgot certain events, or that the reports of precise timing of changes due to GRAZAX® treatment were not accurate. However, it is also likely that patients remembered more significant events and did not recall those that were less important to them, and that the resulting reports are therefore representative of what really mattered to patients.
Some patients reported taking rescue medications at some points during treatment with GRAZAX®. The correlation between reported symptoms or burden and use of other treatments was not examined, as the approach for data collection did not allow this. The use of other treatments was qualified as "coping strategies" and included in the conceptual model.
Further limitations to this study are the smaller sample size of those able to report on an allergy season since completing the course of treatment with GRAZAX®. Also, the sample is not representative of all patients to whom GRAZAX® treatment is prescribed as we did not include patients who had stopped treatment. It was decided to include only patients who had successfully completed at least three years of treatment with GRAZAX® as our aim was to explore the impact of the treatment on symptoms and quality of life both during and after completed treatment, and it was therefore not possible to include patients who had not completed the whole treatment cycle in our sample. Those who dropped out of treatment may have had differences in improvements or side effects compared to those who completed the 3-year treatment course.
This research shows that treatment with GRAZAX® in real life considerably improved allergic rhinitis symptoms and positively impacted on daily life for patients who completed at least three years of treatment. Despite small sample sizes and long recall periods, the consistency of reports between doctors, adolescents and adults and achieving saturation supports the robustness of the study data and that successfully completing a three year course of treatment with GRAZAX® positively impacts quality of life and is consistent with existing data on the efficacy of GRAZAX® treatment.
The authors thank Lisa Elliott at ALK Abello A/S for her input on the manuscript, Magali Desroches at Creativ-Ceutical for her support in drafting the manuscript and Natalia Hawken, previously at Creativ-Ceutical, for setting up the study.
All participants provided written informed consent before participating in this study. The study was submitted to and approved by the following ethics committees: In Germany - Ärztekammer Brandenburg Ethikkommission (Reference AS131 (bB)/2016 ma-fr) ÄrztekammerNordrhein Ethikkommission (Reference 2016397), ÄrztekammerBaden Württemberg Ethikkommission (Reference F-2016-092) and ÄrztekammerHessen Ethikkommission (Reference III/1/ger/sts MC 242/2016). In the Netherlands - Medische EthischeToetsingsCommissie (Reference WT/sl/285330).
The data that supports the findings of this study is not publicly available as the participants did not provide consent for it to be shared with a third party.
Mercedes Romano and Anne Domdey are employed by ALK Abello A/S, manufacturer of GRAZAX® treatment. Nicola Germain, Samuel Aballéa, and Mondher Toumi are employed by Creativ-Ceutical, mandated by ALK Abello A/S to conduct the research. Linda Abetz-Webb was a paid consultant to Creativ-Ceutical for this project.
This study was funded by ALK Abello A/S.
This study was funded by ALK Abello A/S.
Nicola Germain led the study, conducted interviews with German patients, conducted the analyses and contributed to writing the manuscript. Roy Gerth van Wijk participated in a clinician interview and was involved in the manuscript development. Samuel Aballéa designed the study methodology, gave scientific and strategic direction to the project and input into the manuscript. Linda Abetz-Webb took part in the data analysis and writing the manuscript. Mercedes Romano gave input into the study design, study documents and analyses, and reviewed the manuscript. Anne Domdey gave input into the study design, and the manuscript. Mondher Toumi provided scientific guidance and reviewed the manuscript.