Allergic rhinitis (AR) is one of the commonest allergic manifestations affecting approximately 400 million people worldwide. It is associated with a reduced quality of life, low productivity in the workplace, and poor school performance. On the other hand, medical cost increases. Allergic rhinitis has a considerable effect on the quality of life and can have significant consequences if left untreated. Many patients downplay rhinitis symptoms as an inconvenience rather than a disease. The majority of the patients start visiting their local physician, when the problem becomes chronic, especially when it is associated with complications. There is a huge economic burden associated with treatment by pharmacological agents available currently. The pharmacological agents are also associated with various side effects and many severe adverse events. That's why we look for the alternative form of treatment for allergic rhinitis with less financial burden and least adverse reactions associated with medicines presently in use commonly.
Allergic rhinitis, Nonpharmacological treatment, SCIT, Allergoids
Allergic rhinitis is very common in the community affecting all ages, meanwhile, the incidence is highest during the teenage years [1]. However, it is frequently underappreciated and underdiagnosed [1]. The presenting symptoms are sneezing, pruritis, rhinorrhoea, and nasal congestion [2]. Allergic rhinitis can lead to complications such as acute and chronic sinusitis, otitis media, hearing impairment if left untreated [3]. Moreover, the association of allergic rhinitis and asthma control is clinically relevant as it results in poor asthma control [4,5]. Understanding the pathophysiology of allergic rhinitis becomes easy if we remember events are going on in allergic reactions at the cellular level, which is under the autonomic nervous system in genetically susceptible persons.
Avoiding environmental allergens is one of the strategies for the management of allergic rhinitis, however, it might not feasible in daily life [6,7]. Therefore, the majority of allergic rhinitis patients rely on pharmacological treatment to relieve their symptoms [1]. In addition to pharmacotherapy, non-pharmacological treatments have been an interest in the management of allergic rhinitis. Immunotherapy, such as Subcutaneous Allergen Immunotherapy (SCIT) was proven to be effective to treat allergic rhinitis and well-tolerated option for the patients [8]. Helminth therapy by using either human hookworm or pig whipworm was investigated in the clinical trials, however, more evidence is needed to conclude on its efficacy [9]. Acupuncture [10] and homeopathic remedies are also used as a non-pharmacological intervention for allergic rhinitis patients. This review included the pathophysiology of allergic rhinitis for a better understanding of the disease and the current role of non-pharmacological treatment options in allergic rhinitis.
Risk factors associated with allergic rhinitis: Well-recognized risk factors for allergic rhinitis include having atopy, asthma, eczema and other allergic diseases [11,12]. Many recent studies have found that children with a family history of atopic disease or parents with allergic rhinitis, asthma, hay fever, and pollen allergies had a high risk of developing allergic rhinitis [13]. Other confusing risk factors that may contribute to disease risk but are not well understood include vitamin D, obesity, exposure to cigarette smoke, increased total serum IgE, increased blood eosinophils and other environmental exposures common in urban settings [2,14].
It is generally accepted that atopic diseases are the results of inappropriate immunological responses to common environmental allergens in genetically susceptible individuals [15]. Understanding the underlying immune inflammatory mechanisms is central to developing better and more targeted therapies.
Changes at the microscopic level: The pathological changes of nasal mucosa from allergic rhinitis and nonallergic rhinitis have been observed by light and electron microscope. The results showed that the general characteristic was vascular dilatation with increased permeability and glandular proliferation with increased secretory activity. The pathological difference between mucous membranes of allergic and vasomotor rhinitis was mainly cellular infiltration and degree of damage on the vascular wall. In allergic rhinitis, there were more eosinophils, basophils, neutrophils and some mast cells and plasma cells in the nasal mucosa. Gap formation in the capillary wall was found in the allergic nasal mucosa, there was less cellular infiltration in nonallergic rhinitis. Cardinal symptoms and pathophysiologic mechanisms are described graphically in Figure 1.
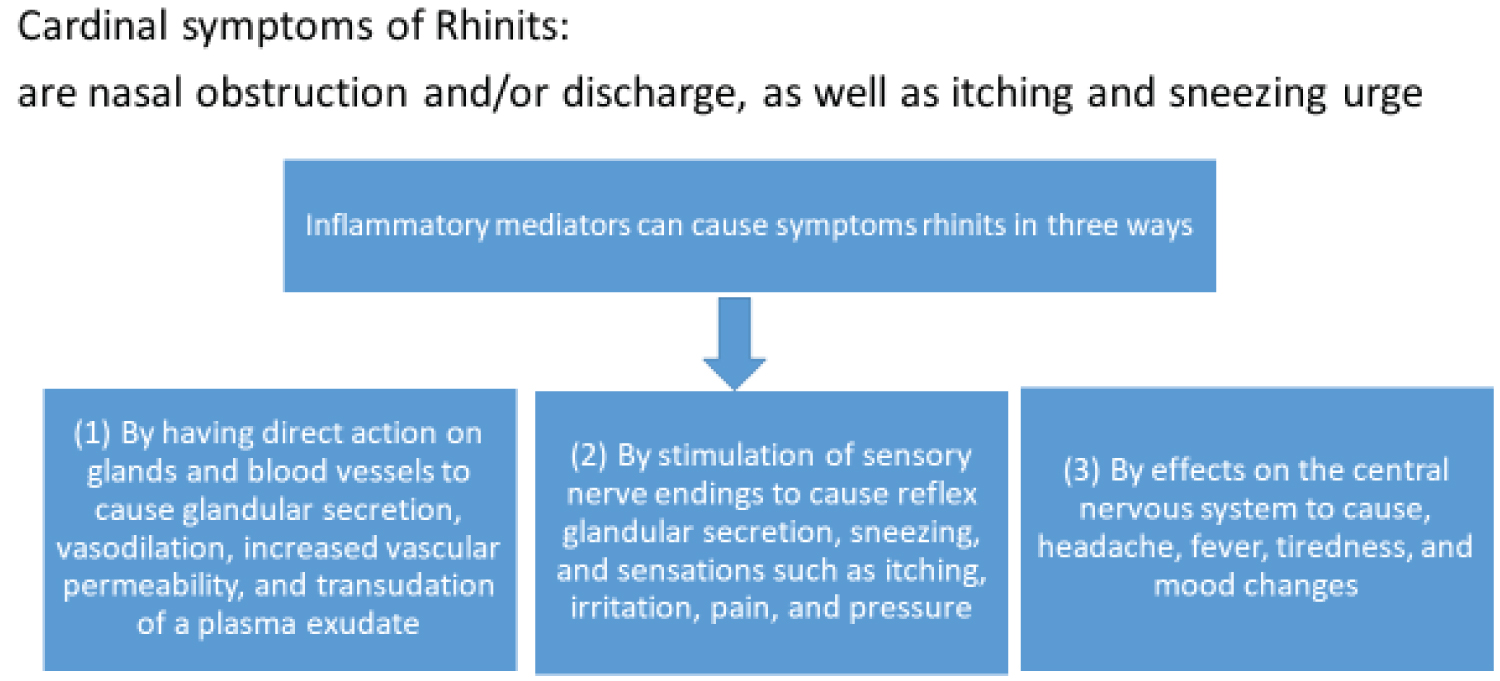 Figure 1: Cardinal symptoms and pathophysiologic mechanism of rhinitis [16]. View Figure 1
Figure 1: Cardinal symptoms and pathophysiologic mechanism of rhinitis [16]. View Figure 1
The inflammatory response in the nasal mucosa in subjects with allergic rhinitis challenged intranasally with an allergen includes an immediate IgE-mediated mast cell response as well as a late-phase response characterized by recruitment of eosinophils, basophils, and T cells expressing Th2 cytokines including IL-4, a switch factor for IgE synthesis, and IL-5, an eosinophil growth factor. Recent advances have suggested that additional pathways may contribute to the pathophysiology of allergic rhinitis including local synthesis of IgE in the nasal mucosa, the epithelial expression of cytokines that regulate Th2 cytokine responses (i.e., thymic stromal lymphopoietin, IL-25, and IL-33), and the activation of histamine receptors other than H1 and H2 such as H4-histamine receptors [17], seasonal increases in the antigen presenting cell (Langerhans' cell) are also evident. Investigations into the mechanisms involved in cell accumulation and activation reveal upregulation of leucocyte endothelial adhesion molecules and an increase in interleukin-4 (IL-4) in naturally occurring rhinitis, while mRNA for IL-4, IL-5 and granulocyte macrophage colony stimulating factor activity and lavage tumour necrosis factor-alpha (TNF alpha) levels are increased following local allergen challenge. These cytokines may be derived from a variety of sources, including mast cells, eosinophils, and T-lymphocytes, and contribute to the underlying inflammatory process in rhinitis. In both seasonal and perennial rhinitis, there is epithelial mast cell accumulation and tissue infiltration by eosinophils. Activation of these cells can be observed by electron microscopy and by elevated levels of tryptase and eosinophil cationic protein in nasal lavage fluid, these levels of eosinophil cationic protein and albumin were also significantly higher than those of the normal non-rhinitis. In the perennial allergic rhinitis, the levels of tryptase, eosinophil cationic protein and albumin were higher than the normal non-rhinitis. The levels of tryptase and histamine were higher in the perennial allergic rhinitis than the seasonal allergic rhinitis. These changes in mediator levels were accompanied by a significant influx of eosinophils into the nasal mucosa of both the symptomatic seasonal rhinitis, compared with asymptomatic and the perennial rhinitis compared to normal controls [18].
Mechanisms underlying the neuronal-based symptoms of allergy: Persons with allergies present with symptoms that often are the result of alterations in the nervous system. Neuronally based symptoms depend on the organ in which the allergic reaction occurs but can include red itchy eyes, sneezing, nasal congestion, rhinorrhea, coughing, bronchoconstriction, airway mucus secretion, dysphagia, altered gastrointestinal motility, and itchy swollen skin. These symptoms occur because mediators released during an allergic reaction can interact with sensory nerves, change processing in the central nervous system, and alter transmission in sympathetic, parasympathetic, and enteric autonomic nerves [19]. There is a definite relation between the symptoms of hyperparasympathetic nervous system and allergic rhinitis as one study results have indicated that patients with intermittent and mild AR have hypervagal activity and hyposympathetic activity, and the predominance lessens in patients with more persistent AR and severe symptoms [20].
Genetic basis for allergic rhinitis: Large-scale genome-wide association strategies are underway to discover new susceptibility variants for allergic rhinitis and allergic phenotypes. Characterization of the underlying genetics provides us with an insight into the potential targets for future studies and the corresponding interventions. The last decade has been marked by the publication of more than 20 Genome-wide association studies (GWASs) GWASs employing common single-nucleotide polymorphism of allergic rhinitis and the associated allergic phenotypes. Allergic diseases and traits have been shown to share a large number of genetic susceptibility loci, of which IL33/IL1RL1, IL-13-RAD50 and C11orf30/LRRC32 appear to be important for more than two allergic phenotypes. GWASs have further reflected the genetic heterogeneity underlying allergic phenotypes [21,22].
The treatment of allergic rhinitis aims to reduce or eliminate the suffering of the patients and to improve the quality of life. Patient counselling is essential to have a better understanding of the disease condition and the medication which are most appropriate to relieve their symptoms.
Subcutaneous allergen immunotherapy subcutaneous immunotherapy (SCIT): Subcutaneous Allergen Immunotherapy Subcutaneous immunotherapy (SCIT) consists of gradual administration of increasing amounts of the allergen to which the individual is sensitive, in order to temper the immune response and to alleviate allergic symptoms. Subcutaneous injection immunotherapy is an established form of treatment and may be considered for individuals with symptoms of allergic rhinitis, allergic conjunctivitis, or allergic asthma with natural exposure to allergens and who demonstrate specific IgE antibodies to the relevant allergen(s). SCIT is usually only recommended for the treatment of allergic respiratory disease following a period of pharmacologic management and observation. Factors to be considered in determining treatment include the severity/duration of symptoms, patient preference/acceptability, adherence, medication requirements, response to avoidance measures, and the adverse effects of medications. The expected response to immunotherapy is antigen specific and depends on the accurate identification and selection of component allergens based on the individual's history, exposure and diagnostic test results (skin testing or serum/in-vitro testing). Subcutaneous venom immunotherapy is recommended for those who have had a systemic reaction to Hymenoptera (e.g., bee, wasp, yellow jacket, hornet, ant) stings and demonstrate Hymenoptera-specific IgE antibodies, especially if the reaction was associated with respiratory symptoms, cardiovascular symptoms, or both. There is insufficient evidence to support the use of allergen immunotherapy for other indications, including atopic dermatitis, food hypersensitivity, chronic urticaria, or angioedema [23-26].
Acupuncture: Acupuncture has been used by allergic patients for the relief of allergic rhinitis, asthma, allergic dermatoses and patients who have other symptoms or medical problems that they consider to be allergic. Despite the report by some patients of temporary benefit, this is an unproven form of allergy therapy due to a lack of published scientific literature [24,26].
Allergoids: Allergoids are allergenic proteins that are treated with formaldehyde to produce larger molecules with decreased ability to react with IgE antibodies. Allergoids are licensed and manufactured for general distribution in Europe but are not available in the United States [23,24].
Autogenous urine injection: Autogenous urine injection revolves around the theory that urine produced by the patient contains unspecified chemicals during an allergic reaction and that injection of these chemicals inhibits or neutralizes future allergic reactions. There is no scientific evidence to support autogenous urine injections. Repeated injections of these antigens could induce autoimmune nephritis.
Detoxification: Detoxification is a method used by individuals who believe that an allergic state can be induced by toxic damage to the immune system from exposure to environmental chemicals. It is believed that certain lipid-soluble chemicals may be stored in body fat for long periods. Detoxification consists of sauna and exercise. The individual ingests high-dose niacin to induce erythema. Body fluids are replenished with water and electrolytes and certain essential oils are consumed, presumably to help replace fat-soluble chemical contaminants. This procedure takes approximately five hours and is repeated daily for 20-30 days. This form of therapy has not been well-studied and is unproven. Environmental chemical avoidance: Individuals with idiopathic environmental intolerance, formerly referred to as multiple chemical sensitivity, have been described as failing to adapt to synthetic chemicals. The 1999 American Academy of Allergy, Asthma, and Immunotherapy (AAAAI) position statement on idiopathic environmental intolerance states that a causal connection between environmental chemicals, foods, and/or drugs and the patient's symptoms continues to be speculative and cannot be based on the results of currently published scientific studies [24-26].
Epicutaneous immunotherapy: Epicutaneous immunotherapy involves the use of patches as a dosage form for allergen specific immunotherapy. An adverse effect of this therapy is patch-induced eczema at the patch site. This allergy treatment method remains unproven due to a lack of supporting evidence published in the peer reviewed scientific literature [25].
Helminth trichuris suis therapy: Treatment with helminth trichuris suis has been proposed as a treatment for allergic rhinitis. A therapeutic approach has been suggested in different experimental models of allergic diseases showing that live ova from trichuris suis, an intestinal helminth of pigs, can protect against allergic reactivity by helminth-induced regulatory T cells and cytokines. Bager, et al. (2010) conducted a double-blind, placebo-controlled study (n = 100) to evaluate the effectiveness of trichuris suis therapy for the treatment of allergic rhinitis. The authors reported that repeated treatment with the helminth trichuris suis induced a substantial clinical and immunologic response but had no therapeutic effect on allergic rhinitis. This allergy treatment method remains unproven due to a lack of supporting evidence published in the peer-reviewed scientific literature [25,26].
Homeopathic remedies: A homeopathic remedy administers a causative agent of a disease and is administered therapeutically in small amounts. There is no scientific evidence to support the homeopathic practice as a method for treating allergies [23,24].
Injection of food extracts: An injection of food extracts consists of a combination of foods based on skin test results or a patient's report of intolerance to foods. No clinical trials support this treatment [24-26].
Peptide therapy: The concept that the clinical response to allergen immunotherapy probably reflects the induction of nonresponsiveness in Th2 lymphocytes led to the concept of immunotherapy with allergen-derived peptides representing T cell activating epitopes that do not react with IgE antibodies [23,24].
Provocation-neutralization therapy: This treatment involves the injection of substances under the skin that are suspected of triggering an allergic reaction in sufficient quantity to cause symptoms similar to the patient's complaints. This is then followed by an immediate injection of a weaker or stronger dilution of the same antigen to relieve the symptoms [23-25].
Rhinophototherapy: Rhinophototherapy uses UV-B, UV-A, and visible light to treat allergic rhinitis. This allergy treatment method remains unproven due to a lack of supporting evidence published in the peer-reviewed scientific literature [23,24].
Rotational and multiple food elimination diets: Proponents of the concept of multiple food allergies sometimes recommend a "rotary diversified diet", in which the patient rotates foods so that the same food is eaten only once every 4-5 days to help identify foods that may cause allergic responses. This allergy treatment method remains unproven due to a lack of supporting evidence published in the peer-reviewed scientific literature [23-26].
Nasal irrigation in the management of allergic rhinitis: Nasal irrigation is in use for symptomatic relief for centuries without any scientific data to determine its efficacy [27]. Cochrane database systemic review found symptomatic relief from inflammatory symptoms in patients with rhinitis [28]. Many studies have proven that nasal saline irrigation is an important adjunctive treatment form in non-pharmacological treatment of rhinitis of different aetiologies including allergic rhinitis [29-31]. Studies have proven that nasal irrigation works better than the placebo in relief of symptoms in patients with allergic rhinitis [31]. Nasal irrigation works by washing out the allergens, thick mucus, as well as infective agents and irritants, which are responsible for inflammatory mechanism origin [28,32,33].
Allergic rhinitis is a chronic problem, inspite of all the advancements in science and new technologies for new researches in the treatment of allergic rhinitis still, we don't have confirmed curative form of treatment. The mainstay of treatment is focused on symptomatic management of different symptoms including sneezing, nasal discharge, and nasal obstruction. We are fully aware of the side effects and adverse events associated with pharmacological treatment methods available currently. Non-pharmacological treatment methods may be helpful in preventing the unwarranted side effects of medicines used in the management of allergic rhinitis. This needs more research and studies to have non-pharmacological treatment methods with better symptomatic relief so that complications of chronic inflammation can be prevented with the least complications. This form of treatment can improve the quality of life without untoward complications associated with pharmacological treatment of allergic rhinitis (AR). One of the most common non-pharmacological treatment method nasal saline irrigation helps in relieving the symptoms of allergic rhinitis, this is a cost-effective and easy to do management option in symptomatic relief of allergic rhinitis. Non-pharmacological treatment methods need further studies so that we have a healthy united airway from nose to lung.